Objective: To provide a critical appraisal of the primary clinical trials exploring the use of glabellar botulinum toxin type A (BTA) injections in the treatment of major depressive disorder (MDD) and propose future directions for research on this topic.
Data Sources: A search in PubMed, Scopus, and Google Scholar databases was performed in September 2017. Search terms included (“botulinum” OR “botox” OR “abobotulinumtoxin” OR “onabotulinum” OR “onabotulinumtoxin” OR “botulinumtoxin”) AND (“antidepressant” OR “depression” OR “depressive” OR “depressed”). No other search parameters were utilized.
Study Selection: Studies were selected for review if they were found to be a primary clinical trial on the use of BTA for the treatment of MDD.
Data Extraction: Six studies were identified and scored by the authors using a 5-point Jadad scoring system.
Results: Three of the 6 studies were found to be of high quality with a Jadad score ≥ 3. The remainder had Jadad scores of 1.
Conclusions: In general, the results from the reviewed studies suggest that BTA may be a promising treatment for MDD. However, these findings need to be interpreted with caution due to several limitations of the reviewed studies such as lack of a priori hypotheses, limited sample sizes, large gender bias, and significant difficulty in ensuring blinding.
Glabellar Botulinum Toxin Injections
in Major Depressive Disorder:
A Critical Review
ABSTRACT
Objective: To provide a critical appraisal of the primary clinical trials exploring the use of glabellar botulinum toxin type A (BTA) injections in the treatment of major depressive disorder (MDD) and propose future directions for research on this topic.
Data Sources: A search in PubMed, Scopus, and Google Scholar databases was performed in September 2017. Search terms included (“botulinum” OR “botox” OR “abobotulinumtoxin” OR “onabotulinum” OR “onabotulinumtoxin” OR “botulinumtoxin”) AND (“antidepressant” OR “depression” OR “depressive” OR “depressed”). No other search parameters were utilized.
Study Selection: Studies were selected for review if they were found to be a primary clinical trial on the use of BTA for the treatment of MDD.
Data Extraction: Six studies were identified and scored by the authors using a 5-point Jadad scoring system.
Results: Three of the 6 studies were found to be of high quality with a Jadad score ≥ 3. The remainder had Jadad scores of 1.
Conclusions: In general, the results from the reviewed studies suggest that BTA may be a promising treatment for MDD. However, these findings need to be interpreted with caution due to several limitations of the reviewed studies such as lack of a priori hypotheses, limited sample sizes, large gender bias, and significant difficulty in ensuring blinding.
Prim Care Companion CNS Disord 2018;20(5):18r02298
To cite: Stearns TP, Shad MU, Guzman GC. Glabellar botulinum toxin injections in major depressive disorder: a critical review. Prim Care Companion CNS Disord. 2018;20(5):18r02298.
To share: https://doi.org/10.4088/PCC.18r02298
© Copyright 2018 Physicians Postgraduate Press, Inc.
aSamaritan Mental Health, Samaritan Health Services, Corvallis, Oregon
bDepartment of Psychiatry, Oregon State Hospital, Salem, Oregon
cDepartment of Psychiatry, Oregon Health & Science University, Portland, Oregon
dDepartment of Psychology, San Francisco State University, San Francisco, California
*Corresponding author: Toren P. Stearns, MD, Samaritan Mental Health, 3509 NW Samaritan Drive Ste 215, Corvallis, OR 97330 ([email protected]).
Depression is the leading cause of disability worldwide, with a 12-month prevalence of around 6.6%.1,2 Despite the large number of biological interventions clinicians have at their disposal, a significant percentage of patients with major depressive disorder (MDD) fail to achieve remission with these treatments.3 Thus, there is a need to develop novel strategies to treat MDD. In recent years, the use of botulinum toxin injections for the treatment of MDD has garnered significant public and scientific interest. In this review, we examined the quality and implications of the primary clinical trials in existence to date for the use of botulinum toxin in the treatment of MDD.
