Safety and Tolerability of Aripiprazole in the Treatment of Irritability Associated With Autistic Disorder in Pediatric Subjects (6-17 Years Old): Results From a Pooled Analysis of 2 Studies
Background: With increasing use of atypical antipsychotics among pediatric patients, detailed information about safety and tolerability is crucial.
Method: Data were pooled from two 8-week, randomized, double-blind, multicenter, parallel-group trials comparing aripiprazole versus placebo in subjects aged 6 to 17 years with irritability associated with DSM-IV-TR-diagnosed autistic disorder: one flexibly dosed (aripiprazole 2-15 mg/d; target of 5, 10, or 15 mg/d), the other fixed-dose (aripiprazole 5, 10, or 15 mg/d). The first was conducted from June 2006-April 2008, and the second, from June 2006-June 2008. Adverse events were characterized with respect to incidence, duration, severity, timing of peak incidence of onset, and dose-response relationship. Extrapyramidal symptoms, drooling, and metabolic parameters were evaluated.
Results: Three hundred thirteen subjects comprised the safety sample (aripiprazole 212, placebo 101). Discontinuations due to adverse events with aripiprazole versus placebo were, overall, 10.4% versus 6.9%; subjects 6-12 years: 10.8% versus 5.1%; and subjects 13-17 years: 8.9% versus 13.6%. Common adverse events with aripiprazole versus placebo included sedation (20.8% vs 4.0%), fatigue (16.5% vs 2.0%), vomiting (13.7% vs 6.9%), increased appetite (12.7% vs 6.9%), somnolence (10.4% vs 4.0%), and tremor (9.9% vs 0.0%). Most adverse events were mild or moderate and occurred early. Only fatigue showed a dose-response relationship (P < .05). Mean body weight change (last observation carried forward, 1.6 vs 0.4 kg) was higher with aripiprazole than placebo (P < .001). There were no between-treatment differences in metabolic changes. The extrapyramidal symptom-related adverse event incidence with aripiprazole versus placebo was, overall, 20.8% vs 9.9%; the incidence of akathisia-related events was 3.3% vs 8.9%.
Conclusions: Aripiprazole was generally safe and well tolerated in subjects (6-17 years) with irritability associated with autistic disorder in these 8-week studies; clinicians should be aware of this clinical profile and strategies to manage adverse events if they occur.
Trial Registration: clinicaltrials.gov Identifiers NCT00332241 and NCT00337571
Prim Care Companion CNS Disord 2011;13(1):e1-e9
© Copyright 2011 Physicians Postgraduate Press, Inc.
Submitted: April 28, 2010; accepted June 17, 2010.
Published online: January 6, 2011 (doi:10.4088/PCC.10m01008gry).
Corresponding author: Adelaide S. Robb, MD, Children’s National Medical Center, 111 Michigan Ave NW, Washington, DC 20010-2970 ([email protected]).
Autistic disorder is a neurodevelopmental disorder with childhood onset that is characterized by atypical development in social interaction and communication and the presence of restricted, repetitive, and stereotyped patterns of behaviors, activities, or interests (DSM-IV-TR).1 Although not part of the diagnostic criteria, a number of associated comorbidities occur; among the most troublesome are serious behavioral disturbances, such as irritability, aggression, tantrums, rapidly changing mood, and deliberate self-injury. These impair social interaction and communication and can disrupt school and family environments.2 A need exists for treatments that are well tolerated that can relieve symptoms of irritability among pediatric patients.
Clinical Points
- All health care professionals and caregivers involved in managing children and adolescents with psychiatric disorders should be aware of potential medication side effects, be able to recognize them, and know how to take appropriate steps to manage them.
- The potential for weight changes and extrapyramidal symptoms with aripiprazole in children and adolescents with irritability associated with autistic disorder should be recognized and managed appropriately.
Atypical antipsychotics are emerging as a treatment option for patients with irritability associated with autistic disorder, with several studies demonstrating clinically relevant efficacy.3,4 However, there remains a clinical need for increased understanding of the potential benefits and risks of atypical antipsychotics and awareness of the overall clinical efficacy and tolerability profile of this treatment option. As with any emerging treatment, it is essential to establish clearly the safety and tolerability profile, especially when the treatment is used in childhood psychiatric disorders, as pediatric patients may be more susceptible to some of the side effects of antipsychotics.5
Recently, results were published from 2 studies evaluating the efficacy and safety of the atypical antipsychotic aripiprazole in the treatment of irritability associated with autistic disorder.6,7 Both trials demonstrated efficacy at endpoint on the primary outcome measure, the Aberrant Behavior Checklist (ABC) irritability subscale8 (P < .05). In order to understand more clearly the safety and tolerability profile of aripiprazole in this population, this pooled post hoc analysis was conducted to evaluate the incidence, time course, severity, and dose response of adverse events (AEs) in the safety sample across the 2 studies.
METHOD
Studies
Two studies were included in this pooled analysis; the study methods have been previously published in detail.6,7 Both were 8-week, randomized, double-blind, multicenter, parallel-group trials that evaluated the efficacy of aripiprazole versus placebo using the caregiver-rated ABC irritability subscale score as the primary outcome measure. The primary difference between the 2 trials is that one was flexibly dosed (2-15 mg/d; target dose of 5, 10, or 15 mg/d [clinicaltrials.gov identifier NCT00332241]),6 and the other had a fixed-dose schedule (5, 10, or 15 mg/d [clinicaltrials.gov identifier NCT00337571]).7 The flexible-dose trial was conducted from June 2006-April 2008, and the fixed-dose, from June 2006-June 2008. The trials were conducted in accordance with the Declaration of Helsinki, and the institutional review board at each site approved the protocol.
Subjects were 6 to 17 years of age; met Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision (DSM-IV-TR) criteria for autistic disorder; and demonstrated behaviors such as irritability, tantrums, aggression, self-injurious behavior, or a combination of these symptoms. Diagnosis was confirmed by the Autism Diagnostic Interview-Revised (ADI-R)9 using modified rater-training criteria. Subjects also were required to have a Clinical Global Impressions-Severity of Illness scale10 score ≥ 4 and an ABC irritability subscale score ≥ 18 at screening and baseline.
