Objective: Sleep disturbances are a feature of attention-deficit/hyperactivity disorder (ADHD) and an adverse event (AE) of methylphenidate treatment. The authors sought to clarify methylphenidate-associated sleep problems and how studies are affected by confounding factors.
Data Sources: Published studies in English collected via online databases and unpublished data from www.clinicaltrials.gov and US Food and Drug Administration websites. Sources were searched from inception to August 2017.
Study Selection: Included were blinded placebo-controlled studies of youth with ADHD conducted in naturalistic settings, leading to 35 studies yielding 75 observations of sleep-related AEs. These studies comprised 3,079 drug-exposed and 2,606 placebo-treated patients.
Data Extraction: Two PhD-level reviewers reviewed each study for inclusion. Four PhD/PharmD-level reviewers extracted data in duplicate. Discrepancies were resolved by discussion or, if needed, by the senior author.
Results: Increased pooled relative risks (RRs) were found for methylphenidate-associated sleep-related AEs for insomnia (general), initial insomnia, middle insomnia, combined insomnia, and sleep disorder. Several sample or study design features were significantly associated with the RR for sleep-related AEs and the methylphenidate formulation studied (P < .05). After correction for confounding variables, significant differences among drugs were found for initial insomnia, insomnia (general), and sleep disorder (P < .0001) as the other categories could not be tested due to insufficient studies. The findings also show that the RR and its interpretation are constrained by the placebo AE rate.
Conclusions: Several types of insomnia and sleep problems are associated with methylphenidate treatment. Study design and sample features influence the RR statistic. By showing that the rate of placebo AEs impacts the RR, this study provides the field with a useful covariate for adjusting RR statistics.
This work may not be copied, distributed, displayed, published, reproduced, transmitted, modified, posted, sold, licensed, or used for commercial purposes. By downloading this file, you are agreeing to the publisher’s Terms & Conditions.


ABSTRACT
Objective: Sleep disturbances are a feature of attention-deficit/hyperactivity disorder (ADHD) and an adverse event (AE) of methylphenidate treatment. The authors sought to clarify methylphenidate-associated sleep problems and how studies are affected by confounding factors.
Data Sources: Published studies in English collected via online databases and unpublished data from www.clinicaltrials.gov and US Food and Drug Administration websites. Sources were searched from inception to August 2017.
Study Selection: Included were blinded placebo-controlled studies of youth with ADHD conducted in naturalistic settings, leading to 35 studies yielding 75 observations of sleep-related AEs. These studies comprised 3,079 drug-exposed and 2,606 placebo-treated patients.
Data Extraction: Two PhD-level reviewers reviewed each study for inclusion. Four PhD/PharmD-level reviewers extracted data in duplicate. Discrepancies were resolved by discussion or, if needed, by the senior author.
Results: Increased pooled relative risks (RRs) were found for methylphenidate-associated sleep-related AEs for insomnia (general), initial insomnia, middle insomnia, combined insomnia, and sleep disorder. Several sample or study design features were significantly associated with the RR for sleep-related AEs and the methylphenidate formulation studied (P < .05). After correction for confounding variables, significant differences among drugs were found for initial insomnia, insomnia (general), and sleep disorder (P < .0001) as the other categories could not be tested due to insufficient studies. The findings also show that the RR and its interpretation are constrained by the placebo AE rate.
Conclusions: Several types of insomnia and sleep problems are associated with methylphenidate treatment. Study design and sample features influence the RR statistic. By showing that the rate of placebo AEs impacts the RR, this study provides the field with a useful covariate for adjusting RR statistics.
J Clin Psychiatry 2019;80(3):18r12210
To cite: Faraone SV, Po MD, Komolova M, et al. Sleep-associated adverse events during methylphenidate treatment of attention-deficit/hyperactivity disorder: a meta-analysis. J Clin Psychiatry. 2019;80(3):18r12210.
