Objective: To examine long-term (11-month) antidepressant efficacy of desvenlafaxine 50 mg/d across a broad range of clinical and functional outcomes in patients with major depressive disorder.
Method: Adult outpatients (≥ 18 years) with major depressive disorder (DSM-IV criteria) and a 17-item Hamilton Depression Rating Scale (HDRS-17) total score ≥ 20 at screening and baseline who responded to 8 weeks of open-label desvenlafaxine 50 mg/d and had a continuing stable response through week 20 were randomly assigned to receive placebo or desvenlafaxine 50 mg/d in a 6-month, double-blind, randomized withdrawal period. Depressive symptoms were evaluated using the HDRS-17, 6-item HDRS, and Clinical Global Impressions-Severity of Ilness and -Improvement (CGI-S, CGI-I). Health outcomes included the Work Productivity and Activity Impairment (WPAI) questionnaire and the World Health Organization 5-Item Well-Being Index (WHO-5). The trial was conducted from June 2009 to March 2011 at 87 study sites in 14 countries worldwide.
Results: Of 874 patients enrolled in open-label treatment, 548 patients were randomly assigned to receive double-blind placebo (n = 276) or desvenlafaxine 50 mg/d (n = 272). At the end of the 6-month double-blind treatment, improvements in depressive symptoms were better maintained among the desvenlafaxine- than placebo-treated patients on all efficacy endpoints (all P ≤ .001); in the desvenlafaxine group, 21.8% (vs 42.9% in the placebo group) had CGI-I ratings of 5, 6, and 7 (minimally worse/much worse/very much worse), and 74.4% met criteria for remission (placebo: 54.2%). WPAI and WHO-5 scores indicated significantly better productivity and well-being with continued desvenlafaxine (vs placebo, P ≤ .001).
Conclusions: Long-term treatment with desvenlafaxine 50 mg/d maintained improvements in major depressive disorder among adult outpatients who exhibited a stable therapeutic response.
Trial Registration: ClinicalTrials.gov identifier: NCT00887224
Efficacy of Desvenlafaxine 50 mg/d Versus Placebo
in the Long-Term Treatment of Major Depressive Disorder:
A Randomized, Double-Blind Trial
ABSTRACT
Objective: To examine long-term (11-month) antidepressant efficacy of desvenlafaxine 50 mg/d across a broad range of clinical and functional outcomes in patients with major depressive disorder.
Method: Adult outpatients (≥ 18 years) with major depressive disorder (DSM-IV criteria) and a 17-item Hamilton Depression Rating Scale (HDRS-17) total score ≥ 20 at screening and baseline who responded to 8 weeks of open-label desvenlafaxine 50 mg/d and had a continuing stable response through week 20 were randomly assigned to receive placebo or desvenlafaxine 50 mg/d in a 6-month, double-blind, randomized withdrawal period. Depressive symptoms were evaluated using the HDRS-17, 6-item HDRS, and Clinical Global Impressions–Severity of Ilness and –Improvement (CGI-S, CGI-I). Health outcomes included the Work Productivity and Activity Impairment (WPAI) questionnaire and the World Health Organization 5-Item Well-Being Index (WHO-5). The trial was conducted from June 2009 to March 2011 at 87 study sites in 14 countries worldwide.
Results: Of 874 patients enrolled in open-label treatment, 548 patients were randomly assigned to receive double-blind placebo (n = 276) or desvenlafaxine 50 mg/d (n = 272). At the end of the 6-month double-blind treatment, improvements in depressive symptoms were better maintained among the desvenlafaxine- than placebo-treated patients on all efficacy endpoints (all P ≤ .001); in the desvenlafaxine group, 21.8% (vs 42.9% in the placebo group) had CGI-I ratings of 5, 6, and 7 (minimally worse/much worse/very much worse), and 74.4% met criteria for remission (placebo: 54.2%). WPAI and WHO-5 scores indicated significantly better productivity and well-being with continued desvenlafaxine (vs placebo, P ≤ .001).
Conclusions: Long-term treatment with desvenlafaxine 50 mg/d maintained improvements in major depressive disorder among adult outpatients who exhibited a stable therapeutic response.
Trial Registration: ClinicalTrials.gov identifier: NCT00887224
Prim Care Companion CNS Disord
2015;17(4):doi:10.4088/PCC.14m01711
© Copyright 2015 Physicians Postgraduate Press, Inc.
Submitted: August 13, 2014; accepted April 7, 2015.
Published online: August 27, 2015.
Corresponding author: Patrice Boyer, MD, PhD, University Paris 7, 100 rue de la Santé, 75014, Paris, France
([email protected]).
Major depressive disorder (MDD) is an often chronic condition with symptoms that can recur over time.1,2 The goal of therapy for MDD is remission, marked clinically by no or minimal depressive symptoms and normalized functioning across multiple settings.3–5 Because relapse is common in the first 6 months after recovery from a depressive episode, a successful acute phase treatment regimen should be continued for 4 to 9 months after symptoms resolve.3–5 For some patients with recurrent MDD, longer-term maintenance therapy (> 1 year or indefinite) may be needed.5
