Beneficial Effects of Adjunctive Aripiprazole in Major Depressive Disorder Are Not Dependent on Antidepressant Therapy History: A Post Hoc Analysis of 3 Randomized, Double-Blind, Placebo-Controlled Trials

ABSTRACT
Objective: To determine whether switching within or between antidepressant therapy (ADT) classes prior to the use of adjunctive antipsychotic treatment is associated with different outcomes in major depressive disorder (MDD).
Method: This was a post hoc analysis of pooled data from 3 similar, multicenter, randomized, double-blind, placebo-controlled registrational studies of aripiprazole adjunctive to ADT conducted between September 2004 and April 2008. The trials comprised the following 3 phases: a 7- to 28-day screening phase, an 8-week single-blind prospective treatment phase, and a 6-week double-blind, randomized phase. Patients were aged 18-65 years and met DSM-IV-TR criteria for MDD. Patients with an inadequate response to ADT during the screening phase entered the prospective treatment phase, during which they were switched to another ADT medication of either the same or a different class. Those patients with an inadequate response were then randomized to double-blind adjunctive aripiprazole or adjunctive placebo and followed for 6 weeks.
Results: Mean improvement in Montgomery-Asberg Depression Rating Scale total score was significantly greater with adjunctive aripiprazole versus adjunctive placebo for both between-class (−9.2 vs −6.2, P < .001) and within-class (−9.8 vs −6.6, P < .001) switch groups. Relative risks for response were 1.6 (95% CI = 1.3-2.1) for those who switched between classes and 1.7 (95% CI = 1.2-2.2) for those who switched within class.
Conclusions: Augmentation with aripiprazole, after either a between-class or within-class switch following initial ADT failure, is an effective option for patients with nonresponsive MDD. In contrast to current strategies employed in clinical practice, these results suggest that adjunctive aripiprazole is a logical strategy in patients unresponsive to ADT.
Trial Registration: ClinicalTrials.gov identifiers: NCT00105196, NCT00095758, NCT00095823
Prim Care Companion CNS Disord 2012;14(6):doi:10.4088/PCC.12m01380
© Copyright 2012 Physicians Postgraduate Press, Inc.
Submitted: March 20, 2012; accepted August 15, 2012.
Published online: November 22, 2012.
Corresponding author: David L. Dunner, MD, FACPsych, Center for Anxiety and Depression, 7525 SE 24th St, Ste 400, Mercer Island, WA ([email protected]).
Depression is a serious mental health condition that is highly prevalent in a broad spectrum of the population in the United States. Major depressive disorder (MDD) has a lifetime prevalence of 16.2% and is estimated to affect approximately 33 to 35 million US adults.1 MDD is seen in all age groups, from the early teens into the elderly years.1 Some degree of social or work-related functional impairment is experienced by about 90% of those with MDD.1 Furthermore, two-thirds of suicides occur in people with depressive disorders, and people with depression are 20 times more likely to commit suicide than those without depression.2,3
The recently revised American Psychiatric Association guidelines for MDD recommend the following 3 phases of treatment: acute (6-12 weeks), with the goals of inducing remission and returning patients to full functioning; continuation (4-9 months) to prevent relapse; and maintenance to protect susceptible patients against recurrence.4 Treatment modalities to achieve these ends include pharmacotherapeutics, psychotherapy, electroconvulsive therapy, transcranial magnetic stimulation, and light therapy, among others.4 Importantly, most depressed subjects will first be treated in a primary care setting, with almost 10% of all primary care visits related to the treatment of depression.5
Several classes of antidepressant medications are available, but the most commonly employed are the selective serotonin reuptake inhibitors (SSRIs).6 Another commonly used class is the serotonin-norepinephrine reuptake inhibitors (SNRIs). For the majority of patients, the effectiveness of these agents as monotherapy is comparable between classes and within classes, with response rates ranging from 50% to 75% during acute phase treatment.4 However, if patients do not respond to the initial treatment choice, clinicians must then adjust therapy. For patients who do not respond to an optimal dose of the first antidepressant therapy (ADT) prescribed, switching to another medication of either the same class or a different class is often the next step. Although the evidence is mixed as to whether switching within or between classes yields better efficacy, it is clear that a large percentage of patients will need additional intervention.7,8 Augmentation with a second-generation antipsychotic is 1 recommendation for patients unresponsive to at least 1 trial of ADT.4 In contrast to switching ADT, atypical antipsychotics constitute the only class of medications approved for difficult-to-treat depression. Quetiapine extended release (XR) and aripiprazole are approved for the adjunctive treatment of patients with MDD who demonstrate an inadequate response to ADT monotherapy, while olanzapine is approved in combination with fluoxetine for the management of treatment-resistant depression. However, different treatment guidelines offer varying advice on when and how to add an atypical antipsychotic agent to the treatment schema of patients with an inadequate response to antidepressants.4,9,10 Therefore, clinicians must carefully individualize treatment to the needs of an individual patient.
Overall, the body of evidence for augmentation with atypical antipsychotics is notably larger than for other augmentation strategies in the treatment of depression.11 Thus, it is important to discern whether switching within or between ADT classes affects future response to augmentation with an antipsychotic agent. To address this question and provide guidance to clinicians, we analyzed pooled data from 3 registrational trials of aripiprazole adjunctive to ADT that led to US Food and Drug Administration (FDA) approval for the use of aripiprazole as adjunctive treatment in MDD.12-14 Aripiprazole is a pharmacologically distinct atypical antipsychotic.15-19 Efficacy of aripiprazole in combination with the SSRIs escitalopram, fluoxetine, paroxetine, and sertraline or the SNRI venlafaxine in patients who had not responded to these ADTs was demonstrated in all 3 trials, as evidenced by significantly greater decreases (improvement) in Montgomery-Asberg Depression Rating Scale (MADRS) and Clinical Global Impressions-severity of illness (CGI-S) and CGI-improvement (CGI-I) scale scores and greater remission and response rates over adjunctive placebo.12-14 For this analysis, patients who had a history of 1 to 3 inadequate responses to ADT, and who demonstrated an inadequate response after switching medications, were evaluated to compare the effects of within-class and between-class switching of ADT prior to initiating augmentation with aripiprazole.

- Augmentation of antidepressant therapy with atypical antipsychotics is an effective strategy to alleviate depressive symptoms in patients who have failed antidepressant monotherapy.