To share: https://doi.org/10.4088/JCP.18r12210
© Copyright 2019 Physicians Postgraduate Press, Inc.
aDepartments of Psychiatry and of Neuroscience and Physiology, SUNY Upstate Medical University, Syracuse, New York
bHighland Therapeutics Inc, Toronto, Ontario, Canada
cCenter for Innovation in Mental Health, Academic Unit of Psychology; University of Southampton, Southampton, United Kingdom
dClinical and Experimental Sciences (CNS and Psychiatry), Faculty of Medicine, University of Southampton, Southampton, United Kingdom
eSolent NHS Trust, Southampton, United Kingdom
fNew York University Child Study Center, New York, New York
gDivision of Psychiatry and Applied Psychology, School of Medicine, University of Nottingham, Nottingham, United Kingdom
*Corresponding author: Stephen V. Faraone, PhD, SUNY Upstate Medical University, 750 East Adams St, Syracuse, NY 13210 ([email protected]).
Methylphenidate has been, for decades, a first-line treatment for attention-deficit/hyperactivity disorder (ADHD), with its relatively high efficacy having been confirmed in numerous randomized controlled clinical trials (RCTs) and meta-analyses.1,2 Currently, when physicians choose among methylphenidate products, they very likely consider duration of action and whether a special route of administration might be preferable (eg, transdermal patch, liquid, pill). There are, however, no definitive comparative data about efficacy or tolerability that would lead physicians to favor one formulation of methylphenidate over another.
Sleep problems are associated with ADHD regardless of the medication status. In a meta-analysis, Cortese et al3 assessed subjective and objective sleep parameters across 16 studies of nonmedicated children with ADHD. For subjective parameters (ie, based on questionnaires), they found youth with ADHD to have significantly higher rates of bedtime resistance, sleep onset difficulties, night awakenings, morning awakenings, sleep-disordered breathing, and daytime sleepiness compared to non-ADHD controls. For objective parameters, they found significantly greater sleep onset latency on actigraphy and the Apnea-Hypopnea Index, more stage shifts, significantly lower sleep efficiency on polysomnography and true sleep time on actigraphy, and significantly shorter times to fall asleep on the Multiple Sleep Latency Test. However, clinical practice suggests that, beyond the effect of ADHD per se, methylphenidate may also contribute to sleep problems, at least in some patients with ADHD.
Although the impact of methylphenidate on sleep-related adverse events (AEs) has been sufficiently established to be noted in the US Food and Drug Administration (FDA) labels for methylphenidate products, to our knowledge, there is no meta-analytic evidence aimed at quantifying the magnitude of this effect. Furthermore, it is unclear which type of sleep problem (insomnia [general]; initial, middle, and/or terminal insomnia; sleep disorder) is specifically associated with methylphenidate use.4 Both the size of the effect and the type of sleep problems are important elements to consider in daily clinical practice when prescribers and patients balance the benefits and the risk associated with methylphenidate.
Additionally, from a research standpoint, the factors that may potentially confound the risk of sleep-related AEs in studies comparing methylphenidate and placebo rates of AEs remain to be elucidated. Gaining insight into these possible confounders is crucial to determine if comparisons of the rates of sleep-related AEs across studies of different methylphenidate formulations are valid.
To address these clinical needs and research gaps, we conducted a systematic review with meta-analysis of RCTs of methylphenidate reporting sleep-related AEs, with the aims to (1) determine the study design-related confounders that significantly impact the relative risk (RR), (2) determine the magnitude of the pooled RR of sleep-related AEs adjusted for confounding variables, and (3) assess if this risk varies according to the type of sleep problem and the methylphenidate formulation studied.
