Objective: Difficult-to-treat depression (eg, depression with atypical or anxious symptoms, treatment-resistant depression, or depression with frequent recurrence) is a challenging real-world health issue. This critical review of the literature focuses on monoamine oxidase inhibitor (MAOI) therapy and difficult-to-treat forms of depression.
Data Sources: A PubMed literature search was performed in November 2012 and refreshed through January 2013 with no date restrictions using key search terms including MAO inhibitor therapy or MAOI and depression and anxiety, atypical, treatment-resistant, recurrent, relapse, or refractory.
Study Selection: Articles were selected to summarize the current needs in difficult-to-treat depression as well as the use of MAOI therapies in this area.
Results: Two strategies have fallen out of favor in the care of patients with major depressive disorder. The first is the use of MAOI therapy and the second is the proactive recognition of difficult-to-treat depression that may not respond as well to more frequently used antidepressants. The infrequent use of MAOIs stems from the perception that other oral therapies for depression are safer and easier to use than oral MAOIs; however, transdermal delivery is one potential strategy to improve the safety of this class of agents. Although food-related interactions with transdermal delivery of MAOI therapy can be lessened, clinicians still need to be vigilant for drug-drug interactions and serotonin syndrome.
Conclusions: Clinicians should consider MAOIs for patients who have had several unsuccessful trials of antidepressants. Guidelines generally reserve MAOIs as third- and fourth-line treatments due to concerns over safety and tolerability; however, transdermal delivery of an MAOI may allay some of the safety and tolerability concerns. Patients should be provided education about MAOIs and their risks.
Prim Care Companion CNS Disord 2013;15(5):doi:10.408/PCC.13r01515
© Copyright 2013 Physicians Postgraduate Press, Inc.
Submitted: March 13, 2013; accepted July 22, 2013.
Published online: October 31, 2013.
Corresponding author: Larry Culpepper, MD, Department of Family Medicine, Boston University School of Medicine, Boston Medical Center, 1 Boston Medical Center Place,
Dowling 5, Boston, MA 02118 ([email protected]).
Reducing the Burden of Difficult-to-Treat Major Depressive Disorder: Revisiting Monoamine Oxidase Inhibitor Therapy
ABSTRACT
Objective: Difficult-to-treat depression (eg, depression with atypical or anxious symptoms, treatment-resistant depression, or depression with frequent recurrence) is a challenging real-world health issue. This critical review of the literature focuses on monoamine oxidase inhibitor (MAOI) therapy and difficult-to-treat forms of depression.
Data Sources: A PubMed literature search was performed in November 2012 and refreshed through January 2013 with no date restrictions using key search terms including MAO inhibitor therapy or MAOI and depression and anxiety, atypical, treatment-resistant, recurrent, relapse, or refractory.
Study Selection: Articles were selected to summarize the current needs in difficult-to-treat depression as well as the use of MAOI therapies in this area.
Results: Two strategies have fallen out of favor in the care of patients with major depressive disorder. The first is the use of MAOI therapy and the second is the proactive recognition of difficult-to-treat depression that may not respond as well to more frequently used antidepressants. The infrequent use of MAOIs stems from the perception that other oral therapies for depression are safer and easier to use than oral MAOIs; however, transdermal delivery is one potential strategy to improve the safety of this class of agents. Although food-related interactions with transdermal delivery of MAOI therapy can be lessened, clinicians still need to be vigilant for drug-drug interactions and serotonin syndrome.
Conclusions: Clinicians should consider MAOIs for patients who have had several unsuccessful trials of antidepressants. Guidelines generally reserve MAOIs as third- and fourth-line treatments due to concerns over safety and tolerability; however, transdermal delivery of an MAOI may allay some of the safety and tolerability concerns. Patients should be provided education about MAOIs and their risks.
Prim Care Companion CNS Disord 2013;15(5):doi:10.408/PCC.13r01515
© Copyright 2013 Physicians Postgraduate Press, Inc.
Submitted: March 13, 2013; accepted July 22, 2013.
Published online: October 31, 2013.
Corresponding author: Larry Culpepper, MD, Department of Family Medicine, Boston University School of Medicine, Boston Medical Center, 1 Boston Medical Center Place,
Dowling 5, Boston, MA 02118 ([email protected]).
Signs of depression may include frequent headaches, backaches, and other pain; lost sense of self-worth and lack of drive; difficulty concentrating; and changes in physical activity, eating habits, and interaction with family. Clinicians in primary care settings are uniquely positioned to help ensure that patients with these types of symptoms, in the absence of other confirmed diagnoses, are screened for major depressive disorder (MDD) and then adequately treated.1 Despite the availability of many new treatments, a number of patients with MDD experience inadequate symptom relief.2 More than half of all patients with depression fail to achieve remission after first-line antidepressant therapy.3,4 This outcome may be especially true for patients with anxious or atypical symptoms or patients who experience recurrent episodes or treatment resistance. Inadequate response to therapy is serious since residual symptoms increase the risk of relapse and recurrence of MDD.3 Risk factors contributing to inadequate response are misdiagnosis, presence of depressive subtypes, and medical and psychiatric comorbidities. Inadequate response may also be the result of medication nonadherence, which may require additional education.5 There is much we can do in primary care to enhance identification and treatment of patients with difficult-to-treat depression.
This article reviews the utility of monoamine oxidase inhibitors (MAOIs) for difficult-to-treat MDD and, in particular, a newer transdermal formulation, which may be a safe and better-tolerated option than oral MAOIs.2
METHOD
This critical review of the literature focused on MAOI therapy and difficult-to-treat forms of depression. A PubMed literature search was performed in November 2012 and refreshed through January 2013 with no date restrictions using key search terms including MAO inhibitor therapy or MAOI and depression and anxiety, atypical, treatment-resistant, recurrent, relapse, or refractory. Articles were selected to summarize the current needs in difficult-to-treat depression as well as the use of MAOI therapies in this area.
RESULTS
Difficult-to-Treat MDD
Recurrent MDD. Most patients with MDD will experience a recurrence following recovery from a depressive episode.6 In one study of 279 patients who recovered but eventually experienced a recurrence, several factors were shown to predict recurrence.6 Women were 43% more likely to experience a recurrence (odds ratio = 1.43) than men. Also, individuals who had never married were 55% more likely to experience a recurrence (odds ratio = 1.55).6 Longer duration of depressive episodes is reported to be associated with a higher risk for recurrence (11% greater likelihood of recurrence for each additional year of depression).6 There is also an 18% increase in risk of recurrence for each additional depressive episode experienced. Overall, the cumulative proportion of recurrence at 15 years of patient follow-up can be up to 85%, despite long periods of wellness.6
Treatment-resistant MDD. It has been estimated that 60%–70% of patients with MDD may have treatment-resistant depression.7 Treatment-resistant depression is unresponsive to adequate standard treatments and is associated with greater severity and longer duration, higher risk of recurrence, and greater likelihood of comorbid conditions. Treatment-resistant depression complicates patient care and increases health care burden.3,8 Although there is no agreed-upon clinical definition for treatment-resistant depression, the working definition is an inadequate response to at least 2 antidepressant trials, with agents of differing classes of adequate dose and duration and treatment adherence.3 Table 1 describes in greater detail a system of staging treatment-resistant depression.9 The primary diagnosis of MDD should be confirmed in patients who appear to have treatment-resistant depression. Confounding factors, such as untreated primary medical conditions, medication side effects, bipolar disorder, and substance abuse problems, should be considered.3

Depression with anxious or atypical symptoms. Clinically relevant subtypes of depression include psychotic depression, severe depression, chronic depression, anxious depression, and atypical depression.3 Psychotic depression is relatively common and is quite likely to be associated with severe depression and longer time to recovery. While it can be difficult, it is important to determine the presence of psychotic symptoms because antidepressant therapy may exacerbate psychotic symptoms and an antipsychotic therapy may be required.3,10 Severe depression is very difficult to treat and is defined by severely intense depressive symptoms, the presence of diagnostic subtypes of MDD, and a severely dysfunctional state. Additional factors in the assessment of severe depression include thoughts of suicidality and inpatient status.3,8,11 Severe depression is associated with a greater number of comorbidities, a longer time to treatment response, greater risk of recurrence, and greater likelihood of suicide.3,8 Chronic depression, defined as depression in which patients are symptomatic more days than not over a 2-year period, is associated with a high rate of treatment resistance.3,11
Patients with MDD are often affected by anxiety symptoms. In the large Sequenced Treatment Alternatives to Relieve Depression (STAR*D) project,12 53.2% of patients had anxious depression. This finding is consistent with previous outpatient reports of patients with MDD.13,14 These patients tend to experience greater severity of depression and longer duration of illness, are slower and less likely to respond to therapy, and have increased risk for suicide.15–19
Atypical depression is the most common depression subtype among depressed outpatients, accounting for 29%–42% of cases.20 Atypical depression is characterized by nonmelancholic, reverse vegetative signs—such as oversleeping, overeating, rejection sensitivity, and reactive mood. It is also believed that age at onset, duration and frequency of episodes, ability to achieve complete remission, and recurrence may differentiate patients with atypical depression from other depressed patients. Patients with atypical depression are also significantly more likely to be obese, which is of relevance in antidepressant treatment selection.21 Levitan et al21 showed that, in comparison to patients with classic depression, patients with atypical depression were 2 times (past history of depression) and 3 times (current history of depression) more likely to be obese.