- There is no evidence that failure to respond to antidepressant monotherapy of different classes is necessary before augmentation with an atypical agent is tried.
METHOD
Design Overview
This was a post hoc analysis of pooled data from 3 nearly identical multicenter, randomized, double-blind, placebo-controlled registrational studies of aripiprazole adjunctive to ADT (ClinicalTrials.gov identifiers: NCT00105196, NCT00095758, NCT00095823).12-14 The 3 studies were conducted between September 2004 and April 2008. Complete accounts of the methods for these studies have been previously described.12-14
Setting and Participants
Patients were aged 18-65 years and met DSM-IV-TR criteria for MDD.20 The studies included patients without psychotic features who had previously shown an incomplete response to an adequate trial of 1 to 3 ADTs during the current episode. An adequate trial was defined as treatment administered at therapeutic doses for more than 6 weeks. The institutional review boards applicable to each site approved the study; all patients participated with written, informed voluntary consent, and possible side effects were fully explained.
Randomization and Interventions
The trial design consisted of 3 phases: a 7- to 28-day screening phase, an 8-week single-blind prospective treatment phase to establish inadequate response to antidepressant monotherapy, and a 6-week double-blind, randomized phase. Patients in the 7- to 28-day screening phase were being treated with a range of ADTs as determined by their physician. Those patients experiencing a major depressive episode—including a total score ≥ 18 on the 17-item Hamilton Depression Rating Scale (HDRS-17)—then qualified for an 8-week prospective treatment phase. Prior to entering the 8-week prospective treatment phase, patients were also required to have reported an inadequate response to previous ADT (1-3 ADT trials) in the current episode, as determined by a < 50% reduction in depressive symptom severity as assessed by the Antidepressant Treatment Response Questionnaire, a validated, self-rated scale shown to accurately assess treatment failure.21,22
During the prospective treatment phase, to document inadequate response to the new ADT, patients were switched to another ADT medication, either of the same class or of a different class from what they had been treated with in the current episode. Patients were classified as between-class switchers if at least 1 of the medications with a reported inadequate response belonged to a different class than the medication received during the prospective monotherapy phase. Other patients were classified as within-class switchers. Medication classes allowed during the screening phase included the SSRIs, SNRIs, norepinephrine-dopamine reuptake inhibitors (NDRIs), tricyclic antidepressants, and others. The choice of the new medication was at the discretion of the investigator but limited to escitalopram 10 mg/d or 20 mg/d, fluoxetine 20 mg/d or 40 mg/d, paroxetine controlled release (CR) 37.5 mg/d or 50 mg/d, sertraline 100 mg/d or 150 mg/d, or venlafaxine XR 150 mg/d or 225 mg/d.
In the double-blind, randomized phase, patients with an inadequate response (< 50% reduction in HDRS-17 score from the baseline visit to the end of the prospective treatment phase, HDRS-17 total score ≥ 14, and CGI-I score ≥ 3 at the end of the prospective treatment phase) to the ADT were then randomized (1:1) to double-blind adjunctive aripiprazole or adjunctive placebo plus the ADT from the prospective treatment phase and followed over the ensuing 6 weeks. The starting dose of aripiprazole was 5 mg that beginning at week 2 could be decreased to 2 mg if the initial dose was not well tolerated or increased to 10 mg, with subsequent weekly dose increases of 5 mg per day to a total of 15 mg if the patient was being treated with paroxetine or fluoxetine and 20 mg if the patient was treated with another ADT. Patients who responded to the ADT monotherapy in the prospective treatment phase were not randomized but continued to receive single-blind placebo plus the ADT and were followed for 6 weeks.
Outcomes and Follow-Up
The primary efficacy endpoint in the 3 studies was the mean change in the MADRS total score from the end of the prospective treatment phase to the end of the double-blind treatment phase using a last observation carried forward (LOCF) analysis. Remission was defined as a MADRS total score ≤ 10 and a reduction of ≥ 50% at endpoint by LOCF analysis. Response was defined as a ≥ 50% reduction in MADRS total score from the beginning of the double-blind phase to endpoint using an LOCF analysis.
Statistical Analysis
Pooled data from the 3 nearly identical aripiprazole registrational studies were analyzed. ADTs used in the screening and prospective treatment phases were summarized on the basis of the randomized sample. By design, all patients in the efficacy analysis had to have switched ADTs, and those who did not were excluded from the analysis. The 2 groups were defined as patients in the efficacy sample who switched ADTs between classes and patients who switched ADTs within class between the screening and prospective treatment phases and were entered into the randomized phase. Mean change from baseline in the MADRS total score was analyzed, stratified by switch-within or switch-between classes, using analysis of covariance (ANCOVA). In the original data sets, double-blind treatment and protocol were main effects and score at the end of the prospective treatment phase assessment was a covariate.12-14 An LOCF analysis of the efficacy sample was used for the primary endpoint in the original studies and this post hoc analysis. The Cochran-Mantel-Haenszel general association test was used for treatment comparisons of remission and response rates, controlling for treatment and protocol with an LOCF analysis of the efficacy sample, stratified by switch-within or switch-between classes. Mean change from baseline in body weight was analyzed, stratified by switch-within or switch-between classes, using ANCOVA, with treatment and protocol as main effects, end of the prospective treatment phase assessment as covariate, and observed cases (OC) analyses of the efficacy sample. The Cochran-Mantel-Haenszel general association test was used for treatment comparisons of clinically relevant weight gain, controlling for treatment and protocol with an OC analysis of the efficacy sample, stratified by switch-within or switch-between classes. The incidence of adverse events was assessed during the double-blind phase for both within-class and between-class ADT switches. All analyses were performed using SAS statistical software V8.2 or above (SAS Institute, Inc, Cary, North Carolina).