Desvenlafaxine (administered as desvenlafaxine succinate) is a serotonin-norepinephrine reuptake inhibitor (SNRI) approved in more than 30 countries for the treatment of MDD in adults.6 Several double-blind, placebo-controlled studies have demonstrated the acute phase (8-week) antidepressant efficacy of desvenlafaxine at the recommended dose of 50 mg/d.7–9 In these trials, 34%–37% of patients given short-term treatment with desvenlafaxine 50 mg/d achieved remission, defined as a 17-item Hamilton Depression Rating Scale (HDRS-17)10 total score ≤ 7.7–9 The short-term trials reported rates of early discontinuation due to adverse events or inadequate efficacy ranging from 3.3%–7.7%, indicating that desvenlafaxine 50 mg/d was effective and generally well-tolerated.7–9
Long-term (> 8 weeks) efficacy and safety of desvenlafaxine 50 mg/d was assessed in an 11-month, phase 3, double-blind, randomized withdrawal study in adults with MDD.11 Patients who responded to 8-week, open-label desvenlafaxine 50-mg/d treatment and continued to have a stable response over 12 additional weeks of open-label desvenlafaxine 50-mg/d treatment were randomly assigned to receive double-blind desvenlafaxine 50 mg/d or placebo for 6 months, with the primary efficacy endpoint of time to relapse. The rate of relapse during the double-blind period was more than 2-fold greater among patients receiving placebo versus desvenlafaxine (30.2% vs 14.3%, P < .001).11
Among patients who respond to treatment for a depressive episode, many who do not relapse nonetheless suffer residual symptoms.12 Thus, for drugs that have demonstrated relapse prevention versus placebo, one of the frequent questions raised by clinicians is “How are the patients who are maintained on a long-term basis with the chosen treatment actually doing?” Prespecified secondary efficacy outcomes of the desvenlafaxine relapse prevention study that were not reported in the primary article11 address this question. The objective of the current report is to describe the antidepressant response of patients receiving desvenlafaxine 50 mg/d versus placebo in an 11-month randomized withdrawal study using a broad set of prespecified outcomes, which are complimentary to the assessment of relapse rate. All safety and tolerability results from the study were reported previously.11
METHOD
Study Design and Overview
Study design and methods were reported previously.11 Briefly, this was a phase 3 multicenter investigation conducted at 87 study sites in 14 countries worldwide from June 2009 to March 2011. The study included a screening period, an open-label treatment period (8-week response phase + 12-week stability phase), and a 6-month, double-blind, placebo-controlled, randomized withdrawal period. Patients who responded to open-label desvenlafaxine 50-mg/d treatment at week 8 (HDRS-17 total score ≤ 11 and Clinical Global Impressions–Improvement [CGI-I]13 score ≤ 2) had a continued stable response through 12 additional weeks of open-label desvenlafaxine 50-mg/d treatment (HDRS-17 total score ≤ 11 and CGI-I score ≤ 2 at study week 20, and no HDRS-17 total score ≥ 16 or CGI-I score ≥ 4 at any visit during the stability phase) were eligible for entry into the double-blind treatment period. Eligible patients were randomly assigned 1:1 to receive 6-month double-blind treatment with placebo or desvenlafaxine 50 mg/d. Patients continued with assigned treatment until study completion or depression relapse, defined as ≥ 1 of the following: HDRS-17 score ≥ 16; discontinuation due to inadequate response, need for additional/alternative antidepressant treatment, or failure to return to the scheduled visit considered related to inadequate efficacy; hospitalized for depression or suicide attempt; or suicide. Patients completing double-blind treatment or discontinuing the study early entered a 7-day taper period (desvenlafaxine dose lowered to 25 mg/d) and a 7-day follow-up.
Study Population
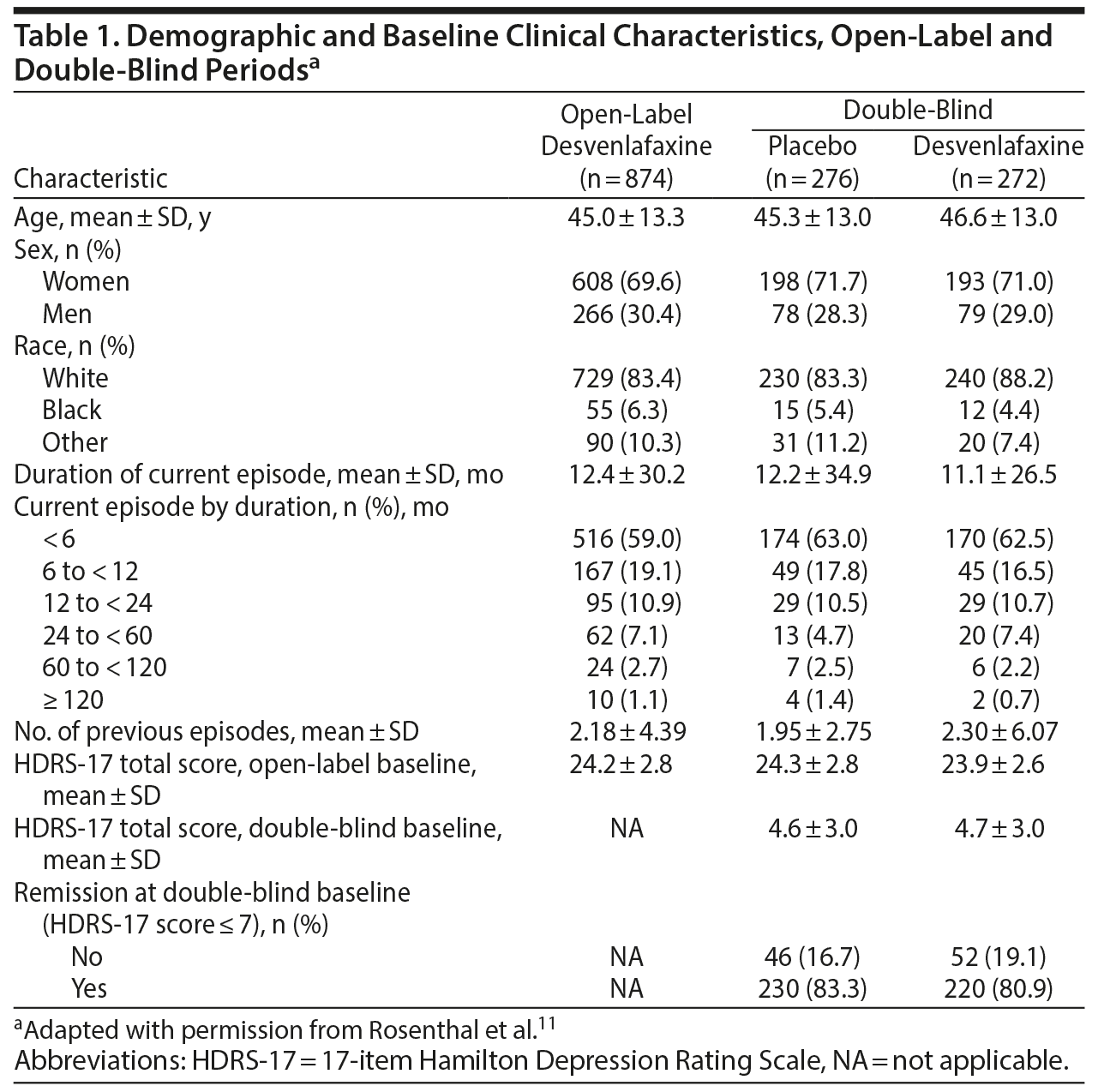
This study enrolled adult outpatients (aged ≥ 18 years) with a primary diagnosis of a single episode of MDD or recurrent MDD without psychotic features consistent with DSM-IV criteria. Depressive symptoms must have been present for ≥ 30 days prior to screening and were evidenced by a HDRS-17 total score ≥ 20, a HDRS-17 item 1 (depressed mood) score ≥ 2, and a CGI–Severity of Illness (CGI-S) score ≥ 4. Comorbid generalized anxiety, panic, or social anxiety disorders were permitted if MDD was the primary diagnosis. Individuals were excluded if they were treated with desvenlafaxine at any time in the past or had a significant risk of suicide based on clinical judgment or a HDRS-17 item 3 (suicide) score > 3 at screening. Additional exclusion criteria included clinically important medical disease, seizure disorder, comorbid substance use disorder, current manic episode, bipolar disorder, psychotic disorder, posttraumatic stress disorder, obsessive-compulsive disorder, or clinically important personality disorder.