METHODS
Literature Search
We conducted a systematic review and meta-analysis following the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement.5 We included original studies that (1) were double- or triple-blind, randomized, placebo-controlled trials with either parallel or crossover designs; (2) enrolled children and/or adolescents (aged 6 to 18 years inclusive) with a diagnosis of ADHD according to Diagnostic and Statistical Manual of Mental Disorders (DSM), Third Edition or subsequent editions, or hyperkinetic disorders according to the International Classification of Diseases and Related Health Problems (ICD), Ninth Revision or subsequent revisions; (3) investigated the efficacy and/or safety of methylphenidate formulations approved or under consideration by the FDA; (4) were conducted in a clinical/naturalistic setting; (5) reported on sleep as determined by spontaneously reported or solicited/elicited AEs (ie, using direct questioning on sleep, rating scale/questionnaire, sleep diary/log, checklist, etc); and (6) were written in English.

- Clinicians should systematically explore sleep issues before starting treatment with methylphenidate and also at each follow-up visit.
- Clinicians should specifically explore initial, middle, and combined insomnia as well as sleep quality and the restorative value of sleep.
- When comparing absolute rates of sleep-related adverse events from different studies, one should keep in mind that comparisons of relative risk statistics cannot be made when the rate of placebo-related adverse events differs between studies.
Studies were collected via PubMed (MEDLINE), Ovid (PsycINFO, EMBASE + EMBASE Classic, Ovid MEDLINE), and Web of Knowledge (Web of Science [Science Citation Index Expanded], Biological Abstracts, BIOSIS, Food Science and Technology Abstracts) databases from start until August 2017 (all searched on August 18, 2017). We also searched for unpublished data on www.clinicaltrials.gov and FDA websites (ie, Medical/Clinical Review and Label documents). The reference sections of eligible full-text reports and relevant reviews were manually searched for more studies. Supplementary Appendix 1 lists the search strategy and syntax for each database.
Screening, Identification of Eligible Records, and Data Extraction
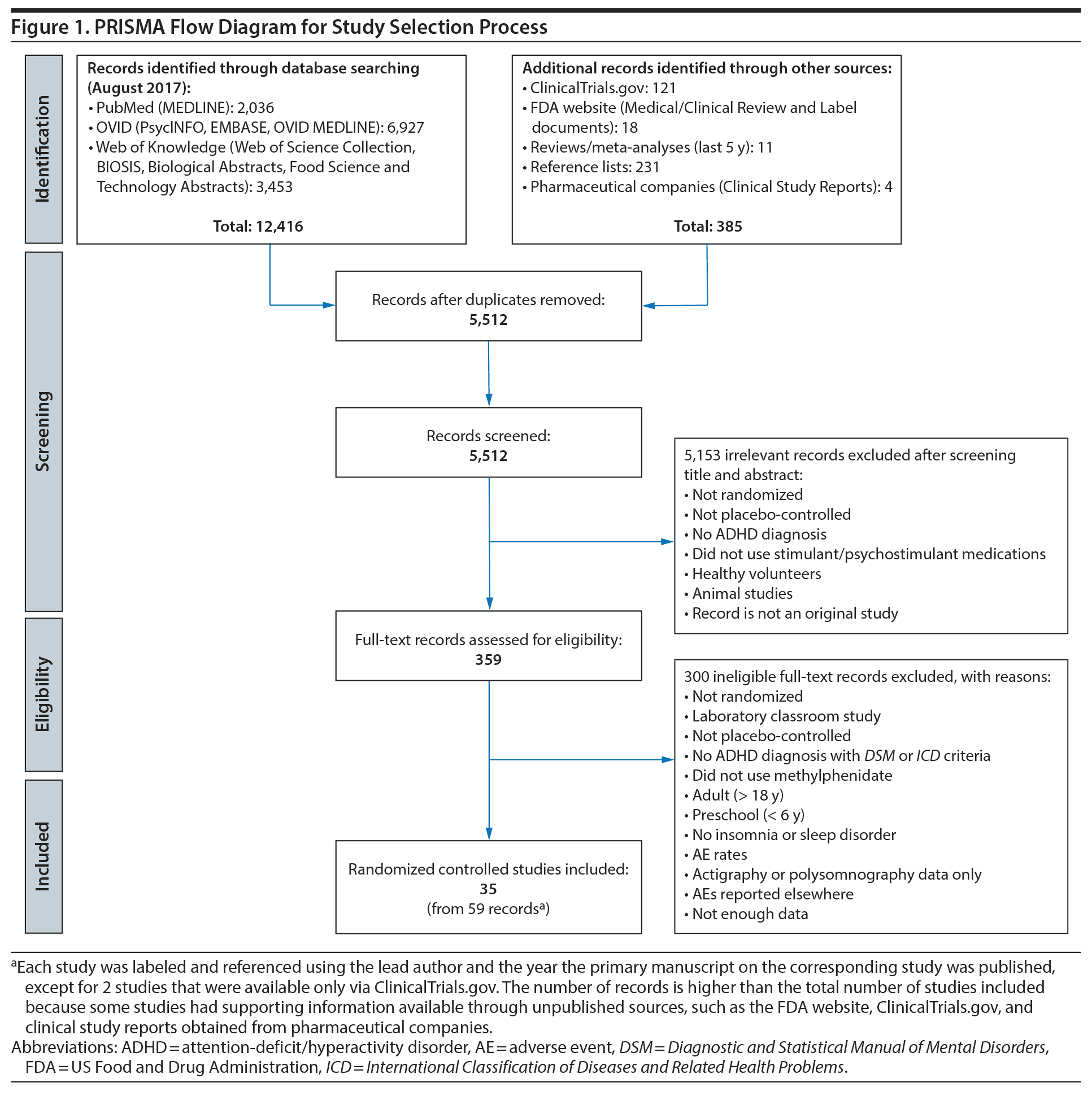