RESULTS
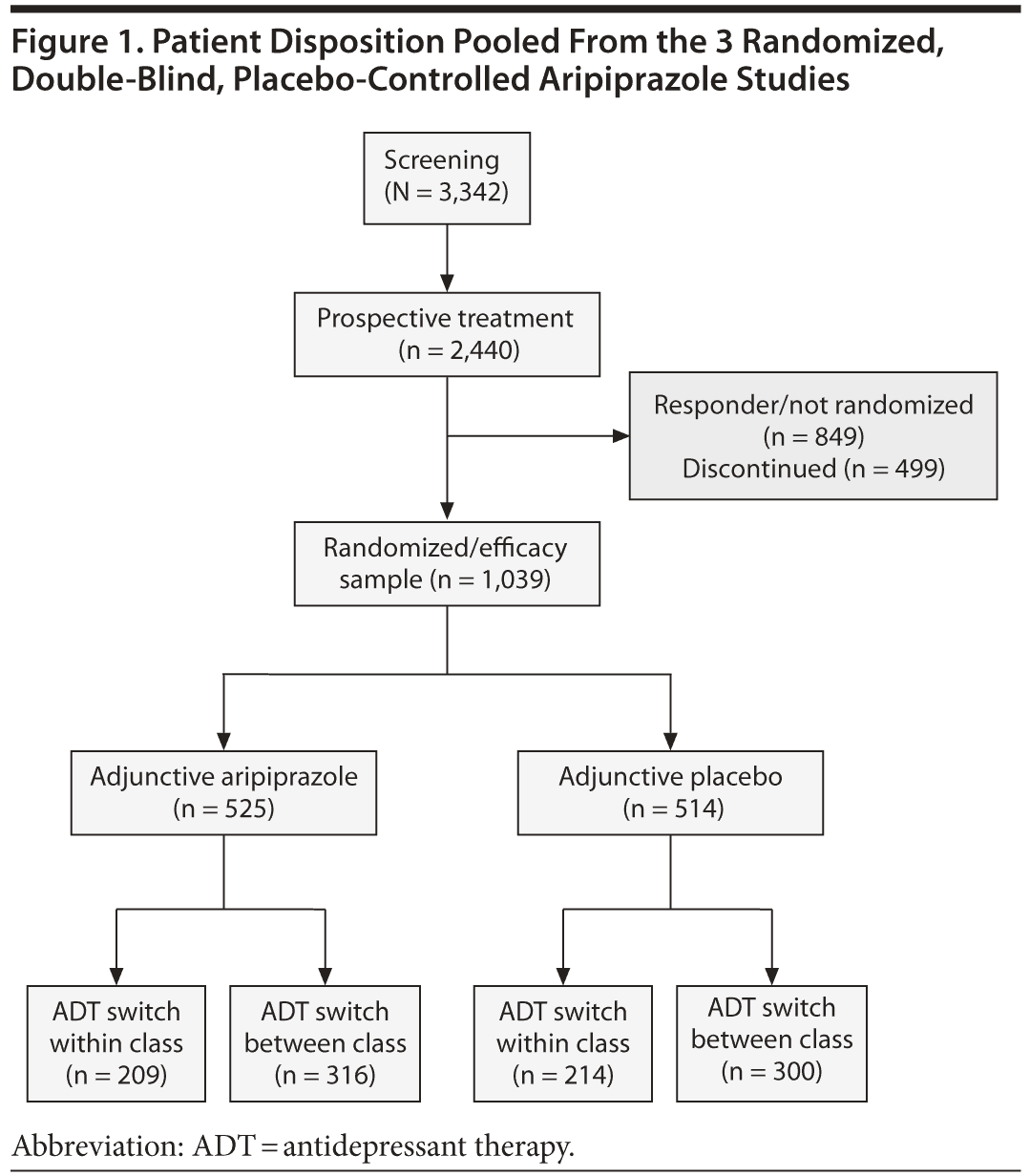
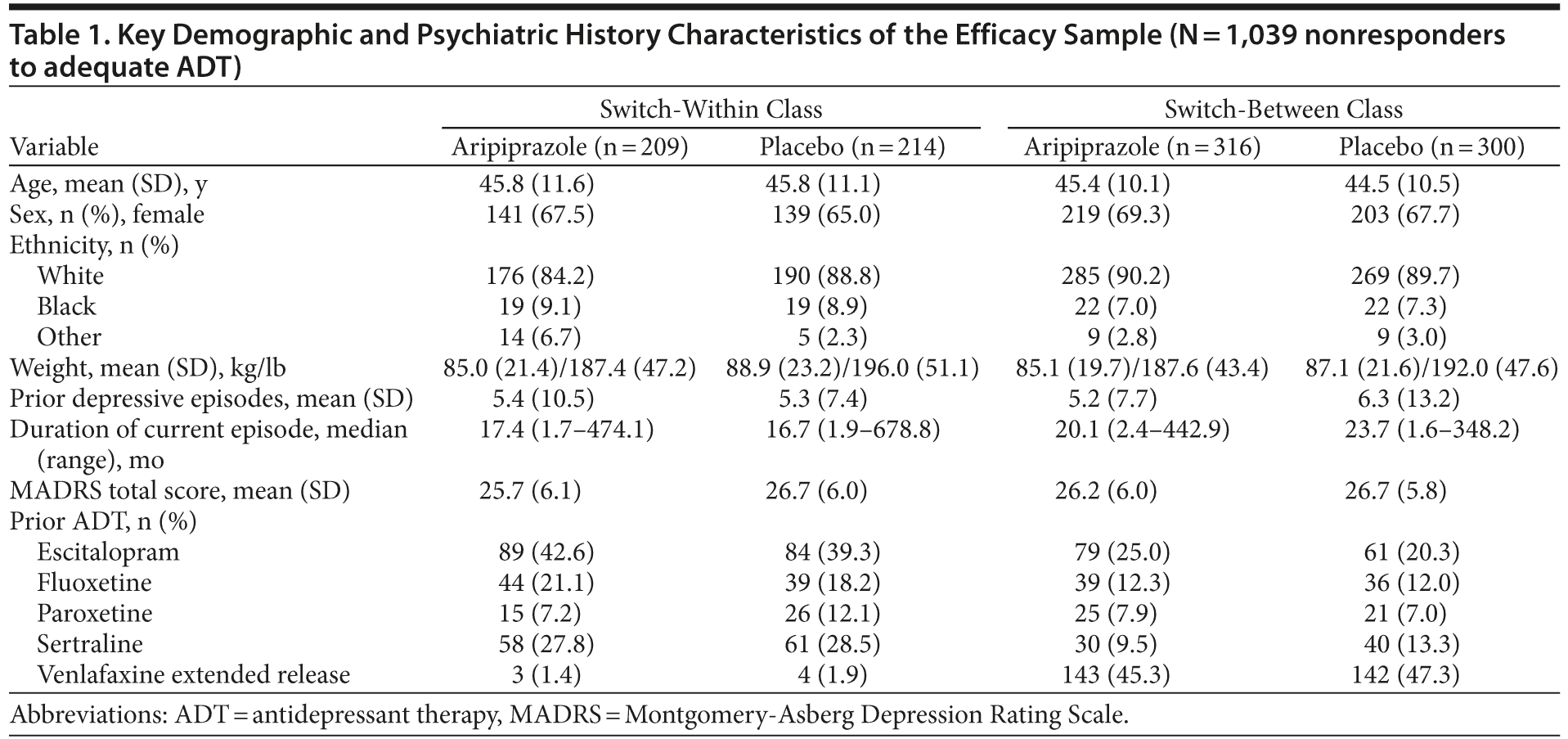
A total of 1,039 nonresponders who were randomized to adjunctive aripiprazole (n = 525) or adjunctive placebo (n = 514), who had confirmed adequacy of previous ADTs, and who had an ADT switch were included in the efficacy sample and is the population included in this analysis (Figure 1). The completion rates of the randomized phase did not differ between the switch-within or switch-between class groups, and a majority of patients in both groups completed the study: 88.6% in the between-class group and 87.7% in the within-class group. Table 1 shows baseline demographic and psychiatric characteristics of the pooled patient population by ADT switch between-class versus within-class groups. Overall, demographic characteristics appeared similar among both those who received adjunctive aripiprazole and adjunctive placebo and those who switched between classes versus within class. Of the randomized patients, 65% to 69% were women, 84% to 90% were white, and the mean weights spanned 187.4 lb (85 kg) to 196.0 lb (89 kg). The mean number of prior depressive episodes ranged from 5.2 to 6.3, the median duration of the