The concept of injecting botulinum toxin type A (BTA) into the glabella, the area of the forehead above and between the eyebrows, was initially based on the principal of the facial feedback hypothesis, which states that facial muscles do not just express emotion but contribute, in part, to its experience. The foundation of the facial feedback hypothesis dates back as early as 1872 when Charles Darwin described “peculiar furrows formed on the forehead” formed by the perpetual contraction of the brow’s “grief muscles” in patients diagnosed with chronic melancholia.4 Now termed the omega sign, the wrinkling of the skin above the nose and between the eyebrows resembles the Greek letter omega and is associated with hyperactivity of the corrugator supercilli and procerus muscles.5 Darwin postulated that the free expression by outward signs of an emotion intensifies its experience, and repression of outward signs of an emotion dampens it.6 In 1911, Swiss neurologist Otto Veraguth described another physical finding seen in individuals diagnosed with melancholia, a triangle-shaped fold formed by the brow and medial half of the upper eyelid termed the Veraguth-fold.7 These early observations helped set the stage for the development of the modern-day facial feedback hypothesis, which stipulates that posing a facial expression should increase the intensity of the emotional experience and inhibiting a facial expression should dampen the intensity of the emotional experience.8
The facial feedback hypothesis has been supported by a wide body of modern-day evidence.9–13 For example, when individuals are asked to imitate facial expressions or body postures seen in photographs, they tend to experience the congruent emotion that is mimicked.9 Feelings of aggressiveness have been reported to be triggered by the involuntary squinting that occurs when subjects are made to face the sun.10 Similarly, elevated electromyographic activity of the corrugator muscles has been shown to correlate with elevated feelings of aggression and sympathetic tone, namely increased pupil size and skin conductance, when subjects actively imitated angry facial expressions or even simply observed them.11 In patients with facial neuromuscular disorders, the severity of depressive symptoms has been found to have a correlation with particular impairments in the ability to smile.12 Interestingly, subjects receiving botulinum toxin injections into the corrugator muscles have been shown to have diminished functional magnetic resonance imaging activation of the amygdala when either mimicking or simply observing angry and sad facial expression seen in photographs.13
Complementary and alternative medical therapies have long had roots in the facial feedback hypothesis. In electroacupuncture, the YingTang site, corresponding to the center of the glabella, is often targeted in the treatment of depression. In a 2004 study, 30 one-hour sessions using a needle delivering current at 80–90 hz to generate visible muscle contractions at the YingTang and Baihui (superior-most point on the scalp) sites demonstrated an equal antidepressant efficacy to that of amitriptyline controls at 6 weeks.14 In another controlled crossover trial,15 25 healthy subjects demonstrated that acupressure on the YingTang site versus a sham site led to a significant decrease in bispectral index and spectral edge frequency on electroencephalogram (EEG), a finding similarly observed with administration of sedatives. Additionally, laughter therapy and laughter yoga have evidence supporting their use in elevating mood and reducing anxiety.16,17 True spontaneous laughter, triggered by either external or internal stimuli, has also been shown to be more efficacious than purposeful, simulated laughter, although both exert a significantly positive effect on mood.18,19
However, not all studies have supported the facial feedback hypothesis. A recent meta-analysis20 of 17 psychological studies failed to support the findings from the original study by Strack et al,21 which originated the facial feedback hypothesis.22 In addition, Söderkvist et al20 demonstrated a feedback effect, but this effect did not persist beyond critical actions of smiling and frowning, thus challenging the long-term effects of BTA on the basis of facial feedback.
Findings from studies using glabellar injection of BTA in MDD were previously analyzed in meta-analyses and reviews.23,24 The main aim of this review is not to synthesize results from a meta-analytic perspective as done previously. Instead, this review is an attempt to critically appraise the methodological limitations and clinical relevance of the findings from BTA clinical trials in an effort to propose future directions in this field of research.