Written informed consent was obtained from all subjects before their enrollment. This study was conducted in accordance with the International Conference on Harmonization Guideline for Good Clinical Practice and the ethical principles that have their origins in the Declaration of Helsinki. The protocol, the investigator’s brochure, and the informed consent form for this clinical study were submitted to an institutional review board or an independent ethics committee for review and written approval (ClinicalTrials.gov identifier: NCT00887224).
Efficacy Endpoints
The primary efficacy outcome of the study was time to relapse following randomization to the double-blind treatment period. Results for the primary outcome were described previously.11
Protocol-specified secondary efficacy endpoints for the study not reported in the primary article were HDRS-17 and 6-item HDRS subscale (HDRS-6) total scores, CGI-I and CGI-S scores, and the proportion of patients achieving remission (HDRS-17 total score ≤ 7). The HDRS-6 was included because of its greater sensitivity to change on core aspects of depression compared with the HDRS-17, to further explore its use as an alternate to the HDRS-17.14 Secondary efficacy assessments were obtained at screening and baseline (except CGI-I) and at scheduled clinic visits throughout the open-label and double-blind periods or at final study visit/early termination. Double-blind efficacy assessments were at double-blind weeks 1, 2, 3, 4, 6, 10, 14, 18, 22, and 26. In the double-blind period, CGI-I score was based on a comparison with double-blind baseline (study week 20). The order of administration of the efficacy assessments was consistent throughout the study, with the HDRS-17 administered first and the CGI administered last. Every effort was made to ensure that the same rater administered efficacy assessments at each study visit. Rater training was conducted at the beginning of the study and repeated while the study was in progress.
Health Outcomes
Patient-perceived work productivity and well-being were assessed using the Work Productivity and Activity Impairment (WPAI) questionnaire and the World Health Organization 5-item Well-Being Index (WHO-5). The WPAI15 is a 6-item instrument designed to measure the effect of symptom severity on work productivity lost and everyday activities over the previous 7 days. Four WPAI scores are generated: absenteeism (amount of work time missed), presenteeism (reduced effectiveness at work), work productivity loss (absenteeism plus presenteeism), and impairment in everyday activities. Higher scores represent a greater decrease in function for each measure. The