Biology of MDD: Elevated MAO-A Levels in the Brain
While the monoamine theory of depression suggests that monoamine levels are lowered in depression, it does not explain how monoamine lowering takes place.22 Because the enzyme MAO-A metabolizes and regulates serotonin, norepinephrine, and dopamine in the brain, it is a logical contributor to the pathophysiology of depression.22,23 Meyer et al22 studied 17 healthy individuals and 17 individuals with MDD to determine if MAO-A levels in the brain are elevated during episodes of untreated depression. They found that the specific distribution volume of MAO-A was significantly (P = .001) elevated in every region of the brain, including the prefrontal cortex, anterior cingulate cortex, posterior cingulate cortex, caudate, putamen, thalamus, anterior temporal cortex, midbrain, hippocampus, and parahippocampus. Furthermore, the specific volume of distribution of MAO-A was elevated an average of 34% throughout the brain during major depression.22 The magnitude of this finding, together with the absence of any other compelling explanation for monoamine loss during major depressive episodes, led the investigators to conclude that elevated MAO-A was in fact the primary cause of monoamine lowering during major depression.22
Mechanism of MAO Inhibition
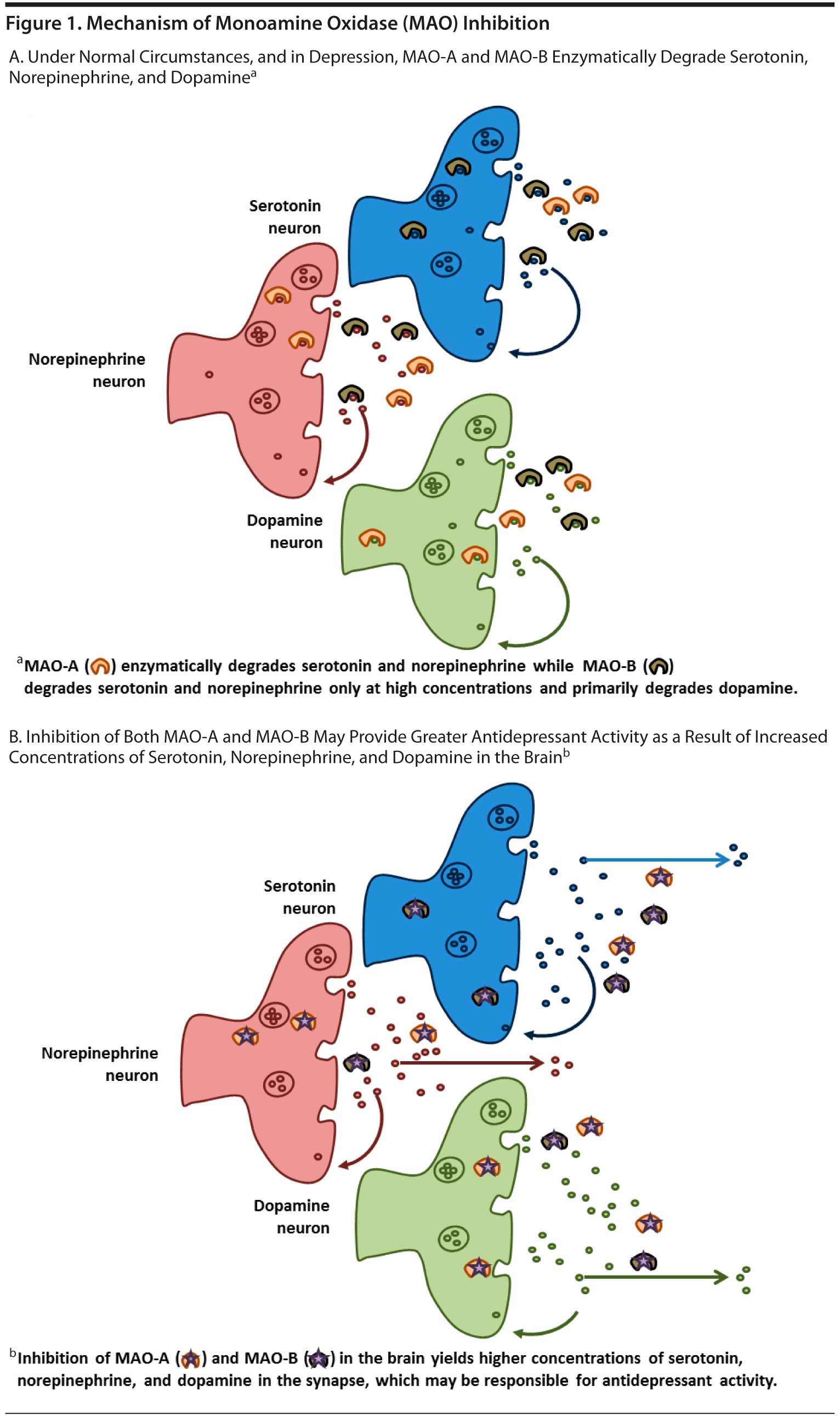
The MAOIs were first developed in the 1950s. Monoamine oxidase is an enzyme that catalyzes the oxidation and inactivation of the monoamine neurotransmitters. The enzyme is found in the brain, gut, and liver in addition to other tissues. There are 2 MAO enzyme isomers, each with different substrates: MAO-A is found primarily in the intestine and in those regions of the brain that have serotonin, norepinephrine, dopamine, and tyramine substrates; MAO-B is found primarily in platelets and in those regions of the brain that are rich in dopaminergic neurons.24 The MAOIs can be selective for either one isomer or the other or nonselective, inhibiting both MAO-A and MAO-B. It is believed, however, that inhibition of MAO-A is necessary for antidepressant effects.24,25 The mechanism of available therapies for MDD is based on the principle of increasing synaptic availability of serotonin, norepinephrine, and/or dopamine—the monoamine neurotransmitters.26,27 The MAOIs inhibit the MAO enzyme, thereby allowing serotonin, norepinephrine, and dopamine to accumulate in the synapse.27,28
The MAO-A isoenzyme metabolizes serotonin, norepinephrine, and dopamine, while MAO-B primarily metabolizes dopamine. While the inhibition of MAO-A in the brain is necessary for an antidepressant effect, inhibition of both MAO-A and MAO-B provides a greater increase in serotonin, norepinephrine, and dopamine than inhibition of either MAOI isomer alone (Figure 1A and B).27,29 Theoretically, inhibition of both MAO-A and MAO-B can lead to greater increases of each neurotransmitter compared with inhibition of either isoenzyme alone.