All identified records were exported into EndNote reference management software. Figure 1 shows the study selection process. The titles and abstracts of all non-duplicated records were screened, and full-text versions of relevant records were acquired. Two PhD-trained reviewers (M.D.P. and M.K.) independently assessed and reviewed each record for eligibility. All discrepancies were resolved through discussion until a consensus was reached. When a consensus was not reached, a senior author (S.V.F.) decided. Working in groups of two, 4 PhD/PharmD-trained reviewers (M.D.P.; M.K.; and S. Curtiss, PharmD, and Z. Henney, PharmD, Ironshore Pharmaceuticals & Development, Inc) extracted data independently from the selected records in duplicate. After completion of data extraction, any discrepancies in data sets were resolved through discussion until a consensus was reached. When a consensus was not reached, a senior author (S.V.F.) acted as an arbitrator.
Sleep AE Definitions
The studies selected used 2 methods to define sleep AEs: Type 1 studies recorded spontaneous reports with the preferred terms defined by the Medical Dictionary for Regulatory Activities (MedDRA) or Coding Symbols for a Thesaurus of Adverse Reaction Terms (COSTART), and Type 2 studies used rating scales or questionnaires to elicit AEs. The AEs from Type 1 studies were classified using these MedDRA codes: 1 = insomnia (general), 2 = initial insomnia, 3 = middle insomnia, 4 = terminal insomnia, 5 = combined insomnia (ie, when the participant endorsed at least 1 type of insomnia measured by the study), 6 = early morning awakening, and 7 = sleep disorder (as a general term encompassing any sleep-related complaints). We recoded “early morning awakening” as “terminal insomnia.” Some studies coded spontaneously reported sleep-related AEs using individual preferred terms for multiple distinct and specific sleep issues (eg, initial insomnia, middle insomnia, terminal insomnia), whereas others collapsed these individual preferred terms into a single combined insomnia term. Another approach was to simply use the insomnia preferred term for general insomnia without indicating or determining distinct subcategories for specific sleep issues. Per the MedDRA system, we coded these as insomnia. Whenever individual preferred terms were provided for distinct sleep-related AEs, these were coded individually, and whenever sleep-related AEs were coded separately but collapsed, they were coded as combined insomnia. However, it is possible that some studies coded multiple distinct sleep-related issues as insomnia (general) or sleep disorder but did not describe that clearly in their Methods sections.
