Efficacy of Duloxetine on Painful Physical Symptoms in Major Depressive Disorder for Patients With Clinically Significant Painful Physical Symptoms at Baseline: A Meta-Analysis of 11 Double-Blind, Placebo-Controlled Clinical Trials

ABSTRACT
Objective: To review efficacy of duloxetine for physical symptoms and depressive illness in patients with at least mild to moderate major depressive disorder (MDD; DSM-IV) and clinically significant painful physical symptoms at baseline.
Data Sources: Global database of duloxetine clinical trials (Eli Lilly and Company).
Study Selection: All 11 acute, double-blind, placebo-controlled studies of duloxetine (7 with duloxetine 60-mg doses and 4 with non-60-mg doses) in the database that used a scale to measure painful physical symptoms and were completed before March 17, 2011.
Data Extraction: For each study, patients with clinically significant pain levels at baseline (Visual Analog Scale overall pain rating ≥ 30, Numerical Rating Scale score ≥ 3, or Brief Pain Inventory 24-hour average pain rating ≥ 3) were selected in order to determine the effect sizes of duloxetine (compared with placebo for each trial) on the pain and depression measures. Overall effect sizes for both painful physical symptoms and MDD were obtained from the mean of individual-trial effect sizes, and each effect size was weighted relative to the number of patients within each study.
Data Synthesis: The overall mean effect sizes were as follows: painful physical symptoms—60-mg trials, 0.29 (95% CI, 0.06 to 0.52); non-60-mg trials, 0.13 (95% CI, −0.19 to 0.45); MDD—60-mg trials, 0.29 (95% CI, 0.18 to 0.40); non-60-mg trials, 0.16 (95% CI, 0.00 to 0.32). Across the 11 studies, the weighted effect size for painful physical symptoms was 0.26 (95% CI, 0.00 to 0.51) and for MDD, 0.25 (95% CI, 0.16 to 0.34).
Conclusions: According to this meta-analysis, duloxetine 60 mg once daily is as effective in improving painful physical symptoms as it is for depression in patients with MDD and clinically significant painful physical symptoms. The results of this meta-analysis indicate that duloxetine has small effect sizes in reducing painful physical symptoms and depressive symptoms in patients with MDD and clinically significant pain levels at baseline. Thus, the results of the study permit one to conclude that duloxetine has a clinically significant impact on painful physical symptoms and in reducing the severity of depressive symptoms. However, the results do not address its efficacy compared to other alternatives, as in all studies the comparator was placebo.
Prim Care Companion CNS Disord 2011;13(6):doi:10.4088/PCC.11r01181
© Copyright 2011 Physicians Postgraduate Press, Inc.
Submitted: March 17, 2011; accepted June 23, 2011.
Published online: December 15, 2011
Corresponding author: Susan G. Ball, PhD, Eli Lilly and Company, Lilly Corporate Center, Indianapolis, IN 46285 ([email protected]).
Although the diagnosis of major depressive disorder (MDD) is based on a number of core symptoms, painful physical symptoms are increasingly recognized as frequently associated symptoms that have clinical relevance for the patient’s outcome.1 In a naturalistic study of 573 outpatients with MDD, pain was reported by more than two-thirds of depressed patients at baseline, with the severity of pain rated as mild in 25% of patients, moderate in 30%, and severe in 14%.2 In the Re-Engineering Systems for Primary Care Treatment of Depression (RESPECT) study, 405 patients with depression were followed in primary care settings while receiving either treatment as usual or enhanced intervention.3 At baseline, painful physical symptoms of sufficient severity to impact daily activities, at least moderately, were present in 42% of the patients. Although there was some improvement in pain, the degree of severity was at least moderate for 32% of the patients at 6 months, and the severity of pain negatively impacted both response and remission rates with treatment.3 Similarly, Leuchter and colleagues4 found that, among patients in the Sequenced Treatment Alternatives to Relieve Depression (STAR*D) trial, 80% of patients reported having painful physical symptoms, and these symptoms were associated with lower remission rates and with longer time to remission. However, in the STAR*D trial, painful physical symptoms were no longer predictive of depression outcomes after adjustment for race, medical comorbidity, and severity of depression.4 Other studies have demonstrated a negative impact of painful physical symptoms on outcome measures such as increase in treatment costs,5 decrease in productivity,6 and poor quality of life.7 Thus, painful physical symptoms associated with depression present an important target for therapeutic intervention.
The neurobiological pathways underlying depression and pain suggest commonalities in the activity of serotonin and norepinephrine transmission.8 Duloxetine is a selective serotonin-norepinephrine reuptake inhibitor (SNRI) that has demonstrated both antidepressant and analgesic efficacy within different conditions including MDD, fibromyalgia, diabetic peripheral neuropathic pain, osteoarthritis, and chronic low back pain.9 With regard to studies of MDD, duloxetine showed an analgesic effect on painful physical symptoms that was partially independent of the improvement in MDD.10,11 In a pooled analysis of 2 identical studies, duloxetine 60 mg once daily significantly improved painful physical symptoms compared with placebo treatment.11 Other pooled analyses of duloxetine trials also indicated efficacy on painful physical symptoms associated with MDD, suggesting analgesic activity.12,13 Duloxetine’s efficacy was demonstrated on painful physical symptoms in elderly patients with MDD14 and in patients with MDD with at least moderate pain.15 However, there have also been other analyses that have questioned whether duloxetine does effectively reduce painful physical symptoms associated with depression. One meta-analysis of duloxetine trials, based on 5 studies, did not support the analgesic efficacy for painful physical symptoms in MDD.16 Another meta-analysis, based on 8 studies, compared duloxetine, paroxetine, or both with placebo in patients with MDD.17 Results of that meta-analysis of pain outcomes from those trials suggested that both duloxetine and paroxetine significantly improved painful physical symptoms compared with placebo, but there was not a significant difference between paroxetine and duloxetine.