WHO-516 measures the frequency of 5 items describing a positive emotional state (cheerful and in good spirits, calm and relaxed, active and vigorous, fresh and rested, daily life has been filled with things that interest me) on a scale from 0 (at no time) to 5 (all the time) over the past 2 weeks. A total score (range, 0–25) is calculated by summing the 5 item scores. Patients completed the WPAI and WHO-5 at baseline, weeks 8 and 20 of open-label treatment, and weeks 14 and 26 of double-blind treatment or at final study visit/early termination.
Statistical Analysis
For analysis of HDRS-17, HDRS-6, and CGI-S efficacy outcomes during the open-label period, descriptive statistics were obtained for the all-enrolled population (all patients who were eligible to enter the open-label period and assigned to treatment) using the last observation carried forward (LOCF) approach for handling missing values. These outcomes were also submitted to a mixed-effects model for repeated measures (MMRM) analysis using data from the all-randomized population (all eligible patients assigned to double-blind treatment) with treatment, visit, treatment-by-visit interaction, and site as factors and the corresponding double-blind baseline score as a covariate. Double-blind WPAI and WHO-5 data were analyzed in the same manner. Remission status at each double-blind study visit (LOCF) was analyzed by a logistic model with treatment and sites as factors and baseline HDRS-17 total score as covariate. For the CGI-I, descriptive statistics were determined for the open-label and double-blind periods in the all-enrolled and all-randomized populations, respectively.
RESULTS
Patients
Among 874 enrolled patients, 548 were randomly assigned to receive placebo (n = 276) or desvenlafaxine 50 mg/d (n = 272) in the double-blind period (Figure 1). Table 1 summarizes demographic and baseline characteristics by treatment group. During the double-blind period, 162 patients discontinued early; in both treatment groups, the most common reason was lack of efficacy (placebo: n = 67, desvenlafaxine: n = 33). In the double-blind withdrawal period, 57.2% of patients in the placebo group and 54.4% of patients in the desvenlafaxine 50-mg/d group reported treatment-emergent adverse events.11
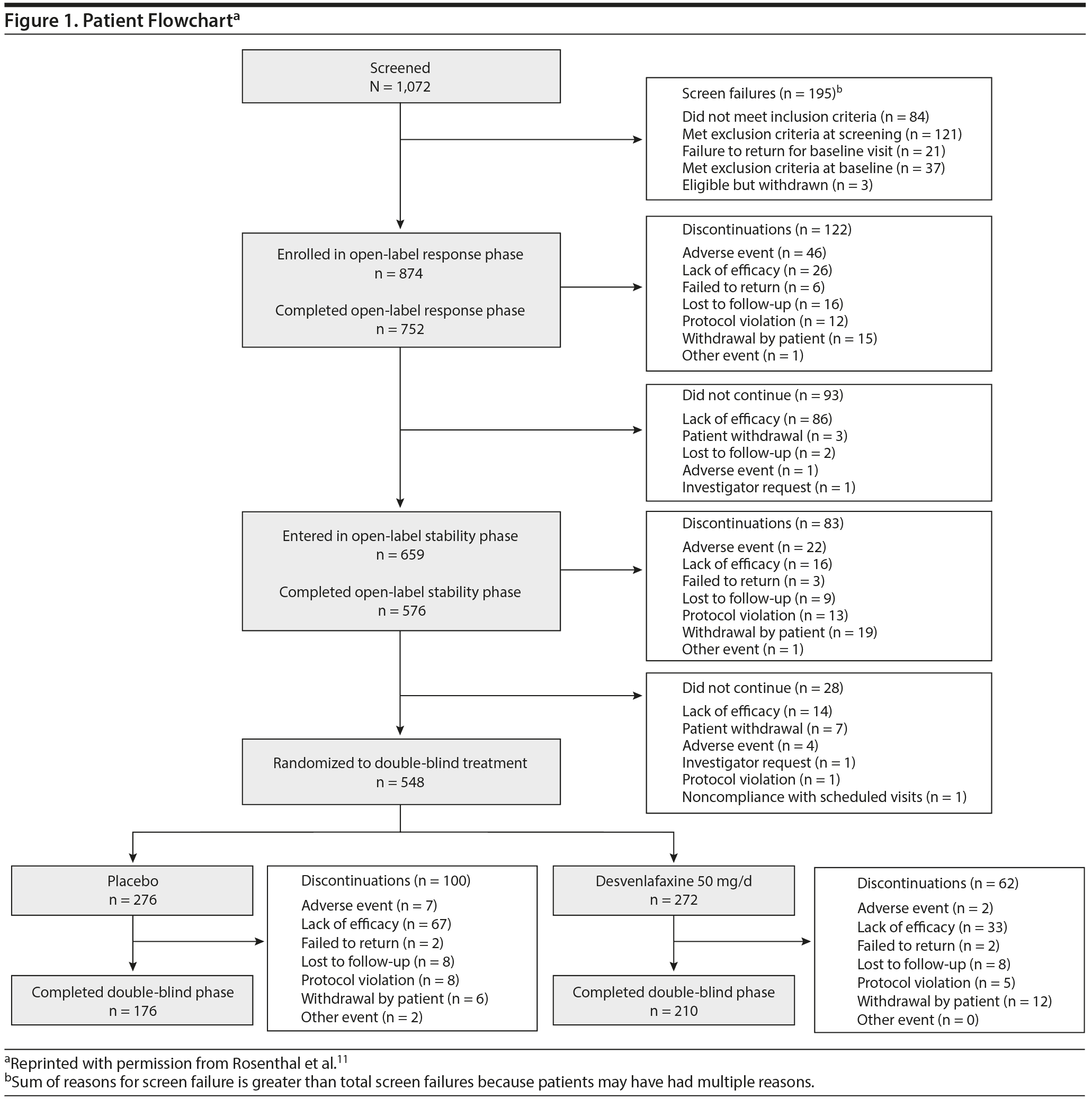
Efficacy
HDRS-17. During the open-label period, HDRS-17 total scores improved from a mean (SD) of 24.21 (2.82) at open-label baseline to 9.29 (6.42) at week 8 (LOCF); improvements in HDRS-17 total score were maintained through week 20 (Figure 2). In the double-blind period, adjusted mean HDRS-17 total scores (MMRM) increased slightly in both treatment groups. This increase was greater in the placebo group than the desvenlafaxine group beginning at double-blind week 2 and continuing through double-blind week 26 (Figure 2). At double-blind week 26, the adjusted mean (SE) increase from double-blind baseline in HDRS-17 total score was statistically significantly greater in the placebo group than the desvenlafaxine group (adjusted mean difference [95% CI]=2.35 [1.39–3.32], P < .001) (Table 2).