Overall Tolerability
Adverse events were assessed at weekly study visits except week 7, when the subject or caregiver was contacted by telephone to assess the patient’s condition. Adverse events were recorded and are reported for events occurring at a rate ≥ 5%. All AEs leading to discontinuation are reported here. Adverse events were also evaluated with respect to their peak incidence of onset by week (the week when most new-onset events occurred), severity, time to resolution, relationship to age and weight, and relationship to dose (fixed-dose study only). Descriptive statistics were generated for by-week and by-severity/by-week AE incidences for the combined group and also by age group (6-12 or 13-17 years). To test for differences in AE profiles across age groups, an odds ratio (OR) estimate was calculated and the ORs were compared across categories using the Breslow-Day test for AEs with incidence ≥ 5% in the pooled aripiprazole group. The Cochran-Armitage test was conducted to assess the dose-response relationship of AEs (ie, increased incidence of AEs with increased dose) for the fixed-dose study only. Median times to resolution of AEs occurring at a rate ≥ 5% were calculated based on Kaplan-Meier maximum likelihood estimates of the survival curves using data from the pooled studies. Subjects for whom the resolution date for a particular AE was missing (ie, AE still continuing at the end of the study or resolution date unknown) were censored at their last day of dosing when analyzing that AE. The safety sample included all subjects randomized to treatment who received at least 1 dose of study medication.
Metabolic Health Outcomes
Changes from baseline in mean weight and median body mass index (BMI) were evaluated using analysis of covariance, with double-blind treatment as a main effect, protocol as a stratification effect, and baseline assessment as a covariate, last observation carried forward (LOCF). Weight change and median BMI change were also evaluated by analysis of covariance using the observed case (OC), or completer, data set. A z score analysis, which takes into account weight gain during development, was also conducted. Median percentage change from baseline in fasting metabolic and glucose laboratory measurements was recorded. The proportion of subjects with a baseline fasting metabolic measure within normal limits who had a subsequent on-treatment abnormal level of a fasting metabolic measurement was evaluated.
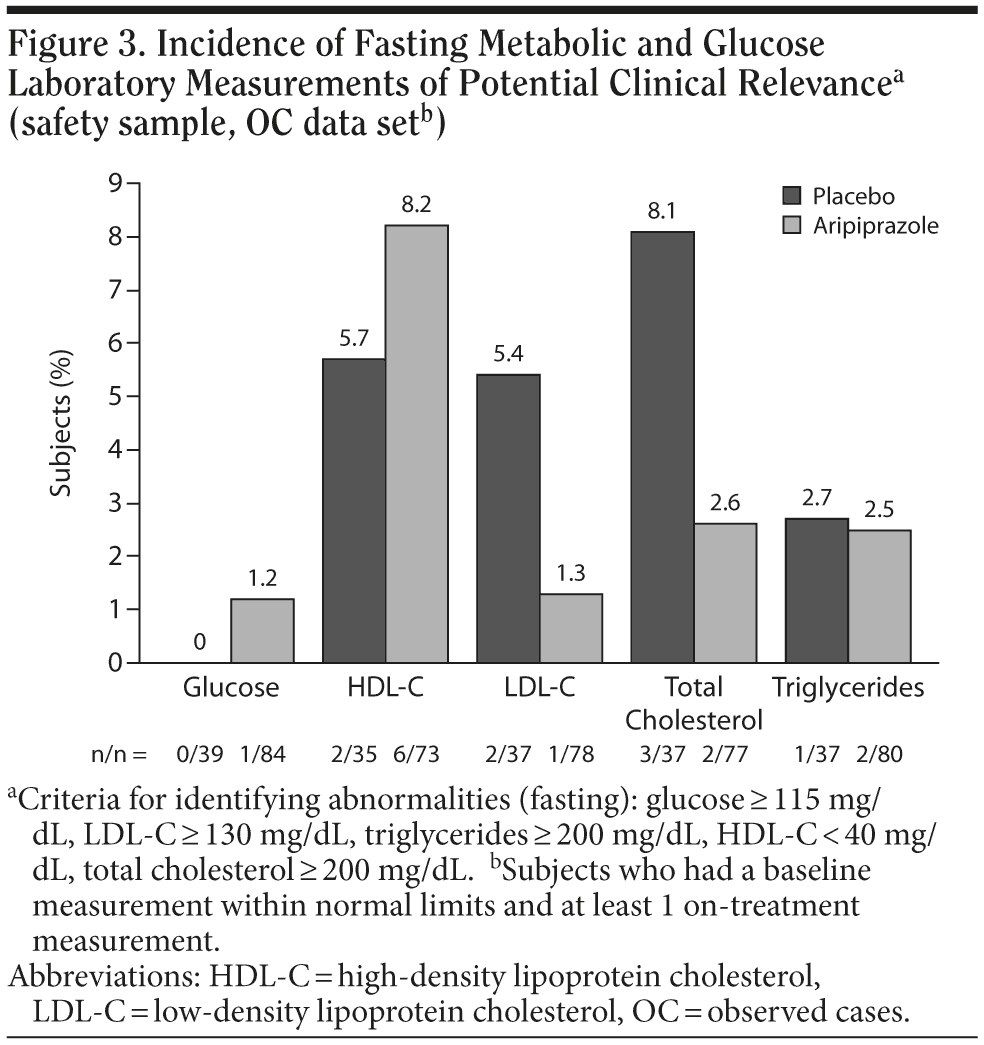
Abnormal metabolic parameters were defined as follows: glucose ≥ 115 mg/dL, low-density lipoprotein cholesterol ≥ 130 mg/dL, triglycerides ≥ 200 mg/dL, high-density lipoprotein cholesterol < 40 mg/dL, and total cholesterol ≥ 200 mg/dL.
Extrapyramidal Symptoms
Extrapyramidal symptoms (EPS) consist of a constellation of side effects. The study protocols defined the following adverse events as extrapyramidal symptoms: generalized rigidity, hyperkinesia, bradykinesia, akinesia, dystonia, hypertonia, akathisia, tremor, flexed posture, involuntary muscle contractions, athetosis, and chorea. If a subject was experiencing 2 or more of these symptoms concurrently, this was considered extrapyramidal syndrome, which was coded by MedDRA as “extrapyramidal disorder.” The EPS-related adverse events were grouped into 5 categories: dystonic events, parkinsonian events, akathisia events, dyskinetic events, and residual events.
Extrapyramidal symptoms (not including drooling) were recorded and characterized with respect to incidence of onset by week, severity, discontinuations, and use of concomitant medications for treatment. Further characterization in terms of their relationship to age, weight, and dose (for any occurring ≥ 5% in either arm of the fixed-dose study only) was also conducted. Severity of EPS was assessed using the Simpson-Angus Scale (SAS),11 the Barnes Akathisia Rating Scale,12 and the Abnormal Involuntary Movement Scale (AIMS).13
Drooling was not classified as an extrapyramidal symptom in each trial’s prespecified definition, but because of a potential relationship between extrapyramidal symptoms and drooling, it was considered separately in this analysis with respect to incidence, incidence of onset by week, severity, and relationship to dose.