current episode was 17 to 24 months, and the mean baseline MADRS total scores at the beginning of the randomized phase ranged from 25.7 to 26.7. Approximately 70% of patients had previously shown an inadequate response to only 1 ADT prior to entering the trial.23
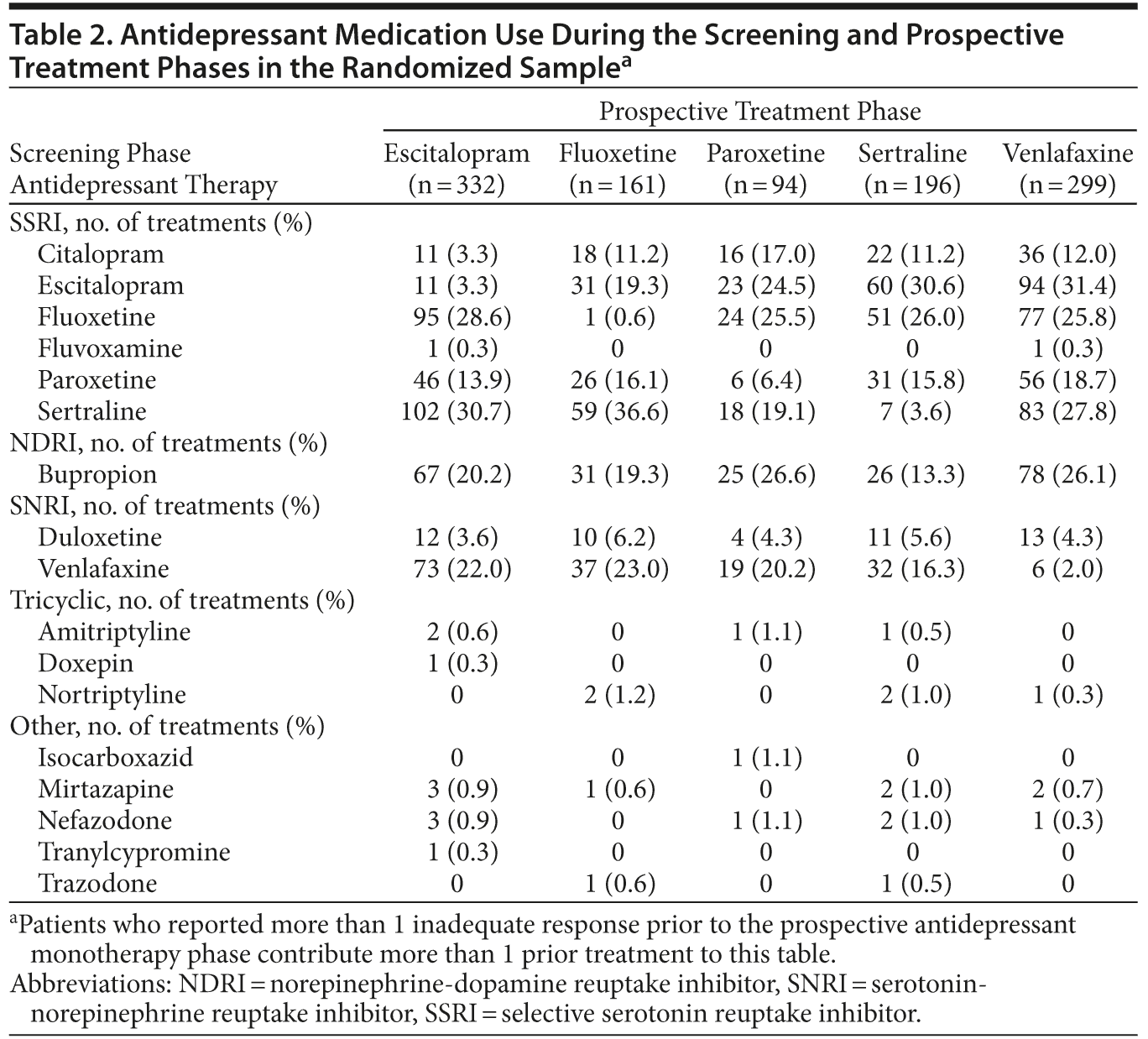
ADT use during the screening and prospective treatment phases for the randomized sample is summarized in Table 2 and includes both patients in the efficacy sample and those who did not switch ADT medication or were not included in the efficacy sample. For the 1,039 patients in the efficacy sample who switched ADT, between the screening treatment and prospective treatment phases, 40.7% (423/1,039) of patients were switched to an ADT in the same class and 59.3% (616/1,039) were switched to an ADT in a different class. Similar proportions of patients in the adjunctive aripiprazole and adjunctive placebo groups were switched within and between classes. A total of 39.8% (209/525) of adjunctive aripiprazole and 41.6% (214/514) of adjunctive placebo patients switched within class, and 60.2% (316/525) of adjunctive aripiprazole and 58.4% (300/514) of adjunctive placebo patients switched between classes.
