Objective: To consolidate the evidence from the literature to evaluate the role of prazosin in the treatment of posttraumatic stress disorder (PTSD).
Data Sources: Major databases, including PubMed, Ovid EMBASE, Ovid Cochrane Central Register of Controlled Trials, Ovid Cochrane Database of Systematic Reviews, Ovid PsycINFO, and Scopus, were searched through August 2015 for studies reporting the role of prazosin in the treatment of PTSD with no language constraints. Keywords included (PTSD OR posttraumatic stress OR posttraumatic stress OR nightmares) AND prazosin.
Study Selection: Of 402 screened articles, 6 studies were included in the systematic review and meta-analysis.
Data Extraction: Two reviewers independently extracted relevant data (study characteristics, type of intervention, outcome measures, and follow-up) from the included studies using a standardized data extraction form. Only randomized controlled trials comparing prazosin to a placebo or control group in patients with PTSD were included.
Results: The patients with PTSD receiving prazosin showed significant improvement in nightmares (standardized mean difference [SMD] = 1.01; 95% CI, 0.72-1.30), overall PTSD symptoms (SMD = 0.77; 95% CI, 0.48-1.06), and clinical global improvement (SMD = 0.94; 95%, CI 0.6-1.29) compared to the placebo/control group. Prazosin improved sleep quality (SMD = 0.87; 95% CI, 0.55-1.19), hyperarousal symptoms (SMD = 1.04; 95% CI, 0.23-1.84), dream content (SMD = 1.33; 95% CI, 0.69-1.97), and total sleep time (60.98 minutes; 95% CI, 18.69-103.26). Prazosin was fairly well tolerated. Minor side effects were reported, which were similar between the prazosin and placebo groups.
Conclusions: This study suggests that prazosin improves nightmares and overall PTSD symptoms including hyperarousal, sleep disturbances, total sleep time, and sleep quality.
Efficacy of Prazosin in Posttraumatic Stress Disorder:
A Systematic Review and Meta-Analysis
ABSTRACT
Objective: To consolidate the evidence from the literature to evaluate the role of prazosin in the treatment of posttraumatic stress disorder (PTSD).
Data Sources: Major databases, including PubMed, Ovid EMBASE, Ovid Cochrane Central Register of Controlled Trials, Ovid Cochrane Database of Systematic Reviews, Ovid PsycINFO, and Scopus, were searched through August 2015 for studies reporting the role of prazosin in the treatment of PTSD with no language constraints. Keywords included (PTSD OR posttraumatic stress OR posttraumatic stress OR nightmares) AND prazosin.
Study Selection: Of 402 screened articles, 6 studies were included in the systematic review and meta-analysis.
Data Extraction: Two reviewers independently extracted relevant data (study characteristics, type of intervention, outcome measures, and follow-up) from the included studies using a standardized data extraction form. Only randomized controlled trials comparing prazosin to a placebo or control group in patients with PTSD were included.
Results: The patients with PTSD receiving prazosin showed significant improvement in nightmares (standardized mean difference [SMD] = 1.01; 95% CI, 0.72–1.30), overall PTSD symptoms (SMD = 0.77; 95% CI, 0.48–1.06), and clinical global improvement (SMD = 0.94; 95%, CI 0.6–1.29) compared to the placebo/control group. Prazosin improved sleep quality (SMD = 0.87; 95% CI, 0.55–1.19), hyperarousal symptoms (SMD = 1.04; 95% CI, 0.23–1.84), dream content (SMD = 1.33; 95% CI, 0.69–1.97), and total sleep time (60.98 minutes; 95% CI, 18.69–103.26). Prazosin was fairly well tolerated. Minor side effects were reported, which were similar between the prazosin and placebo groups.
Conclusions: This study suggests that prazosin improves nightmares and overall PTSD symptoms including hyperarousal, sleep disturbances, total sleep time, and sleep quality.
Prim Care Companion CNS Disord 2016;18(4):doi:10.4088/PCC.16r01943
© Copyright 2016 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry and Behavioral Science, University of North Dakota School of Medicine and Health Sciences, Fargo
bDepartment of Psychiatry, Oregon Health and Science University, Portland
cDepartment of Public Health, School of Medicine, University of North Dakota, Grand Forks
dMayo Clinic Libraries, Rochester, Minnesota
eDepartment of Psychiatry and Behavioral Sciences, University of Texas Medical School, Houston
*Corresponding author: Balwinder Singh, MD, MS, Department of Psychiatry and Behavioral Science, University of North Dakota School of Medicine and Health Sciences, 1919 Elm St N, Fargo, ND 58102 ([email protected]).
Trauma can be any impactful event inclusive of imminent threat to life, serious injury, or sexual defilement. Posttraumatic stress disorder (PTSD) is a mental health illness triggered by experiencing, perceiving, or witnessing a trauma.1 With the recent changes in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5),1 the diagnostic criteria for PTSD seem to have become stricter, presumably leading to lower detection of its prevalence compared to DSM-IV criteria.2,3 However, the cross-sectional studies of representative samples of US adults reported increasing lifetime prevalence of PTSD, ranging between 8.3%3 and 10.4%.2 Similarly, in a convenience sample of US veterans, the prevalence was reported to be 30.5% for current diagnosis and 67.5% for lifetime diagnosis of PTSD.2 Therefore, even with the refined criteria, the disease burden of PTSD seems to be rising. PTSD not only affects the quality of life and incurs financial burden on its victims, but also makes them prone to depression, drug abuse, anger issues, marital discord, suicide attempts, and other physical and mental disabilities.4 Thus, newer and more effective management of PTSD is crucial.