METHODS
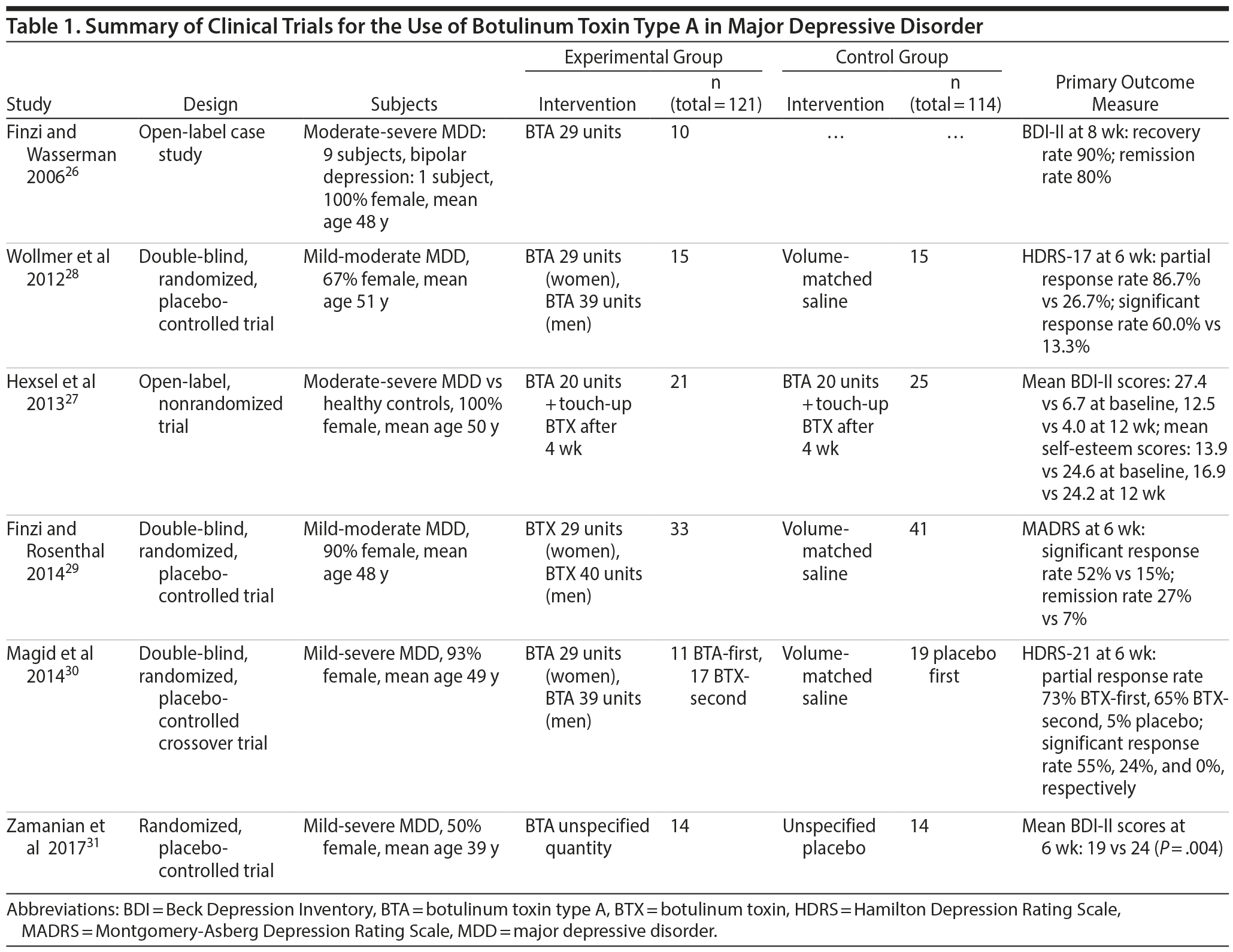
In September 2017, a nonsystematic literature search was made in the databases Pubmed, Scopus, and Google Scholar for primary clinical trials involving the use of botulinum toxin in the treatment of MDD, with quality assessed by the authors using a 5-point Jadad scoring system.25 Search terms included (“botulinum” OR “botox” OR “abobotulinumtoxin” OR “onabotulinum” OR “onabotulinumtoxin” OR “botulinumtoxin”) AND (“antidepressant” OR “depression” OR “depressive” OR “depressed”). No other search parameters were utilized. This literature search resulted in a total of 6 clinical trials26–31 that examined the use of botulinum toxin injections for the treatment of MDD (Table 1). All of these studies employed US Food and Drug Administration (FDA)–approved BTA, namely BOTOX (onabotulinumtoxin A), because it is the only BTA product approved by the FDA to reduce forehead lines for cosmetic reasons. Jadad scores were given as follows with a maximum total of 5 points: randomization: not randomized (0), described as randomized (1), randomization described and appropriate (2); blindness: not blind (0), described as double-blind (1), double-blind described and appropriate (2); and dropouts: not described (0), dropouts described (1). Although all 6 studies are included, this review more heavily focused on studies with higher Jadad scores. In addition, 2 pooled meta-analyses utilizing data from a subset of these 6 primary trials28–30 were identified, namely the published works of Parsaik et al23 and Magid et al.24 These 2 publications23,24 are not included in the scoring given that the aim of this article is to explore primary clinical trials, although they are incorporated in the narrative review.