Type 2 studies used a rating scale cut-point to define clinically significant problems, which defined the proportion of research participants experiencing each event. We coded these events using MedDRA codes with a few modifications. We used code 1 for exceeding the clinical cut-point on ratings of insomnia, code 2 for exceeding the clinical cut-point on ratings of sleep onset latency, code 3 for exceeding the clinical cut-point on ratings of awakening during the night, and code 7 for exceeding the clinical cut-point on ratings of severe sleep issues. We added code 8 for exceeding the clinical cut-point on ratings of sleep duration. We excluded 2 studies that used a global sleep rating not convertible to a MedDRA category. Supplementary Table 1 gives the details of how each study assessed sleep AEs.
Potentially Confounding Variables
From each study we extracted the following covariates, if available: type of AE reporting (spontaneous or elicited), year of study, number randomized to drug and placebo groups, mean age, percentage of males in sample, percent of treatment-naive patients in sample, frequency of dosing (1, 2 or 3 times each day), treatment duration (days), number of study periods, number of sites, percent of white participants, exclusions for psychiatric comorbidity, diagnostic system used, percent of sample previously treated with stimulants, percent of sample previously treated with nonstimulants, release type of drug (immediate vs long-acting), FDA registration (yes/no), study sponsor (government, foundation, industry), study design (parallel, crossover), and nature of the blind (double vs triple). We also classified studies into 4 dosing strategy categories based on 2 variables: (1) whether the dose had been stabilized or optimized prior to the study and (2) whether forced titration was used during the study period.
Statistical Analyses
Our meta-analysis used the random-effects model of DerSimonian and Laird,6 which computes a pooled RR weighted by sample size. We used the Egger method7 to assess for publication biases. Some research groups contributed more than one data set to the meta-analysis. Because measures reported from the same research group may not be statistically independent of one another, standard statistical procedures will produce inaccurate P values. To address this intrastudy clustering, variance estimates for the meta-analysis regression were adjusted using the Huber formula8 as implemented in STATA.9 This formula is a “theoretical bootstrap” that produces robust statistical tests. The method works by entering the cluster scores (ie, sum of scores within families) into the formula for the estimate of variance. The Huber estimate is also called the “sandwich” estimate because it is calculated as the product of 3 matrices: the matrix formed by taking the outer product of the observation-level likelihood score vectors is in the middle, and this matrix is pre- and post-multiplied by the usual model-based variance matrix. The resulting P values are valid even when observations are not statistically independent. The meta-analytic regressions were weighted by the reciprocal of the variance of the effect size. Because the RRs were markedly skewed, we used the log-transformed RR in all regression analyses.
To determine if sample and study design features confounded the association of drugs with sleep-related AEs, we conducted 2 sets of meta-analytic regression. One set assessed the association of each feature with the drug studied. The other assessed the association of each feature with the RR for sleep-related AEs. We defined a variable as a confounder if it was associated with both drug name and the RR for sleep-related AEs. To adjust for these confounding variables, we executed the following steps for each drug (referred to as the target drug) separately: (1) in the subset of data that excludes the target drug, estimate a multivariate model predicting the log-transformed RR from all confounders; (2) for the target drug only, use the estimated model from step 1 to predict the log-transformed RR; and (3) compute the adjusted log-transformed RR as the actual RR minus the log-transformed RR adjusted for confounding variables.
Because there were many missing data points and these were not missing at random (ie, some studies had many missing data points, others had fewer missing data points), we analyzed “missing” as a separate category for all variables. For categorical variables, we simply added a “missing” category. For continuous variables, we first created a categorical variable by partitioning them into quintiles and then added a “missing” category.
We also capitalized on the high correlation between drug and placebo AE rates to predict the RR from the placebo AE rate. To do this, we first transformed the drug and placebo rates using the logit transformation, which makes proportions suitable for linear regression. We then regressed the logit drug rate onto the logit placebo rate (z = 8.7, P < .001) and the square of that rate (z = 3.1, P = .002). We then used coefficients from the resulting regression equation to predict AE RRs from placebo rates in the range observed in the studies we reviewed. The equation was
RR = invlogit(1.45*logit(x) + 0.131*logit(x)*logit(x) + 1)/x
in which logit is the logit function and invlogit is the inverse logit function.