MAOIs Available in the United States
for the Treatment of Depression
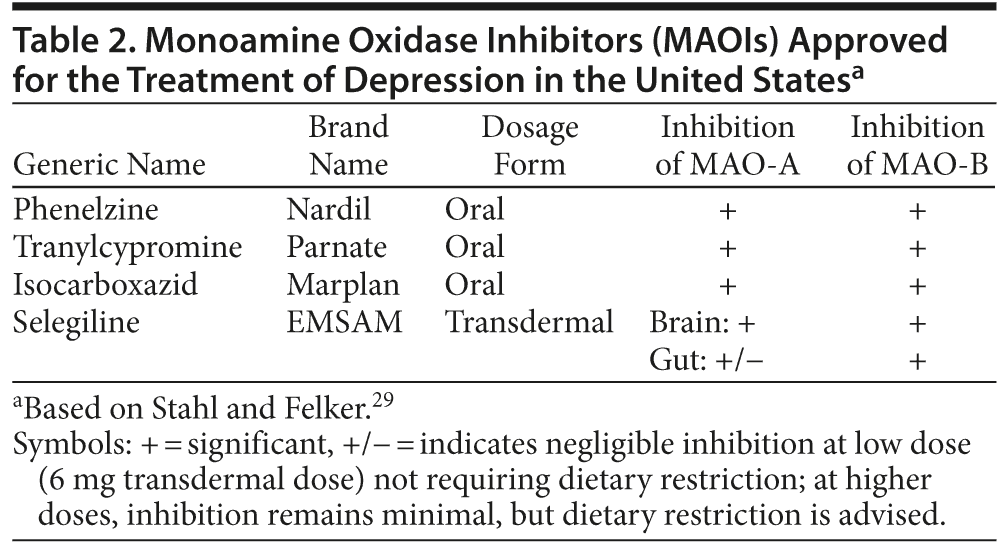
There are currently 3 oral MAOIs and 1 transdermal MAOI available in the United States for the treatment of MDD. These include isocarboxazid, phenelzine, and tranylcypromine and the selegiline transdermal system (STS), respectively (Table 2).29 Isocarboxazid, phenelzine, and tranylcypromine, the classic oral MAOIs approved for use as antidepressants, are all nonselective and inhibit both MAO isoenzymes. The transdermal formulation of selegiline is the only transdermal antidepressant approved to treat MDD. All MAOIs approved for use in the United States for the treatment of depression bind irreversibly to the MAO for the life of the enzyme. A washout of 7 to 10 days is required for a new MAO to be generated and enzymatic catalysis of monoamine neurotransmitters to resume.27,28
Guideline Recommendations for MAOIs
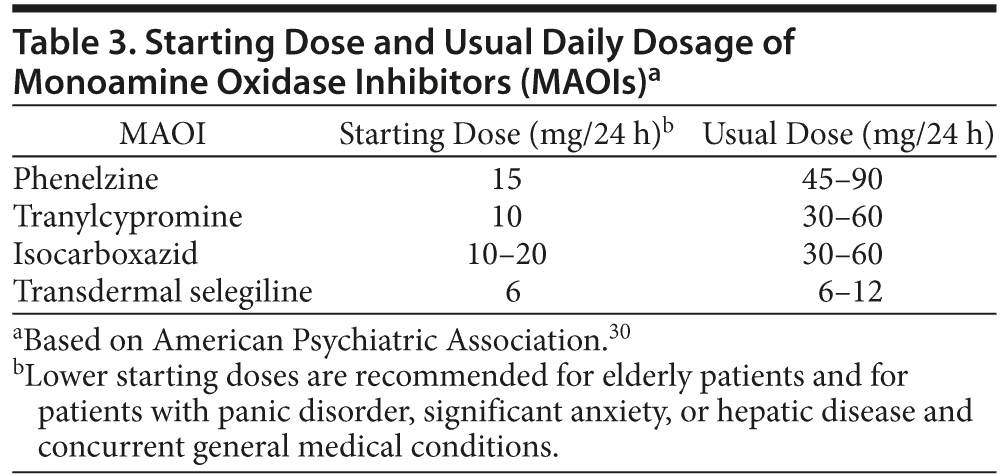
While these agents are no longer recommended by depression treatment guidelines as first-line agents, they are, however, recommended in certain situations.24 For the most part, current guidelines from the American Psychiatric Association reserve MAOIs for third- or fourth-line treatment due to concerns over tolerability and safety, the most serious of which are hypertensive crisis and serotonin syndrome.24,30 The American Psychiatric Association guidelines note in particular the efficacy of this class for patients with atypical depression. The guidelines also point out their appropriate use in patients who were successfully treated previously with an MAOI and would like to resume therapy.30 Among the preferred uses of the classical MAOIs are the treatment of atypical depression, treatment-refractory patients, patients without compliance problems, and those with associated panic attacks.31 The MAOIs should be started at a low dose and titrated upward to the usual daily dosage (Table 3).24,30
Minimizing Safety and Tolerability Concerns
Use of oral MAOIs is most commonly associated with early side effects such as insomnia, sedation, orthostatic hypotension, dizziness, and nausea, while later side effects often include weight gain, edema, muscle pain, myoclonus, paresthesia, and sexual dysfunction.24,32 Hypertensive crisis and serotonin syndrome (commonly related to medication nonadherence, dietary violations, or unwanted drug-drug interactions), both of which can occur as a result of the mechanism of action of MAOIs, are much more serious.24,29
Hypertensive crisis. The MAOI-A isoenzyme metabolizes not only serotonin, norepinephrine, and dopamine, but also tyramine found in the gut and liver. This action restricts the uptake of tyramine into the circulatory system during digestion. When high levels of tyramine enter the bloodstream as a result of oral MAOI use, this can lead to the release of norepinephrine, which can cause a rapid increase in blood pressure. This “hypertensive crisis” can result in stroke or death from cerebral hemorrhage.24 For this reason, patients taking oral MAOIs must limit their intake of dietary tyramine and therefore avoid aged cheeses; cured or aged meats; meats, fish, or poultry that might be spoiled; broad bean pods (such as fava bean pods); concentrated yeast extract; sauerkraut; tofu; soy bean condiments including soy sauce; and tap beer.24,33 Concomitant use of MAOIs with adrenergic medications—including amphetamines, sympathomimetic vasoconstrictors, and over-the-counter decongestants—is also believed to increase the risk of hypertensive crisis.
Serotonin syndrome. This rare but potentially fatal syndrome can occur as the result of an interaction between MAOIs and drugs that have strong serotonergic effects, for example, other antidepressants, synthetic opioids, and some triptan migraine medications.24,34 To avoid serotonin syndrome, contraindicated medications should not be taken for 1 to 2 weeks before initiating therapy with the MAOI or for 2 weeks after stopping MAOI therapy. An exception is the SSRI fluoxetine, which, because of its long half-life, requires a 5-week washout period or longer (eg, if significant liver impairment is present, up to 2–3 months is prudent) prior to initiating an MAOI.24,30
Responsiveness to MAOIs
in Difficult-to-Treat Depression
Evidence of responsiveness to MAOIs in treatment-resistant depression. There is mixed evidence about the utility of MAOIs in cases of treatment-resistant depression. A review of the literature identified a few strategies with MAOIs, some successful and others not. Some evidence suggests that MAOI therapy may be effective in patients with early-stage treatment-resistant depression (up to 3 prior antidepressant trials).35 In elderly patients with treatment-resistant depression who had failed tricyclic antidepressant (TCA) or venlafaxine extended-release treatment, a population with many comorbid conditions, remission was not achieved in any patient switched to phenelzine but was achieved by one-third of patients augmented with lithium.36 Tolerance of both treatments was very good considering the patients’ health status and demands for other medications. An analysis of STAR*D showed that treatment with either tranylcypromine or extended-release venlafaxine plus mirtazapine yielded modest remission rates that were not statistically different between the 2 treatment groups.37 Although both treatment groups were similar in quality of response, the lower side effect profile, lack of dietary restrictions, and ease of use of venlafaxine and mirtazapine suggest that the combination may be preferred over the oral MAOI tranylcypromine for patients with treatment-resistant depression.
