Aripiprazole-Induced Gray Matter Growth in a Patient With Major Depressive Disorder With Panic Disorder
To the Editor: Aripiprazole, a D2 partial agonist antipsychotic, was recently approved by the US Food and Drug Administration for major depressive disorder (MDD) add-on therapy. Its effects are believed to be related to its 5-HT1A partial agonist, 5-HT2C antagonist, and 5-HT2A antagonist properties.1 Here, I present a case of MDD with panic disorder in which improvement of symptoms was seen after 6 weeks of aripiprazole treatment. Magnetic resonance imaging (MRI) structural analysis also revealed gray matter growth and brain volume increase within 6 weeks.
Case report. Ms A is a 39-year-old female patient with DSM-IV-diagnosed single-episode MDD with panic disorder for 2 years (in March 2007, Hamilton Depression Rating Scale [HDRS]2 score = 44; Panic Disorder Severity Scale [PDSS]3 score = 23). She received several kinds of antidepressant therapy, such as fluoxetine, paroxetine, venlafaxine, and escitalopram, each for 4 to 5 months, without much improvement (lowest HDRS score = 38, lowest PDSS score = 21). She also could not tolerate the nausea, vomiting, sedation, body weight gain, muscle weakness, fatigue, and irritability side effects of these antidepressants. No specific physical illness, psychotic features, past manic episodes, nonpsychiatric medication use, or substance abuse were mentioned.
In consideration of Ms A’s tendency to experience side effects, in April 2009 treatment was started with aripiprazole 5 mg/d initially, with abrupt switching from escitalopram 20 mg/d (in April 2009, HDRS score = 41; PDSS score = 22), and titrated to 10 mg/d within 2 weeks with no significant side effects except mild excitement. After 6 weeks of aripiprazole therapy, her MDD and panic disorder symptoms improved (HDRS score = 25, PDSS score = 12).
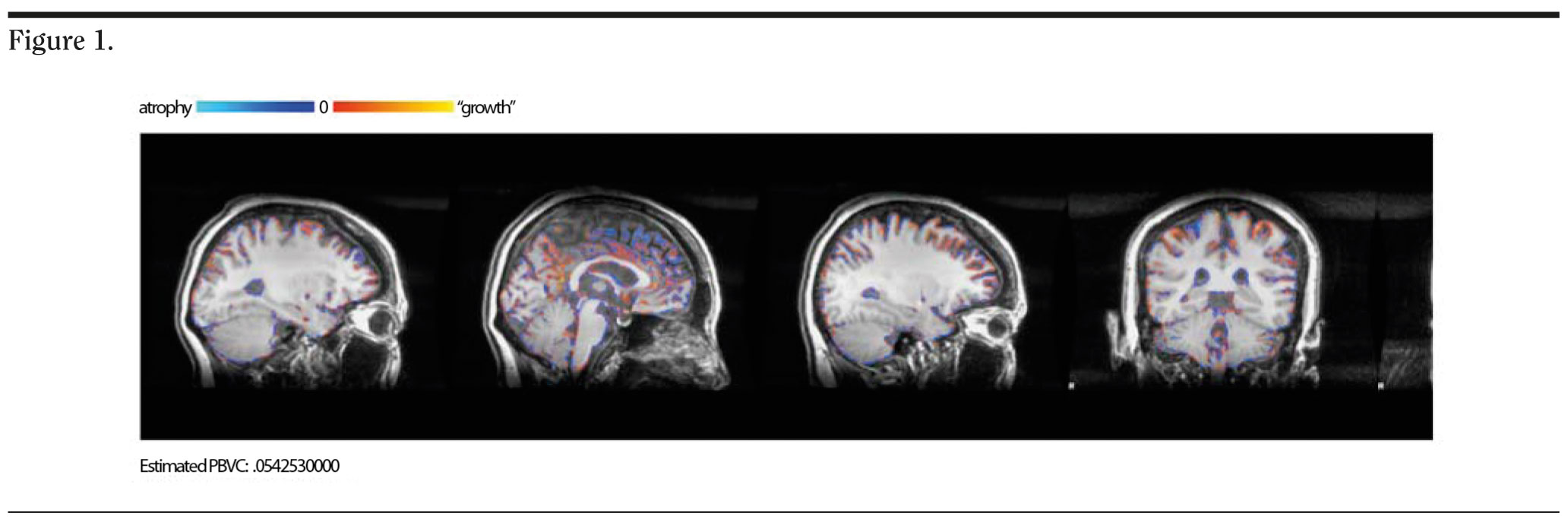
Structural brain magnetic resonance imaging (MRI) scans were obtained with a 3T scanner (GE Healthcare, Taipei City, Taiwan) housed at Buddhist Tzu-Chi Hospital, Taipei Branch, Taiwan. Scans with 3-dimensional fast spoiled gradient-echo recovery (3D-FSPGR) T1W1 (repetition time [TR] = 11.2 msec, echo time [TE] = 5.2 msec, matrix = 256 × 256, field of view = 260 mm, number of excitation = 1, slice thickness = 1 mm, 180 slices, no gap) were performed at the first visit and the 6-week visit. Ms A’s body weight was 69.5 kg at baseline and 69.8 kg at the sixth week. Her hydration status remained similar, without significant differences in water or food intake between the 2 visits. Structural MRI was preprocessed with the structural image evaluation, using normalization, of atrophy, cross sectional version (SIENAX) function of FSL (FMRIB Software Library, Oxford, United Kingdom) to calculate single time point gray matter, white matter, and brain volume after registering and normalizing to template. The brain morphology change was estimated by SIENA function to calculate percentage brain volume change.