RESULTS
We kept 35 studies (see PRISMA diagram in Figure 1 for details) for the meta-analysis, yielding 75 observations of sleep-related AEs. There are more observations than studies because some studies reported more than one type of sleep AE or more than one method for collecting AEs. The 35 studies comprised 3,079 drug-exposed patients and 2,606 placebo-treated patients. After pooling data across all studies, we found significantly increased RRs for methylphenidate-associated sleep-related AEs for general insomnia (RR = 1.61, z = 9.2, P < .001), initial insomnia (RR = 2.78, z = 6.6, P < .001), middle insomnia (RR = 2.97, z = 183, P < .001), combined insomnia (RR = 2.97, z = 6.4, P < .001), and sleep disorder (RR = 1.99, z = 4.2, P < .001). All insomnia categories are MedDRA categories as described in the Methods section.
Assessment of Potentially Confounding Variables
To find potential confounding variables, we first tested if any sample or study design features significantly predicted the RR for sleep-related AEs. The following variables did not predict the RR for sleep-related AEs: ethnicity, exclusions for psychiatric comorbidity, enrollment of treatment-naive patients, use of crossover versus parallel design, number randomized, treatment duration, elicited versus spontaneous reporting of AEs, study sponsorship (industry vs National Institutes of Health vs other) and whether the study was FDA registered or not. The following variables were associated with both the relative risk for sleep AEs and the type of drug studied and hence were considered potential confounders: mean age of sample, percentage of males in sample, percentage of stimulant responders in sample, year of publication, number of study sites, use of stabilization/optimization during dosing period, rater of AEs, and use of rating scale to elicit AEs. See Supplementary Table 2 for details about these confounding variables.
A potential confounder that is not a design feature is the rate of AEs in the placebo group. This rate could be influenced by either sample features (eg, some types of patients with ADHD are more prone to insomnia) or design features (eliciting AEs will yield higher rates than relying on spontaneous AEs). Because the placebo AE rate could be influenced by these and other potential confounders, we tested it as a potential confounder and found it to be significantly associated with drug (χ2 = 21.0, P = .01) and with the RR for sleep-related AEs (z = −3.3, P = .001). When we added the placebo AE rate to the multivariate model, it significantly predicted the RR. In that model, age and year of publication lost statistical significance. When we then added drug to the multivariate confounder model, it significantly predicted the RR (χ29 = 49, P < .0001); all other covariates, except for age, year of publication, and dosing method, stayed significant (all P < .05).
In addition to these confounding variables, 2 correlated drug features were associated with both the RR and the drug studied. Extended-release formulations had higher RRs for sleep-related AEs than immediate-release formulations (4.8 vs 1.9, P = .03), and drugs that were dosed once each day had a higher mean RR (4.6, P < .0001) than those dosed twice (1.7) or 3 times (2.5) each day. We did not consider these to be confounders because they are features of the drugs being compared.