RESULTS
A total of 316 subjects were randomized (aripiprazole 213, placebo 103), and a total of 313 subjects comprised the safety sample (aripiprazole 212, placebo 101). The mean age of subjects exposed to aripiprazole was 9.6 years, and the majority (78.8%) of subjects were between the ages of 6 and 12 years. Subjects were predominantly male (88.7%) and Caucasian (71.2%) (Table 1). The disposition of subjects across the study is shown in Figure 1.
Overall Tolerability
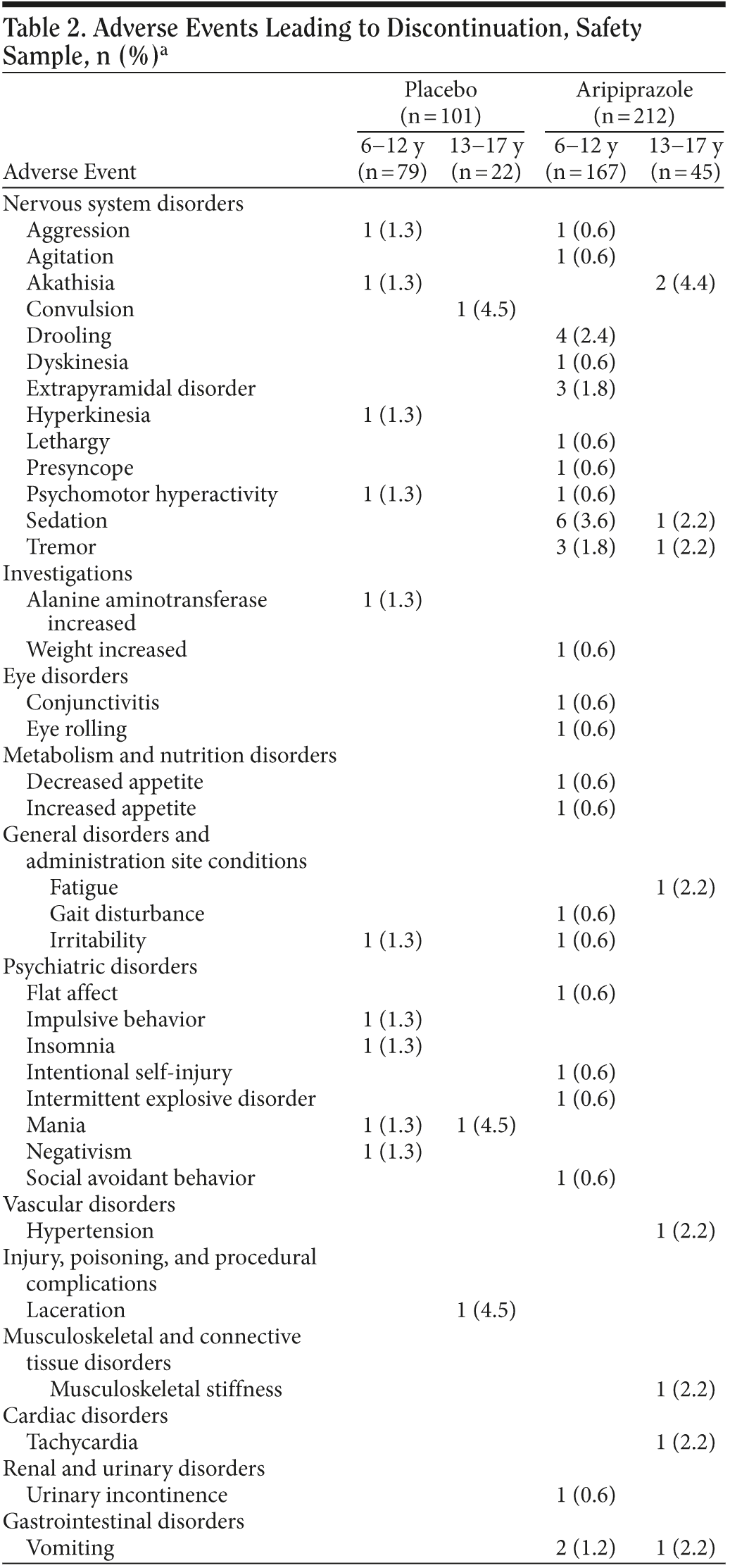
Discontinuations due to adverse events. Adverse events that led to discontinuation in the safety sample of 313 patients (those subjects receiving at least 1 dose of study medication) are shown in Table 2. The percentages of subjects who discontinued due to at least 1 AE with aripiprazole versus placebo were, overall, 10.4% versus 6.9%. For the 2 age categories, the percentages were as follows: subjects aged 6-12 years, 10.8% versus 5.1%; subjects aged 13-17 years, 8.9% versus 13.6%.
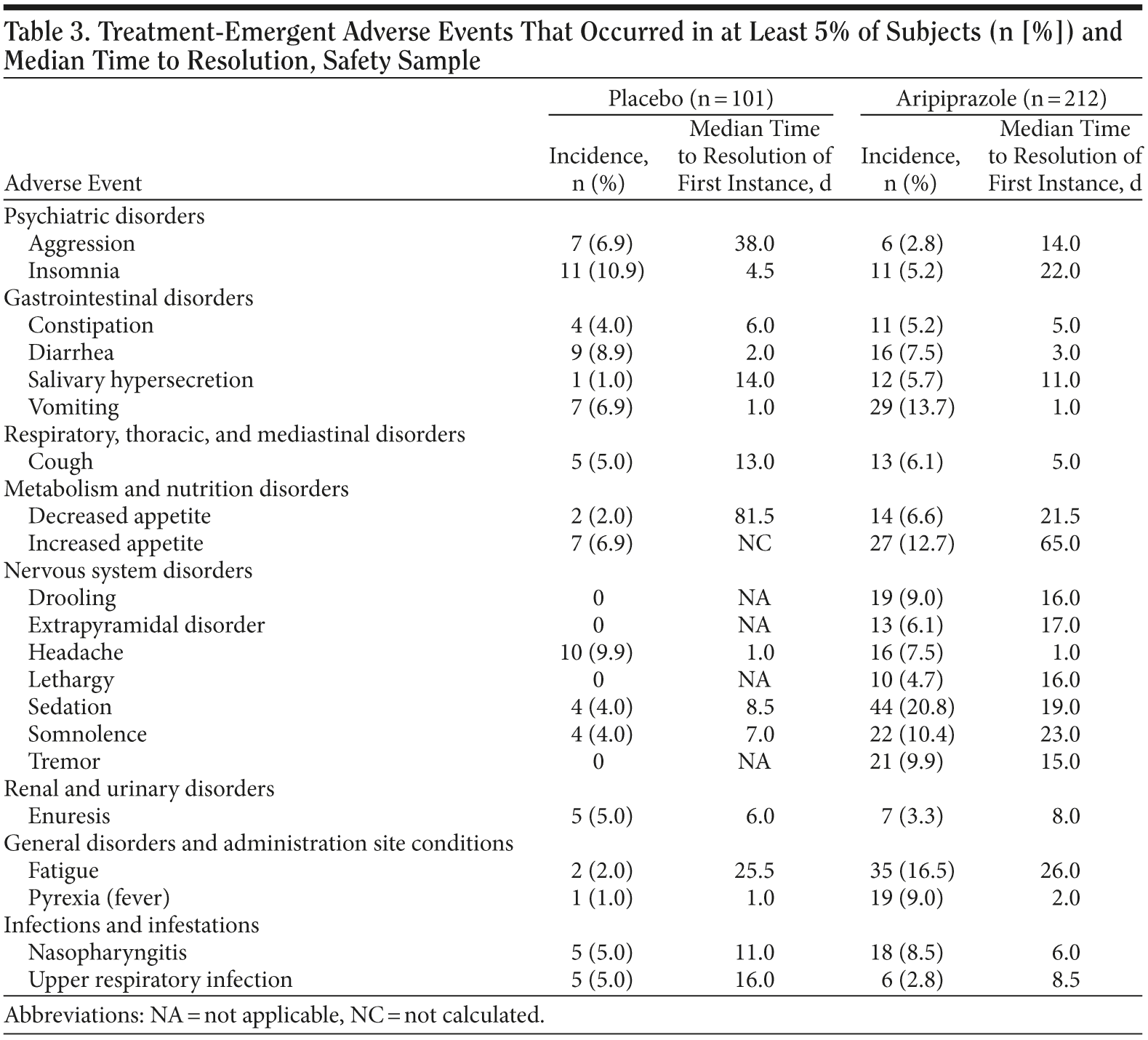
Incidence of adverse events, timing of peak incidence of onset, time to resolution, and age group comparison. Adverse events with an incidence of ≥ 5% are shown in Table 3. The most common AEs seen in the aripiprazole-treated group (vs placebo) were as follows: sedation (20.8% vs 4.0%), fatigue (16.5% vs 2.0%), vomiting (13.7% vs 6.9%), increased appetite (12.7% vs 6.9%), somnolence (10.4% vs 4.0%), and tremor (9.9% vs 0.0%). There was a statistically significant difference between the age groups for the event of salivary hypersecretion (ages 6-12 years: aripiprazole, 6.6%; placebo, 0%; ages 13-17 years: aripiprazole, 2.2%; placebo, 4.5%; P = .016). Similar statistical testing (by age) was not possible for tremor, drooling, extrapyramidal disorder, or lethargy because there were no reported cases in both placebo groups and was not possible for pyrexia or constipation because there were no cases in the 13- to 17-year age group.