Given the heterogeneity of the above analyses with regard to studies and outcomes, and the differences in analytic methods used to evaluate the efficacy of painful physical symptoms associated with MDD, the current meta-analysis was undertaken to examine the efficacy of duloxetine for both painful physical symptoms and depressive illness based on the Eli Lilly and Company global database of duloxetine clinical trials, with 11 double-blind, placebo-controlled studies selected for the analyses. Because the majority of these studies were not designed to specifically address painful physical symptoms, patients with MDD in these studies were generally not required to have these symptoms at baseline, which would necessarily limit conclusions about efficacy for painful physical symptoms in the absence of the symptoms. Therefore, the present meta-analysis also specifically examined the efficacy of duloxetine for painful physical symptoms in patients with MDD who had clinically significant painful physical symptoms at baseline.

- Many patients with depression also experience painful physical symptoms.
- In patients with painful physical symptoms, duloxetine 60 mg once daily had a clinically significant impact on those symptoms and in reducing the severity of depressive symptoms.
METHOD
Trials
The 11 clinical trials used in this individual trial-level meta-analysis are listed in Table 1 along with the study codes, citations, and clinical trial registry information (if available). The dosing groups examined included duloxetine 40 mg (20 mg twice daily), 60 mg once daily, 80 mg (40 mg twice daily), and 120 mg (60 mg twice daily). For convenience and clarity, hereafter, total daily doses will be expressed throughout the article. All studies were randomized, double-blind, and placebo-controlled, with a duration of 8 to 12 weeks for the acute therapy. Baseline demographic and clinical characteristics of the patients in each trial were described in the respective publications listed in Table 1. These 11 studies were selected as they each used a scale to measure painful physical symptoms. The cutoff to include these studies in this meta-analysis was its first submission date of March 17, 2011. As such, the unpublished data from 2 other recently concluded studies (HMGR and HMGU) were not included.
Table 1. Duloxetine Clinical Trials and Pain Measures Used in the Effect Size Analysisa
60-mg/d dose
HMFS-B
Non-60-mg/d dosec
aFor major depressive disorder effect sizes, the 17-item Hamilton Depression Rating Scale total score was used.
bThese are the company study codes.
cDoses include 40 mg/d, 80 mg/d, and 120 mg/d.
Abbreviations: BPI = Brief Pain Inventory, NRS = Numerical Rating Scale,
VAS = Visual Analog Scale.
Patients
The entry criteria were similar across all the studies. Study participants were outpatients, including men and women at least 18 years of age who met the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV)25 criteria for MDD. Patients must have signed an informed consent form prior to participating in the respective study. To be eligible for enrollment, patients must have scored ≥ 4 on the Clinical Global Impressions-Severity of Illness scale (CGI-S)26 and had a total score of at least ≥ 15 on the 17-item Hamilton Depression Rating Scale (HDRS17)27 or a total score of ≥ 20 on the Montgomery-Asberg Depression Rating Scale (MADRS)28 when appropriate. In addition, for studies HMDH and HMCB, patients were required to have pain ratings of at least mild to moderate on the average pain item of the Brief Pain Inventory29 (BPI; score of ≥ 2 in HMCB; ≥ 3 in HMDH). Exclusion criteria included the following: primary Axis I disorder (other than major depression), an Axis II disorder that could interfere with study compliance, treatment-resistant depression, serious medical illness, substance abuse or dependence within the prior year or positive drug screen test, and current use of central nervous system medication.
Measures
Overall pain was assessed using different tools, including a Visual Analog Scale (VAS) for overall pain, BPI average pain severity, and a Numerical Rating Scale (NRS) for average pain. For the current effect size analysis, only the subset of patients from each of the trials that met the threshold for clinically significant pain were included for both analyses of painful physical symptoms and MDD. For each study, patients with clinically significant levels of pain at baseline (a VAS overall pain rating ≥ 30, an NRS score ≥ 3, or a BPI 24-hour average pain rating ≥ 3) were selected to determine the individual trial effect sizes on the pain outcome measure. For MDD assessment, HDRS17 total score was used in all studies with the exception of study HMDH, in which MADRS total score was used.
Analysis of Effect Size
The effect sizes for change in pain and depression scales were determined using the Glass estimation methods.30,31 Briefly, least squares means and standard errors for main effect of treatment (each individual dose of duloxetine vs placebo) were calculated using the mixed-model repeated-measures (MMRM) analysis method. An unstructured covariance structure was used to model the within-patient errors. Kenward-Roger correction32 was used to estimate denominator degrees of freedom. In the MMRM analysis, the model included the fixed categorical effects of treatment, pooled investigative site, visit, and treatment-by-visit interaction, as well as the continuous, fixed covariates of the baseline value of the variable being analyzed and baseline value of the variable being analyzed-by-visit interaction. The baseline value of the variable being analyzed and the baseline-by-visit interaction are included to account for the differing influence over time of the baseline score on the postbaseline scores.
Effect size of each duloxetine dose compared with placebo for each individual study and weighted effect size from several studies were calculated using least squares means and standard deviations for main effect of treatment.30 Effect size for each individual study was calculated as the difference in least squares mean change between the duloxetine group (each dose group) and the placebo group divided by the standard deviation and effect size:

where m1 and m2 are the least squares means and S is the standard deviation from the MMRM analysis. Hedges33 showed that if both the experimental and the control population distributions are normal and N1 and N2 are moderate to large (at least equal to 10), the sampling distribution of effect size is approximately normal under null hypothesis with the mean of zero and variance of

To estimate the effect size for pooled duloxetine doses across different studies, a meta-analysis approach was applied.34 In this approach, the weighted effect size is computed as a weighted mean of the effect sizes from all studies. The weight for each study is equal to the precision, which is the inverse of the variance of the effect size for each study divided by the sum of the inverse of the variance of effect size for each study. It is shown that the asymptotic variance of this pooled effect size is 1/(sum of [1/variance of effect size of each study]).