Since its first identification in the DSM-III (early 1980s), PTSD has been managed with various combinations of medication and behavioral therapy. The role of monoamine oxidase inhibitors, benzodiazepines, tricyclic antidepressants, and selective serotonin reuptake inhibitors cannot be denied in the management of depression, hypervigilance, reexperiencing, and emotional numbing aspects of PTSD.5 However, the side effects of these drugs could make them less rewarding. Moreover, the symptoms of nightmares, sleep disturbances, and hyperarousal are not adequately addressed by these drugs.6,7 Prazosin (a sympatholytic α-1 adrenergic receptor blocker) is an emerging treatment modality for these debilitating symptoms of PTSD. The evidence of its effectiveness and efficacy is promising, however, not definitive.7–9 Therefore, we conducted this systematic review and meta-analysis to consolidate the existing evidence from the literature to evaluate the efficacy of prazosin in the management of PTSD.
METHOD
This systematic review and meta-analysis followed the PRISMA (preferred reporting items for systematic reviews and meta-analyses) guidelines.10
Data Sources and Search Strategies
A comprehensive search (available on request) was conducted in PubMed, Ovid PsycINFO, Ovid EMBASE, Ovid Cochrane Central Register of Controlled Trials, Ovid Cochrane Database of Systematic Reviews, and Scopus through August 2015 (initial search dated September 11, 2013, updated on August 2015: added 126 more studies). The search strategy was conducted by an experienced librarian (P.J.E.) using a combination of controlled vocabulary and keywords: (PTSD OR posttraumatic stress OR posttraumatic stress OR nightmares) AND prazosin. To broaden the search for potentially eligible studies, we also reviewed the references of possibly eligible articles.
Eligibility Criteria
Only randomized controlled trials (RCTs) including patients with PTSD and comparing prazosin to a placebo or control group were included. The study’s eligibility criteria for the meta-analysis were (1) RCTs, (2) subjects with PTSD enrolled, and (3) reports of mean change in PTSD score between prazosin and placebo/control.
Study Selection
Two reviewers (B.S. and A.J.H.) independently identified studies meeting inclusion criteria that enrolled subjects of any age with PTSD who were treated with prazosin and reported the primary outcome as change in PTSD symptoms or nightmares. Studies were selected without imposing any restrictions on the basis of language of publication. We did not restrict any studies in which subjects were taking other medications for PTSD as is the case in real-life situations.
Any disagreement between the reviewers was resolved by consensus, and interreviewer agreement was assessed using k statistics.11 Studies from which data could not be extracted for meta-analysis but fulfilled inclusion criteria were included in the systematic review. Cohort, case-control, and cross-sectional studies and case series or case reports were excluded from the systematic review.
Data Collection
Two reviewers (B.S. and A.J.H.) independently extracted relevant data (study characteristics: author, year, country, study design, sample size, demographic characteristics of study participants, inclusion and exclusion criteria, type of intervention, outcome measures, and follow-up) from the included studies using a standardized data extraction form.
Outcome Measures
Improvement in sleep quality and global clinical status were measured by the Pittsburgh Sleep Quality Index (PSQI)12 and the Clinical Global Impression of Change (CGI-C),13 respectively. Improvement in the PTSD symptoms was measured by change in Clinician-Administered PTSD Scale (CAPS)14 scores or the PTSD Checklist–Civilian version (PCL-C).15 The PSQI is a self-report scale assessing sleep quality and sleep disturbance. The CGI-C is an investigator/clinician–rated assessment of change in global clinical status. Improvement in other sleep parameters such as sleep duration and dream content if given were also measured.
Quality Assessment
To assess the methodological quality of included studies, we used Cochrane Collaboration’s Risk of Bias Tool.16 Two reviewers independently and in duplicate assessed the quality of studies for random sequence generation; allocation concealment; blinding of participants, caregivers, or study personnel who assessed outcomes; incomplete outcome data; selective reporting; and other biases including funding source and type of funding provided. Responses for each criterion were reported as low risk of bias, high risk of bias, and unclear risk of bias by 2 investigators. Any disagreement was resolved by mutual consensus.