After analyzing MRI brain structure by SIENAX, we found that gray matter had increased from 674455.53 mm3 to 720632.73 mm3 within 6 weeks, and the percentage brain volume change was 0.054253 mm3, which suggested that Ms A’s brain volume increased after aripiprazole treatment (Figure 1).
MDD and panic disorder have been reported to be associated with gray matter deficits in the amygdala, hippocampus, prefrontal cortex, and anterior cingulate cortex in past brain structure analysis reports.4,5 These regional gray matter deficits are related to symptom severity and even represent a kind of "trait marker" for these diseases. The relationship between gray matter and antipsychotics has been discussed in the recent 2 decades. Some second-generation antipsychotics, such as olanzapine, risperidone, and clozapine, were reported to increase gray matter volume or prevent gray matter atrophy in patients with schizophrenia within 1 to 2 years.6,7 Also, recent time-lapse mapping analysis of brain structure in patients with schizophrenia suggested that those in an olanzapine treatment group would have less frontoparietal gray matter volume reduction than those in a haloperidol treatment group.8
Reasons for gray matter growth effects might include the following: synaptic remodeling and neurogenesis9 from the stimulation of neurotrophic factors by antipsychotics10; prevention of oxidative stress or 6-OH-dopamine lesioning, with subsequent increased glial cell proliferation in frontal cortex11; and modulation of glutamate receptor function.12 These results were mostly based on schizophrenia or first-episode psychotic patients. Therapy duration was usually more than 12 weeks to 1 year.
In the present case, a patient with MDD and panic disorder received aripiprazole treatment and had response of symptoms and brain morphology change within 6 weeks. I hypothesize that aripiprazole’s D2 partial agonism might help the dopamine system stabilize and modulate neuronal activity through an inverted-U relationship.13 Serotonin systems might stabilize as a result of the 5-HT1A partial agonism and 5-HT2C antagonism effects of aripiprazole. Serotonergic and dopaminergic actions of aripiprazole might help MDD and panic disorder symptoms improve. A role for aripiprazole as adjunctive therapy is already established for atypical or anxious MDD,14 especially with antidepressants with serotonergic action.15 An effect of aripiprazole as augmentation therapy in panic disorder is suggested,16 but more randomized and well-controlled studies are needed to confirm the therapeutic effect.
Regarding the gray matter growth phenomenon, aripiprazole’s D2 partial agonism and 5-HT2A and 5-HT2C antagonism might produce gray matter growth that is similar to other atypical antipsychotics’ effects in patients with schizophrenia. Aripiprazole was reported to modulate glutamate transporter gene expression17 and promote brain-derived neurotrophic factor activity within 48 hours,18 which might contribute to gray matter growth or brain volume increase. The relatively short duration (48 hours) of aripiprazole treatment in the animal model17,18 might explain this patient’s 6-week findings of brain structure.
Apart from neurotransmitter theory, glial cell metabolic activity might also be relevant. A clozapine neuroimaging study showed that brain would express different metabolic activity patterns with clozapine than with haloperidol in patients with schizophrenia.19 Different brain metabolic activity with aripiprazole treatment was seen in healthy volunteers who had the dopamine D2 receptor polymorphism versus those who did not have this polymorphism.20 When the modulating phenomenon of glial metabolism is combined with "neuron-glia" coupling activity seen in positron emission tomography21 and glial cell proliferation evidence,11 we can hypothesize that aripiprazole would modulate the brain glial cell activity of this patient and relate to the gray matter growth results.
To my knowledge, this is the first report of aripiprazole monotherapy in MDD with panic disorder. Neuroimaging evidence might help us explain the therapeutic effect of aripiprazole in MDD with panic disorder. Hopefully, this preliminary report will be confirmed by further research on aripiprazole monotherapy in MDD with panic disorder.
References
1. Pae CU, Serretti A, Patkar AA, et al. Aripiprazole in the treatment of depressive and anxiety disorders: a review of current evidence. CNS Drugs. 2008;22(5):367-388. PubMed doi:10.2165/00023210-200822050-00002
2. Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23:56-62.
3. Shear MK, Rucci P, Williams J, et al. Reliability and validity of the Panic Disorder Severity Scale: replication and extension. J Psychiatr Res. 2001;35(5):293-296.