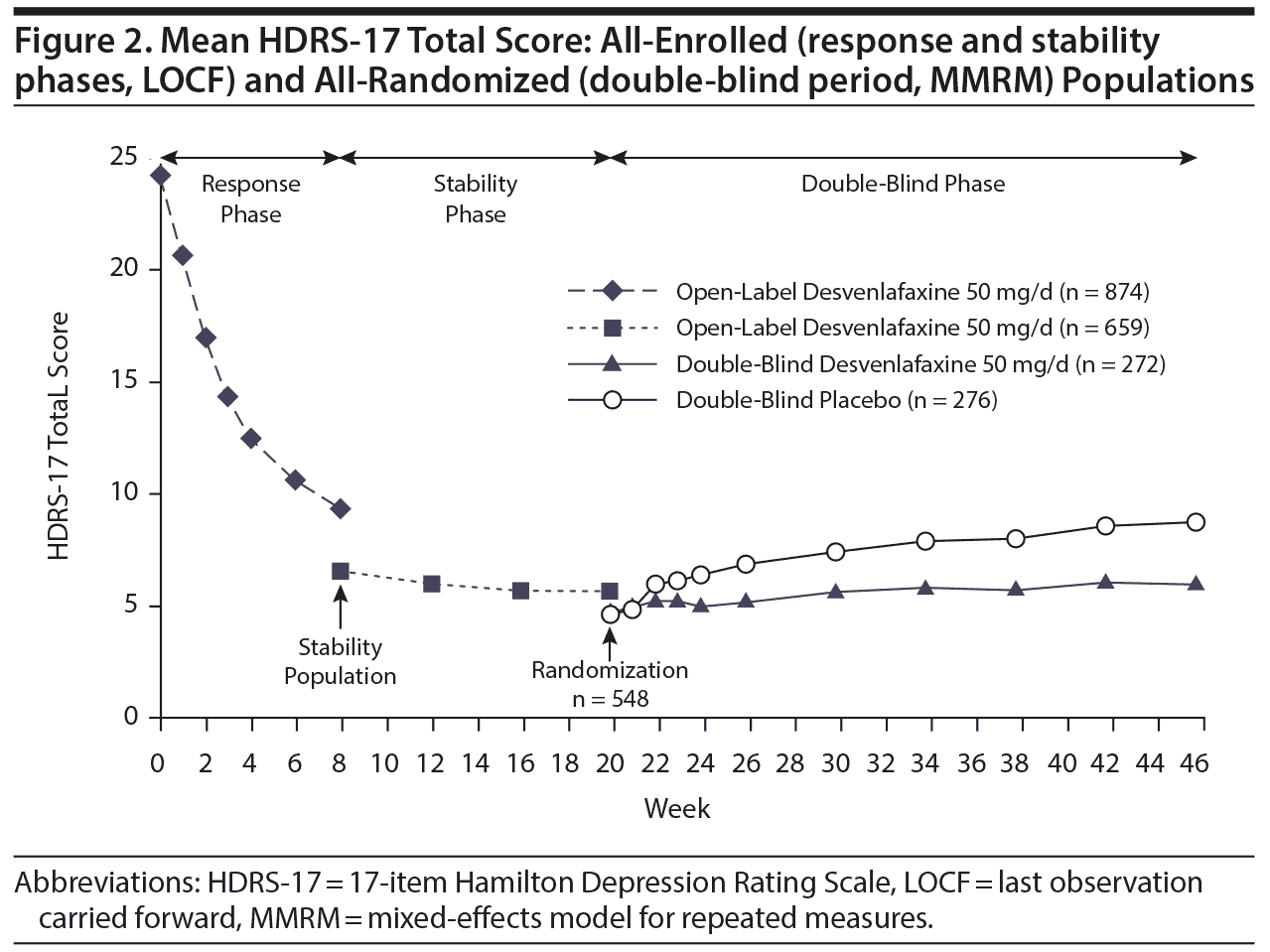
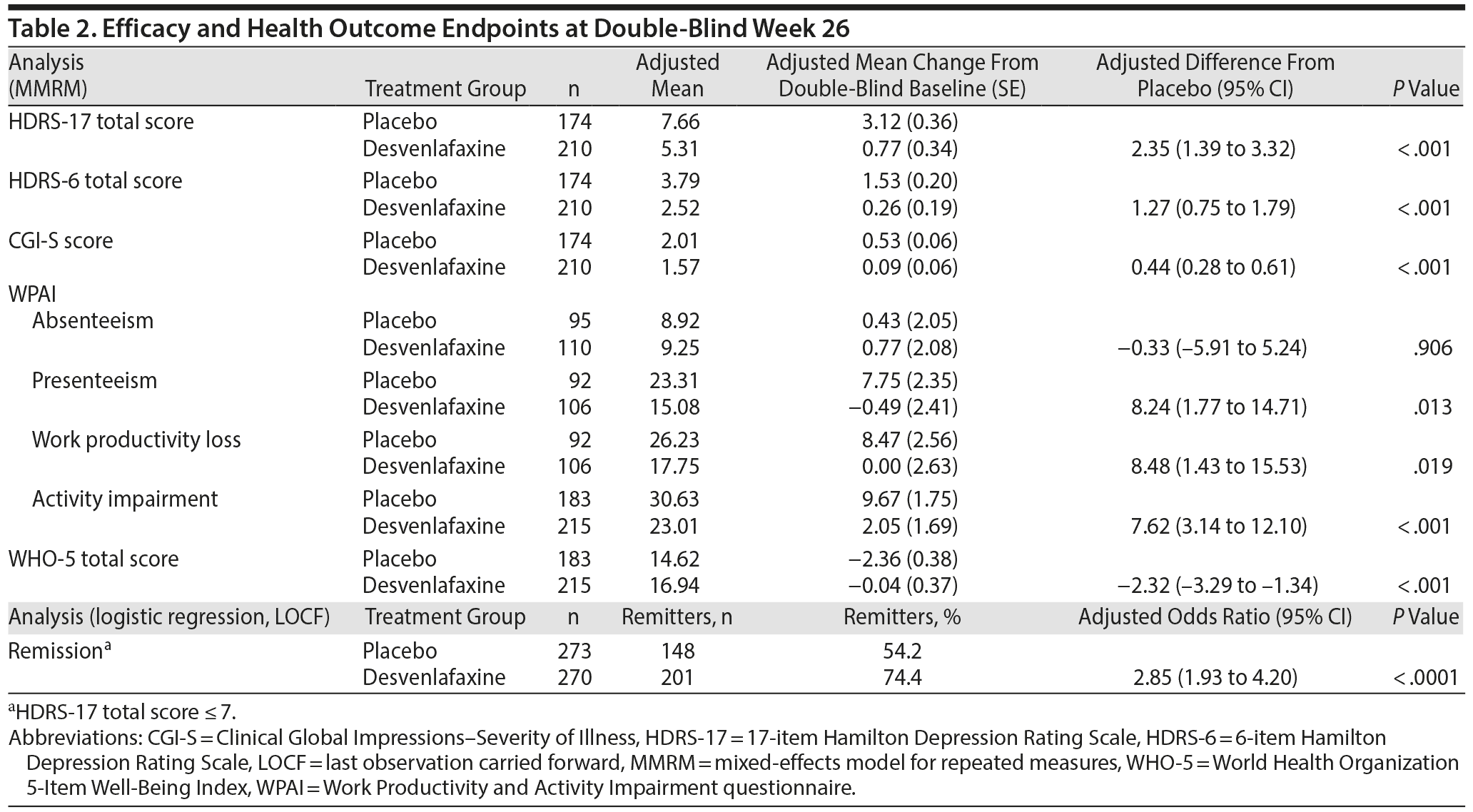
HDRS-6 total score. Mean (SD) HDRS-6 total score decreased from 12.83 (1.63) at open-label baseline to 4.77 (3.64) at open-label week 8, LOCF (change from open-label baseline: −8.06 [3.81]); decreases were maintained during the open-label stability phase (change from open-label baseline at open-label week 20: −9.95 [2.97]). During the double-blind period, mean (SD) HDRS-6 total scores remained largely unchanged in the desvenlafaxine group but increased in the placebo group. A statistically significant difference between groups was observed beginning at double-blind week 2 and continuing through double-blind week 26 (MMRM, all P ≤ .007). At double-blind week 26, the adjusted mean (SE) increase from double-blind baseline in HDRS-6 total score was statistically significantly greater in the placebo group than in the desvenlafaxine group (adjusted mean difference [95% CI]=1.27 [0.75–1.79], P < .001) (Table 2).
Remission. The proportion of patients achieving remission (HDRS-17 total score ≤ 7) was assessed in each study period. At the end of open-label week 8 (LOCF), 46.8% of patients met the criteria for remission; the proportion of patients who had remitted increased to 75.9% at the end of the open-label stability phase (week 20, LOCF). At double-blind baseline, similar proportions of patients in the placebo and desvenlafaxine groups were in remission (83.3% and 80.9%, respectively). During double-blind treatment, there was a significantly greater decrease in the remission rate for the placebo group compared with the desvenlafaxine group beginning at double-blind week 2 (all P ≤ .0497). At double-blind week 26 (LOCF), the proportion of patients in remission was significantly greater in the desvenlafaxine group than in the placebo group (P < .0001) (Table 2).
CGI Assessments. During the open-label response phase, mean (SD) CGI-S scores improved from a baseline of 4.48 (0.58) to 2.26 (1.11) at open-label week 8 (LOCF). CGI-S scores were maintained during the open-label stability phase, with a mean (SD) CGI-S score of 1.68 (0.87) at open-label week 20 (LOCF). During the double-blind period, CGI-S scores remained largely unchanged from double-blind baseline in the desvenlafaxine group but increased significantly in the placebo group compared with desvenlafaxine beginning at double-blind week 3. At the end of the double-blind period (week 26, LOCF), the adjusted mean increase in CGI-S score from double-blind baseline was statistically significantly greater in the placebo group than in the desvenlafaxine group (P < .001) (Table 2).