Meta-Analysis of Odds Ratios Adjusted for Confounders
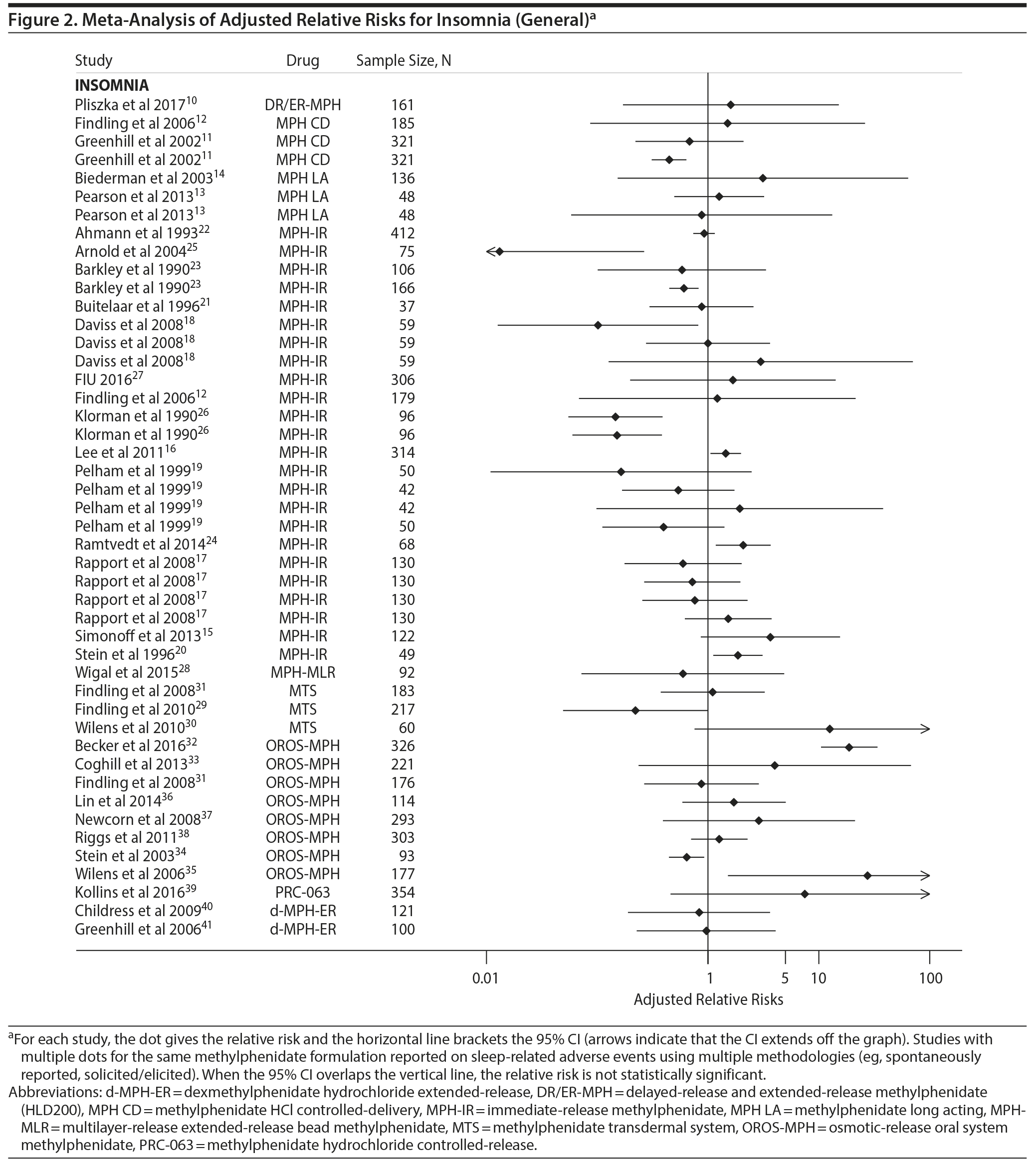
Figure 2 gives the adjusted RRs for the MedDRA insomnia (general) category. This category was used when it was not possible to determine the specific type of insomnia. Figure 2 shows a wide range of RRs with some significantly less than 1, many (> 75%) not significantly different from 1, and some significantly greater than 1. For the studies in Figure 2, we found no evidence of publication bias (t = 0.37, P = .7).