Clinical Assessments
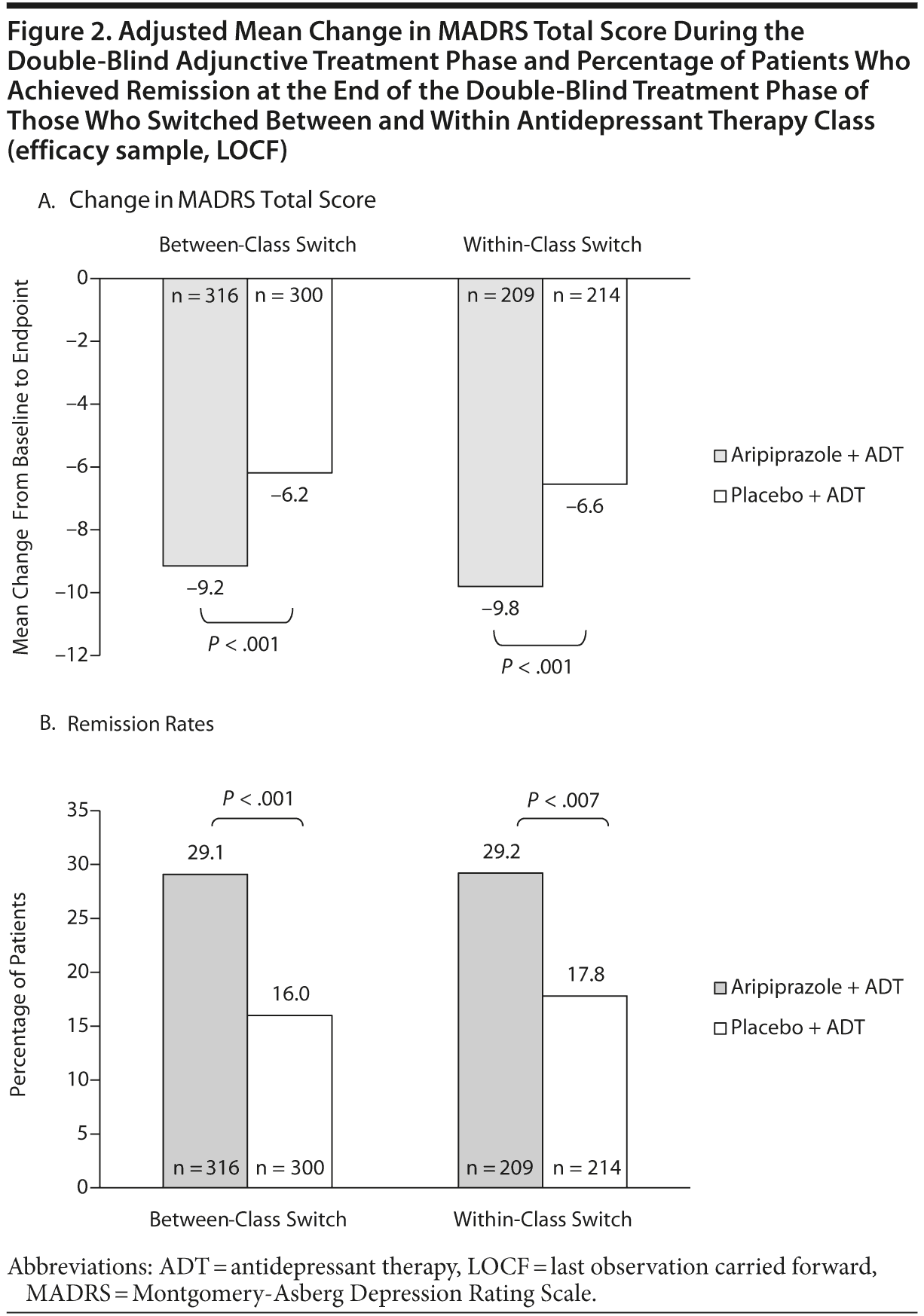
As shown in Figure 2A, the mean change from baseline in MADRS total score was significant for adjunctive aripiprazole compared with adjunctive placebo for patients who switched within ADT class, as well as for patients who switched between ADT classes (P < .001). The treatment difference in mean change from baseline for MADRS total score between adjunctive aripiprazole and adjunctive placebo for between-class switching was −3.0 (95% CI = −4.3 to -1.7) and for within-class switching was −3.3 (95% CI = −4.8 to −1.7). The magnitudes of the symptom improvement for between-class and within-class switches were equivalent for aripiprazole augmentation versus placebo augmentation (Figure 2A).
Response rates were 37.3% (118/316) for aripiprazole versus 23.0% (69/300) for placebo among patients who switched between classes and 37.8% (79/209) for aripiprazole versus 22.4% (48/214) for placebo among patients who switched within class. The relative risks for response were 1.6 (95% CI = 1.3-2.1) for those patients who switched between classes, and 1.7 (95% CI = 1.2-2.2) for those who switched within class. As seen in Figure 2B, the remission rates (defined as MADRS total score ≤ 10 at endpoint and at least 50% reduction from baseline in MADRS total score) for switching between classes were 29.1% (92/316) for aripiprazole versus 16.0% (48/300) for placebo and for switching within class were 29.2% (61/209) for aripiprazole and 17.8% (38/214) for placebo. The relative risk for remission for those patients who switched between classes was 1.8 (95% CI = 1.3-2.5) and for those who switched within class was 1.6 (95% CI = 1.1-2.3). There were no differences in the percentage of patients who achieved remission between the aripiprazole group who switched between classes or within class or between the placebo groups who switched between classes or within class.