Evidence of responsiveness to MAOIs in subtypes of MDD. There is evidence that atypical depression is a distinct biological entity.20,38,39 Gold and associates38 suggested that, unlike melancholic depression, atypical depression is characterized by hypersomnolence, hyperphagia, and fatigue. From a clinical perspective, researchers at Columbia University (New York, New York) considered mood reactivity, the ability to feel at least 50% better when exposed to a positive event, to be a crucial defining feature of atypical depression.20,40 In addition to mood reactivity, the Columbia criteria required patients to have at least 2 of the recognized symptoms of atypical depression. The results of almost 20 years of study by the Columbia group confirmed what they described as the pharmacologic dissection of depressive subtypes: that is, that while patients with typical melancholic depression respond to a range of antidepressant agents, those with atypical depression respond preferentially to MAOIs and fail to respond to TCAs.20,40,41 Since then, other studies have shown that patients with atypical depression appear to be more likely to respond to MAOIs.40,42–44
Liebowitz et al40 conducted the largest of these trials in 119 patients with depression, mood reactivity, and at least 2 other symptoms suggestive of atypical depression (eg, overeating, oversleeping, extreme fatigue while depressed, oversensitivity to rejection). Patients who showed no response to 10 days of placebo treatment were randomized in a double-blind fashion to receive an MAOI (phenelzine), a TCA (imipramine), or placebo for 6 weeks. Response rates were 71% for the MAOI, 50% for the TCA, and 28% for placebo. Both active treatments were significantly more effective than placebo (P < .05) and, while the difference between the 2 active drugs did not reach statistical significance, the MAOI did exhibit a trend for better efficacy over the TCA. The investigators concluded that their findings supported the idea that responsiveness to MAOI therapy was a factor that differentiated atypical from melancholic depression.40
Quitkin et al45 conducted a study of identical design in 90 patients with probable atypical depression. At 6 weeks, response rates were 83% for phenelzine, 50% for imipramine, and 19% for placebo. Both active treatments were significantly superior to placebo (P < .001), and the MAOI was significantly superior to the TCA (P = .005). In addition, the MAOI was shown to be significantly superior to the TCA as measured by the Hamilton Depression Rating Scale (HDRS), the 90-item Hopkins Symptom Checklist, and the Clinical Global Impressions scale.45
STS: Efficacy of a Transdermal MAOI
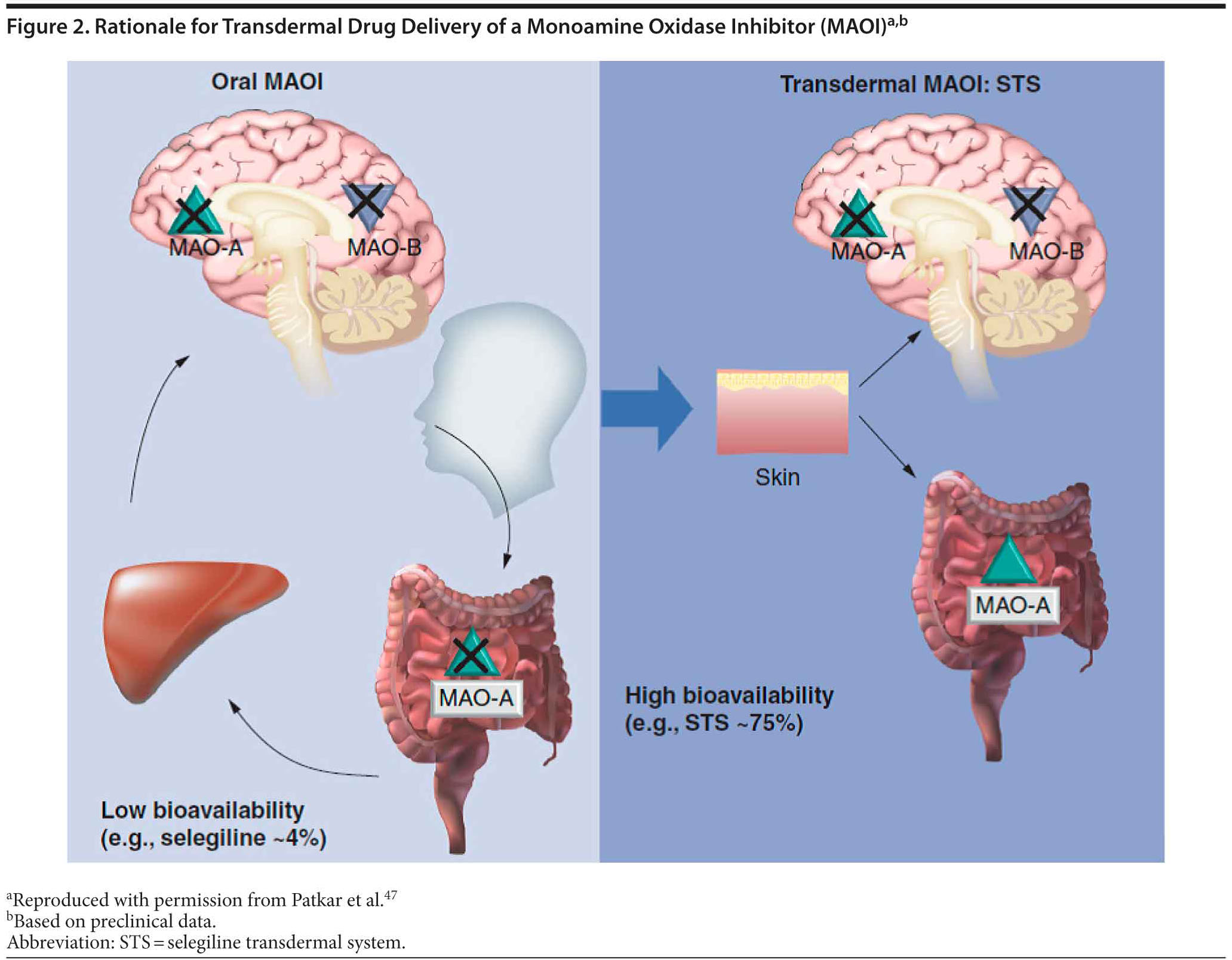
The transdermal form of selegiline was developed with pharmacokinetic and pharmacodynamic properties that would allow inhibition of MAO-A and MAO-B in the central nervous system while largely avoiding inhibition of MAO-A in the liver and intestine.46 The bioavailability of selegiline is approximately 75% following administration of the transdermal formulation compared with 4.4% after oral administration. This difference is due to absorption of drug from the patch directly into the systemic circulatory system, while orally administered preparations are absorbed into the gastrointestinal and portal circulatory system and exposed to first-pass metabolism before entry into the systemic circulatory system and becoming available in the central nervous system (Figure 2).47 Subsequently, STS results in higher and more sustained steady-state levels of selegiline compared with the oral formulation. The transdermal system allows for the administration of much lower doses than oral therapy to achieve therapeutic levels in the brain and also reduces the risk of the tyramine reaction and hypertensive crisis.24,48,49 A summary of 4 randomized clinical trials and 1 meta-analysis of STS is presented in Table 4.50–53
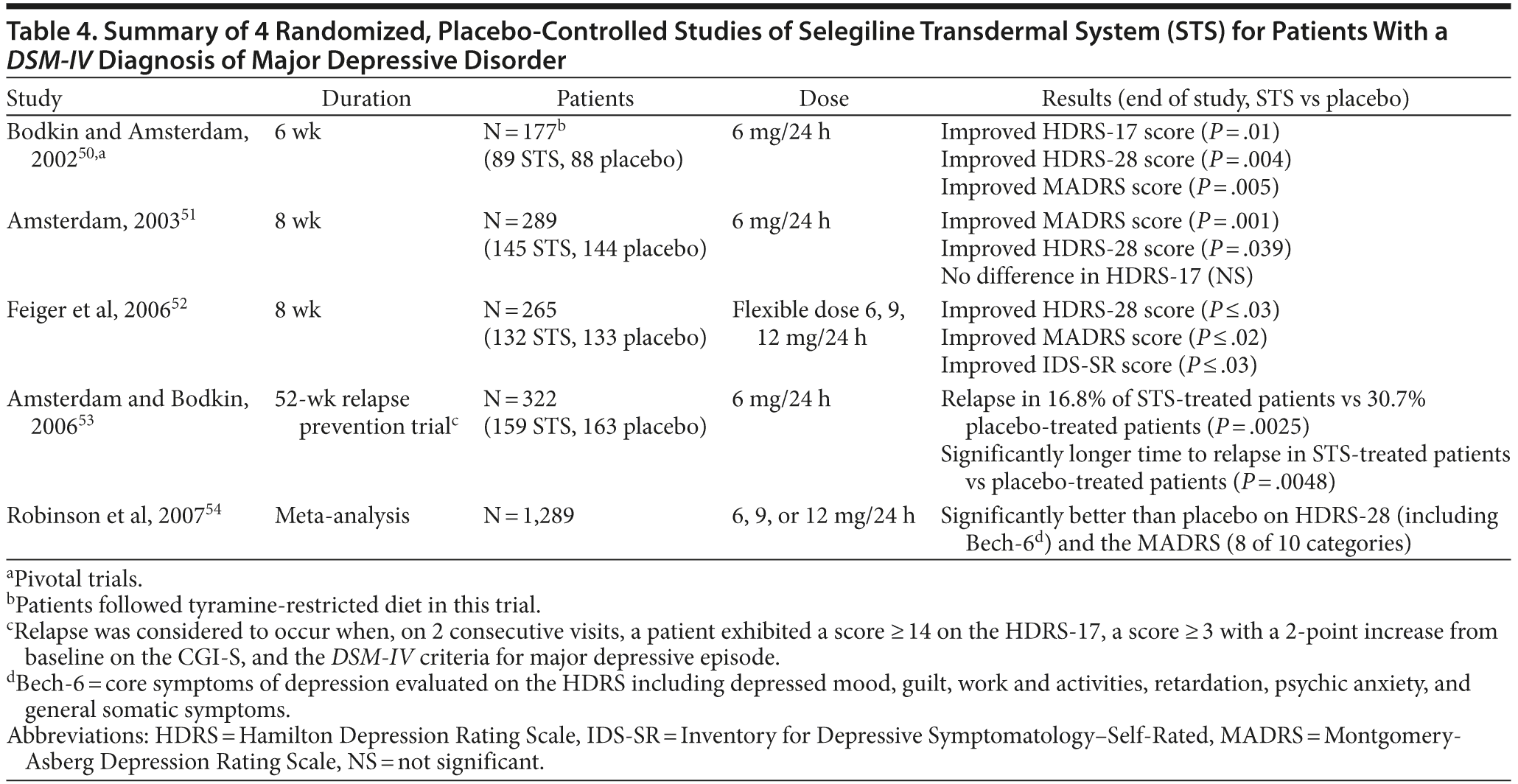
Meta-analysis on the efficacy of STS in short-term studies. Robinson et al54 conducted a meta-analysis of short-term, placebo-controlled efficacy trials of STS in the treatment of MDD. The sample included 1,289 patients with a mean ± SD age of 42 ± 11.3 years. The majority were women (62%), were white (85%), and had recurrent depression (64%). Mean HDRS-28 and Montgomery-Asberg Depression Rating Scale (MADRS) scores at baseline were 29 and 28, respectively.54 Transdermal selegiline was broadly effective for depressive symptoms on the basis of HDRS item analysis, including all Bech-6 core depression symptoms of depressed mood; guilt; work and activities; slowed thought, speech, or motor activity (retardation); psychic anxiety; and general somatic symptoms such as fatigue and aches. The effects of treatment with STS were also significant for HDRS ratings for suicidality, reverse vegetative symptoms such as hypersomnia and overeating, motor retardation, and decreased libido (in women). Of all the treatment effects of STS, the most prominent were related to interest and motivation in work and activities. Treatment effects of STS were also significant for 8 of 10 MADRS symptom categories with improvement in lassitude and concentration and in reverse vegetative symptoms, fatigue, and retardation, which suggests a utility for STS in minimizing common residual symptoms in depressed patients.54
Safety and Tolerability of STS