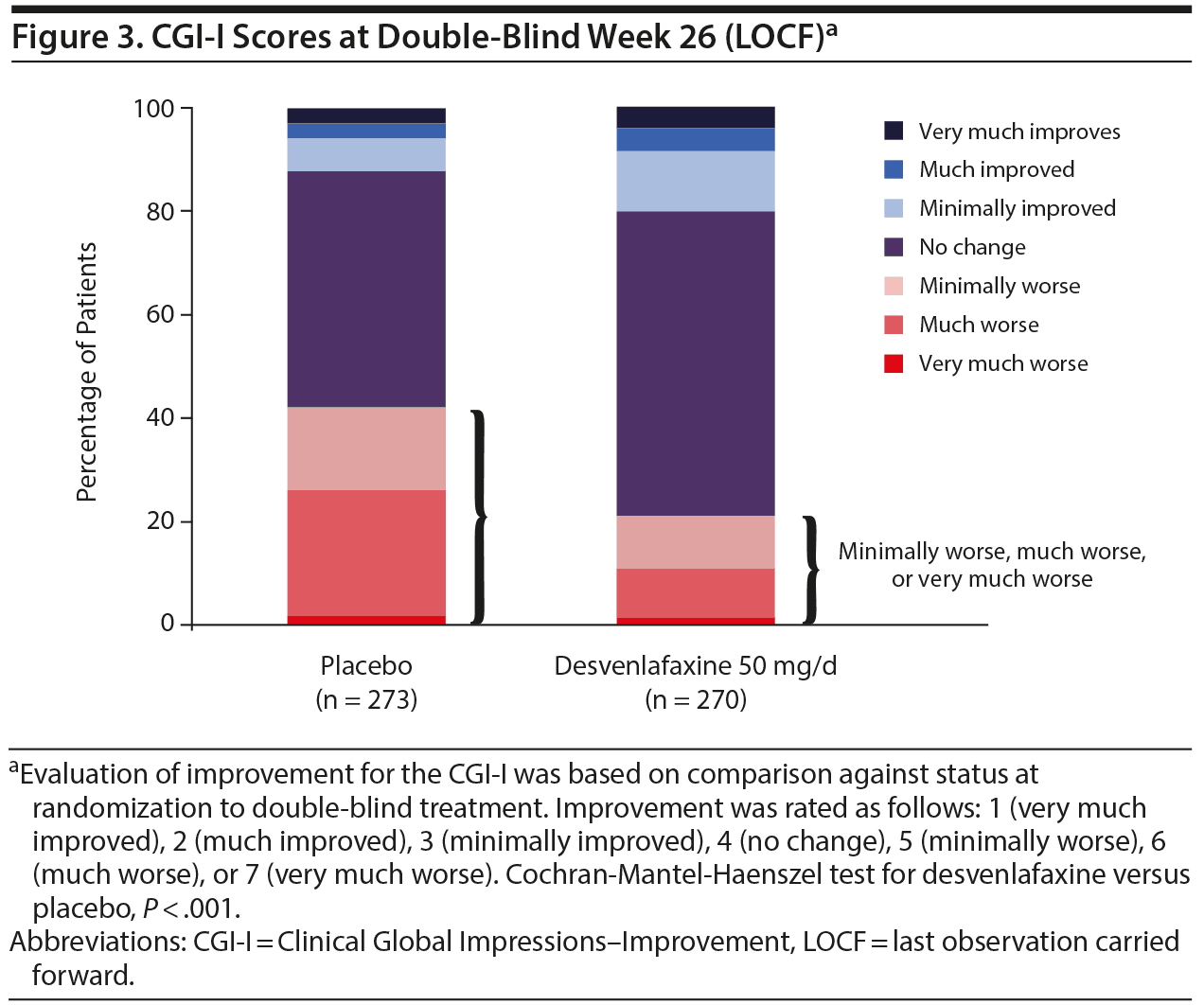
At the end of the open-label response phase (open-label week 8, LOCF), 55% and 27.7% of patients had a CGI-I rating of 1 (very much improved) or 2 (much improved), respectively; those proportions were 77.5% and 16.1%, respectively, at the end of the open-label stability phase (open-label week 20, LOCF). At the end of the double-blind period (week 20, LOCF), 78.1% of patients in the desvenlafaxine group and 57.1% of patients in the placebo group had CGI-I ratings of 1 to 4. The percentages of patients with CGI-I ratings of 5 (minimally worse), 6 (much worse), or 7 (very much worse) were higher in the placebo group than in the desvenlafaxine group (P < .001) (Figure 3).
Health Outcomes
WPAI. During the open-label response phase, improvements in all 4 WPAI domains (absenteeism, presenteeism, work productivity loss [absenteeism plus presenteeism], and impairment in everyday activities) were observed; at open-label week 8 (LOCF), the largest mean (SD) change from baseline was seen on the activity impairment domain (–31.0 [30.5]). These improvements were maintained throughout the open-label stability phase. During the double-blind period, a significant advantage of desvenlafaxine over placebo was seen at double-blind weeks 14 and 26 (LOCF) in the WPAI domains of presenteeism, work productivity loss, and activity impairment (all P ≤ .041), but not in absenteeism (Table 2).
WHO-5. During the open-label phase, WHO-5 scores improved and were maintained; mean (SD) change from baseline in WHO-5 total score was 8.35 (6.25) at open-label week 8 (LOCF) and 10.65 (6.20) at open-label week 20 (LOCF). During the double-blind period, significant between-group differences in WHO-5 total score favored desvenlafaxine (vs placebo) at double-blind weeks 14 and 26, LOCF (both P < .001). Mean WHO-5 total score declined from double-blind baseline for placebo-treated patients but showed little change from double-baseline baseline at double-blind week 26 for desvenlafaxine-treated patients (Table 2).
DISCUSSION
The results of the current analysis characterize the long-term (20 weeks open-label + 6 months double-blind) antidepressant response to desvenlafaxine 50-mg/d treatment using a broad set of outcomes that are complementary to the assessment of the relapse rate. In adult outpatients receiving desvenlafaxine 50 mg/d who had responded to 8-week open-label treatment and then maintained a stable response for an additional 12 weeks with continued open-label treatment, antidepressant efficacy was demonstrated compared with placebo through 6 months of double-blind treatment. The primary analysis for the study demonstrated that desvenlafaxine 50 mg/d significantly delayed time to relapse versus placebo, and the estimated probability of relapse was approximately twice as high for placebo compared with the desvenlafaxine group (30.2% vs 14.3%).11 Rosenthal and colleagues11 reported only those primary efficacy results, together with safety data, in the primary article. The prespecified secondary efficacy outcomes for the study reported here extend the results of the analysis of relapse to show that continued administration of desvenlafaxine 50 mg/d after 20 weeks of open-label treatment maintained improvement in depressive symptoms and functional outcomes versus placebo.