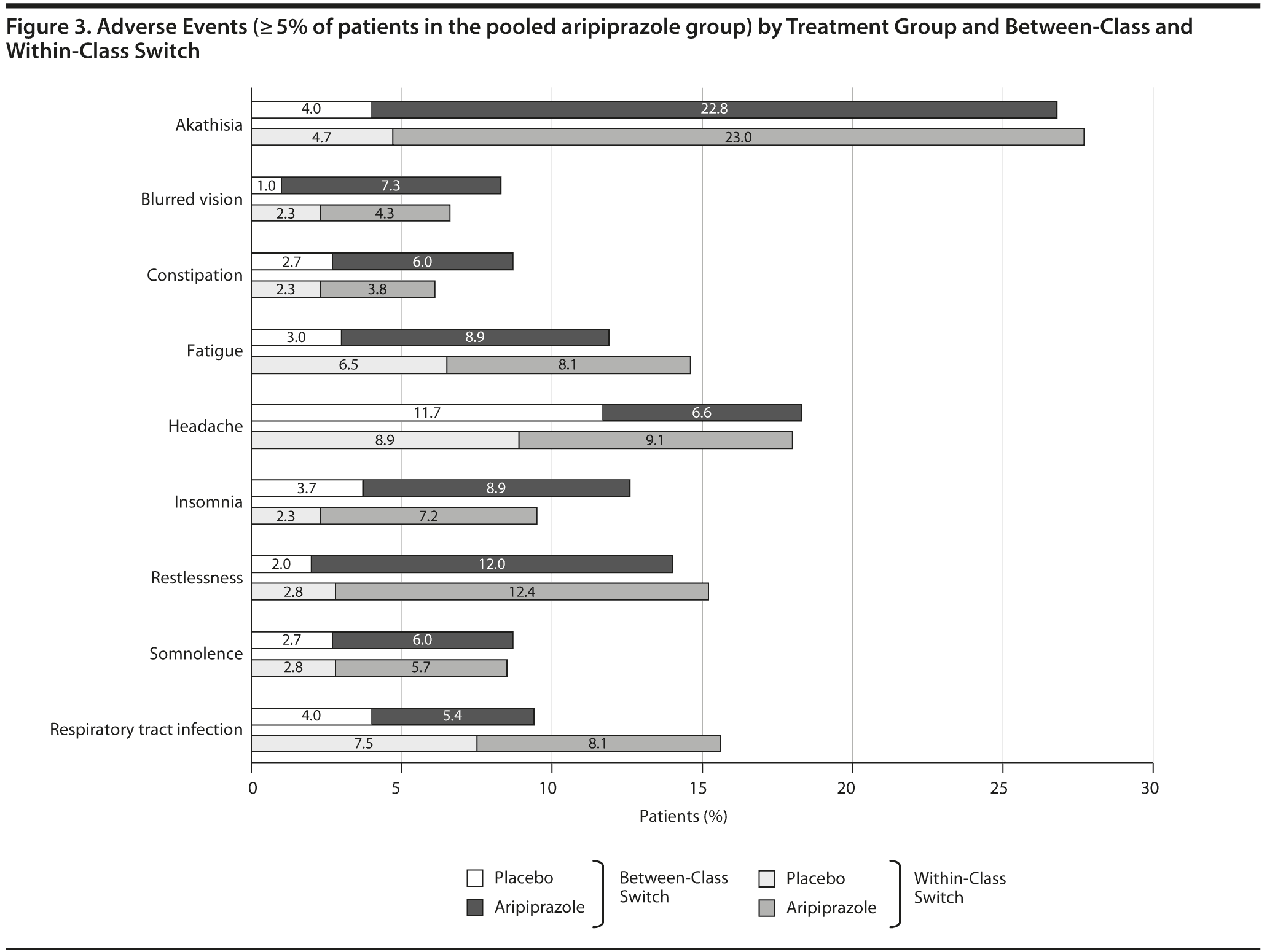
Adverse Events
Adverse events were assessed during the double-blind phase, and those occurring in ≥ 5% of patients in the pooled aripiprazole group are summarized by within-class and between-class switch in Figure 3. The most frequent adverse events in those treated with aripiprazole, regardless of whether patients switched within or between classes, were akathisia, restlessness, fatigue, insomnia, and headache. The adverse event profiles of aripiprazole in this analysis were similar to the overall populations in the primary data sets.12-14
At the end of the randomized phase, patients who received aripiprazole gained a mean of 1.9 lb (0.86 kg) more than placebo recipients in the within-class switch group (P = .001) and 2.5 lb (1.15 kg) more than placebo in the between-class switch group (P < .001, efficacy sample, OC analysis). Clinically relevant weight gain (≥ 7% from baseline) was seen in 5.1% and 1.1% of the aripiprazole and placebo groups, respectively, in those who switched within class and in 5.6% and 0.8% of the aripiprazole and placebo groups, respectively, in those who switched between classes (efficacy sample, OC analysis).
DISCUSSION
The results of this study show that augmentation with aripiprazole, after either a between-class or within-class switch following initial ADT failure, is an effective option for patients with nonresponsive MDD. Atypical antipsychotic agents work quickly as augmenting agents in patients who have not responded to ADT in contrast to the 6 to 12 weeks required for a second trial of ADT.24,25 Casey et al26 utilized the same data set as the current study and showed that more than twice as many patients taking adjunctive aripiprazole compared with adjunctive placebo achieved an early response by week 2 and that this early response was a significant predictor of endpoint remission. Further research in this area would be of interest.
This post hoc analysis is the only study to investigate the effects of switching ADT prior to augmentation with aripiprazole and answers an important question regarding the sequencing of treatment for patients with MDD. The Sequenced Treatment Alternatives to Relieve Depression (STAR*D) trial provides the most robust information to date about efficacy of sequential treatments but is limited in the treatments employed.7 The patients analyzed here most closely compare with step 3 of STAR*D, which included a mix of patients who switched within and between ADT medication class.25 The remission rates reported here with adjunctive aripiprazole (29.1%-29.2%) compare favorably with the reported remission rates during step 3 of STAR*D (13.7%).25 The atypical antipsychotics were excluded from STAR*D, although they are the only augmentation options for the treatment of MDD that are approved by the FDA. Aripiprazole, quetiapine, and the combination of olanzapine with fluoxetine are all FDA approved for this purpose,11,27 but no head-to-head studies of the atypical agents as augmentation therapy for depression have been conducted. A large meta-analysis of 16 trials comprising 3,480 patients concluded that this strategy was significantly more effective than adjunctive placebo, and there were no clinically relevant differences in efficacy among the various agents.11 However, since the methods of each of the trials varied, definitive conclusions could not be drawn regarding comparative efficacy.
In addition, studies of adjunctive quetiapine XR did not include a prospective phase, and studies of adjunctive olanzapine did not permit the investigator flexibility when selecting the antidepressant used during the prospective ADT monotherapy phase.11 Thus, the robust aripiprazole clinical trial program, which included patients who showed an inadequate response to a wide range of ADTs both historically and prospectively, provides an ideal platform to examine the effects of prior ADT history on subsequent treatment outcomes with an adjunctive atypical antipsychotic.