Statistical Analyses
We calculated standardized mean difference (SMD) with 95% confidence interval (CI) for each study. SMD is used as a summary statistic in meta-analysis when the studies assess the same outcome using different measures.16,17 Data are reported as mean and standard deviation (SD) for continuous variables and frequency and proportions for categorical variables. To account for between-study heterogeneity, we used the random DerSimonian and Laird18 effects model for analysis. Heterogeneity among the studies included in the meta-analysis was assessed using χ2 statistic and the Cochran Q statistic.19 A P value < .10 on the Cochran Q test suggests that heterogeneity is due to between-study factors rather than sampling errors.19 All other P values were considered statistically significant at P < .05. Review Manager (RevMan) Version 5.3 (The Nordic Cochrane Centre, The Cochrane Collaboration, Copenhagen, Denmark) was used to perform all statistical analyses. Missing data were imputed using the standardized imputation techniques.16
RESULTS
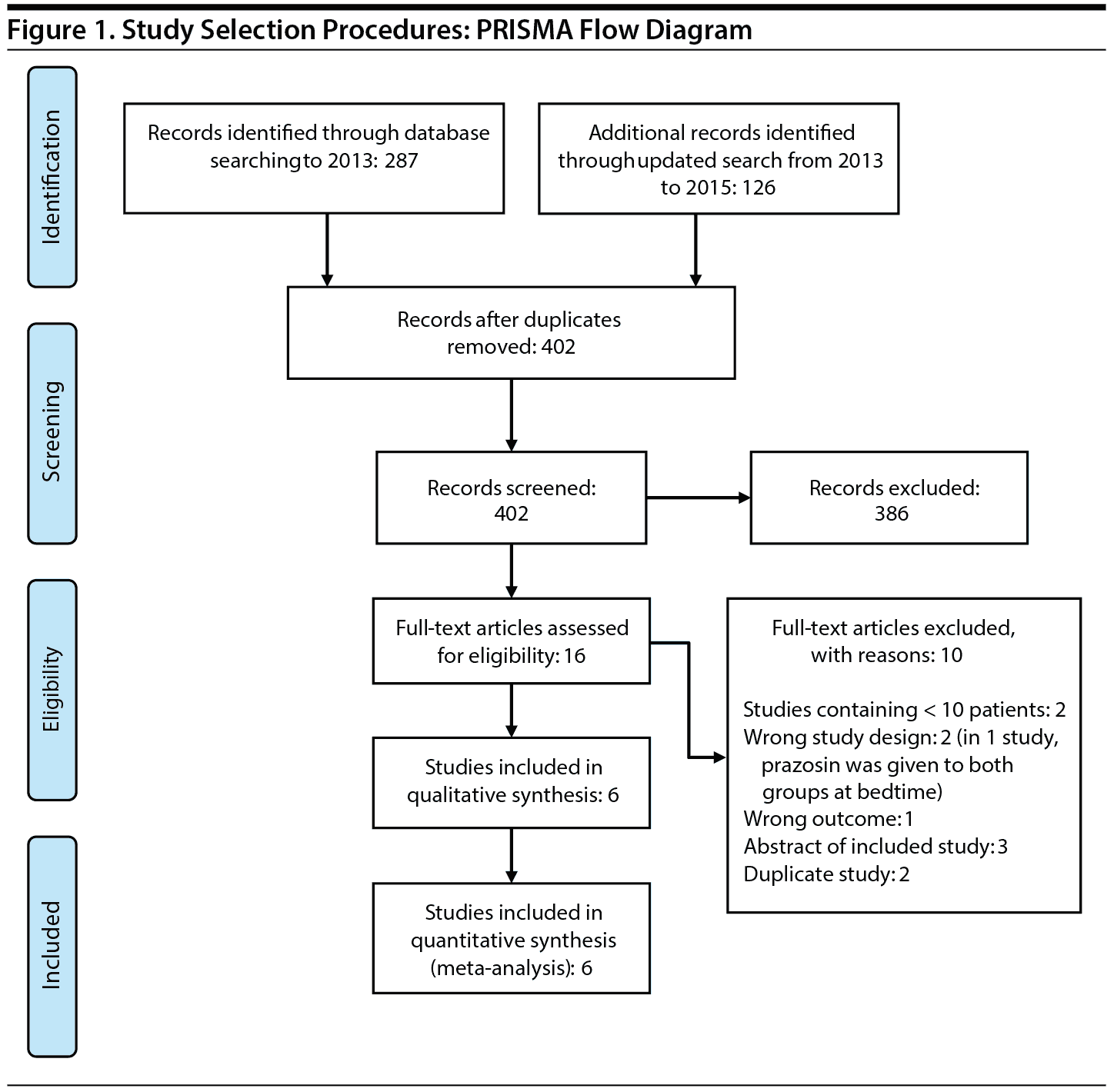
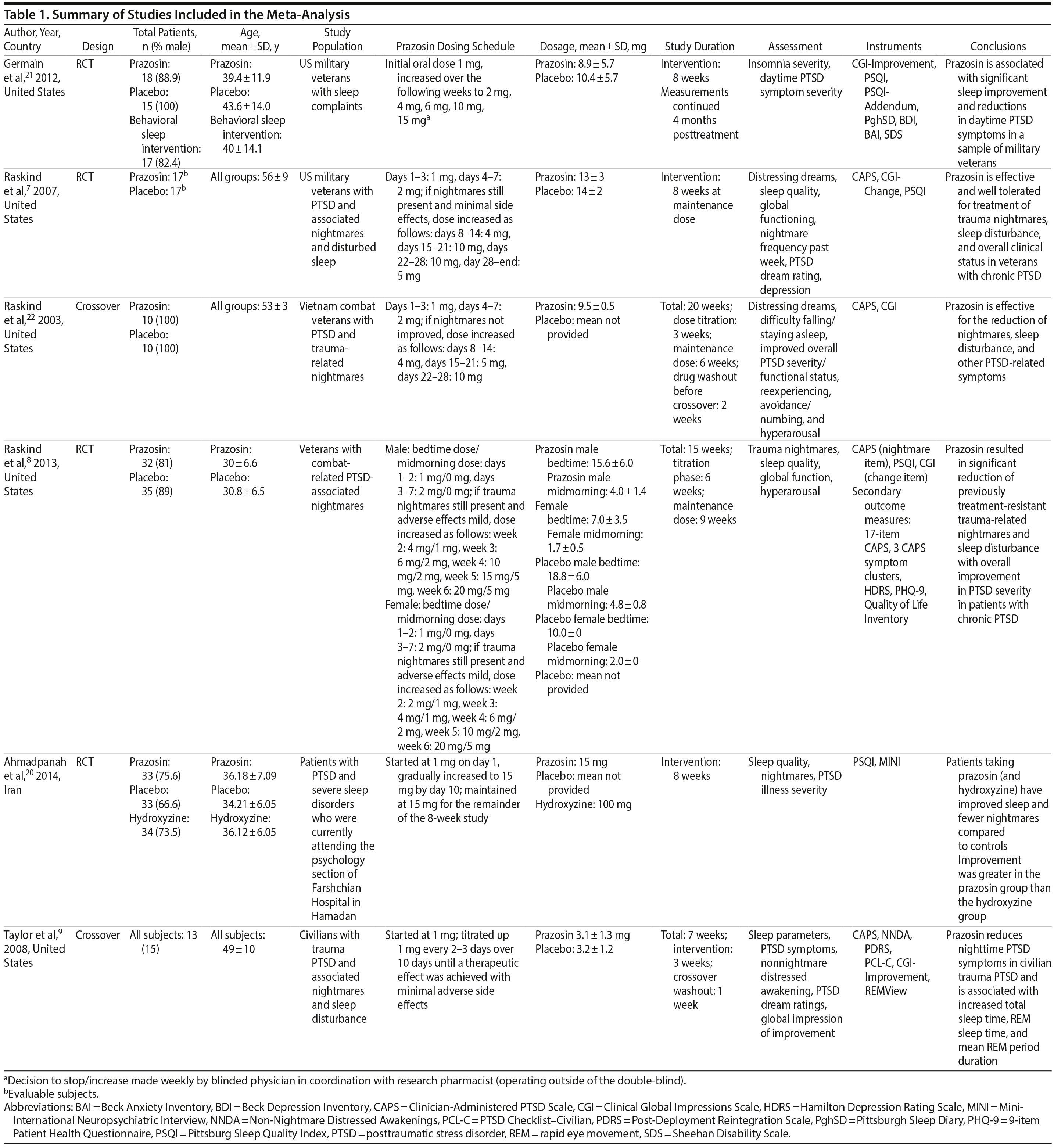
Figure 1 shows the study selection flow diagram. A total of 402 articles were identified through the initial detailed search. After the initial screening of abstracts, 16 studies were included for full-text review. A total of 6 RCTs7–9,20–22 enrolling 280 subjects (80% were male) were included in the systematic review and meta-analysis. The interreviewer agreement during both phases of study selection was excellent (k = 0.94, 95% agreement). Table 1 describes the details of the study characteristics.
Overall PTSD Score
Five studies were included in the PTSD analysis, including both civilian (1 study9) and veteran (4 studies7,8,20,22) populations. Compared to placebo, prazosin significantly decreased overall PTSD symptoms (SMD = 0.77; 95% CI, 0.48–1.06; P < .001) in both the civilian and veteran populations (Figure 2A).