Long-term (> 8 weeks) efficacy at higher desvenlafaxine dosages (200–400 mg/d) has been established in previous open-label17,18 and placebo-controlled19 trials. In the current study, desvenlafaxine 50-mg/d efficacy was robust in the open-label phase, with 75.9% of patients considered in remission at week 20. This result compares favorably to prior flexible-dose, 9 to 12–month open-label trials of desvenlafaxine 200 to 400 mg/d in which remission rates ranged from 37%–58%.17,18 The higher remission rate at double-blind baseline in the current study most likely reflects the selection of an “enriched” patient population, that had demonstrated both acute-phase response to desvenlafaxine 50 mg/d at 8 weeks and a continued stable response through 12 additional weeks of open-label treatment to qualify for enrollment in the double-blind period. Remission rates at the end of double-blind treatment were significantly higher for the desvenlafaxine 50-mg/d group (74.4%) compared with the placebo group (54.2%), in which rates had fallen substantially. The 20 percentage point difference in remission rates at study endpoint is consistent with results from randomized withdrawal studies of the SNRIs duloxetine20 and venlafaxine.21 Patients in the desvenlafaxine group in the current trial also maintained significantly lower scores on the HDRS-17, HDRS-6, and CGI-S through the double-blind period compared with patients in the placebo group, indicating that desvenlafaxine 50-mg/d–treated patients had fewer depressive symptoms through the 6 months of double-blind treatment.
In addition to the lower burden of depressive symptoms, longer-term treatment with desvenlafaxine 50 mg/d versus placebo was associated with better functional outcomes compared with placebo in this study. Decreased work productivity is a major functional impairment related to MDD, with depressed individuals losing an average of roughly 27 work days per year due to presenteeism and absenteeism.22 However, evidence from a short-term (12-week) placebo-controlled trial of desvenlafaxine 50-mg/d gainfully employed patients with depression suggested that antidepressant treatment can positively affect productivity: patients who were moderately ill at baseline had significantly improved WPAI scores (vs placebo) after 12 weeks of treatment.9 In the current study, WPAI and WHO-5 scores improved from baseline in the response phase and were maintained during the stability phase of open-label treatment. During double-blind treatment, patients in the placebo group had a significant increase from double-blind baseline in presenteeism (indicating a decline in effectiveness at work), a significant increase in absenteeism plus presenteeism (work productivity loss), and a significant increase in impairment in everyday activities compared with those who continued desvenlafaxine 50-mg/d treatment. Thus, the WPAI results indicated that placebo-treated patients continued to go to work, but their function both at work and in their everyday activities declined over the double-blind period, whereas desvenlafaxine-treated patients generally maintained the productivity gained during 20-week open-label treatment with desvenlafaxine 50 mg/d. Further, WHO-5 scores showed a significant decrease in perceived well-being in patients assigned to placebo in the double-blind period compared with those continuing desvenlafaxine; for patients who received desvenlafaxine treatment in the double-blind period, there was little change from double-blind baseline in positive emotional state. Relapse prevention studies for other antidepressant drugs have often included secondary efficacy outcomes to evaluate depressive symptoms during longer-term treatment,20,23–27 but few have included assessments of patient functioning.20,23 Treatment guidelines state that one of the goals of acute treatment of a depressive episode is to return the patient to full baseline level of functioning, and that patient functioning should be monitored during continuation treatment.3 Given the potentially large clinical and economic impact of such improvements, long-term functional outcomes warrant further study.
The current investigation has a number of limitations that should be noted. The study population consisted of generally healthy adult outpatients across multiple countries with a primary diagnosis of MDD and limited comorbidities; results may not generalize to a broader MDD patient population. Results for the double-blind period are for patients who had a stable response to desvenlafaxine and may not generalize to patients with a shorter duration of response. The study duration was 11 months; the efficacy of desvenlafaxine 50 mg/d with treatment durations > 1 year has not been assessed. Strengths of the current study include the 12-week open-label stability phase, which likely served to reduce in the double-blind period the number of patients who were not “true” desvenlafaxine responders. In addition, patients were not required to meet MDD remission criteria to enter the double-blind period, and, thus, patients enrolled in the double-blind treatment period may reflect a clinically relevant patient population.
CONCLUSION
Antidepressant efficacy was maintained for patients who continued treatment with desvenlafaxine 50 mg/d for an additional 6 months after achieving a stable response, whereas patients assigned to placebo showed a worsening of depressive symptoms. Desvenlafaxine 50 mg/d maintained patient functioning, as measured by WPAI and WHO-5 scores, at significantly higher levels compared with placebo treatment during the 6-month double-blind period.
Drug names: desvenlafaxine (Pristiq), duloxetine (Cymbalta), venlafaxine (Effexor and others).
Author affiliations: University of Ottawa, Ottawa, Canada, and University Paris 7, Paris, France (Dr Boyer); Pfizer Global Research and Development, Paris, France (Ms Vialet); and Pfizer, Collegeville, Pennsylvania (Drs Hwang and Tourian). Dr Tourian is now with inVentiv Health Clinical, Princeton, New Jersey.
Potential conflicts of interest: Dr Boyer has participated as a speaker or on an advisory board for Laboratoires Servier. Ms Vialet is an employee of Pfizer Global Research and Development. Dr Hwang is an employee of and stock shareholder in Pfizer. Dr Tourian was an employee of Pfizer at the time of the study and currently is a stock shareholder in Pfizer.
Funding/support: This study was sponsored by Pfizer.
Role of the sponsor: The sponsor contributed to the design and conduct of the study, collection and analysis of data, the final content of the manuscript, and the decision to submit for publication.
Previous presentation: Study data previously presented at the 165th Annual Meeting of the American Psychiatric Association; May 5–9, 2012; Philadelphia, Pennsylvania.
Acknowledgments: Medical writing support was provided by Karen Dougherty, PhD, of Peloton Advantage (Parsippany, New Jersey) and was funded by Pfizer Inc. Dr Dougherty reports no other conflicts of interest related to the subject of this article.
REFERENCES
1. Vuorilehto MS, Melartin TK, Isometsä ET. Course and outcome of depressive disorders in primary care: a prospective 18-month study. Psychol Med. 2009;39(10):1697–1707. doi:10.1017/S0033291709005182 PubMed
2. Judd LL, Akiskal HS, Maser JD, et al. Major depressive disorder: a prospective study of residual subthreshold depressive symptoms as predictor of rapid relapse. J Affect Disord. 1998;50(2–3):97–108. doi:10.1016/S0165-0327(98)00138-4 PubMed
3. Work Group on Major Depressive Disorder. Practice guideline for the treatment of patients with major depressive disorder. American Psychiatric Association website. http://psychiatryonline.org/pracGuide/pracGuideTopic_7.aspx. Accessed April 29, 2015.