An estimated 95% confidence interval (CI) for the effect size and weighted effect size was created using these estimators and the critical values from a standard normal distribution. If the lower bound of 95% of the effect size is greater than zero, it indicates that duloxetine is statistically significantly superior to placebo. The approach described here for calculating weighted effect size across studies is a fixed-effect meta-analysis approach, where we assume that the true effects investigated in the set of studies are regarded as the effects of interests that may not be generalized to the larger population.
RESULTS
Trial Characteristics
The 11 selected clinical trials were conducted by Eli Lilly and Company, Indianapolis, Indiana, and/or Boehringer Ingelheim GmbH, Ingelheim am Rhein, Germany. All were placebo-controlled, double-blind, multicenter studies investigating the efficacy of duloxetine for the treatment of MDD with treatment duration of 8 to 12 weeks. All of the studies (except HMDH) used HDRS17 total score as the primary endpoint for assessing MDD. In study HMDH, MADRS total score was the primary endpoint for measuring MDD. Except in studies HMDH and HMCB, the patients were not required to have clinically significant pain as an entry criterion at baseline. Most of these studies were disclosed and/or registered on ClinicalTrials.gov (Table 1). Additional details of the trials and patients can be found in the publications and clinical trials registry links listed in Table 1.
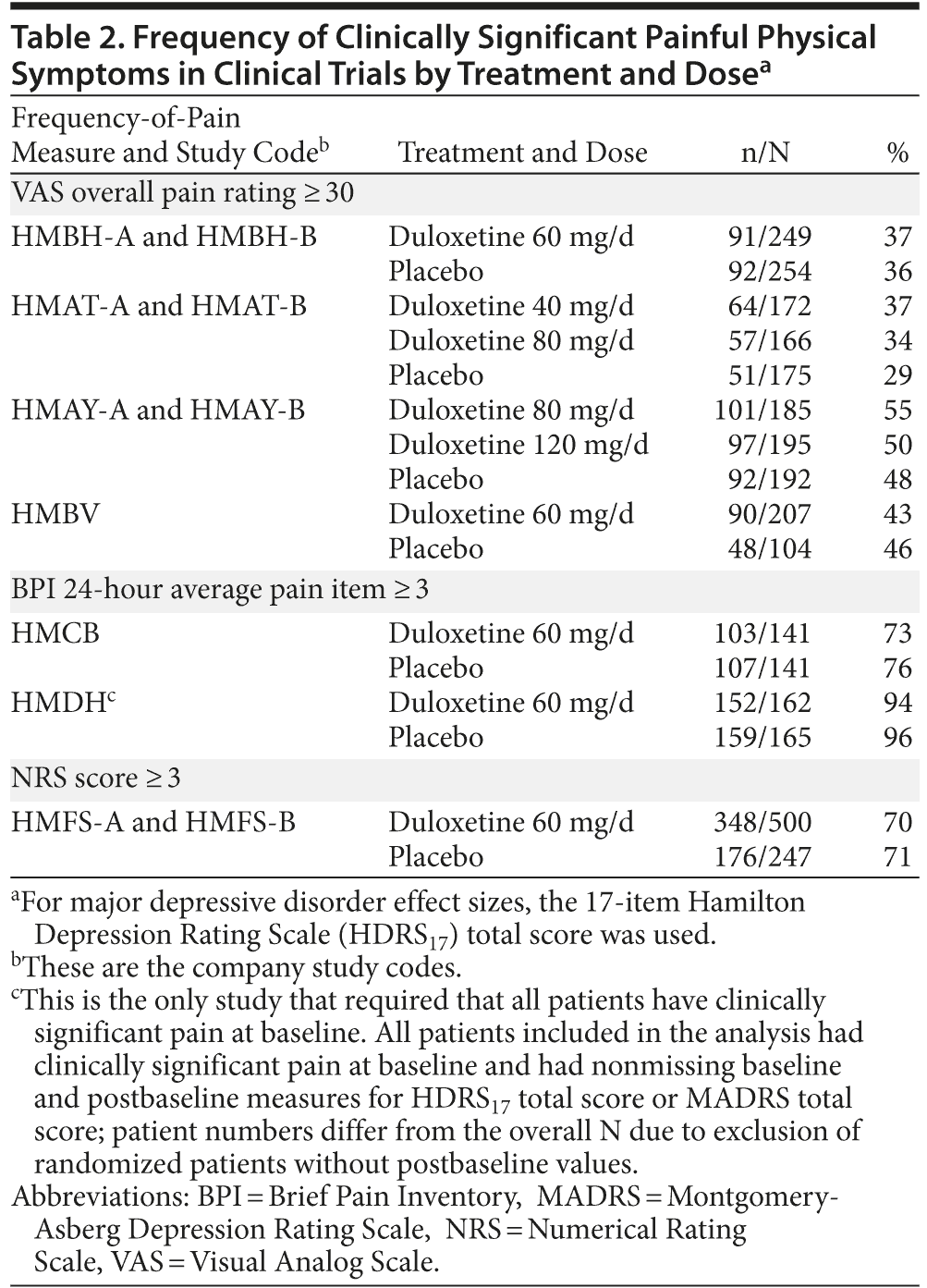
Frequency of Clinically Significant Painful Physical Symptoms
The proportions of patients with clinically significant painful physical symptoms at baseline in these trials are presented in Table 2. For trials that used the VAS measure, the proportions of patients ranged from 29% to 55% with ≤ 7% difference between duloxetine and placebo groups in these 7 trials. In the 2 trials that used BPI average pain measure, and in the 1 trial that used NRS, frequency of pain was much higher than that in the trials that used VAS, ranging between 70% and 96% (Table 2). However, because the 2 trials that used the BPI required patients to have a BPI score ≥ 2 or ≥ 3 at baseline, these higher rates are not unexpected.