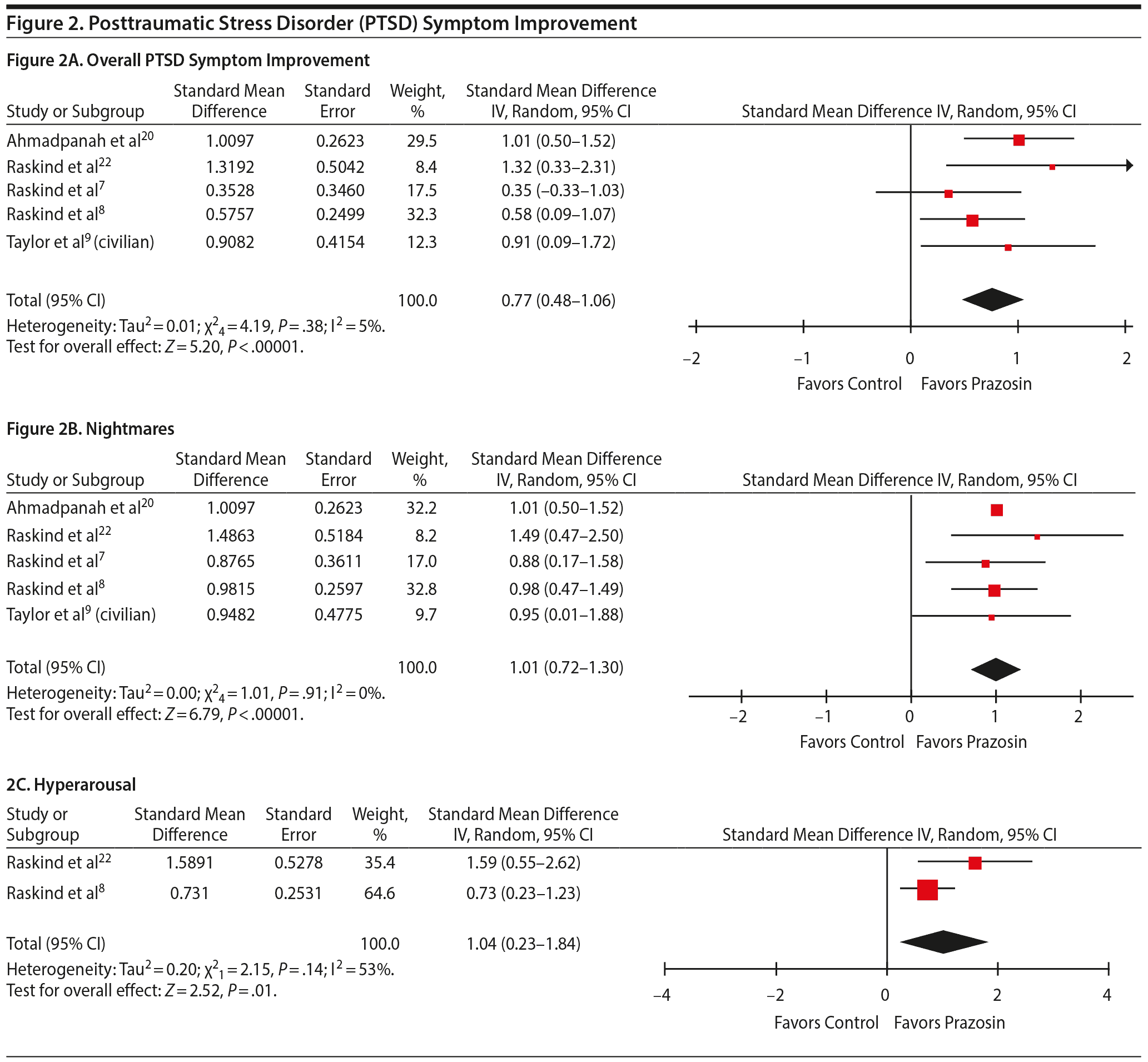
Nightmares and Distressing Dreams
Five studies7–9,20,22 were included in the nightmares and distressing dreams analysis. Prazosin significantly improved nightmares in patients with PTSD compared to placebo (SMD = 1.01; 95% CI, 0.72–1.30; P < .001) (Figure 2B).
Hyperarousal
Two studies8,22 were included in the hyperarousal analysis. Compared to placebo, prazosin significantly improved hyperarousal symptoms in patients with PTSD (SMD = 1.04; 95% CI, 0.23–1.84; P = .01) (Figure 2C).
Difficulty Falling or Staying Asleep
Only 2 studies9,22 reported outcome data for this component. Prazosin did appear to help with difficulty falling or staying asleep, but the effect did not reach statistical significance (P = .08), most likely due to the small number of studies.
Reexperiencing/Intrusion
Limited data from 2 studies8,22 suggested no difference between prazosin and placebo in improving these symptoms, most likely secondary to the small sample size (P = .18).
Avoidance/Numbing
The improvement in avoidance symptoms was similar between the prazosin and placebo groups (P = .11).
CGI-C Scale
Four studies7–9,22 reported the CGI change for overall PTSD severity and functional status. Prazosin showed favorable change in the global clinical status compared to placebo (SMD = 0.94; 95% CI, 0.60–1.29; P < .001) (Figure 3A).
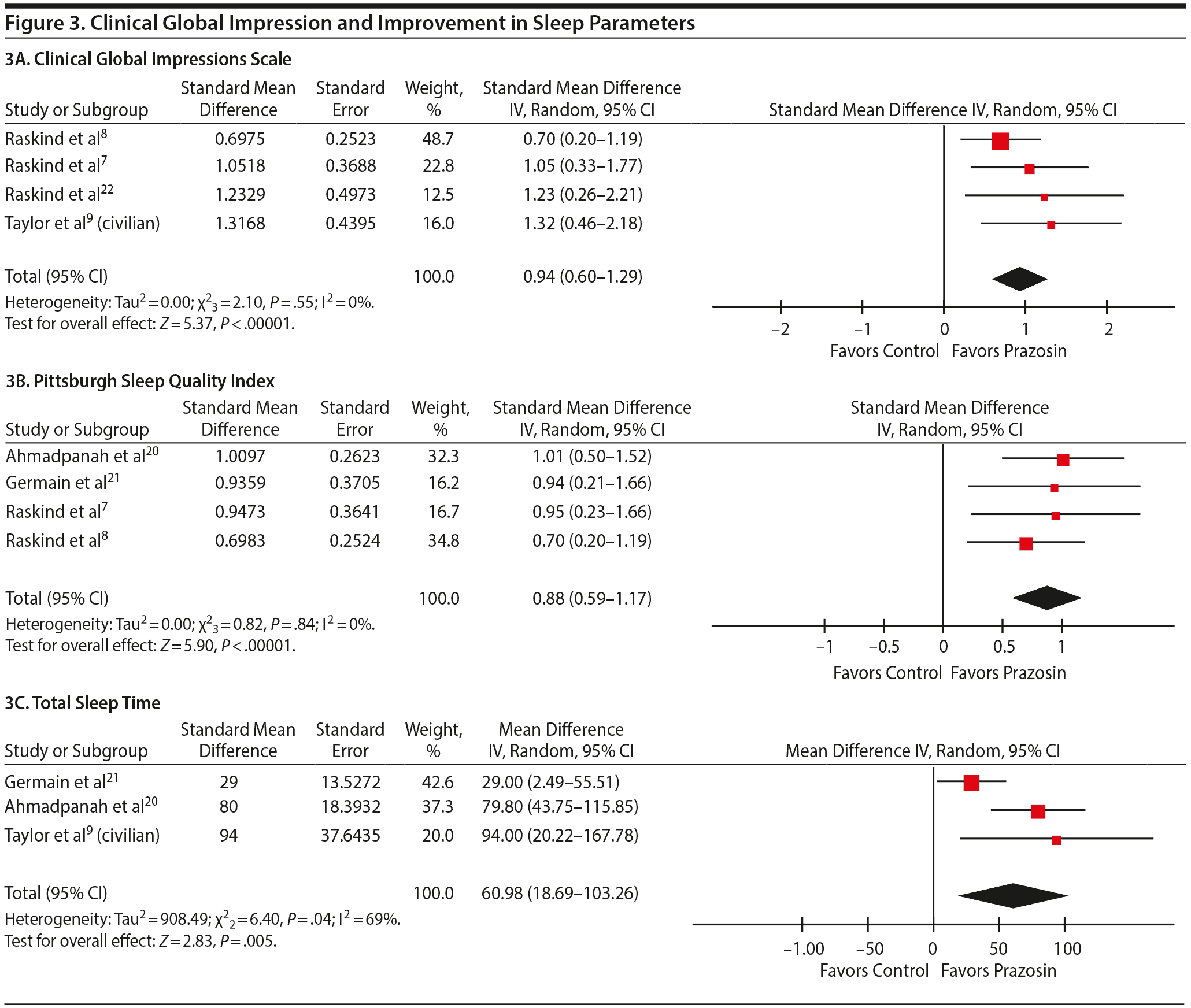
Improvement in Sleep
PSQI. Four studies7,8,20,21 were included in this analysis. Prazosin produced significantly greater improvement in the sleep quality than placebo (SMD = 0.88; 95% CI, 0.59–1.17; P < .001) (Figure 3B).
PTSD Dream Rating Scale. Two studies7,9 reported data on the PTSD Dream Rating Scale,23 which is an interviewer-administered instrument for assessing pathological contents of trauma-associated dreams. On the basis of pooled data from 2 studies, prazosin was more effective in reducing trauma nightmares to normal dreams than placebo (SMD = 1.33; 95% CI, 0.69–1.97; P < .001).