RESULTS
Jadad scoring of the 6 studies is provided in Table 2, with the simple mean score of these 6 studies being 2.33. Of 6 studies reviewed, 3 studies had Jadad scores of 1. The first of these was Finzi and Wasserman’s 2006 case series26 with 10 female patients who were followed for 8 weeks after administration of BTA. This case series26 reported the highest recovery and remission rates (90% and 80%, respectively) of all 6 BTA trials. The second study with a Jadad score of 1 was published in 2013 by Hexsel et al27 and was a 12-week open-label uncontrolled trial with a total of 25 depressed versus 25 nondepressed subjects, with the depressed group showing more than a 50% reduction in Beck Depression Inventory scores at 12 weeks and no mention of remission rates. This was the only study that administered an additional BTA touch-up dose after 4 weeks. The authors27 did not clarify the rationale for having a depressed versus a nondepressed group rather than a placebo group, which would have provided for a better research design overall. Another notable limitation of the first 2 studies26,27 was the use of a subjective scale to assess depression (ie, Beck Depression Inventory) rather than a more objective assessment tool.
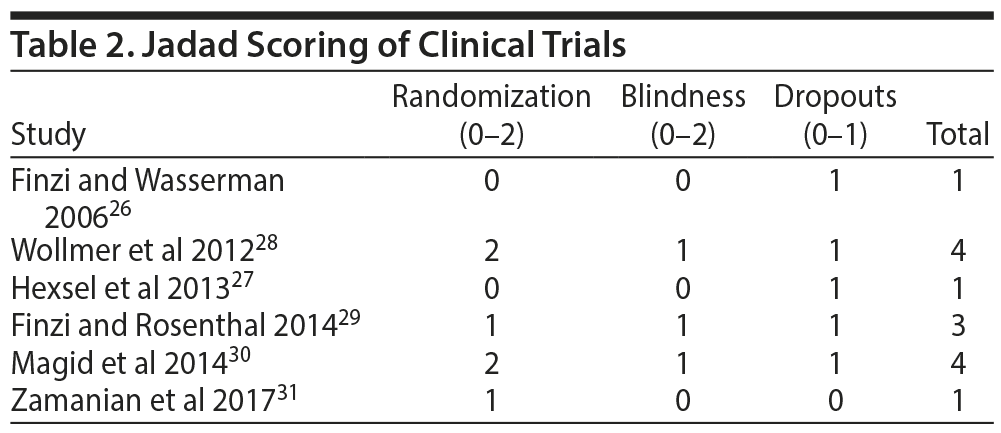
The remaining 4 studies were randomized controlled trials (RCTs). The most recent RCT, published by Zamanian et al in 2017,31 was a 6-week placebo-controlled trial of BTA in depressed versus nondepressed subjects that also received a Jadad score of 1 given a lack of a description of what placebo was used as well as no mention of blinding. However, this was the only study conducted with equal numbers of male and female subjects (ie, 14 males and 14 females), while the other clinical trials were heavily biased toward female subjects (case series by Finzi and Wasserman26 with 100% female, Hexsel et al27 with 100% female, Wollmer et al28 with 67% female, Finzi and Rosenthal29 with 90% female, and Magid et al30 with 93% female).
The remaining 3 studies scored more favorably, with Jadad scores ≥ 3. The 2012 study by Wollmer et al,28 notably the only RCT to have an a priori hypothesis, was conducted in 30 subjects with mild-moderate MDD and showed a response rate of 60% and remission rate of 33% for the BTA group versus a 13% response and remission rate in the placebo group. The 6-week double-blind, placebo-controlled RCT by Finzi and Rosenthal29 revealed a response rate of 52% and a remission rate of 27% versus 15% and 7% for placebo, respectively. This study was followed up by a double-blind, placebo-controlled crossover study by Magid et al30 in 2014 in which mild to moderately depressed subjects demonstrated a 55% response rate in the BTA-first group, 24% in the BTA-second group, and 0% in the placebo group. The placebo-second group was not analyzed in this study due to carry-over effects from BTA in the first phase of the study. In addition, there was an uneven distribution of subjects in the 3 subject groups analyzed (ie, 11 subjects in the BTA-first group, 19 subjects in the placebo-first group, and 17 subjects in the BTA-second group). This study30 may have been underpowered, as the power analysis was conducted with only a 3-point difference on the 21-item Hamilton Depression Rating Scale and not the 17-item version.
The reviewed studies found BTA to be a well-tolerated treatment for depression, and only minimal adverse effects were reported. The most common adverse effects found to be significantly different from placebo included local irritation at the injection site and transient headache. It is unusual to find fewer adverse effects in studies that have such low placebo responses as in the reviewed studies.
DISCUSSION
Although the results from the reviewed studies appear promising, the clinical evidence to support the use of BTA in patients with MDD is still preliminary. In addition to reviewing some of the limitations of the BTA studies, several important questions need to be addressed to confirm BTA’s efficacy and enable optimal use of this novel antidepressant strategy.