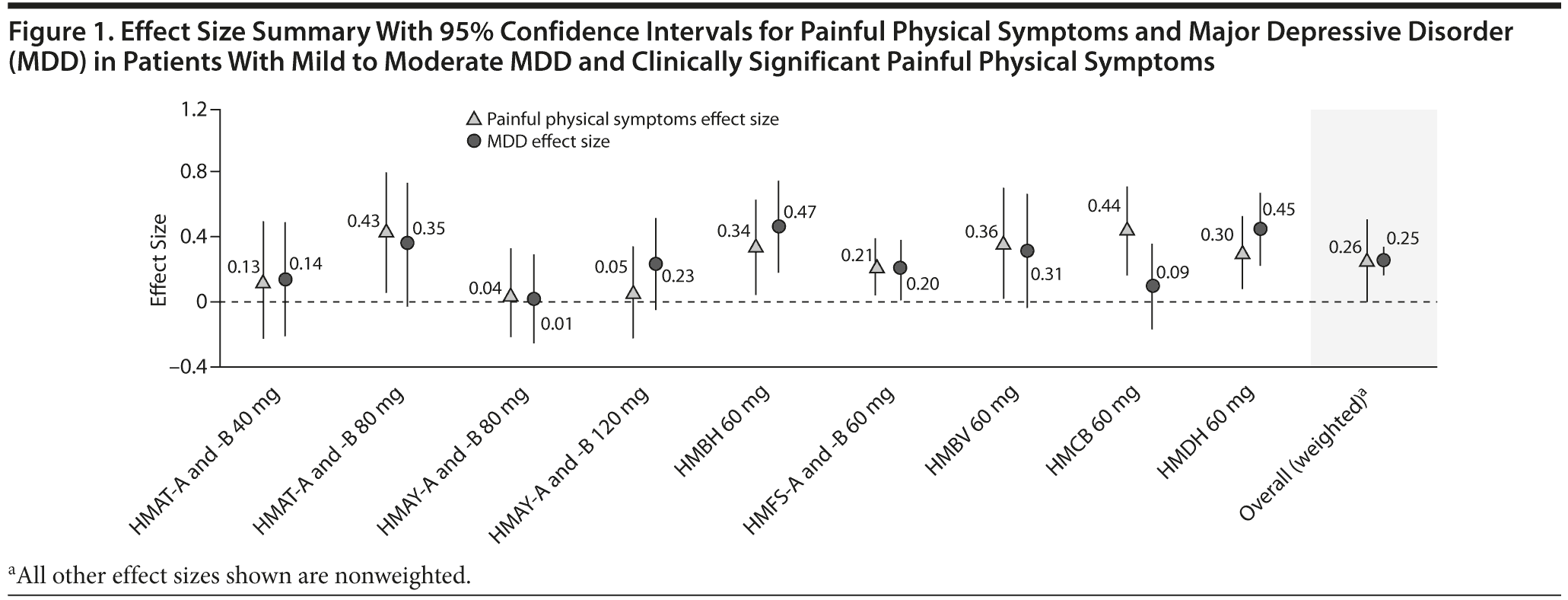
Effect Size by Individual and Pooled Trials
Analyses of the effect sizes (95% CI), or the treatment effectiveness, of duloxetine in each trial in comparison with placebo (for both painful physical symptoms and depression outcomes) are presented in Figure 1. For convenience, identical trials with the same protocol (such as studies HMBH-A and HMBH-B) were pooled. As shown in Figure 1, individually, most of the studies had effect sizes significantly different from zero, ranging from 0.21 to 0.44 for painful physical symptoms and 0.20 to 0.47 for depression. The pooled weighted effect size for all duloxetine doses was significant for efficacy in MDD (Figure 1). Although the pooled weighted effect size for efficacy of duloxetine across all doses on painful physical symptoms associated with MDD was not significant, it was closer to significance with a lower bound of 95% CI equal to −0.002 (Figure 1); this observed effect size was similar in magnitude to that of depression.