Total sleep time. On the basis of pooled analysis from 3 studies, prazosin increased the total sleep time by 60.98 minutes (95% CI, 18.69–103.26) compared to placebo (P = .005) (Figure 3C).
Minimal heterogeneity was observed between the studies for the majority of outcomes. On the basis of 1 study,21 behavioral sleep intervention was comparable to prazosin in reducing daytime PTSD symptoms and improving sleep.
Quality Assessment
Table 2 shows the quality assessment of RCTs included in the meta-analysis. The majority of the studies were of good quality with low or uncertain risk of bias. Only 1 study21 had higher risk of attrition bias, which was excluded from the primary outcome analysis of improvement in PTSD symptoms.

Adverse Effects
Prazosin was fairly well tolerated. The most commonly reported side effects were orthostatic hypotension, nasal congestion, and headache, which were comparable between the prazosin and placebo groups (P > .05).
Subgroup Analysis
No difference was observed in the major outcomes according to the study location (US vs non-US), population (veteran vs civilian), and study design (crossover vs parallel).
DISCUSSION
In this study, we conducted a thorough systematic review and meta-analysis of available RCTs in an effort to explore the evidence supporting efficacy of prazosin in the treatment of PTSD. In the majority of patients, PTSD is strongly characterized by nightmares and sleep disturbances. In fact, these sleep disturbances tend to originate in psychiatric distress and are often termed psychiatric insomnia.24 Current management modalities for PTSD rely mainly on exposure-based cognitive-behavioral therapy (CBT), such as cognitive-processing therapy and prolonged exposure therapy, and eye movement