A significant limitation of the reviewed studies is whether blinding can be maintained with BTA given that it has significant cosmetic effects easily discernable both by the raters and the study subjects. Unblinding is likely one of the major reasons that the reviewed studies reported significantly low placebo and high response rates. In the Wollmer et al study,28 despite several efforts to ensure blinding, nearly 90% of study subjects could accurately guess their group at the end of the study. With the low placebo response rates across the board (ie, 13.3%,28 15%,29 and 0%–5%30), these trials appear to have much lower placebo rates than those usually observed in the majority of clinical trials with antidepressants. A 2002 review32 of 75 double-blind placebo-control trials of antidepressants for MDD demonstrated that the mean ± SD placebo response rate is 29.7% ± 8.3%. It has been suggested that the actual mean placebo response rate in MDD is closer to the 35%–45% range, as most negative trials are typically unpublished.33 Interestingly, one of the reviewed studies29 still reported low placebo response and remission rates (ie, 15% and 7%, respectively) despite lack of any significant association between changes in Montgomery-Asberg Depression Rating Scale (MADRS)34 scores and the percent of subjects who could correctly guess their group allocation. The study by Magid et al30 reported a 0% response rate in the placebo group, which raises even bigger concerns for unblinding at least among the raters if not the study subjects. This same study30 additionally reported that most raters correctly guessed the active treatment group as opposed to the placebo group.
To an extent, the informed consent procedure itself contributes to some degree of unblinding. Given that subjects become highly attentive to the somatic and psychological changes disclosed in the informed consent procedure, they frequently use this information to see through the double-blind.35,36 Therefore, it is imperative that future trials employ a more convincing placebo than normal saline to more closely mimic all or at least some of the unblinding properties and adverse effects of BTA without, of course, causing its own adverse effects. One such alternative would be the use of collagen filler as a placebo, as it visibly reduces glabellar lines without impacting corrugator and procerus tone. One may consider pursuing a 3-armed study comparing the antidepressant efficacy of collagen monotherapy versus BTA monotherapy versus BTA plus collagen combination therapy. It is worth noting that combination therapy of BTA and collagen filler has been found to result in a significantly greater objective improvement in glabellar frown scores over BTA monotherapy without increasing patient discomfort or procedure-related complications.37 If subjective cosmetic improvement truly is not a factor in the antidepressant action of BTA, one would expect the antidepressant effect of combination and BTA monotherapy groups to be similar. Given that these trials have shown antidepressant activity in around 6 weeks, such a study design seems feasible. In future studies, unblinding should be evaluated at various points in the trial by assessing the group allocation perceptions. Other possibilities to help reduce the effects of unblinding in future trials include utilizing a crossover study design and long treatment durations given that the placebo effect, regardless of the treatment arm, tends to somewhat diminish over a prolonged duration.
One clinically relevant question to ask is if there is a relationship between BTA dose and antidepressant response. On the basis of currently available data, it is rather difficult to answer this question because 426–29 of the 626–31 studies reviewed employed almost the same dose of BTA that ranged from 29 to 40 units for female subjects and 39 to 40 units for male subjects. One study31 did not report BTA dose, and the study by Hexsel et al27 used 20 units at baseline followed by a booster dose at week 4 of the study. In the future, it will be important to use a fixed-dose design with multiple doses to find out if any other BTA doses will be more (or less) effective than those used in the reviewed studies. In addition, since most reviewed studies used a single-injection strategy, it will be extremely useful to conduct longer-duration studies to see how long the efficacy of a single injection lasts and when, if ever, a maintenance BTA dose will be required to maintain the response after 24 weeks (which, according to the Magid et al study,30 was the longest period that subjects in a crossover study maintained the response after a single BTA injection). As stated earlier, the only study that included a booster dose at 4 weeks27 actually used a lower initial dose (20 units) in comparison to the other studies, so the results should be interpreted accordingly.
An intriguing question, which is somewhat linked with the mechanism of action of BTA, is whether there is a relationship between a change in baseline glabellar frown line scores and antidepressant response. Most studies that assessed glabellar frown lines saw no correlation between the frown scores and treatment response. Hexsel et al27 reported no baseline differences in glabellar frown line scores at rest or with maximum frowning between the depressed versus nondepressed subjects. Resting and maximum frowning scores were also assessed in the 24-week crossover trial by Magid et al30 and showed that while antidepressant response was maintained through this study period in the BTA-first group, the frown scores came back to the baseline value, further supporting a lack of relationship between frown score and antidepressant response. Finzi and Rosenthal’s study29 was only able to demonstrate a trend toward significance in an association between frown scores and treatment response, although this study did not require an observable frown at rest as an inclusion criterion. Notably, in this study 5 of the 13 patients with no baseline frown at rest still experienced full remission after BTA treatment. On the other hand, the study by Wollmer et al28 did report a significant association between frown line scores and BTA response. Interestingly, 1 patient in this study remitted despite the misperception of being on placebo, possibly due to a lack of any noticeable cosmetic changes in frown lines. A notable point related to the relationship between frown line scores and BTA response is that most reviewed studies based their frown scores on maximal voluntary frowning effort, which may vary between patients based on the extent of their voluntary effort, muscle strength and its innervation, and frequency of using these frown muscles.