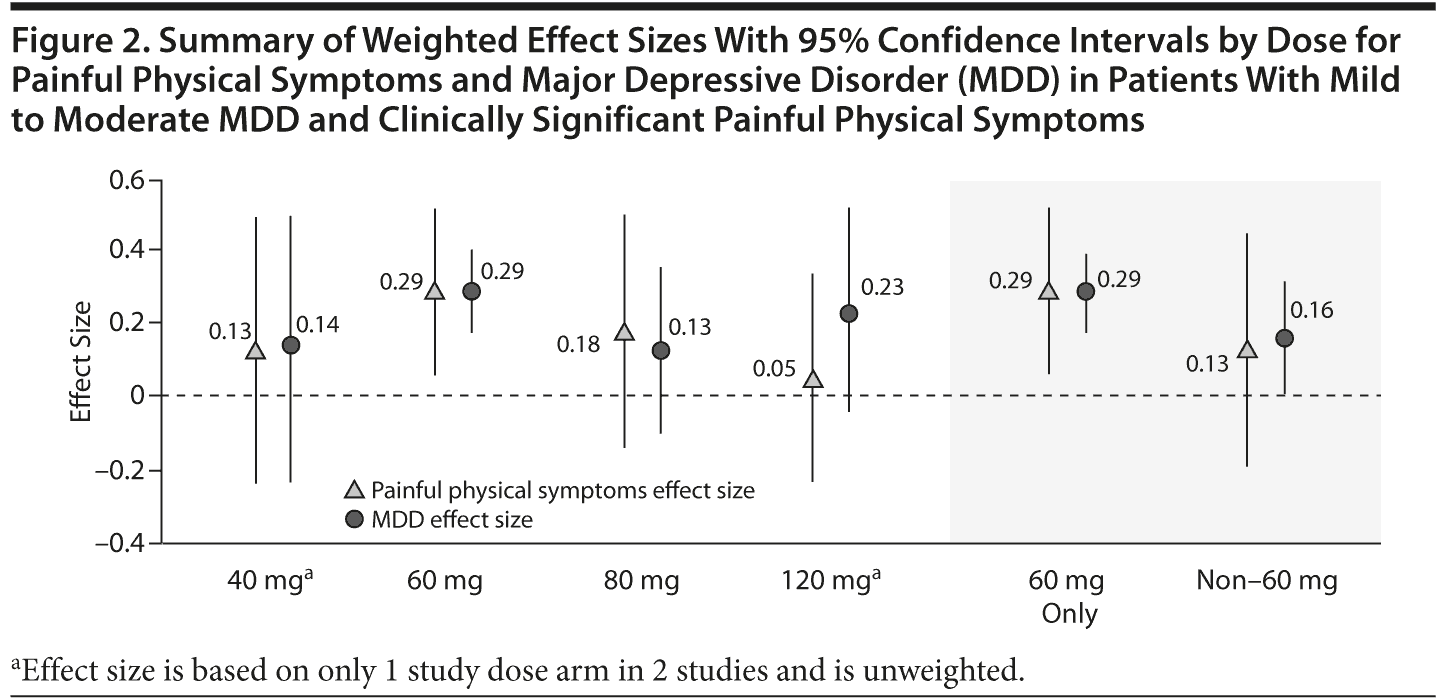
Effect Size by Dose
Because the doses varied across trials, a meta-analysis was also undertaken to examine the effect sizes based on dose. Figure 2 presents the results of the effectiveness of duloxetine in comparison with placebo by dose across multiple studies as assessed by pooled effect sizes (95% CIs). The dose showing the greatest effect size was 60 mg/d. The overall weighted effect sizes for the duloxetine 60-mg dose for painful physical symptoms and depression were similar (0.29) and both were statistically significant, whereas the overall weighted effect size for non-60-mg doses of duloxetine for painful physical symptoms (0.13) was slightly lower than that for depression (0.16).
DISCUSSION
In this trial-level meta-analysis of 11 placebo-controlled trials, patients who had mild or moderate baseline painful physical symptoms associated with MDD demonstrated a clinically meaningful reduction as assessed by effect size in both depression symptoms and painful physical symptoms. Similar findings were reported earlier in the pooled analyses of similarly designed placebo-controlled duloxetine trials.11,21 The effect sizes for both painful physical symptoms and MDD were similar for most trials. The weighted effect sizes for overall studies were 0.26 for painful physical symptoms and 0.25 for MDD, suggesting that duloxetine is equally effective on both painful physical symptoms and MDD.
This finding is different from the meta-analysis results reported by Spielmans,16 as described earlier in this article. In the meta-analysis by Spielmans, only 5 studies were used (HMBH-A, HMBH-B, HMAT-A, HMAT-B, and HMCB). In addition to these 5 studies, 6 more studies were included in the current analysis. Spielmans’ analysis included the patients with or without clinically significant painful physical symptoms at baseline from Eli Lilly’s online database. Data on painful physical symptoms published in Eli Lilly’s online database included all randomized patients with baseline and nonmissing postbaseline measures of painful physical symptoms; for studies HMBH-A, HMBH-B, HMAT-A, and HMAT-B, the VAS results from both analysis of covariance (ANCOVA) with the last observation carried forward (LOCF) and MMRM at endpoint were presented. In study HMCB, the BPI results from MMRM at endpoint were presented. The meta-analysis by Spielmans16 does not clearly state whether the LOCF or MMRM results from the first 4 studies were used for calculating the effect size. However, in the present analysis, we had selected the subset of patients with clinically significant painful physical symptoms at baseline, and we also used the MMRM analysis method. The MMRM method has been recognized in the literature as a method with key theoretical advantages over LOCF.35-39 The LOCF method imputes the endpoint using the last nonmissing observation, thus assuming that the last nonmissing observation will remain unchanged up to study endpoint; MMRM uses all the data across all the time points and extrapolates the information from observed data to infer the missing data mechanism. Based on the research conducted by Siddiqui et al,35 the MMRM analysis appears to be a superior approach in controlling Type I error rates and minimizing biases, as compared with the LOCF ANCOVA analysis. In addition, within our MMRM analysis method, the results were summarized using the main effect of treatment averaged across all the time points, instead of using the treatment effect at the last visit.
Main effect of treatment was deemed most appropriate because it evaluates the overall treatment effect across all time points. In addition, pain improvement is often characterized by rapid onset, with drug versus placebo differences remaining fairly constant over the course of treatment.