desensitization and reprocessing (EMDR). There is obvious long-term benefit to CBT and EMDR; however, slower action and patient compliance is a major problem with one-on-one behavioral therapy.25 Research shows that these sleep disturbances are related to a disrupted rapid eye movement (REM) phase of the sleep cycle.26 Our meta-analysis indicates that patients with PTSD receiving prazosin had significant improvement in their sleep quality, dream content, sleep duration, and nightmares. In addition, prazosin also improved hyperarousal, global functional status, and overall PTSD symptoms. Therefore, prazosin seems to improve many symptoms in addition to nightmares and sleep disturbances in patients with PTSD.
Prior studies27 have shown that activation of adrenergic α-1 receptors in the central nervous system can lead to increased cognitive processing and fear response, ultimately resulting in cortisol secretion as a buffer for stress. Additionally, these activated receptors disrupt the REM cycle and increase the non-REM phase, resulting in easily disturbed sleep.27 Prazosin crosses the blood-brain barrier and specifically inhibits the α-1 receptors, thus potentially contributing to improved sleep and decreased nightmares.7,8,22
Another aspect of sleep disorders commonly encountered in PTSD is difficulty falling asleep or staying asleep. These patients tend to sleep lightly and are easily awoken,28 resulting in inadequate body rest. Pooled data from our meta-analysis indicates that patients taking prazosin stayed asleep longer (61 min) compared to placebo. Interestingly, there were no carryover effects of prazosin. Soon after discontinuing the prazosin, the disrupted sleep pattern reemerged, reinforcing the efficacy of the drug.22 Additionally, 3 of the 5 clinical trials7,8,21 used the PSQI to compare the results of prazosin to placebo; and all observed significantly improved sleep quality.