That only 1 study reported any statistically significant correlation between BTA response and frown line scores brings up the possibility that BTA may possibly work independently of frown lines and outside the context of the facial feedback hypothesis. An interesting and relatively recent hypothesis that may explain the mechanism of antidepressant response with BTA is a possibility of retrograde axonal flow of BTA. This hypothesis is supported by the retrograde transport of BTA by central neurons and motor neurons to the afferent synapses in an animal study.38 In contrast to facial feedback theory, this hypothesis provides a more plausible explanation for a lack of relationship between furrow lines and antidepressant response of BTA. In future clinical trials, it would be interesting to investigate the placebo response in depressed subjects without frown lines so that there is minimal risk for unblinding. Future trials should have at least 2 active groups, 1 with frown lines and the other without, which will answer the question of whether frown lines are a marker of BTA response or not.
Another clinically relevant question that needs to be investigated is whether BTA can be used as monotherapy, as most study subjects in these trials were treated with antidepressants concurrently. Although Finzi and Rosenthal29 demonstrated higher remission rates among study subjects taking antidepressants concurrently than those on BTA as monotherapy, a remission rate of 21% was reported with BTA monotherapy. Future studies will require larger samples that are adequately powered, not only to investigate a differential BTA response in combination with various antidepressant agents, but also to differentiate between the clinical and demographic characteristics of study subjects in BTA augmentation versus monotherapy groups. The information garnered from this study design may be highly relevant to understanding BTA’s mechanism of action in various subtypes of depressed patients, especially in reference to different neurotransmitter systems that may mediate its differential response.
Furthermore, future research will need to address the effect of sex on BTA response. With the exception of the study by Zamanian et al,31 most studies were conducted predominantly in female subjects, ranging from 67% in the Wollmer et al study28 to 100% in the Hexsel et al study.27 Thus, the 3 RCTs with high Jadad scores28–30 were done predominantly with female subjects. It is possible that this research attracts female participants because of the cosmetic value of BTA injection for them. Large clinical trials will be required with a balanced representation of male and female subjects to rule out any sex effects with BTA treatment.
It will also be clinically useful to see if BTA will be effective in alleviating depressive symptoms when given in nonglabellar facial muscles that confer sadness, such as the depressor anguli oris, or if BTA given in multiple facial muscles is more effective than glabellar injection itself. Further, clinical trials exploring the efficacy of BTA in bipolar depression are warranted. Thus far, only 1 patient, from Finzi and Wasserman’s study,26 has been trialed on BTA for the treatment of bipolar depression and has demonstrated a favorable response.
Likewise, research investigating methods of activating and increasing muscular tone in facial musculature involved in smiling for the purposes of mood enhancement is also an intriguing area of exploration. With efficacy demonstrated in the treatment of epilepsy, a 2011 pilot study39 on the use of external trigeminal nerve stimulation in the treatment of MDD yielded promising results. A 2013 study40 suggested that modulating emotion through external functional electrical stimulation may be possible, although more research is needed in this area. There have been some anecdotal reports suggesting that individuals receiving glabellar paralysis may experience a reduction in their ability to express and relate the emotions of fear, anger, and sadness with others in socially appropriate circumstances.41 While this is a concerning potential side effect of BTA therapy, emotional and affective blunting has been reported with selective serotonin reuptake inhibitors (SSRIs) as well, along with the development of an “SSRI-induced apathy syndrome”42–44 However, antidepressant effects of BTA are potentially mediated by apparent emotional blunting caused by blockade of muscles used in the expression of sad or despondent emotions.
CONCLUSION
Although the results from the reviewed studies provide evidence supporting the use of glabellar BTA injections for the treatment of MDD, future studies with larger and more balanced sample populations, more convincing placebo controls, and improved assurance of blinding are required to further elucidate the role of BTA in the treatment of MDD.
Submitted: March 14, 2018; accepted June 8, 2018.
Published online: October 4, 2018.