In the current meta-analysis, the effect sizes did not follow a pattern of dose-response in the efficacy of duloxetine on either painful physical symptoms or MDD. However, there was a greater increase in effect sizes from 40 mg/d to 60 mg/d followed by decrease at 80 mg/d or 120 mg/d. The lack of a dose-response may be due to lack of enough power at 40 mg/d, 80 mg/d, and 120 mg/d as compared with 60 mg/d because 7 of 11 studies were conducted to assess the efficacy of duloxetine 60 mg/d relative to placebo, whereas duloxetine 40 mg/d and 120 mg/d treatment were represented in only 2 studies and 80 mg/d was represented in only 4 studies. Similar findings were observed with our earlier effect-size analysis for the dose-response relationship for duloxetine efficacy in depression.40 Antidepressants, in general, have a relatively flat dose-response relationship on improvement in depression.41 The lack of dose-response may also be due to high variability in duloxetine pharmacokinetics, in that different dose levels may be required to maintain blood levels.42,43 Antidepressant therapy is also challenged by high rates of placebo response in randomized controlled trials, a phenomenon that has been shown to influence the clinical outcomes.44,45 Higher placebo response is associated with lower effect size and vice versa.46
In this analysis, the overall effect sizes were small for both painful physical symptoms and MDD. However, the individual study effect size was as high as 0.47, suggesting that some patients may benefit from the treatment more than other patients. In addition, the small effect size is also partly due to the fact that the sample size was reduced by the inclusion of only the subset of patients with clinically significant pain level at baseline. When describing effect sizes, we used the terminology suggested by Cohen47 in his nontechnical guide for interpreting the clinical impact of one of the most commonly expressed types of effect size in the behavioral sciences (ie, the correlation coefficient, or r). Per standards used in the behavioral sciences (as described by Cohen47), correlation values within the 0.10 to 0.29 ranges were considered “small” effects; values from 0.30 to 0.49, “medium” effects; and correlations exceeding 0.50, “large.” These definitions roughly parallel the conversion of correlation value into coefficients of determination (r2) to represent the approximate r values necessary to account for 1%, 10%, and 25% of the variance of the dependent variable, respectively.48 In a recent review of all Eli Lilly chronic pain studies, effect sizes of 0.2 to 0.6 were reported49; these findings are comparable with those reported in the current analysis for painful physical symptoms.
Descending serotonin and norepinephrine pathways have been suggested as modulators of pain perception, and duloxetine has demonstrated efficacy in nondepressed patients with diabetic peripheral neuropathic pain, osteoarthritis, and chronic low back pain and in both depressed and nondepressed patients with fibromyalgia.9 The current findings of equal efficacy as evidenced by similar effect sizes for painful physical symptoms and MDD further support the involvement of the dual-reuptake mechanism of serotonin and norepinephrine in depression and pain. However, it should be noted that a meta-analysis of 8 studies (7 studies from Eli Lilly and Company and 1 from GlaxoSmithKline) did not suggest superiority of the SNRI compared with SSRI interventions.17 Those authors note that their meta-analysis results do not support the hypothesis that SNRIs may have special analgesic effects in addition to their antidepressant effects because of dual-reuptake inhibition. The analysis demonstrated that duloxetine was superior to placebo in treating painful physical symptoms in depression. Although paroxetine was also shown to be effective, this finding does not disprove the potential role for dual-reuptake inhibition actions in treating the painful physical symptoms associated with depression. Regardless of the mechanism, it has been postulated that improvement in both depression and pain improves treatment outcomes for patients with MDD, such as response and long-term remission.11
The limitations of this study include the fact that the results are based on a post hoc fixed-effect meta-analysis and thus may not be applicable to the general population. Additionally, the trials did not specifically enroll patients with pain at baseline except in 2 studies. Also, a few studies used non-60-mg doses, and thus our study is underpowered to find differences at different doses of duloxetine on painful physical symptoms. Strengths of the study include the use of the analytically appropriate MMRM method, the use of the main effect of treatment to provide estimate of effect across all time points, and the selection of patients with clinically significant painful physical symptoms at baseline that would warrant clinical intervention.
In summary, the trial-level meta-analysis results presented here further support the efficacy of duloxetine for the treatment of both depressive symptoms and painful physical symptoms associated with MDD. However, the results do not address its efficacy compared to other alternatives, as in all studies the comparator was placebo.
Drug names: duloxetine (Cymbalta), paroxetine (Paxil, Pexeva, and others).
Author affiliations: Lilly Research Laboratories, Eli Lilly and Company, Indianapolis, Indiana (Drs Ball, Desaiah, Spann, Russell, and Robinson and Ms Zhang); and University Psychiatric Center-Campus Gasthuisberg, Leuven, Belgium (Dr Demyttenaere). Dr Spann is currently employed by Alcon Laboratories, Fort Worth, Texas.
Potential conflicts of interest: Drs Ball, Desaiah, Russell, and Robinson and Ms Zhang are employees of Eli Lilly, and Drs Ball and Desaiah are stock shareholders in Eli Lilly. Dr Spann was an employee of Eli Lilly from March 2006 to March 2010. Dr Demyttenaere has been a consultant for, received grant/research support or honoraria from, or been on the speakers/advisory boards of AstraZeneca, Boehringer Ingelheim, Eli Lilly, GlaxoSmithKline, Lundbeck, Servier, Takeda, and Wyeth.
Funding/support: This meta-analysis was sponsored by Eli Lilly and Company, Indianapolis, Indiana.
Previous presentation: Presented at the 50th Anniversary Meeting of NCDEU (New Research Approaches for Mental Health Interventions); June 16, 2010; Boca Raton, Florida.
Acknowledgment: The authors gratefully acknowledge the statistical assistance of Adam Meyers, MS, of Eli Lilly and Company, Indianapolis, Indiana.