The pooled data also showed a decrease in overall PTSD symptoms as assessed by the CAPS. Thus, the benefits of prazosin are not restricted to sleep disorders alone, but flow over into the general well-being and early recovery from PTSD symptoms. Such improvement could be due to relaxing sleep cycles, which lead to adequate rest, improved mood, and decreased anger outbursts.29,30 Similarly, 47,8,9,22 of the 5 studies tested the effect of drug intervention on daytime and overall functionality using the CGI. Patients with PTSD taking prazosin reported significantly improved scores compared to the placebo group. Our results echoed prior literature reviews31–35 in establishing prazosin as an effective and efficacious drug that has comparatively less economic and compliance issues. The common side effects of prazosin include hypotension, syncope, headaches, and drowsiness.36 As such, it would be prudent to consider possible drug interactions while adding prazosin as an adjunct therapy to the current pharmacologic approach.
This study does have some limitations. First, the analyses were restricted to a limited number of studies despite conducting an extensive literature search. We included only completed, published RCTs that compared prazosin to placebo in the presence or absence of any other pharmacologic treatment. In most of the studies, patients with PTSD were taking other psychotropic medications. Therefore, we cannot effectively comment on the efficacy of prazosin in isolation for PTSD symptoms. Also, our results are only as accurate as those reported by the clinical trials. Publication bias could not be assessed due to the small number of studies.37
In conclusion, this study suggests that prazosin not only helps with nightmares and sleep disturbances, but also improves overall PTSD symptoms and functionality. These results could pave the way for future research into the optimum dosage of prazosin as well as effective combination therapies aiming to decrease the burden of PTSD.
Submitted: February 22, 2016; accepted April 26, 2016.
Published online: July 28, 2016.
Drug name: prazosin (Minipress and others).
Potential conflicts of interest: None.
Funding/support: None.
REFERENCES
1. American Psychiatric Association. Diagnostic and Statistical Manual for Mental Disorders. Fifth Edition. Washington, DC: American Psychiatric Association; 2013.
2. Miller MW, Wolf EJ, Kilpatrick D, et al. The prevalence and latent structure of proposed DSM-5 posttraumatic stress disorder symptoms in US national and veteran samples. Psychol Trauma. 2013;5(6):501–512. doi:10.1037/a0029730
3. Kilpatrick DG, Resnick HS, Milanak ME, et al. National estimates of exposure to traumatic events and PTSD prevalence using DSM-IV and DSM-5 criteria. J Trauma Stress. 2013;26(5):537–547. doi:10.1002/jts.21848 PubMed
4. Sareen J, Cox BJ, Stein MB, et al. Physical and mental comorbidity, disability, and suicidal behavior associated with posttraumatic stress disorder in a large community sample. Psychosom Med. 2007;69(3):242–248. doi:10.1097/PSY.0b013e31803146d8 PubMed
5. Cohen BJ. Theory and Practice of Psychiatry. New York, NY: Oxford University Press; 2003.
6. Belkin MR, Schwartz TL. Alpha-2 receptor agonists for the treatment of posttraumatic stress disorder. Drugs Context. 2015;4:212286. doi:10.7573/dic.212286 PubMed
7. Raskind MA, Peskind ER, Hoff DJ, et al. A parallel group placebo controlled study of prazosin for trauma nightmares and sleep disturbance in combat veterans with posttraumatic stress disorder. Biol Psychiatry. 2007;61(8):928–934. doi:10.1016/j.biopsych.2006.06.032 PubMed
8. Raskind MA, Peterson K, Williams T, et al. A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returned from Iraq and Afghanistan. Am J Psychiatry. 2013;170(9):1003–1010. doi:10.1176/appi.ajp.2013.12081133 PubMed
9. Taylor FB, Martin P, Thompson C, et al. Prazosin effects on objective sleep measures and clinical symptoms in civilian trauma posttraumatic stress disorder: a placebo-controlled study. Biol Psychiatry. 2008;63(6):629–632. doi:10.1016/j.biopsych.2007.07.001 PubMed
10. Moher D, Liberati A, Tetzlaff J, et al; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. 2009;339:b2535. doi:10.1136/bmj.b2535 PubMed
11. Cohen J. A coefficient of agreement for nominal scales. Educ Psychol Meas. 1960;20(1):37–46. doi:10.1177/001316446002000104
12. Buysse DJ, Reynolds CF 3rd, Monk TH, et al. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;28(2):193–213. doi:10.1016/0165-1781(89)90047-4 PubMed
13. Guy W. Clinical Global Impressions (028-CGI). In: ECDEU Assessment Manual for Psychopharmacology. Rockville, MD: National Institute of Mental Health; 1976:216–222. >
14. Blake DD, Weathers FW, Nagy LM, et al. The development of a Clinician-Administered PTSD Scale. J Trauma Stress. 1995;8(1):75–90. doi:10.1002/jts.2490080106 PubMed
15. Weathers FW, Litz BT, Herman JA, et al. The PTSD Checklist: Reliability, validity, and diagnostic utility. Presented at the annual meeting of the International Society for Traumatic Stress Studies; October 24–27, 1993; San Antonio, TX.