Robinson and Amsterdam55 conducted a similar review of the key safety and tolerability data accrued during all clinical trials of STS in patients with MDD.55 Controlled and uncontrolled studies included 2,036 patients treated with STS and 668 who had received placebo patches. Subjects averaged 44.4 years of age, with a range of 17 to 95 years, and the majority of patients were female and white. Dosing ranged from 3 mg/24 hours to 12 mg/24 hours. The maximum STS dose group of STS 9 mg/24 hours and STS 12 mg/24 hours included more elderly patients because the open-label flexible-dose study had no upper age limit and because the dose was titrated up to the maximum dose tolerated in the trial. Exposure to STS was up to 12 weeks for 1,238 patients, 12 to 14 weeks for 538 patients, 24 to 48 weeks for 205 patients, and over 48 weeks for 55 patients.55
The investigators reported on adverse events with an incidence ≥ 5% in placebo-controlled efficacy trials.55 A total of 72% of patients who had received placebo and 76% of patients treated with STS experienced at least 1 adverse event. In patients in the placebo-controlled studies of STS 6 mg/24 hours, the only adverse event with an incidence of twice that of the placebo group was application site reaction.55 In higher-dose studies (9 mg/24 hours or 12 mg/24 hours), adverse events with an incidence of at least twice that of placebo included insomnia (32% vs 7%), dry mouth (13% vs 6%), dizziness (14% vs 5%), nervousness (10% vs 4%), and abnormal dreams (5% vs 2%).55 Most adverse events were considered mild or moderate. In placebo-controlled studies, only 0.6% of patients treated with STS and 0.1% of patients treated with placebo had adverse events rated as severe. Most of these adverse events occurred at higher 9 mg/24 hours or 12 mg/24 hours doses. Severe insomnia was seen in 2.2% of patients treated with STS and 4.0% of patients treated with placebo.55 Except for the very first study conducted with STS,50 patients were not required to follow a tyramine-restricted diet. Still, no hypertensive crises occurred during STS therapy. While on STS therapy, 63 of 2,867 patients (2.2%) had reports of elevated blood pressure, most of whom had preexisting hypertension. Blood pressure elevation was judged to be unrelated to STS therapy.55
Two cases of serotonin syndrome were reported in patients treated with STS in clinical studies.55 The first involved a patient who experienced 2 acute episodes of severe akathisia 1 month apart. The first case occurred 1 week after discontinuing STS, immediately following ingestion of a single dose of venlafaxine. A similar event occurred a month later within an hour of a rechallenge with venlafaxine. Both events required medical intervention to treat symptoms. The final assessment of these serious adverse events described them as “akathisia secondary to venlafaxine treatment” and not serotonin syndrome.55 The second case was a patient who was hospitalized for “drug overdose” while receiving STS in an open-label extension study following double-blind therapy.55 The patient had been taking several contraindicated medications, including nortriptyline, bupropion, and a sympathomimetic agent for weight loss, and was wearing 2 STS patches simultaneously (12 mg/24 hours each). The patient recovered uneventfully.
Effect on weight. In short-term, placebo-controlled trials, patients who had received treatment with STS had a mean weight loss of 1.2 lb compared to a gain of 0.3 lb in patients who had received the placebo patch.55 A total of 2.1% of patients treated with STS and 2.4% of patients on placebo experienced a clinically significant (≥ 5%) weight gain during therapy; 5.0% of patients on STS and 2.8% of patients on placebo experienced clinically significant (≥ 5%) weight loss. In 676 patients who had received longer-term treatment (≥ 3 months) with STS, there was a mean weight loss of 1.6 lb from baseline.55
Sexual functioning. The incidence of adverse events related to sexual function across all trials was 2.7% (55/2,036) in patients treated with STS and 0.8% (7/831) in patients who received placebo.55 Clayton et al56 used a patient-rated questionnaire to examine the impact of treatment with STS 6 mg/24 hours on sexual function in patients with MDD. In this meta-analysis of four 6- to 8-week randomized, double-blind, placebo-controlled studies, the Medex Sexual Dysfunction Subscale (MED-D-SD) was used to assess sexual interest, arousal, maintenance of interest, orgasm, and satisfaction (n = 389, STS; n = 400, placebo).56 The analysis suggests that STS does not induce sexual dysfunction or lack of sexual satisfaction in female or male patients based on the results from the MED-D-SD questionnaire.56 Additional data from this analysis suggest that STS may improve sexual satisfaction based on improvements in interest, maintaining interest, and satisfaction reported in female patients.
STS: postmarketing safety. A postmarketing safety analysis of adverse events reported with the use of STS in the real world was recently published.57 In this analysis, a total of 29,141 patients were exposed to STS between 2006 and 2010. A total of 3,155 adverse events in 1,516 of 29,141 (5.2%) patients were reported, regardless of cause. Those most frequently reported adverse events are captured in Table 5. The adverse event profile for STS in the real world appears to be consistent with safety data on STS from randomized, placebo-controlled clinical trials. Therapeutic doses of STS appear to be tolerable in routine clinical practice.