For all of the AEs evaluated in the aripiprazole group across all ages, the peak incidence of onset was at week 1 or 2, except for nasopharyngitis and tremor (week 3), extrapyramidal disorder and aggression (week 4), drooling (week 5), diarrhea (weeks 2 and 5), enuresis and upper respiratory tract infection (week 6), and cough (week 7). When evaluated by age group, the peak incidence of onset for drooling was at weeks 2, 3, and 5 in the 6- to 12-year age group (4 cases each) and was at weeks 1, 4, 5, and 7 in the 13- to 17-year age group (1 case each). Nasopharyngitis (6-12 years: weeks 3, 5, and 6; 13-17 years: week 3), extrapyramidal disorder (6-12 years: week 4; 13-17 years: week 4), and cough (6-12 years: week 7; 13-17 years: no incidence) also seemed to peak after week 2 of treatment. Additional AEs that showed a late peak in the 6- to 12-year subgroup were diarrhea (peaks at weeks 2 and 5), tremor (week 3), headache (weeks 1 and 3), aggression (week 4), upper respiratory tract infection (week 6), and insomnia (weeks 1, 5, and 7). Additional AEs that showed a late peak in the 13- to 17-year subgroup were salivary hypersecretion (week 3), sedation (weeks 1 and 3), increased appetite (week 3), and tremor and lethargy (week 5).
The median times to resolution of the first instance of AEs that occurred at a rate ≥ 5% are also shown in Table 3. As would be expected, there is substantial variability in the time to resolution for the AEs that were evaluated.
Severity of adverse events. Most AEs were classified as mild or moderate. Adverse events occurring in ≥ 5% of subjects and classified as severe in subjects receiving aripiprazole were (1 case each) fatigue, headache, increased appetite, and vomiting in week 1; drooling, lethargy, tremor, and sedation in week 3. Of the AEs classified as severe, most occurred in the younger age subgroup (6-12 years); the only severe AE that occurred in the 13- to 17-year age group was fatigue.
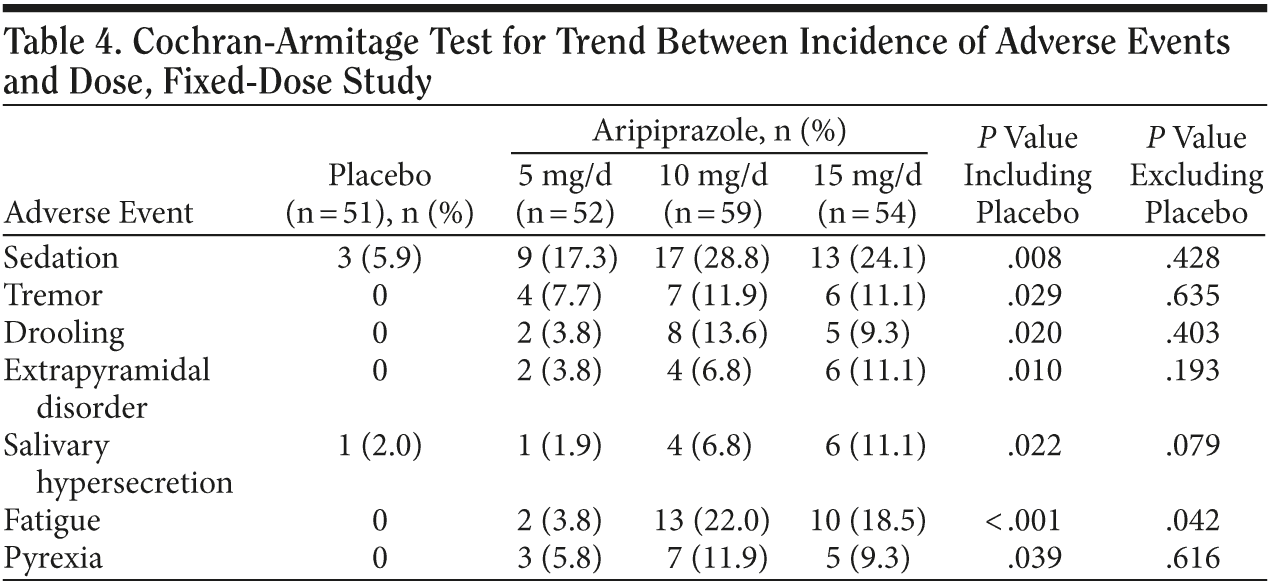
Dose-response relationship of adverse events. The Cochran-Armitage test was conducted to assess the dose-response relationship of AEs in the fixed-dose study. When placebo was included in the analysis, 7 AEs appeared to show a dose-response relationship; however, only 2 events—sedation in 3 subjects and salivary hypersecretion in 1 subject—occurred in the placebo group. When placebo was excluded from the analysis, only the AE of fatigue still showed a statistically significant dose-response relationship: placebo: 0%; aripiprazole: 5 mg/d, 3.8%; 10 mg/d, 22.0%; 15 mg/d, 18.5%; P < .05 (Table 4).