4. Frodl TS, Koutsouleris N, Bottlender R, et al. Depression-related variation in brain morphology over 3 years: effects of stress? Arch Gen Psychiatry. 2008;65(10):1156-1165. PubMed doi:10.1001/archpsyc.65.10.1156
5. Uchida RR, Del-Ben CM, Busatto GF, et al. Regional gray matter abnormalities in panic disorder: a voxel-based morphometry study. Psychiatry Res. 2008;163(1):21-29. PubMed doi:10.1016/j.pscychresns.2007.04.015
6. Lieberman JA, Tollefson GD, Charles C, et al. HGDH Study Group. Antipsychotic drug effects on brain morphology in first-episode psychosis. Arch Gen Psychiatry. 2005;62(4):361-370. PubMed doi:10.1001/archpsyc.62.4.361
7. Molina V, Reig S, Sanz J, et al. Increase in gray matter and decrease in white matter volumes in the cortex during treatment with atypical neuroleptics in schizophrenia. Schizophr Res. 2005;80(1):61-71. PubMed doi:10.1016/j.schres.2005.07.031
8. Thompson PM, Bartzokis G, Hayashi KM, et al. HGDH Study Group. Time-lapse mapping of cortical changes in schizophrenia with different treatments. Cereb Cortex. 2009;19(5):1107-1123. PubMed doi:10.1093/cercor/bhn152
9. Konradi C, Heckers S. Antipsychotic drugs and neuroplasticity: insights into the treatment and neurobiology of schizophrenia. Biol Psychiatry. 2001;50(10):729-742. PubMed doi:10.1016/S0006-3223(01)01267-7
10. Parikh V, Khan MM, Mahadik SP. Olanzapine counteracts reduction of brain-derived neurotrophic factor and TrkB receptors in rat hippocampus produced by haloperidol. Neurosci Lett. 2004;356(2):135-139. PubMed doi:10.1016/jneulet.2003.10.07
11. Wang HD, Dunnavant FD, Jarman T, et al. Effects of antipsychotic drugs on neurogenesis in the forebrain of the adult rat. Neuropsychopharmacology. 2004;29(7):1230-1238. PubMed doi:10.1038/sj.npp.1300449
12. Duncan GE, Miyamoto S, Leipzig JN, et al. Comparison of the effects of clozapine, risperidone, and olanzapine on ketamine-induced alterations in regional brain metabolism. J Pharmacol Exp Ther. 2000;293(1):8-14. PubMed
13. Seamans JK, Yang CR. The principal features and mechanisms of dopamine modulation in the prefrontal cortex. Prog Neurobiol. 2004;74(1):1-58. PubMed doi:10.1016/j.pneurobio.2004.05.006
14. Trivedi MH, Thase ME, Fava M, et al. Adjunctive aripiprazole in major depressive disorder: analysis of efficacy and safety in patients with anxious and atypical features. J Clin Psychiatry. 2008;69(12):1928-1936. PubMed doi:10.4088/JCP.v69n1211
15. Bourin M, Chenu F, Prica C, et al. Augmentation effect of combination therapy of aripiprazole and antidepressants on forced swimming test in mice. Psychopharmacology (Berl). 2009;206(1):97-107.
16. Hoge EA, Worthington JJ 3rd, Kaufman RE, et al. Aripiprazole as augmentation treatment of refractory generalized anxiety disorder and panic disorder. CNS Spectr. 2008;13(6):522-527. PubMed
17. Segnitz N, Schmitt A, Gebicke-Harter PJ, et al. Differential expression of glutamate transporter genes after chronic oral treatment with aripiprazole in rats. Neurochem Int. 2009;55(7):619-628.
18. Park SW, Lee JG, Ha EK, et al. Differential effects of aripiprazole and haloperidol on BDNF-mediated signal changes in SH-SY5Y cells. Eur Neuropsychopharmacol. 2009;19(5):356-362. PubMed doi:10.1016/j.euroneuro.2008.12.012
19. Molina V, Gispert JD, Reig S, et al. Cerebral metabolic changes induced by clozapine in schizophrenia and related to clinical improvement. Psychopharmacology (Berl). 2005;178(1):17-26. PubMed doi:10.1007/s00213-004-1981-9
20. Kim E, Kwon JS, Shin YW, et al. Taq1A polymorphism in the dopamine D2 receptor gene predicts brain metabolic response to aripiprazole in healthy male volunteers. Pharmacogenet Genomics. 2008;18(2):91-97. PubMed doi:10.1097/FPC.0b013e3282f3ef8c
21. Magistretti PJ. Cellular bases of functional brain imaging: insights from neuron-glia metabolic coupling. Brain Res. 2000;886(1-2):108-112. PubMed doi:10.1016/S0006-8993(00)02945-0
Author affiliations: Department of Psychiatry, Buddhist Tzu-Chi General Hospital, Taipei Branch; and Institute of Brain Science, National Yang Ming University, Taipei City, Taiwan. Potential conflicts of interest: None reported. Funding/support: Buddhist Tzu-Chi General Hospital, Taipei Branch hospital project TCRD-TPE-97-02. Acknowledgment: The author thanks Dr Yuan-Yu Hsu (Department of Medical Imaging, Buddhist Tzu-Chi General Hospital, Taipei Branch) for MRI acquisition help and technical assistance.
doi:10.4088/JCP.09l05459gre
© Copyright 2010 Physicians Postgraduate Press, Inc.