Not surprisingly, there was an increase in adverse events for the aripiprazole treatment groups versus placebo in this study, but discontinuations due to adverse events in the original data sets were low.12-14 Although rates of discontinuations due to adverse events have not been shown to differ among the atypical agents,11 the specific side effects that contribute to discontinuation have not been analyzed and may vary for each agent. Therefore, the adverse event profile of each agent should be taken into consideration when selecting therapy for an individual patient. The adverse event profiles in this study show that aripiprazole augmentation, regardless of whether patients switched ADTs within or between classes, is relatively well tolerated and safe during the course of treatment. It should be noted that the doses of atypical antipsychotics used as augmentation therapy for depression are generally lower than those used to treat mania or schizophrenia, and the emergence of adverse events is generally dose related.4,28 In addition, the atypical antipsychotics have been used long term in these settings without additional sequelae.29 Nevertheless, there is risk of treatment-emergent extrapyramidal symptoms and metabolic disturbances, which vary depending on the specific agent, and must be continuously monitored.29
A limitation of this study was that the patients were not randomized to either the initial treatment during the screening phase or the switch medication during the prospective phase and instead were assigned on the basis of the original physician’s preference and at the discretion of the investigator, respectively, as would be seen in clinical practice. Nevertheless, the numbers of patients in both the switch-within and -between class groups were large enough to provide power to detect a difference and were evenly distributed between aripiprazole and placebo as would have been achieved with randomization. Furthermore, the results obtained show no difference, confirming our original assumption. A similar approach was done by Shelton et al,30 who investigated the role of augmentation of ADT after switching classes, as well as in a study of ziprasidone augmentation of sertraline. 31 Both studies found that antipsychotic augmentation was associated with greater clinical effect than continued ADT monotherapy (sertraline).30,31
Overall, these results support the use of adjunctive aripiprazole in the treatment of unresponsive MDD. Furthermore, they suggest that this strategy is a viable option for patients unresponsive to ADT due to the rapid effects of low-dose aripiprazole in this population.
Drug names: aripiprazole (Abilify), bupropion (Wellbutrin, Aplenzin, and others), citalopram (Celexa and others), doxepin (Silenor and others), duloxetine (Cymbalta), escitalopram (Lexapro and others), fluoxetine (Prozac and others), fluvoxamine (Luvox and others), isocarboxazid (Marplan), mirtazapine (Remeron and others), nortriptyline (Pamelor, Aventyl, and others), olanzapine (Zyprexa), paroxetine (Paxil, Pexeva, and others), quetiapine (Seroquel), sertraline (Zoloft and others), tranylcypromine (Parnate and others), trazodone (Oleptro and others), venlafaxine (Effexor and others), ziprasidone (Geodon).
Author affiliations: Center for Anxiety and Depression, Mercer Island, Washington (Dr Dunner); Otsuka Pharmaceutical Development and Commercialization, Inc, Princeton, New Jersey (Drs Baker and Forbes); and Bristol-Myers Squibb, Wallingford, Connecticut (Drs Berman, Laubmeier, and Manos). Dr Laubmeier is currently an employee of Otsuka, and Dr Forbes is currently employed by Genentech.
Author contributions: All of the authors had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Potential conflicts of interest: Dr Dunner, in the past year, has served as a consultant to or on the advisory boards of Cervel, Cyberonics, Eli Lilly, Jazz, Neuronetics, and Pfizer; has received grant/research support from Cyberonics and Neuronetics; has served on the speakers’ bureaus of AstraZeneca, Bristol-Myers Squibb, Neuronetics, and Pfizer; and owns a Neurostar device to treat patients with repetitive transcranial magnetic stimulation. Drs Berman and Manos are employees of Bristol-Myers Squibb. Dr Laubmeier is a former employee of Bristol-Myers Squibb and a current employee of Otsuka. Dr Baker is an employee of Otsuka. Dr Forbes is a former employee of Otsuka and a current employee of Genentech.
Funding/support: The design and conduct of the study was supported by Bristol-Myers Squibb, Princeton, New Jersey, and Otsuka Pharmaceutical Co, Ltd, Tokyo, Japan, and editorial assistance was provided by Bristol-Myers Squibb.
Previous presentation: Preliminary results of these data were presented at the Annual Meeting of the American College of Neuropsychopharmacology; December 4-9, 2010; Miami Beach, Florida.
Acknowledgment: The authors would like to thank Meredith Rogers, MS, of Bristol-Myers Squibb, for providing writing and editorial assistance. Ms Rogers has no other conflicts of interest related to the subject of this article.
REFERENCES
1. Kessler RC, Berglund P, Demler O, et al; National Comorbidity Survey Replication. The epidemiology of major depressive disorder: results from the National Comorbidity Survey Replication (NCS-R). JAMA. 2003;289(23):3095-3105. PubMed doi:10.1001/jama.289.23.3095
2. Sartorius N. The economic and social burden of depression. J Clin Psychiatry. 2001;62(suppl 15):8-11. PubMed
3. Osby U, Brandt L, Correia N, et al. Excess mortality in bipolar and unipolar disorder in Sweden. Arch Gen Psychiatry. 2001;58(9):844-850. PubMed doi:10.1001/archpsyc.58.9.844
4. Gelenberg AJ, Freeman MP, Markowitz JC, et al. Practice Guideline for the Treatment of Patients With Major Depressive Disorder, Third Edition. http://psychiatryonline.org/guidelines.aspx. Accessed August 30, 2012.
5. Stafford RS, Ausiello JC, Misra B, et al. National patterns of depression treatment in primary care. Prim Care Companion J Clin Psychiatry. 2000;2(6):211-216. PubMed doi:10.4088/PCC.v02n0603
6. Marcus SC, Olfson M. National trends in the treatment for depression from 1998 to 2007. Arch Gen Psychiatry. 2010;67(12):1265-1273. PubMed doi:10.1001/archgenpsychiatry.2010.151
7. Rush AJ, Trivedi MH, Wisniewski SR, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163(11):1905-1917. PubMed doi:10.1176/appi.ajp.163.11.1905
8. Papakostas GI, Fava M, Thase ME. Treatment of SSRI-resistant depression: a meta-analysis comparing within- versus across-class switches. Biol Psychiatry. 2008;63(7):699-704. PubMed doi:10.1016/j.biopsych.2007.08.010
9. Trangle M, Dieperink B, Gabert T, et al. Institute for Clinical Systems Improvement. Depression, Major, in Adults in Primary Care (Guideline). http://www.icsi.org/guidelines_and_more/gl_os_prot/behavioral_health/depression_5/depression__major__in_adults_in_primary_care_4.html.Updated May 31, 2012. Accessed August 30, 2012.