Serious adverse events. Serious AEs were reported in 2 (0.9%) aripiprazole-treated subjects. The first was an event of presyncope in an 11-year-old Caucasian male subject receiving aripiprazole 5 mg/d. This was characterized by an unsteady gait, eyes rolling back, and being near syncope. The AE was considered by the investigator to be mild in intensity and possibly related to treatment; the study drug was discontinued, and the event resolved on the same day it occurred. The second was an event of aggression in a 9-year-old African American female subject receiving aripiprazole 10 mg/d. Study medication had been discontinued the day prior to the serious adverse event due to increased agitation. The aggression was severe/very severe, resulted in an inpatient psychiatric admission, was considered not likely to be related to treatment, and resolved 13 days later.
Metabolic Health Outcomes
The adjusted mean change (LOCF) in body weight (1.6 vs 0.4 kg; P < .001) and median change in BMI (0.7 vs 0.2 kg/m2; P = .001) at endpoint were higher in the aripiprazole group than the placebo group. The adjusted mean ± SE change from baseline to endpoint in body weight z score was higher in the aripiprazole group compared with the placebo group (0.116 ± 0.02 vs -0.014 ± 0.03; P < .001), as was the median change from baseline to endpoint in BMI z score (0.11 vs 0.01; P < .05). An OC (completer) analysis showed weight gain of 1.6 kg (n = 176) with aripiprazole and 0.5 kg (n = 71) with placebo and median change in BMI of 0.7 kg/m2 (n = 173) versus 0.2 kg/m2 (n = 71), respectively (P ≤ .001 for both). Also in the OC analysis, the adjusted mean ± SE change from baseline to endpoint in body weight z score was higher in the aripiprazole group compared with the placebo group (0.133 ± 0.02 vs -0.024 ± 0.03; P < .001), as was the median change from baseline to endpoint in BMI z score (0.11 vs 0.01; P < .01).
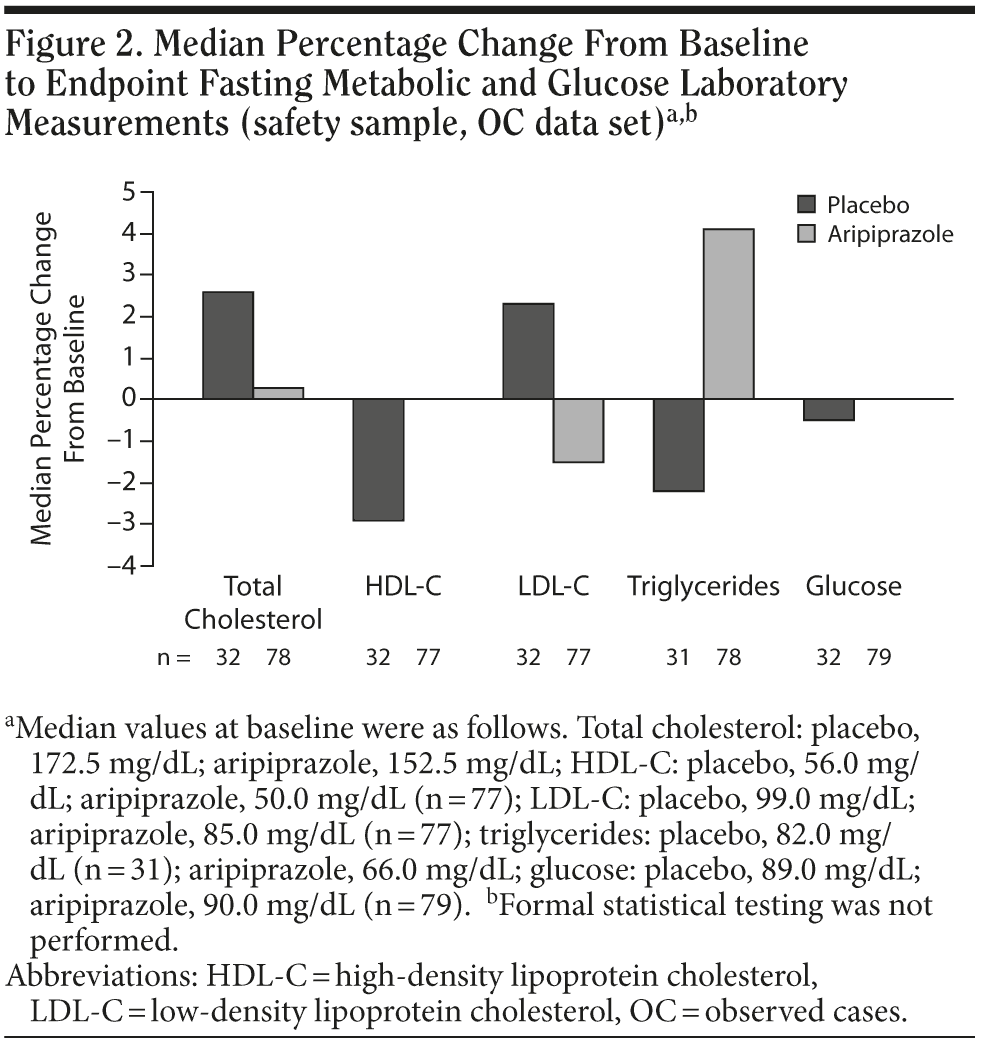
The median percentage changes from baseline to endpoint in fasting metabolic and glucose laboratory measurements among subjects receiving aripiprazole are shown in Figure 2. There were no meaningful differences between the aripiprazole and placebo groups in these metabolic parameters. Although statistical testing was not performed on the pooled sample, a nonparametric evaluation of fasting metabolic measures in the individual studies demonstrated no statistically significant differences between aripiprazole or placebo, except for a slight increase in high-density lipoprotein cholesterol in the 5-mg/d arm of the fixed-dose study (P = .035). Furthermore, the proportion of subjects with a baseline metabolic measurement within normal limits who had an abnormal on-treatment metabolic measurement is shown in Figure 3.