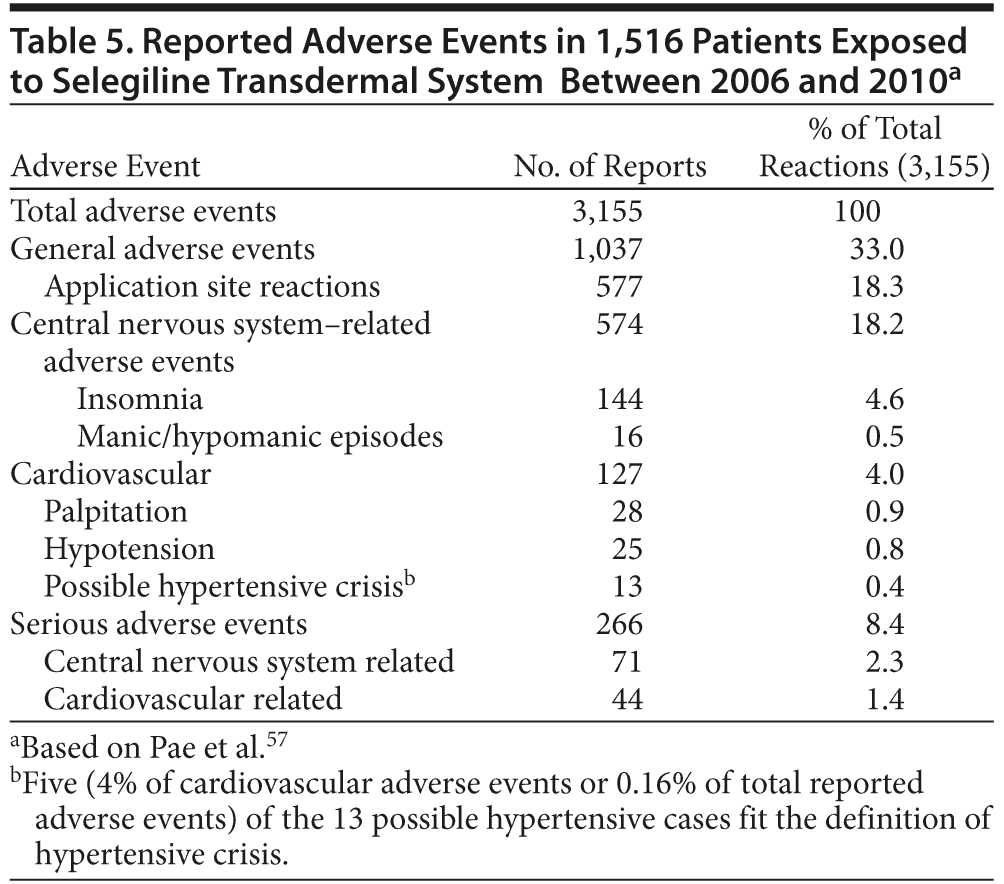
Recommended Dietary Modifications
and Contraindications With STS
The transdermal formulation of selegiline delivers the MAOI directly into the circulation and leaves the MAO-A in the gut largely unaffected.5 At the lowest dose of STS, 6 mg/24 hours, dietary restrictions are not required. This lack of restrictions may improve the likelihood of adherence to therapy. It should be noted, however, that dietary restrictions are still required with higher doses due to the lack of sufficient safety data and that, just as with any MAOI, precautions against drug interactions must be observed with STS at all doses. Certain foods and beverages should be avoided beginning on the first day of therapy with STS 9 mg/24 hours or 12 mg/24 hours and should continue to be avoided for 2 weeks after a dose reduction to STS 6 mg/24 hours or following the discontinuation of STS 9 mg/24 hours or 12 mg/24 hours (Table 6).58
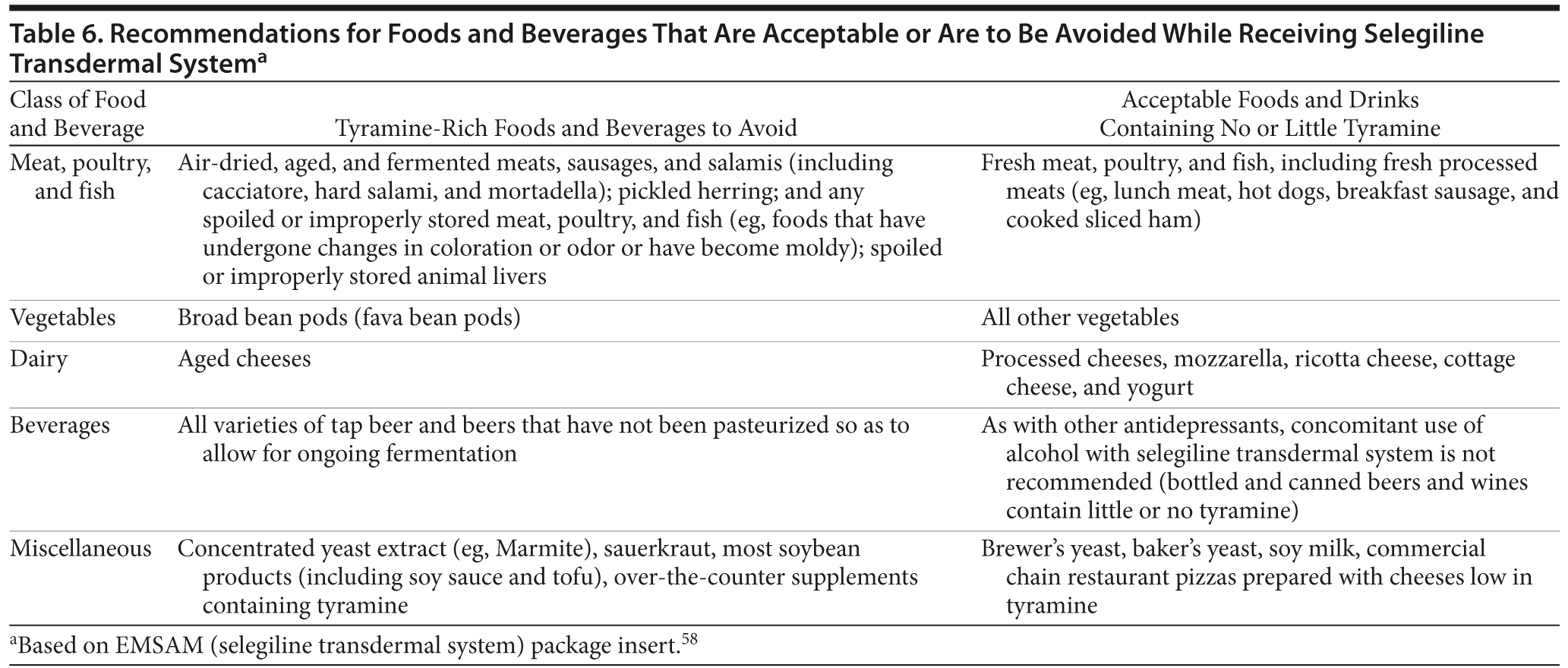
Transdermal selegiline is contraindicated with dextromethorphan and other sympathomimetic amines, including amphetamines as well as cold products and weight-reducing preparations that contain vasoconstrictors (eg, pseudoephedrine, phenylephrine, phenylpropanolamine, and ephedrine). In primary care, it is common for patients to use over-the-counter medications in these classes, and patients should question their use and be provided with adequate cautionary education by any clinician prior to and during administration of MAOI therapy.
CONCLUSION
The long-term goals of treatment for depression are to achieve disease remission and restore functioning and quality of life to our patients.7,59 However, a substantial number of patients with MDD do not achieve these goals even with repeated courses of long-term therapy. Difficult-to-treat MDD can be found in patients with residual depressive symptoms, atypical or anxious symptoms, chronic depression, and multiple recurrent episodes. Given the significant number of patients who do not completely respond to first-line therapy, it makes good clinical sense to revisit other strategies that have be shown to be effective but that may not necessarily be used as frequently as they were in the past. In fact, the best documented treatments such as lithium augmentation, switching to an MAOI, and electroconvulsive therapy are among the least utilized.60
Some evidence suggests that patients with atypical depression or treatment-resistant depression seem to respond well to MAOI therapy. The unique therapeutic benefits of MAOIs across different domains of mood and anxiety disorders may be the result of their modulating effects on all 3 monoamine neurotransmitters involved in the pathophysiology of depression (ie, serotonin, norepinephrine, and dopamine). The lack of effect on dopamine transmission with other types of antidepressants may be a possible cause of residual symptoms such as impaired motivation, concentration, and pleasure, even in patients who initially respond to other antidepressants.