10. National Collaborating Centre for Mental Health. Depression. The treatment and management of depression in adults (National Institute for Health and Clinical Excellence clinical guideline 90). http://www.nice.org.uk/nicemedia/live/12329/45888/45888.pdf. Updated October 2009. Accessed August 30, 2012.
11. Nelson JC, Papakostas GI. Atypical antipsychotic augmentation in major depressive disorder: a meta-analysis of placebo-controlled randomized trials. Am J Psychiatry. 2009;166(9):980-991. PubMed doi:10.1176/appi.ajp.2009.09030312
12. Berman RM, Marcus RN, Swanink R, et al. The efficacy and safety of aripiprazole as adjunctive therapy in major depressive disorder: a multicenter, randomized, double-blind, placebo-controlled study. J Clin Psychiatry. 2007;68(6):843-853. PubMed doi:10.4088/JCP.v68n0604
13. Marcus RN, McQuade RD, Carson WH, et al. The efficacy and safety of aripiprazole as adjunctive therapy in major depressive disorder: a second multicenter, randomized, double-blind, placebo-controlled study. J Clin Psychopharmacol. 2008;28(2):156-165. PubMed doi:10.1097/JCP.0b013e31816774f9
14. Berman RM, Fava M, Thase ME, et al. Aripiprazole augmentation in major depressive disorder: a double-blind, placebo-controlled study in patients with inadequate response to antidepressants. CNS Spectr. 2009;14(4):197-206. PubMed
15. Burris KD, Molski TF, Xu C, et al. Aripiprazole, a novel antipsychotic, is a high-affinity partial agonist at human dopamine D2 receptors. J Pharmacol Exp Ther. 2002;302(1):381-389. PubMed doi:10.1124/jpet.102.033175
16. Jordan S, Koprivica V, Chen R, et al. The antipsychotic aripiprazole is a potent, partial agonist at the human 5-HT1A receptor. Eur J Pharmacol. 2002;441(3):137-140. PubMed doi:10.1016/S0014-2999(02)01532-7
17. Shapiro DA, Renock S, Arrington E, et al. Aripiprazole, a novel atypical antipsychotic drug with a unique and robust pharmacology. Neuropsychopharmacology. 2003;28(8):1400-1411. PubMed doi:10.1038/sj.npp.1300203
18. Stark AD, Jordan S, Allers KA, et al. Interaction of the novel antipsychotic aripiprazole with 5-HT1A and 5-HT 2A receptors: functional receptor-binding and in vivo electrophysiological studies. Psychopharmacology (Berl). 2007;190(3):373-382. PubMed doi:10.1007/s00213-006-0621-y
19. Kikuchi T, Tottori K, Uwahodo Y, et al. 7-(4-[4-(2,3-Dichlorophenyl)-1-piperazinyl]butyloxy)-3,4-dihydro-2(1H)-quinolinone (OPC-14597), a new putative antipsychotic drug with both presynaptic dopamine autoreceptor agonistic activity and postsynaptic D2 receptor antagonistic activity. J Pharmacol Exp Ther. 1995;274(1):329-336. PubMed
20. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision. Washington, DC: American Psychiatric Association; 2000.
21. Fava M. Diagnosis and definition of treatment-resistant depression. Biol Psychiatry. 2003;53(8):649-659. PubMed doi:10.1016/S0006-3223(03)00231-2
22. Chandler GM, Iosifescu DV, Pollack MH, et al. RESEARCH: validation of the Massachusetts General Hospital Antidepressant Treatment History Questionnaire (ATRQ). CNS Neurosci Ther. 2010;16(5):322-325. PubMed doi:10.1111/j.1755-5949.2009.00102.x
23. Thase ME, Trivedi MH, Nelson JC, et al. Examining the efficacy of adjunctive aripiprazole in major depressive disorder: a pooled analysis of 2 studies. Prim Care Companion J Clin Psychiatry. 2008;10(6):440-447. PubMed doi:10.4088/PCC.v10n0603
24. Chen J, Gao K, Kemp DE. Second-generation antipsychotics in major depressive disorder: update and clinical perspective. Curr Opin Psychiatry. 2011;24(1):10-17. PubMed doi:10.1097/YCO.0b013e3283413505
25. Trivedi MH, Rush AJ, Wisniewski SR, et al; STAR*D Study Team. Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: implications for clinical practice. Am J Psychiatry. 2006;163(1):28-40. PubMed doi:10.1176/appi.ajp.163.1.28
26. Casey DE, Laubmeier K, Vogel Marler S, et al. Efficacy of adjunctive aripiprazole in major depressive disorder: a pooled response quartile analysis and the predictive value of week 2 early response. Prim Care Companion CNS Disord. 2012;14(3):e1-e7 doi:10.4088/PCC.11m01251
27. McElroy SL, Guerdjikova A, Mori N, et al. Therapeutic potential of new second generation antipsychotics for major depressive disorder. Expert Opin Investig Drugs. 2010;19(12):1527-1544. PubMed doi:10.1517/13543784.2010.538048
28. Lam RW, Kennedy SH, Grigoriadis S, et al; Canadian Network for Mood and Anxiety Treatments (CANMAT). Canadian Network for Mood and Anxiety Treatments (CANMAT) clinical guidelines for the management of major depressive disorder in adults, 3: pharmacotherapy. J Affect Disord. 2009;117(suppl 1):S26-S43. PubMed doi:10.1016/j.jad.2009.06.041
29. McDonagh MS, Peterson K, Carson S, et al. Drug class review: atypical antipsychotic drugs: final update 3 report [Internet]. Portland, Oregon: Oregon Health and Science University; 2010. PubMed
30. Shelton RC, Tollefson GD, Tohen M, et al. A novel augmentation strategy for treating resistant major depression. Am J Psychiatry. 2001;158(1):131-134. PubMed doi:10.1176/appi.ajp.158.1.131
31. Dunner DL, Amsterdam JD, Shelton RC, et al. Efficacy and tolerability of adjunctive ziprasidone in treatment-resistant depression: a randomized, open-label, pilot study. J Clin Psychiatry. 2007;68(7):1071-1077. doi:10.4088/JCP.v68n0714 PubMed