4. Nutt DJ, Davidson JR, Gelenberg AJ, et al. International consensus statement on major depressive disorder. J Clin Psychiatry. 2010;71(suppl E1):e08. doi:10.4088/JCP.9058se1c.08gry PubMed
5. Davidson JR. Major depressive disorder treatment guidelines in America and Europe. J Clin Psychiatry. 2010;71(suppl E1):e04. doi:10.4088/JCP.9058se1c.04gry PubMed
6. Pristiq [package insert]. Philadelphia, PA: Wyeth Pharmaceuticals, Inc, a subsidiary of Pfizer Inc; 2011.
7. Boyer P, Montgomery S, Lepola U, et al. Efficacy, safety, and tolerability of fixed-dose desvenlafaxine 50 and 100 mg/day for major depressive disorder in a placebo-controlled trial. Int Clin Psychopharmacol. 2008;23(5):243–253. doi:10.1097/YIC.0b013e32830cebed PubMed
8. Liebowitz MR, Manley AL, Padmanabhan SK, et al. Efficacy, safety, and tolerability of desvenlafaxine 50 mg/day and 100 mg/day in outpatients with major depressive disorder. Curr Med Res Opin. 2008;24(7):1877–1890. doi:10.1185/03007990802161923 PubMed
9. Dunlop BW, Reddy S, Yang L, et al. Symptomatic and functional improvement in employed depressed patients: a double-blind clinical trial of desvenlafaxine versus placebo. J Clin Psychopharmacol. 2011;31(5):569–576. doi:10.1097/JCP.0b013e31822c0a68 PubMed
10. Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23(1):56–62. doi:10.1136/jnnp.23.1.56 PubMed
11. Rosenthal JZ, Boyer P, Vialet C, et al. Efficacy and safety of desvenlafaxine 50 mg/d for prevention of relapse in major depressive disorder:a randomized controlled trial. J Clin Psychiatry. 2013;74(2):158–166. doi:10.4088/JCP.12m07974 PubMed
12. Nierenberg AA, Husain MM, Trivedi MH, et al. Residual symptoms after remission of major depressive disorder with citalopram and risk of relapse: a STAR*D report. Psychol Med. 2010;40(1):41–50. doi:10.1017/S0033291709006011 PubMed
13. Guy W. ECDEU Assessment Manual for Psychopharmacology–Revised. Rockville, MD: National Institute of Health, Department of Health, Education, and Welfare; 1976.
14. Bech P, Boyer P, Germain JM, et al. HAM-D-17 and HAM-D-6 sensitivity to change in relation to desvenlafaxine dose and baseline depression severity in major depressive disorder. Pharmacopsychiatry. 2010;43(7):271–276. doi:10.1055/s-0030-1263173 PubMed
15. Reilly MC, Zbrozek AS, Dukes EM. The validity and reproducibility of a work productivity and activity impairment instrument. Pharmacoeconomics. 1993;4(5):353–365. doi:10.2165/00019053-199304050-00006 PubMed
16. World Health Organization. Wellbeing measures in primary health care/the DEPCARE Project. World Health Organization Regional Office for Europe. http://www.euro.who.int/__data/assets/pdf_file/0016/130750/E60246.pdf. Accessed April 30, 2015.
17. Ferguson JM, Tourian KA, Rosas GR. High-dose desvenlafaxine in outpatients with major depressive disorder. CNS Spectr. 2012;17(3):121–130. doi:10.1017/S1092852912000508 PubMed
18. Tourian KA, Pitrosky B, Padmanabhan SK, et al. A 10-month, open-label evaluation of desvenlafaxine in outpatients with major depressive disorder. Prim Care Companion CNS Disord. 2011;13:e1–e10.
19. Rickels K, Montgomery SA, Tourian KA, et al. Desvenlafaxine for the prevention of relapse in major depressive disorder: results of a randomized trial. J Clin Psychopharmacol. 2010;30(1):18–24. doi:10.1097/JCP.0b013e3181c94c4d PubMed
20. Perahia DG, Gilaberte I, Wang F, et al. Duloxetine in the prevention of relapse of major depressive disorder: double-blind placebo-controlled study. Br J Psychiatry. 2006;188(4):346–353. doi:10.1192/bjp.188.4.346 PubMed
21. Kocsis JH, Thase ME, Trivedi MH, et al. Prevention of recurrent episodes of depression with venlafaxine ER in a 1-year maintenance phase from the PREVENT Study. J Clin Psychiatry. 2007;68(7):1014–1023. doi:10.4088/JCP.v68n0706 PubMed
22. Kessler RC, Akiskal HS, Ames M, et al. Prevalence and effects of mood disorders on work performance in a nationally representative sample of US workers. Am J Psychiatry. 2006;163(9):1561–1568. doi:10.1176/ajp.2006.163.9.1561 PubMed
23. Robert P, Montgomery SA. Citalopram in doses of 20–60 mg is effective in depression relapse prevention: a placebo-controlled 6 month study. Int Clin Psychopharmacol. 1995;10(suppl 1):29–35. doi:10.1097/00004850-199503001-00006 PubMed
24. Schmidt ME, Fava M, Robinson JM, et al. The efficacy and safety of a new enteric-coated formulation of fluoxetine given once weekly during the continuation treatment of major depressive disorder. J Clin Psychiatry. 2000;61(11):851–857. doi:10.4088/JCP.v61n1107 PubMed
25. Simon JS, Aguiar LM, Kunz NR, et al. Extended-release venlafaxine in relapse prevention for patients with major depressive disorder. J Psychiatr Res. 2004;38(3):249–257. doi:10.1016/j.jpsychires.2003.10.004 PubMed
26. Rapaport MH, Bose A, Zheng H. Escitalopram continuation treatment prevents relapse of depressive episodes. J Clin Psychiatry. 2004;65(1):44–49. doi:10.4088/JCP.v65n0107 PubMed
27. Gorwood P, Weiller E, Lemming O, et al. Escitalopram prevents relapse in older patients with major depressive disorder. Am J Geriatr Psychiatry. 2007;15(7):581–593. doi:10.1097/01.JGP.0000240823.94522.4c PubMed
Enjoy this premium PDF as part of your membership benefits!