Extrapyramidal Symptoms
Characterization of extrapyramidal symptoms: incidence, timing, discontinuations, management. Overall, 20.8% of subjects receiving aripiprazole reported EPS-related AEs compared with 9.9% in the placebo group. The incidence of non-akathisia-related EPS-related AEs was 17.9% in aripiprazole-treated subjects and 2.0% in placebo-treated subjects. The incidence of akathisia-related AEs (akathisia, hyperkinesia, psychomotor hyperactivity, hyperkinesia, and psychomotor agitation) was 3.3% in the aripiprazole-treated group and 8.9% in the placebo group. The most frequently reported EPS-related AEs in the aripiprazole-treated group were tremor (9.9% vs 0% for placebo) and extrapyramidal disorder (defined as 2 or more simultaneous extrapyramidal symptoms; 6.1% vs 0% for placebo). The peak incidence of onset for tremor occurred at week 3 and at week 4 for extrapyramidal disorder.
Discontinuation due to EPS occurred in 4.2% of subjects receiving aripiprazole and 3.0% of subjects receiving placebo. The most common EPS leading to discontinuation were tremor (aripiprazole: 4 cases [1.9%]—1 case each at weeks 2 and 3 and 2 cases at week 5; placebo: 0 cases) and extrapyramidal disorder (aripiprazole: 3 cases [1.4%], 1 each at weeks 2, 3, and 4; placebo: 0 cases). There were 4 cases (1.9%) of drooling leading to discontinuation (aripiprazole: 2 cases each at weeks 2 and 3; placebo: 0 cases). The incidence of onset of all EPS generally peaked at or between week 3 and week 5.
In the flexible-dose study, 1 subject in the aripiprazole-treated group received benztropine for the treatment of EPS-related AEs. In the fixed-dose study, 2 subjects in the 5-mg/d arm received propranolol; 2 subjects in the 5-mg/d arm, 1 subject in the 10-mg/d arm, and 5 subjects in the 15-mg/d arm received benztropine; and 1 placebo-treated subject and 2 aripiprazole 5-mg/d-treated subjects received propranolol.
Of all of the drooling, extrapyramidal disorder, and tremor events that occurred, 38 were classified as mild, 13 as moderate, and 2 as severe (1 drooling and 1 tremor). There was a significant difference between aripiprazole and placebo on the mean change from baseline to endpoint (LOCF) in SAS (0.1 vs -0.4, P = .03) scores and a significantly greater decrease (improvement) than placebo on the AIMS (-0.4 vs 0, P = .005) scores, but no difference on the Barnes (0 vs 0.1, P = .106) scores.
Drooling: incidence and timing. The incidence of drooling among subjects receiving aripiprazole versus placebo was 9.0% vs 0%. For drooling, 14 events were classified as mild, 4 as moderate, and 1 as severe. The incidence of onset of drooling peaked at week 5.
Dose-response relationship of extrapyramidal symptoms. Tremor, drooling, and extrapyramidal disorder showed a statistically significant (P < .05) dose-response relationship when placebo was included in the analysis. However, when placebo was excluded from the analysis, no statistically significant dose-response relationship was observed (Table 4).
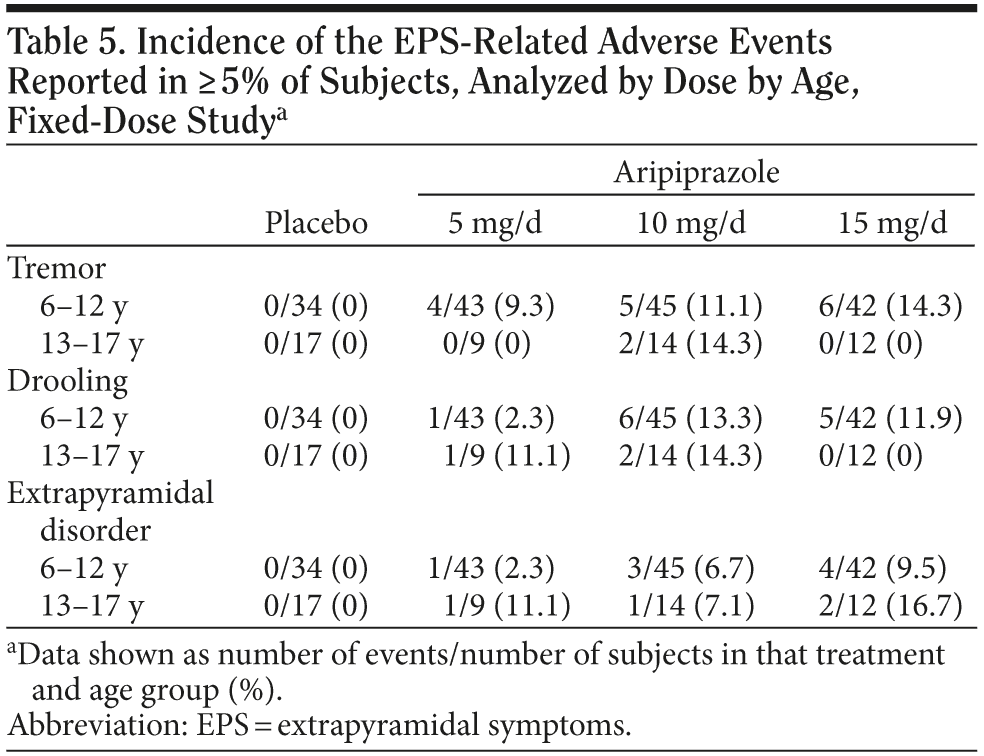
Further characterization of extrapyramidal symptoms by dose, age group, and body weight. The percentage of subjects reporting any EPS-related event was similar between the 6- to 12-year-olds (21.6%) and the 13- to 17-year-olds (17.8%). For the EPS-related AEs that occurred in ≥ 5% of subjects—tremor, drooling, and extrapyramidal disorder—Table 5 shows the incidence by dose and age in the fixed-dose trial. Tremor appeared more commonly in younger subjects than in older subjects. Also in younger subjects, tremor, drooling, and extrapyramidal disorder all appear to be more common at doses ≥ 10 mg/d than at 5 mg/d.