Potential conflicts of interest: None.
Funding/support: None.
REFERENCES
1. Kessler RC, Berglund P, Demler O, et al; National Comorbidity Survey Replication. The epidemiology of major depressive disorder: results from the National Comorbidity Survey Replication (NCS-R). JAMA. 2003;289(23):3095–3105. PubMed CrossRef
2. World Health Organization (WHO) fact sheet on depression. WHO website. http://www.who.int/news-room/fact-sheets/detail/depression. Accessed August 29, 2018.
3. Rush AJ, Trivedi MH, Wisniewski SR, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163(11):1905–1917. PubMed CrossRef
4. Shorter E. Darwin’s contribution to psychiatry. Br J Psychiatry. 2009;195(6):473–474. PubMed CrossRef
5. Greden JF, Genero N, Price HL. Agitation-increased electromyogram activity in the corrugator muscle region: a possible explanation of the “Omega sign”? Am J Psychiatry. 1985;142(3):348–351. PubMed CrossRef
6. James W. What is an emotion? Mind. 1884;9(34):188–205. CrossRef
7. Veraguth O. Die klinische Untersuchung Nervenkranker. Wiesbaden: Germany: Verlagvon JF Bergmann; 1911.
8. Davis JI, Senghas A, Brandt F, et al. The effects of BOTOX injections on emotional experience. Emotion. 2010;10(3):433–440. PubMed CrossRef
9. Duclos SE, Laird JD, Schneider E, et al. Emotion-specific effects of facial expressions and postures on emotional experience. J Pers Soc Psychol. 1989;57(1):100–108. CrossRef
10. Marzoli D, Custodero M, Pagliara A, et al. Sun-induced frowning fosters aggressive feelings. Cogn Emotion. 2013;27(8):1513–1521. PubMed CrossRef
11. Lee IS, Yoon SS, Lee SH, et al. An amplification of feedback from facial muscles strengthened sympathetic activations to emotional facial cues. Auton Neurosci. 2013;179(1–2):37–42. PubMed CrossRef
12. VanSwearingen JM, Cohn JF, Bajaj-Luthra A. Specific impairment of smiling increases the severity of depressive symptoms in patients with facial neuromuscular disorders. Aesthetic Plast Surg. 1999;23(6):416–423. PubMed CrossRef
13. Hennenlotter A, Dresel C, Castrop F, et al. The link between facial feedback and neural activity within central circuitries of emotion: new insights from botulinum toxin-induced denervation of frown muscles. Cereb Cortex. 2009;19(3):537–542. PubMed CrossRef
14. Yan M, Mao X, Wu JL. Comparison of electroacupuncture and amitriptyline in treating depression. Zhongguo Linchuang Kangfu. 2004;18:3548–3549.
15. Litscher G. Effects of acupressure, manual acupuncture and Laserneedle acupuncture on EEG bispectral index and spectral edge frequency in healthy volunteers. Eur J Anaesthesiol. 2004;21(1):13–19. PubMed CrossRef
16. Ko HJ, Youn CH. Effects of laughter therapy on depression, cognition and sleep among the community-dwelling elderly. Geriatr Gerontol Int. 2011;11(3):267–274. PubMed CrossRef
17. Shahidi M, Mojtahed A, Modabbernia A, et al. Laughter yoga versus group exercise program in elderly depressed women: a randomized controlled trial. Int J Geriatr Psychiatry. 2011;26(3):322–327. PubMed CrossRef
18. Mora-Ripoll R. The therapeutic value of laughter in medicine. Altern Ther Health Med. 2010;16(6):56–64. PubMed
19. Mora-Ripoll R. Potential health benefits of simulated laughter: a narrative review of the literature and recommendations for future research. Complement Ther Med. 2011;19(3):170–177. PubMed CrossRef
20. Söderkvist S, Ohlén K, Dimberg U. How the experience of emotion is modulated by facial feedback. J Nonverbal Behav. 2018;42(1):129–151. PubMed CrossRef
21. Strack F, Martin LL, Stepper S. Inhibiting and facilitating conditions of the human smile: a nonobtrusive test of the facial feedback hypothesis. J Pers Soc Psychol. 1988;54(5):768–777. PubMed CrossRef
22. Acosta A, Adams RB Jr, Albohn DN, et al. Registered replication report: Strack, Martin, & Stepper (1988). Perspect Psychol Sci. 2016;11(6):917–928. PubMed CrossRef
23. Parsaik AK, Mascarenhas SS, Hashmi A, et al. Role of botulinum toxin in depression. J Psychiatr Pract. 2016;22(2):99–110. PubMed CrossRef