Misinformation about MAOIs and the understanding on how to avoid potential dietary and drug-drug interactions has limited their use. A transdermal formulation of an MAOI may provide a treatment option that may mitigate some of the risks associated with oral MAOIs, including certain adverse events as well as dietary interactions. Clinicians in primary care settings may choose to reconsider MAOI therapy, particularly STS, as an option for patients with difficult-to-treat MDD. However, before prescribing an MAOI, clinicians must be mindful of drug-drug interactions that can occur with other medications their patients may be taking. Selegiline transdermal system may also provide clinicians with an option for patients who have experienced intolerable side effects such as weight gain or sexual dysfunction with other antidepressant therapies. With the appropriate education and vigilant use, clinicians can extend a valuable treatment option to patients with difficult-to-treat MDD.29
Drug names: bupropion (Wellbutrin, Aplenzin, and others), fluoxetine (Prozac and others), imipramine (Tofranil and others), isocarboxazid (Marplan), lithium (Lithobid and others), mirtazapine (Remeron and others), nortriptyline (Pamelor, Aventyl, and others), phenelzine (Nardil), selegiline transdermal system (EMSAM), tranylcypromine (Parnate and others), venlafaxine (Effexor and others).
Author affiliation: Department of Family Medicine, Boston University School of Medicine, Boston Medical Center, Boston, Massachusetts.
Potential conflicts of interest: Dr Culpepper has served as an advisor to or consultant for Boehringer-Ingelheim, Forest, Janssen, Jazz, H. Lundbeck A/S, Merck, Pfizer, Reckitt Benckiser, Sunovion, and Takeda and has made presentations regarding a federally funded study of methods to reduce hospital readmissions (with no mention of pharmaceutical agents) supported through Merck’s speaker’s bureau.
Funding/support: Funding for the medical writing and editorial assistance was provided by Mylan Specialty LP.
Acknowledgments: Medical writing and editorial assistance on the manuscript was provided by Sean M. Gregory, PhD, HYBRID Healthcare Communications, Red Bank, New Jersey. Dr Gregory reports no conflicts of interest related to the subject of this article.
Role of the sponsor: The sponsor had no role in the development of this article. Design of the manuscript, interpretation of the data, and final approval were provided by Dr Culpepper.
REFERENCES
1. Greenberg PE, Kessler RC, Birnbaum HG, et al. The economic burden of depression in the United States: how did it change between 1990 and 2000? J Clin Psychiatry. 2003;64(12):1465–1475. doi:10.4088/JCP.v64n1211 PubMed
2. Culpepper L. The use of monoamine oxidase inhibitors in primary care. J Clin Psychiatry. 2012;73(suppl 1):37–41. doi:10.4088/JCP.11096su1c.06 PubMed
3. Gaynes BN. Identifying difficult-to-treat depression: differential diagnosis, subtypes, and comorbidities. J Clin Psychiatry. 2009;70(suppl 6):10–15. doi:10.4088/JCP.8133su1c.02 PubMed
4. Rush AJ, Trivedi MH, Wisniewski SR, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163(11):1905–1917. doi:10.1176/appi.ajp.163.11.1905 PubMed
5. Cohen LJ, Sclar DA. Issues in adherence to treatment with monoamine oxidase inhibitors and the rate of treatment failure. J Clin Psychiatry. 2012;73(suppl 1):31–36. doi:10.4088/JCP.11096su1c.05 PubMed
6. Mueller TI, Leon AC, Keller MB, et al. Recurrence after recovery from major depressive disorder during 15 years of observational follow-up. Am J Psychiatry. 1999;156(7):1000–1006. PubMed
7. Trivedi MH, Daly EJ. Treatment strategies to improve and sustain remission in major depressive disorder. Dialogues Clin Neurosci. 2008;10(4):377–384. PubMed
8. Thase ME. Treatment of severe depression. J Clin Psychiatry. 2000;61(suppl 1):17–25. PubMed
9. Thase ME, Rush AJ. When at first you don’t succeed: sequential strategies for antidepressant nonresponders. J Clin Psychiatry. 1997;58(suppl 13):23–29. PubMed
10. Kantrowitz JT, Tampi RR. Risk of psychosis exacerbation by tricyclic antidepressants in unipolar major depressive disorder with psychotic features. J Affect Disord. 2008;106(3):279–284. doi:10.1016/j.jad.2007.07.012 PubMed
11. Kornstein SG, Schneider RK. Clinical features of treatment-resistant depression. J Clin Psychiatry. 2001;62(suppl 16):18–25. PubMed
12. Fava M, Rush AJ, Alpert JE, et al. Difference in treatment outcome in outpatients with anxious versus nonanxious depression: a STAR*D report. Am J Psychiatry. 2008;165(3):342–351. doi:10.1176/appi.ajp.2007.06111868 PubMed
13. Sanderson WC, Beck AT, Beck J. Syndrome comorbidity in patients with major depression or dysthymia: prevalence and temporal relationships. Am J Psychiatry. 1990;147(8):1025–1028. PubMed
14. Fava M, Rankin MA, Wright EC, et al. Anxiety disorders in major depression. Compr Psychiatry. 2000;41(2):97–102. doi:10.1016/S0010-440X(00)90140-8 PubMed
15. Joffe RT, Bagby RM, Levitt A. Anxious and nonanxious depression. Am J Psychiatry. 1993;150(8):1257–1258. PubMed
16. VanValkenburg C, Akiskal HS, Puzantian V, et al. Anxious depressions: clinical, family history, and naturalistic outcome—comparisons with panic and major depressive disorders. J Affect Disord. 1984;6(1):67–82. PubMed
17. Tollefson GD, Holman SL, Sayler ME, et al. Fluoxetine, placebo, and tricyclic antidepressants in major depression with and without anxious features. J Clin Psychiatry. 1994;55(2):50–59. PubMed
18. Clayton PJ, Grove WM, Coryell W, et al. Follow-up and family study of anxious depression. Am J Psychiatry. 1991;148(11):1512–1517. PubMed
19. Davidson JR, Meoni P, Haudiquet V, et al. Achieving remission with venlafaxine and fluoxetine in major depression: its relationship to anxiety symptoms. Depress Anxiety. 2002;16(1):4–13. doi:10.1002/da.10045 PubMed
20. Nierenberg AA, Alpert JE, Pava J, et al. Course and treatment of atypical depression. J Clin Psychiatry. 1998;59(suppl 18):5–9. PubMed
21. Levitan RD, Davis C, Kaplan AS, et al. Obesity comorbidity in unipolar major depressive disorder: refining the core phenotype. J Clin Psychiatry. 2012;73(8):1119–1124. doi:10.4088/JCP.11m07394 PubMed
22. Meyer JH, Ginovart N, Boovariwala A, et al. Elevated monoamine oxidase a levels in the brain: an explanation for the monoamine imbalance of major depression. Arch Gen Psychiatry. 2006;63(11):1209–1216. doi:10.1001/archpsyc.63.11.1209 PubMed
23. Shih JC, Chen K, Ridd MJ. Monoamine oxidase: from genes to behavior. Annu Rev Neurosci. 1999;22(1):197–217. doi:10.1146/annurev.neuro.22.1.197 PubMed
24. Thase ME. The role of monoamine oxidase inhibitors in depression treatment guidelines. J Clin Psychiatry. 2012;73(suppl 1):10–16. doi:10.4088/JCP.11096su1c.02 PubMed
25. Thase ME, Trivedi MH, Rush AJ. MAOIs in the contemporary treatment of depression. Neuropsychopharmacology. 1995;12(3):185–219. doi:10.1016/0893-133X(94)00058-8 PubMed
26. Owens MJ. Selectivity of antidepressants: from the monoamine hypothesis of depression to the SSRI revolution and beyond. J Clin Psychiatry. 2004;65(suppl 4):5–10. PubMed
27. VanDenBerg CM. The transdermal delivery system of monoamine oxidase inhibitors. J Clin Psychiatry. 2012;73(suppl 1):25–30. doi:10.4088/JCP.11096su1c.04 PubMed
28. Krishnan KR. Revisiting monoamine oxidase inhibitors. J Clin Psychiatry. 2007;68(suppl 8):35–41. PubMed
29. Stahl SM, Felker A. Monoamine oxidase inhibitors: a modern guide to an unrequited class of antidepressants. CNS Spectr. 2008;13(10):855–870. PubMed
30. American Psychiatric Association. Practice guideline for the treatment of patients with major depressive disorder, 3rd ed. Published 2010. http://psychiatryonline.org/content.aspx?bookid=28§ionid=1667485#654001. Accessed September 24, 2012.