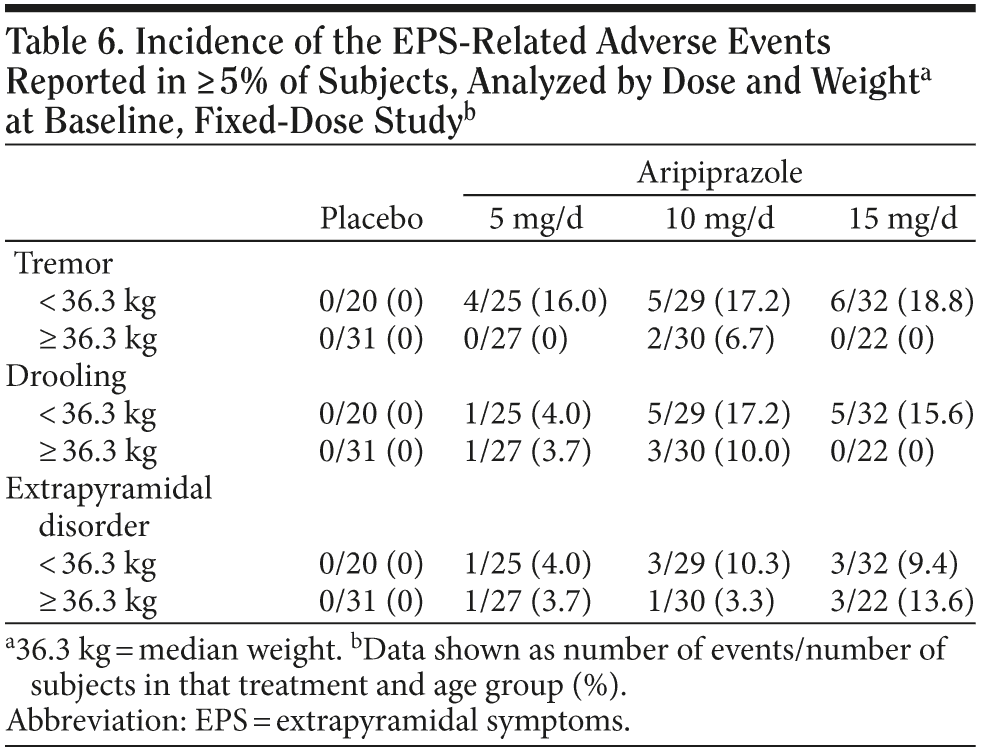
Table 6 demonstrates the incidence by dose and median weight for tremor, drooling, and extrapyramidal disorder for subjects in the fixed-dose trial. Tremor and drooling were more common in subjects weighing less than the median. There did not appear to be a strong dose-response relationship for tremor, but drooling appeared more commonly at ≥ 10 mg/d in subjects weighing less than the median weight.
In an analysis by dose in the fixed-dose study, there was a statistically significant difference between the 10-mg/d aripiprazole dose group and placebo on the mean change from baseline to endpoint (LOCF) in SAS score (5 mg/d: 0, P = .336; 10 mg/d: 0.7, P = .007; 15 mg/d: 0.2, P = .151 [vs placebo -0.4]). There were significantly greater decreases with the 5-mg/d, 10-mg/d, and 15-mg/d aripiprazole dose groups versus placebo on the AIMS scores (5 mg/d: -0.2, P = .002; 10 mg/d: -0.1, P = .022; 15 mg/d: -0.2, P = .002 [vs placebo 0.2]), but no difference on the Barnes scores (5 mg/d: 0, P = .213; 10 mg/d: 0, P = .108; 15 mg/d: 0, P = .068 [vs placebo 0.2]).
DISCUSSION
This analysis provides a comprehensive overview of the safety and tolerability profile of aripiprazole in subjects with irritability associated with autistic disorder, which may help in understanding how best to use this treatment option. It is essential for clinicians to recognize and manage AEs, especially in pediatric populations treated with atypical antipsychotics, as it has been suggested that children may be at higher risk than adults for a number of AEs—in particular, extrapyramidal symptoms and metabolic abnormalities.14 This post hoc, pooled analysis of two 8-week trials of aripiprazole for the treatment of irritability in pediatric subjects (6-17 years) with autistic disorder demonstrates that, for the most part, AEs, when they occur, are largely mild to moderate in severity, are manageable, and, with the potential exception of weight gain, resolve over time. Common AEs of note seen with aripiprazole treatment were sedation, fatigue, vomiting, increased appetite, somnolence, and tremor. Peak incidence of onset of most AEs occurred early during treatment, with the exception of extrapyramidal symptoms, which tended to occur later and frequently after the stable dose had been achieved. Tremor appeared to occur more often in younger subjects, and both tremor and drooling were seen more frequently in subjects weighing less than the median. It is also important to note the dose-response relationship observed with respect to fatigue.
Extrapyramidal symptom-related AEs should be distinguished from dyskinesia that may occur due to switching between medications. It is important to recognize extrapyramidal symptoms as a constellation of side effects, usually seen in broad clinical terms as akathisia, dystonia, parkinsonism, or, occasionally, these events in combination. In addition to their effects on function, they are thought to have a significant impact on subjective tolerability and adherence to medication.15 Early recognition, understanding of expected time course, and management can reduce the burden of these potential side effects.
In these short-term studies, there was a significant increase in weight, although subjects’ lipid and glucose parameters remained stable. Although triglyceride changes seemed to differ between placebo and aripiprazole groups, it should be noted that the baseline values for the 2 groups differed, limiting interpretation of this finding. Clinical interpretation of the weight and metabolic findings should also consider that subjects entering the studies were on average overweight at baseline, approximately 1 standard deviation above the sex- and age-adjusted average weight, and this was more pronounced in the placebo group. Thus, at least in the short term, it seems unlikely that there is any clinically meaningful difference in metabolic effects between aripiprazole and placebo in this population. However, a detailed analysis of changes in metabolic parameters for those subjects who gained significant weight is warranted, as are longer-term data. It is recommended that clinicians and caregivers increase their awareness of potential metabolic effects of antipsychotics and minimize risk through careful subject selection, healthy lifestyle counseling, and close physical health monitoring.16 Recent evidence confirms that atypical antipsychotics are associated with weight gain in pediatric subjects; however, these agents do differ in their effects on metabolic parameters.17 Weight increases can be easily monitored, as weight is an easily conducted assessment; if an increase is present, the clinician should discuss with the patient and the patient’s caregivers whether or not to continue treatment.
Strengths of the study include the large overall subject population from this pooled analysis. The findings are, however, limited by their post hoc nature. Also, the results are not generalizable to other conditions, as these clinical studies were designed for registrational purposes and were therefore limited to those subjects who met the specific entry criteria. Finally, although these analyses can provide some insight into the short-term expectations around AEs, it will be necessary to have high-quality long-term data in order to more fully understand the safety and tolerability profile of aripiprazole in this population.
