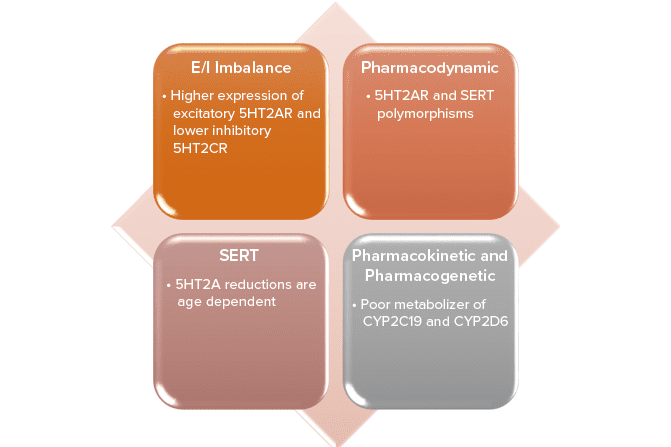
Because this piece has no abstract, we have provided for your benefit the first 3 sentences of the full text.
Sequenced Treatment Alternatives toRelieve Depression (STAR*D), the largestprospective, randomized antidepressanttreatment trial to date in outpatients withmajor depressive disorder (MDD) recruitedfrom real-world clinical settings, enrolling4011 outpatients aged 18 to 75 years withnonpsychotic MDD. Designed to determinewhich treatments are most effective afternonremission or intolerance to an initial selectiveserotonin reuptake inhibitor (SSRI),or to any of a series of subsequent randomizedtreatments, STAR*D was conductedin 18 primary and 23 psychiatric care settingsacross the United States. This reviewsummarizes unique features of the study,initial remission rates and associated participantcharacteristics, remission rates forsubsequent treatment steps, relapse ratesduring follow-up, and clinical implications.’ ‹
Enjoy this premium PDF as part of your membership benefits!