16. Cochrane Handbook for Systematic Reviews of Interventions, Version 5.1.0. Cochrane Web site. www.cochrane-handbook.org. Updated March 2011.
17. Parsaik AK, Singh B, Khosh-Chashm D, et al. Efficacy of ketamine in bipolar depression: systematic review and meta-analysis. J Psychiatr Pract. 2015;21(6):427–435. doi:10.1097/PRA.0000000000000106 PubMed
18. DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7(3):177–188. doi:10.1016/0197-2456(86)90046-2 PubMed
19. Higgins JP, Thompson SG, Deeks JJ, et al. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557–560. doi:10.1136/bmj.327.7414.557 PubMed
20. Ahmadpanah M, Sabzeiee P, Hosseini SM, et al. Comparing the effect of prazosin and hydroxyzine on sleep quality in patients suffering from posttraumatic stress disorder. Neuropsychobiology. 2014;69(4):235–242. doi:10.1159/000362243 PubMed
21. Germain A, Richardson R, Moul DE, et al. Placebo-controlled comparison of prazosin and cognitive-behavioral treatments for sleep disturbances in US military veterans. J Psychosom Res. 2012;72(2):89–96. doi:10.1016/j.jpsychores.2011.11.010 PubMed
22. Raskind MA, Peskind ER, Kanter ED, et al. Reduction of nightmares and other PTSD symptoms in combat veterans by prazosin: a placebo-controlled study. Am J Psychiatry. 2003;160(2):371–373. doi:10.1176/appi.ajp.160.2.371 PubMed
23. Esposito K, Benitez A, Barza L, et al. Evaluation of dream content in combat-related PTSD. J Trauma Stress. 1999;12(4):681–687. doi:10.1023/A:1024725319777
24. Nowell PD, Buysse DJ, Reynolds CF 3rd, et al. Clinical factors contributing to the differential diagnosis of primary insomnia and insomnia related to mental disorders. Am J Psychiatry. 1997;154(10):1412–1416. doi:10.1176/ajp.154.10.1412 PubMed
25. Green B. Prazosin in the treatment of PTSD. J Psychiatr Pract. 2014;20(4):253–259. doi:10.1097/01.pra.0000452561.98286.1e PubMed
26. Woodward SH, Arsenault NJ, Murray C, et al. Laboratory sleep correlates of nightmare complaint in PTSD inpatients. Biol Psychiatry. 2000;48(11):1081–1087. doi:10.1016/S0006-3223(00)00917-3 PubMed
27. Boehnlein JK, Kinzie JD. Pharmacologic reduction of CNS noradrenergic activity in PTSD: the case for clonidine and prazosin. J Psychiatr Pract. 2007;13(2):72–78. doi:10.1097/01.pra.0000265763.79753.c1 PubMed
28. Pillar G, Malhotra A, Lavie P. Posttraumatic stress disorder and sleep—what a nightmare! Sleep Med Rev. 2000;4(2):183–200. doi:10.1053/smrv.1999.0095 PubMed
29. Yoo S-S, Gujar N, Hu P, et al. The human emotional brain without sleep—a prefrontal amygdala disconnect. Curr Biol. 2007;17(20):R877–R878. doi:10.1016/j.cub.2007.08.007 PubMed
30. Gujar N, McDonald SA, Nishida M, et al. A role for REM sleep in recalibrating the sensitivity of the human brain to specific emotions. Cereb Cortex. 2011;21(1):115–123. doi:10.1093/cercor/bhq064 PubMed
31. Berger W, Mendlowicz MV, Marques-Portella C, et al. Pharmacologic alternatives to antidepressants in posttraumatic stress disorder: a systematic review. Prog Neuropsychopharmacol Biol Psychiatry. 2009;33(2):169–180. doi:10.1016/j.pnpbp.2008.12.004 PubMed
32. Bajor LA, Ticlea AN, Osser DN. The Psychopharmacology Algorithm Project at the Harvard South Shore Program: an update on posttraumatic stress disorder. Harv Rev Psychiatry. 2011;19(5):240–258. doi:10.3109/10673229.2011.614483 PubMed
33. Miller LJ. Prazosin for the treatment of posttraumatic stress disorder sleep disturbances. Pharmacotherapy. 2008;28(5):656–666. doi:10.1592/phco.28.5.656 PubMed
34. Nappi CM, Drummond SPA, Hall JMH. Treating nightmares and insomnia in posttraumatic stress disorder: a review of current evidence. Neuropharmacology. 2012;62(2):576–585. doi:10.1016/j.neuropharm.2011.02.029 PubMed
35. Kung S, Espinel Z, Lapid MI. Treatment of nightmares with prazosin: a systematic review. Mayo Clin Proc. 2012;87(9):890–900. doi:10.1016/j.mayocp.2012.05.015 PubMed
36. Koola MM, Varghese SP, Fawcett JA. High-dose prazosin for the treatment of posttraumatic stress disorder. Ther Adv Psychopharmacol. 2014;4(1):43–47. doi:10.1177/2045125313500982 PubMed
37. Sterne JA, Gavaghan D, Egger M. Publication and related bias in meta-analysis: power of statistical tests and prevalence in the literature. J Clin Epidemiol. 2000;53(11):1119–1129. doi:10.1016/S0895-4356(00)00242-0 PubMed
Enjoy this premium PDF as part of your membership benefits!