CONCLUSIONS
Aripiprazole was generally safe and well tolerated in the treatment of pediatric subjects (6-17 years) with irritability associated with autistic disorder. It is vital that all health care professionals and caregivers for children and adolescents with irritability associated with autistic disorder are aware of, can recognize, and know how to take appropriate steps to manage medication side effects. During treatment of subjects with irritability associated with autistic disorder with aripiprazole, weight changes and extrapyramidal symptoms deserve close attention by the prescribing physician; however, it should be considered that early-onset side effects most likely will not, in general, persist over time. Further long-term data would add to the clinical evidence base for this agent as a treatment option for children with irritability associated with autistic disorder.
Drug names: aripiprazole (Abilify), benztropine (Cogentin and others), propranolol (Inderal, InnoPran, and others).
Author affiliations: Center for Clinical and Community Research, Children’s National Medical Center, Washington, DC (Dr Robb); Bristol-Myers Squibb Company, Plainsboro, New Jersey (Drs Andersson, Bellocchio, and Mankoski); Bristol-Myers Squibb Company, Wallingford, Connecticut (Drs Manos, Marcus, and Owen); and Otsuka America Pharmaceutical Inc, Rockville, Maryland (Dr Rojas-Fernandez and Ms Mathew).
Potential conflicts of interest: Dr Robb is a consultant to Lundbeck, Otsuka, Forest, Bristol-Myers Squibb, and Eli Lilly; has research contracts with Janssen, Supernus, Sepracor, GlaxoSmithKline, National Institute of Child Health and Human Development, Pfizer, and Bristol-Myers Squibb; is a member of the speakers bureau for Forest, McNeil, Bristol-Myers Squibb, and Eli Lilly; is a member of the advisory board for Otsuka and Eli Lilly; and receives royalties from Epocrates. Drs Andersson, Bellocchio, and Mankoski are employees of and stock shareholders in Bristol-Myers Squibb, Drs Manos and Marcus are employees of Bristol-Myers Squibb, and Dr Owen is a former employee of Bristol-Myers Squibb. Dr Rojas-Fernandez and Ms Mathew are employees of Otsuka America.
Funding/support: These studies were supported by Bristol-Myers Squibb (Princeton, New Jersey) and Otsuka Pharmaceutical Co, Ltd (Tokyo, Japan). Editorial support for the preparation of this manuscript was provided by Ogilvy Healthworld Medical Education; funding was provided by Bristol-Myers Squibb.
Previous presentations: Presented at the 56th Annual Meeting of the American Academy of Child and Adolescent Psychiatry, October 27-November 1, 2009, Honolulu, Hawaii; the 61st Annual Meeting of the American Academy of Neurology, April 25-May 2, 2009, Seattle, Washington; and the Congress of Neurological Surgeons, October 24-29, 2009, New Orleans, Louisiana.
REFERENCES
1. American Psychiatric Association. Diagnostic and Statistical Manual for Mental Disorders, Fourth Edition, Text Revision. Washington, DC: American Psychiatric Association; 2000.
2. Volkmar F, Cook E Jr, Pomeroy J, et al; American Academy of Child and Adolescent Psychiatry. Summary of the practice parameters for the assessment and treatment of children, adolescents, and adults with autism and other pervasive developmental disorders. J Am Acad Child Adolesc Psychiatry. 1999;38(12):1611-1616. PubMed doi:10.1097/00004583-199912000-00028
3. McDougle CJ, Stigler KA, Erickson CA, et al. Atypical antipsychotics in children and adolescents with autistic and other pervasive developmental disorders. J Clin Psychiatry. 2008;69(suppl 4):15-20. PubMed
4. Malone RP, Waheed A. The role of antipsychotics in the management of behavioural symptoms in children and adolescents with autism. Drugs. 2009;69(5):535-548. PubMed doi:10.2165/00003495-200969050-00003
5. Correll CU. Assessing and maximizing the safety and tolerability of antipsychotics used in the treatment of children and adolescents. J Clin Psychiatry. 2008;69(suppl 4):26-36. PubMed
6. Owen R, Sikich L, Marcus R, et al. Aripiprazole in the treatment of irritability in children and adolescents with autistic disorder. Pediatrics. 2009;124(6):1533-1540. PubMed doi:10.1542/peds.2008-3782
7. Marcus R, Owen R, Kamen L, et al. A placebo-controlled, fixed-dose study of aripiprazole in children and adolescents with irritability associated with autistic disorder. J Am Acad Child Adolesc Psychiatry. 2009;48(11):1110-1119. PubMed doi:10.1097/CHI.0b013e3181b76658
8. Aman MG, Singh NN. Aberrant Behavior Checklist: Manual. East Aurora, NY: Slosson Educational Publications; 1986.
9. Lord C, Rutter M, Le Couteur A. Autism Diagnostic Interview-Revised: a revised version of a diagnostic interview for caregivers of individuals with possible pervasive developmental disorders. J Autism Dev Disord. 1994;24(5):659-685. PubMed doi:10.1007/BF02172145
10. Guy W. ECDEU Assessment Manual for Psychopharmacology. US Department of Health, Education, and Welfare publication (ADM) 76-338. Rockville, MD: National Institute of Mental Health; 1976: 218-222.
11. Simpson GM, Angus JW. A rating scale for extrapyramidal side effects. Acta Psychiatr Scand suppl. 1970;45(S212):11-19. PubMed doi:10.1111/j.1600-0447.1970.tb02066.x
12. Barnes TR. A rating scale for drug-induced akathisia. Br J Psychiatry. 1989;154(5):672-676. PubMed doi:10.1192/bjp.154.5.672
13. Guy W. Abnormal Involuntary Movement Scale (AIMS). ECDEU Assessment Manual for Psychopharmacology. US Department of Health, Education, and Welfare publication (ADM) 76-338. Rockville, MD: National Institute of Mental Health; 1976: 534-537.
14. Vitiello B, Correll C, van Zwieten-Boot B, et al. Antipsychotics in children and adolescents: increasing use, evidence for efficacy and safety concerns. Eur Neuropsychopharmacol. 2009;19(9):629-635. PubMed doi:10.1016/j.euroneuro.2009.04.008
15. Pierre JM. Extrapyramidal symptoms with atypical antipsychotics: incidence, prevention and management. Drug Saf. 2005;28(3):191-208. PubMed doi:10.2165/00002018-200528030-00002
16. Correll CU, Carlson HE. Endocrine and metabolic adverse effects of psychotropic medications in children and adolescents. J Am Acad Child Adolesc Psychiatry. 2006;45(7):771-791. PubMed
17. Correll CU, Manu P, Olshanskiy V, et al. Cardiometabolic risk of second-generation antipsychotic medications during first-time use in children and adolescents. JAMA. 2009;302(16):1765-1773. PubMed doi:10.1001/jama.2009.1549