31. Stahl SM. Selecting an antidepressant by using mechanism of action to enhance efficacy and avoid side effects. J Clin Psychiatry. 1998;59(suppl 18):23–29. PubMed
32. Fiedorowicz JG, Swartz KL. The role of monoamine oxidase inhibitors in current psychiatric practice. J Psychiatr Pract. 2004;10(4):239–248. doi:10.1097/00131746-200407000-00005 PubMed
33. Gardner DM, Shulman KI, Walker SE, et al. The making of a user friendly MAOI diet. J Clin Psychiatry. 1996;57(3):99–104. PubMed
34. Sternbach H. The serotonin syndrome. Am J Psychiatry. 1991;148(6):705–713. PubMed
35. Amsterdam JD, Shults J. MAOI efficacy and safety in advanced stage treatment-resistant depression—a retrospective study. J Affect Disord. 2005;89(1–3):183–188. doi:10.1016/j.jad.2005.06.011 PubMed
36. Kok RM, Vink D, Heeren TJ, et al. Lithium augmentation compared with phenelzine in treatment-resistant depression in the elderly: an open, randomized, controlled trial. J Clin Psychiatry. 2007;68(8):1177–1185. doi:10.4088/JCP.v68n0803 PubMed
37. McGrath PJ, Stewart JW, Fava M, et al. Tranylcypromine versus venlafaxine plus mirtazapine following three failed antidepressant medication trials for depression: a STAR*D report. Am J Psychiatry. 2006;163(9):1531–1541, quiz 1666. doi:10.1176/appi.ajp.163.9.1531 PubMed
38. Gold PW, Licinio J, Wong ML, et al. Corticotropin releasing hormone in the pathophysiology of melancholic and atypical depression and in the mechanism of action of antidepressant drugs. Ann N Y Acad Sci. 1995;771
(1 Stress):716–729. doi:10.1111/j.1749-6632.1995.tb44723.x PubMed
39. Anisman H, Ravindran AV, Griffiths J, et al. Endocrine and cytokine correlates of major depression and dysthymia with typical or atypical features. Mol Psychiatry. 1999;4(2):182–188. doi:10.1038/sj.mp.4000436 PubMed
40. Liebowitz MR, Quitkin FM, Stewart JW, et al. Antidepressant specificity in atypical depression. Arch Gen Psychiatry. 1988;45(2):129–137. doi:10.1001/archpsyc.1988.01800260037004 PubMed
41. Pande AC, Birkett M, Fechner-Bates S, et al. Fluoxetine versus phenelzine in atypical depression. Biol Psychiatry. 1996;40(10):1017–1020. doi:10.1016/0006-3223(95)00628-1 PubMed
42. Quitkin FM, Harrison W, Stewart JW, et al. Response to phenelzine and imipramine in placebo nonresponders with atypical depression: a new application of the crossover design. Arch Gen Psychiatry. 1991;48(4):319–323. doi:10.1001/archpsyc.1991.01810280035005 PubMed
43. Quitkin FM, Stewart JW, McGrath PJ, et al. Phenelzine versus imipramine in the treatment of probable atypical depression: defining syndrome boundaries of selective MAOI responders. Am J Psychiatry. 1988;145(3):306–311. PubMed
44. Quitkin FM, Stewart JW, McGrath PJ, et al. Columbia atypical depression: a subgroup of depressives with better response to MAOI than to tricyclic antidepressants or placebo. Br J Psychiatry suppl. 1993;(21):30–34. PubMed
45. Quitkin FM, McGrath PJ, Stewart JW, et al. Atypical depression, panic attacks, and response to imipramine and phenelzine: a replication. Arch Gen Psychiatry. 1990;47(10):935–941. doi:10.1001/archpsyc.1990.01810220051006 PubMed
46. Preskorn SH. Why the transdermal delivery of selegiline (6 mg/24 hr) obviates the need for a dietary restriction on tyramine. J Psychiatr Pract. 2006;12(3):168–172. doi:10.1097/00131746-200605000-00006 PubMed
47. Patkar AA, Portland KB, Pae CU. Selegiline transdermal system in major depressive disorder. Neuropsychiatry. 2012;2(2):125–134. doi:10.2217/npy.12.9
48. Cohen LJ, Sclar DA, Culpepper L, et al. Discussion: a fresh look at monoamine oxidase inhibitors for depression. J Clin Psychiatry. 2012;73(suppl 1):42–45. doi:10.4088/JCP.11096su1c.07 PubMed
49. Wecker L, James S, Copeland N, et al. Transdermal selegiline: targeted effects on monoamine oxidases in the brain. Biol Psychiatry. 2003;54(10):1099–1104. doi:10.1016/S0006-3223(02)01892-9 PubMed
50. Bodkin JA, Amsterdam JD. Transdermal selegiline in major depression: a double-blind, placebo-controlled, parallel-group study in outpatients. Am J Psychiatry. 2002;159(11):1869–1875. doi:10.1176/appi.ajp.159.11.1869 PubMed
51. Amsterdam JD. A double-blind, placebo-controlled trial of the safety and efficacy of selegiline transdermal system without dietary restrictions in patients with major depressive disorder. J Clin Psychiatry. 2003;64(2):208–214. doi:10.4088/JCP.v64n0216 PubMed
52. Feiger AD, Rickels K, Rynn MA, et al. Selegiline transdermal system for the treatment of major depressive disorder: an 8-week, double-blind, placebo-controlled, flexible-dose titration trial. J Clin Psychiatry. 2006;67(9):1354–1361. doi:10.4088/JCP.v67n0905 PubMed
53. Amsterdam JD, Bodkin JA. Selegiline transdermal system in the prevention of relapse of major depressive disorder: a 52-week, double-blind, placebo-substitution, parallel-group clinical trial. J Clin Psychopharmacol. 2006;26(6):579–586. doi:10.1097/01.jcp.0000239794.37073.70 PubMed
54. Robinson DS, Gilmor ML, Yang Y, et al. Treatment effects of selegiline transdermal system on symptoms of major depressive disorder: a meta-analysis of short-term, placebo-controlled, efficacy trials. Psychopharmacol Bull. 2007;40(3):15–28. PubMed
55. Robinson DS, Amsterdam JD. The selegiline transdermal system in major depressive disorder: a systematic review of safety and tolerability. J Affect Disord. 2008;105(1–3):15–23. doi:10.1016/j.jad.2007.04.024 PubMed
56. Clayton AH, Campbell BJ, Favit A, et al. Symptoms of sexual dysfunction in patients treated for major depressive disorder: a meta-analysis comparing selegiline transdermal system and placebo using a patient-rated scale. J Clin Psychiatry. 2007;68(12):1860–1866. doi:10.4088/JCP.v68n1205 PubMed
57. Pae CU, Bodkin JA, Portland KB, et al. Safety of selegiline transdermal system in clinical practice: analysis of adverse events from postmarketing exposures. J Clin Psychiatry. 2012;73(5):661–668. doi:10.4088/JCP.12m07648 PubMed
58. EMSAM (selegiline transdermal system) [package insert]. Napa, CA: Dey Pharma LP; 2011.
59. Gaynes BN, Rush AJ, Trivedi MH, et al. The STAR*D study: treating depression in the real world. Cleve Clin J Med. 2008;75(1):57–66. doi:10.3949/ccjm.75.1.57 PubMed
60. Thase ME. Therapeutic alternatives for difficult-to-treat depression: a narrative review of the state of the evidence. CNS Spectr. 2004;9(11):808–816, 818–821. PubMed
Enjoy this premium PDF as part of your membership benefits!