REFERENCES
1. Greden JF. Physical symptoms of depression: unmet needs. J Clin Psychiatry. 2003;64(suppl 7):5-11. PubMed
2. Bair MJ, Robinson RL, Eckert GJ, et al. Impact of pain on depression treatment response in primary care. Psychosom Med. 2004;66(1):17-22. PubMed doi:10.1097/01.PSY.0000106883.94059.C5
3. Kroenke K, Shen J, Oxman TE, et al. Impact of pain on the outcomes of depression treatment: results from the RESPECT trial. Pain. 2008;134(1-2):209-215. PubMed doi:10.1016/j.pain.2007.09.021
4. Leuchter AF, Husain MM, Cook IA, et al. Painful physical symptoms and treatment outcome in major depressive disorder: a STAR*D (Sequenced Treatment Alternatives to Relieve Depression) report. Psychol Med. 2010;40(2):239-251. PubMed doi:10.1017/S0033291709006035
5. Gameroff MJ, Olfson M. Major depressive disorder, somatic pain, and health care costs in an urban primary care practice. J Clin Psychiatry. 2006;67(8):1232-1239. PubMed doi:10.4088/JCP.v67n0809
6. Demyttenaere K, Bonnewyn A, Bruffaerts R, et al. Comorbid painful physical symptoms and depression: prevalence, work loss, and help seeking. J Affect Disord. 2006;92(2-3):185-193. PubMed doi:10.1016/j.jad.2006.01.007
7. Kroenke K, Bair MJ, Damush TM, et al. Optimized antidepressant therapy and pain self-management in primary care patients with depression and musculoskeletal pain: a randomized controlled trial. JAMA. 2009;301(20):2099-2110. PubMed doi:10.1001/jama.2009.723
8. Giesecke T, Gracely RH, Williams DA, et al. The relationship between depression, clinical pain, and experimental pain in a chronic pain cohort. Arthritis Rheum. 2005;52(5):1577-1584. PubMed doi:10.1002/art.21008
9. Robinson MJ, Edwards SE, Iyengar S, et al. Depression and pain. Front Biosci. 2009;14(1):5031-5051. PubMed doi:10.2741/3585
10. Perahia DG, Pritchett YL, Desaiah D, et al. Efficacy of duloxetine in painful symptoms: an analgesic or antidepressant effect? Int Clin Psychopharmacol. 2006;21(6):311-317. PubMed doi:10.1097/01.yic.0000224782.83287.3c
11. Fava M, Mallinckrodt CH, Detke MJ, et al. The effect of duloxetine on painful physical symptoms in depressed patients: do improvements in these symptoms result in higher remission rates? J Clin Psychiatry. 2004;65(4):521-530. PubMed doi:10.4088/JCP.v65n0411
12. Mallinckrodt CH, Goldstein DJ, Detke MJ, et al. Duloxetine: a new treatment for the emotional and physical symptoms of depression. Prim Care Companion J Clin Psychiatry. 2003;5(1):19-28. PubMed doi:10.4088/PCC.v05n0105
13. Goldstein DJ, Lu Y, Detke MJ, et al. Effects of duloxetine on painful physical symptoms associated with depression. Psychosomatics. 2004;45(1):17-28. PubMed doi:10.1176/appi.psy.45.1.17
14. Raskin J, Wiltse CG, Siegal A, et al. Efficacy of duloxetine on cognition, depression, and pain in elderly patients with major depressive disorder: an 8-week, double-blind, placebo-controlled trial. Am J Psychiatry. 2007;164(6):900-909. PubMed doi:10.1176/appi.ajp.164.6.900
15. Brecht S, Courtecuisse C, Debieuvre C, et al. Efficacy and safety of duloxetine 60 mg once daily in the treatment of pain in patients with major depressive disorder and at least moderate pain of unknown etiology: a randomized controlled trial. J Clin Psychiatry. 2007;68(11):1707-1716. PubMed doi:10.4088/JCP.v68n1110
16. Spielmans GI. Duloxetine does not relieve painful physical symptoms in depression: a meta-analysis. Psychother Psychosom. 2008;77(1):12-16. PubMed doi:10.1159/000110055
17. Krebs EE, Gaynes BN, Gartlehner G, et al. Treating the physical symptoms of depression with second-generation antidepressants: a systematic review and metaanalysis. Psychosomatics. 2008;49(3):191-198. PubMed doi:10.1176/appi.psy.49.3.191
18. Detke MJ, Lu Y, Goldstein DJ, et al. Duloxetine, 60 mg once daily, for major depressive disorder: a randomized double-blind placebo-controlled trial. J Clin Psychiatry. 2002;63(4):308-315. PubMed doi:10.4088/JCP.v63n0407
19. Detke MJ, Lu Y, Goldstein DJ, et al. Duloxetine 60 mg once daily dosing versus placebo in the acute treatment of major depression. J Psychiatr Res. 2002;36(6):383-390. PubMed doi:10.1016/S0022-3956(02)00060-2
20. Brannan SK, Mallinckrodt CH, Brown EB, et al. Duloxetine 60 mg once-daily in the treatment of painful physical symptoms in patients with major depressive disorder. J Psychiatr Res. 2005;39(1):43-53. PubMed doi:10.1016/j.jpsychires.2004.04.011
21. Nemeroff CB, Schatzberg AF, Goldstein DJ, et al. Duloxetine for the treatment of major depressive disorder. Psychopharmacol Bull. 2002;36(4):106-132. PubMed
22. Goldstein DJ, Lu Y, Detke MJ, et al. Duloxetine in the treatment of depression: a double-blind placebo-controlled comparison with paroxetine. J Clin Psychopharmacol. 2004;24(4):389-399. PubMed doi:10.1097/01.jcp.0000132448.65972.d9
23. Detke MJ, Wiltse CG, Mallinckrodt CH, et al. Duloxetine in the acute and long-term treatment of major depressive disorder: a placebo- and paroxetine-controlled trial. Eur Neuropsychopharmacol. 2004;14(6):457-470. PubMed doi:10.1016/j.euroneuro.2004.01.002
24. Perahia DG, Wang F, Mallinckrodt CH, et al. Duloxetine in the treatment of major depressive disorder: a placebo- and paroxetine-controlled trial. Eur Psychiatry. 2006;21(6):367-378. PubMed doi:10.1016/j.eurpsy.2006.03.004
25. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition. Washington, DC: American Psychiatric Association; 1994.