24. Magid M, Finzi E, Kruger THC, et al. Treating depression with botulinum toxin: a pooled analysis of randomized controlled trials. Pharmacopsychiatry. 2015;48(6):205–210. PubMed CrossRef
25. Jadad AR, Moore RA, Carroll D, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials. 1996;17(1):1–12. PubMed CrossRef
26. Finzi E, Wasserman E. Treatment of depression with botulinum toxin A: a case series. Dermatol Surg. 2006;32(5):645–649, discussion 649–650. PubMed
27. Hexsel D, Brum C, Siega C, et al. Evaluation of self-esteem and depression symptoms in depressed and nondepressed subjects treated with onabotulinumtoxinA for glabellar lines. Dermatol Surg. 2013;39(7):1088–1096. PubMed CrossRef
28. Wollmer MA, de Boer C, Kalak N, et al. Facing depression with botulinum toxin: a randomized controlled trial. J Psychiatr Res. 2012;46(5):574–581. PubMed CrossRef
29. Finzi E, Rosenthal NE. Treatment of depression with onabotulinumtoxinA: a randomized, double-blind, placebo controlled trial. J Psychiatr Res. 2014;52:1–6. PubMed CrossRef
30. Magid M, Reichenberg JS, Poth PE, et al. Treatment of major depressive disorder using botulinum toxin A: a 24-week randomized, double-blind, placebo-controlled study. J Clin Psychiatry. 2014;75(8):837–844. PubMed CrossRef
31. Zamanian A, Ghanbari Jolfaei A, Mehran G, et al. Efficacy of botox versus placebo for treatment of patients with major depression. Iran J Public Health. 2017;46(7):982–984. PubMed
32. Walsh BT, Seidman SN, Sysko R, et al. Placebo response in studies of major depression: variable, substantial, and growing. JAMA. 2002;287(14):1840–1847. PubMed CrossRef
33. Bridge JA, Birmaher B, Iyengar S, et al. Placebo response in randomized controlled trials of antidepressants for pediatric major depressive disorder. Am J Psychiatry. 2009;166(1):42–49. PubMed CrossRef
34. Montgomery SA, Asberg M. A new depression scale designed to be sensitive to change. Br J Psychiatry. 1979;134(4):382–389. PubMed CrossRef
35. Swartzman LC, Burkell J. Expectations and the placebo effect in clinical drug trials: why we should not turn a blind eye to unblinding, and other cautionary notes. Clin Pharmacol Ther. 1998;64(1):1–7. PubMed CrossRef
36. Rutherford BR, Sneed JR, Roose SP. Does study design influence outcome? the effects of placebo control and treatment duration in antidepressant trials. Psychother Psychosom. 2009;78(3):172–181. PubMed CrossRef
37. Patel MP, Talmor M, Nolan WB. Botox and collagen for glabellar furrows: advantages of combination therapy. Ann Plast Surg. 2004;52(5):442–447, discussion 447. PubMed CrossRef
38. Antonucci F, Rossi C, Gianfranceschi L, et al. Long-distance retrograde effects of botulinum neurotoxin A. J Neurosci. 2008;28(14):3689–3696. PubMed CrossRef
39. Schrader LM, Cook IA, Miller PR, et al. Trigeminal nerve stimulation in major depressive disorder: first proof of concept in an open pilot trial. Epilepsy Behav. 2011;22(3):475–478. PubMed CrossRef
40. Zariffa J, Hitzig SL, Popovic MR. Neuromodulation of emotion using functional electrical stimulation applied to facial muscles. Neuromodulation. 2014;17(1):85–92, discussion 92. PubMed CrossRef
41. Havas DA, Glenberg AM, Gutowski KA, et al. Cosmetic use of botulinum toxin-a affects processing of emotional language. Psychol Sci. 2010;21(7):895–900. PubMed CrossRef
42. Opbroek A, Delgado PL, Laukes C, et al. Emotional blunting associated with SSRI-induced sexual dysfunction: do SSRIs inhibit emotional responses? Int J Neuropsychopharmacol. 2002;5(2):147–151. PubMed CrossRef
43. Bolling MY, Kohlenberg RJ. Reasons for quitting serotonin reuptake inhibitor therapy: paradoxical psychological side effects and patient satisfaction. Psychother Psychosom. 2004;73(6):380–385. PubMed CrossRef
44. Barnhart WJ, Makela EH, Latocha MJ. SSRI-induced apathy syndrome: a clinical review. J Psychiatr Pract. 2004;10(3):196–199. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!