26. Guy W. ECDEU Assessment Manual for Psychopharmacology, revised. Publication ADM 76-338. Bethesda, MD: Department of Health, Education, and Welfare; 1976:218-222.
27. Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23(1):56-62. PubMed doi:10.1136/jnnp.23.1.56
28. Montgomery SA, Asberg M. A new depression scale designed to be sensitive to change. Br J Psychiatry. 1979;134(4):382-389. PubMed doi:10.1192/bjp.134.4.382
29. Cleeland CS, Ryan KM. Pain assessment: global use of the Brief Pain Inventory. Ann Acad Med Singapore. 1994;23(2):129-138. PubMed
30. Hedges LV, Olkin I. Statistical Methods for Meta-Analysis. New York, NY: Academic Press; 1985.
31. Van Den Noortgate W, Onghena P. Estimating the mean effect size in meta-analysis: bias, precision, and mean squared error of different weighting methods. Behav Res Methods Instrum Comput. 2003;35(4):504-511. PubMed doi:10.3758/BF03195529
32. Kenward MG, Roger JH. Small sample inference for fixed effects from restricted maximum likelihood. Biometrics. 1997;53(3):983-997. PubMed doi:10.2307/2533558
33. Hedges LV. Distribution theory for Glass’s estimator of effect size and related estimators. J Educ Stat. 1981;6(2):107-128. doi:10.2307/1164588
34. Hedges LV. Estimation of effect size from a series of independent experiments. Psychol Bull. 1982;92(2):490-499. doi:10.1037/0033-2909.92.2.490
35. Siddiqui O, Hung HM, O’ Neill R. MMRM vs LOCF: a comprehensive comparison based on simulation study and 25 NDA datasets. J Biopharm Stat. 2009;19(2):227-246. PubMed doi:10.1080/10543400802609797
36. Lane P. Handling drop-out in longitudinal clinical trials: a comparison of the LOCF and MMRM approaches. Pharm Stat. 2008;7(2):93-106. PubMed doi:10.1002/pst.267
37. Mallinckrodt CH, Clark WS, David SR. Accounting for dropout bias using mixed-effects models. J Biopharm Stat. 2001;11(1-2):9-21. PubMed doi:10.1081/BIP-100104194
38. Mallinckrodt CH, Kaiser CJ, Watkin JG, et al. Type I error rates from likelihood-based repeated measures analyses of incomplete longitudinal data. Pharm Stat. 2004;3(3):171-186. doi:10.1002/pst.131
39. Mallinckrodt CH, Lane PW, Schnell D, et al. Recommendations for the primary analysis of continuous endpoints in longitudinal clinical trials. Drug Inf J. 2008;42:303-319.
40. Pritchett YL, Marciniak MD, Corey-Lisle PK, et al. Use of effect size to determine optimal dose of duloxetine in major depressive disorder. J Psychiatr Res. 2007;41(3-4):311-318. PubMed doi:10.1016/j.jpsychires.2006.06.013
41. Khan A, Khan SR, Walens G, et al. Frequency of positive studies among fixed and flexible dose antidepressant clinical trials: an analysis of the Food and Drug Administration summary basis of approval reports. Neuropsychopharmacology. 2003;28(3):552-557. PubMed doi:10.1038/sj.npp.1300059
42. Sharma A, Goldberg MJ, Cerimele BJ. Pharmacokinetics and safety of duloxetine, a dual-serotonin and norepinephrine reuptake inhibitor. J Clin Pharmacol. 2000;40(2):161-167. PubMed doi:10.1177/00912700022008810
43. Westanmo AD, Gayken J, Haight R. Duloxetine: a balanced and selective norepinephrine- and serotonin-reuptake inhibitor. Am J Health Syst Pharm. 2005;62(23):2481-2490. PubMed doi:10.2146/ajhp050006
44. Sinyor M, Levitt AJ, Cheung AH, et al. Does inclusion of a placebo arm influence response to active antidepressant treatment in randomized controlled trials? results from pooled and meta-analyses. J Clin Psychiatry. 2010;71(3):270-279. PubMed doi:10.4088/JCP.08r04516blu
45. Papakostas GI, Fava M. Does the probability of receiving placebo influence clinical trial outcome? a meta-regression of double-blind, randomized clinical trials in MDD. Eur Neuropsychopharmacol. 2009;19(1):34-40. PubMed doi:10.1016/j.euroneuro.2008.08.009
46. Nunes EV, Levin FR. Treatment of depression in patients with alcohol or other drug dependence: a meta-analysis. JAMA. 2004;291(15):1887-1896. PubMed doi:10.1001/jama.291.15.1887
47. Cohen J. Statistical Power Analysis for the Behavioral Sciences. 2nd ed. Hillsdale, NJ: Lawrence Erlbaum Associates; 1988.
48. Rutledge T, Loh C. Effect sizes and statistical testing in the determination of clinical significance in behavioral medicine research. Ann Behav Med. 2004;27(2):138-145. PubMed doi:10.1207/s15324796abm2702_9
49. Mease PJ, Walker DJ, Alaka K. Evaluation of duloxetine for chronic pain conditions. Pain Manag. 2011;1(2):159-170. doi:10.2217/pmt.11.4