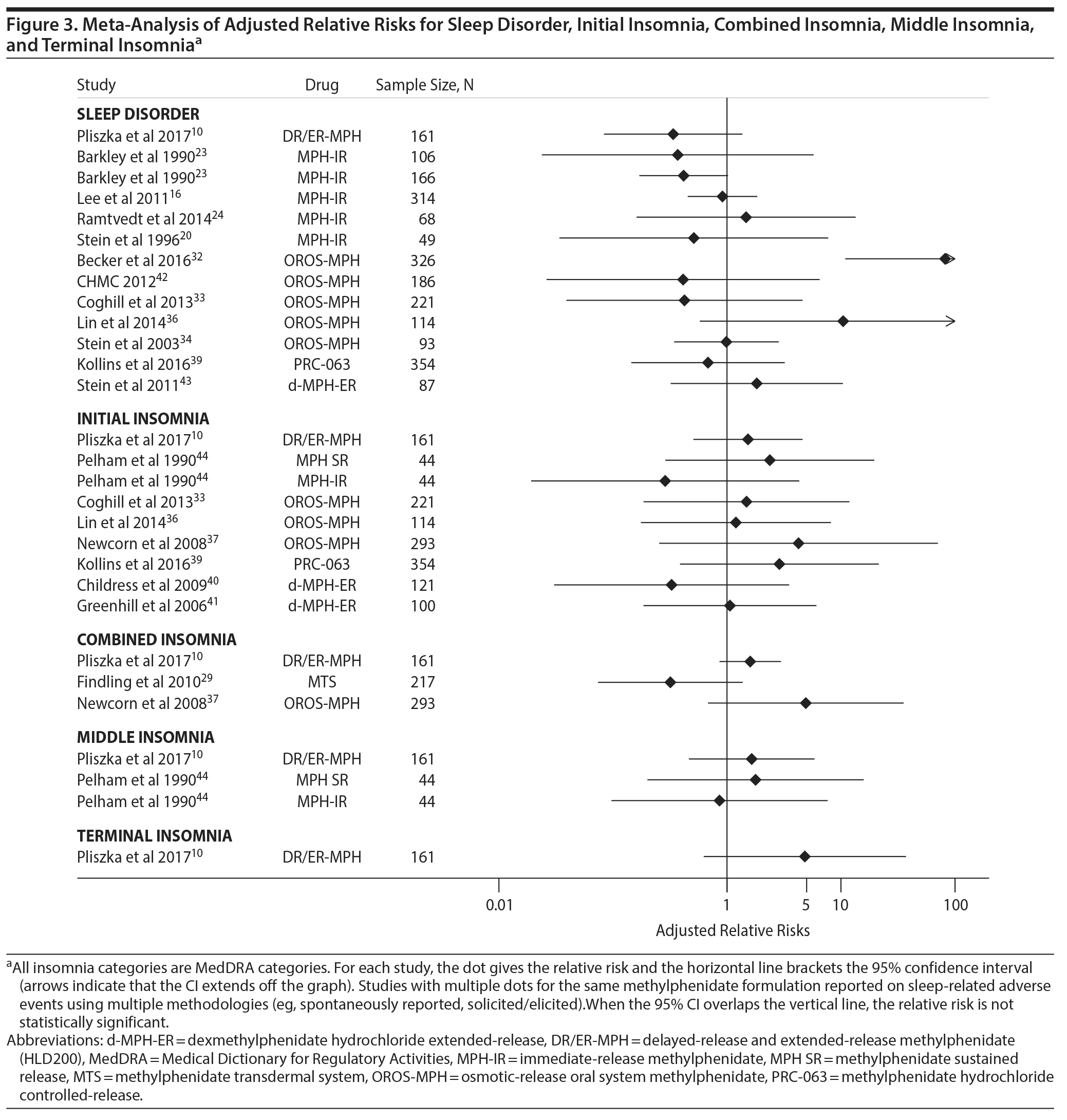
Figure 3 gives a similar plot for sleep disorder and for 3 MedDRA subtypes of insomnia (initial insomnia, middle insomnia, and terminal insomnia) and combined insomnia. All but 1 of the RRs in Figure 4 overlap with 1, indicating a nonsignificant effect. The term combined insomnia means that the study assessed at least 2 subtypes of insomnia and the participant endorsed at least 1 of these. By contrast, the MedDRA term insomnia (Figure 2) was used when subtypes of insomnia were not evaluated. For the studies in Figure 3, we found no evidence of publication bias (t = 0.37, P = .7).
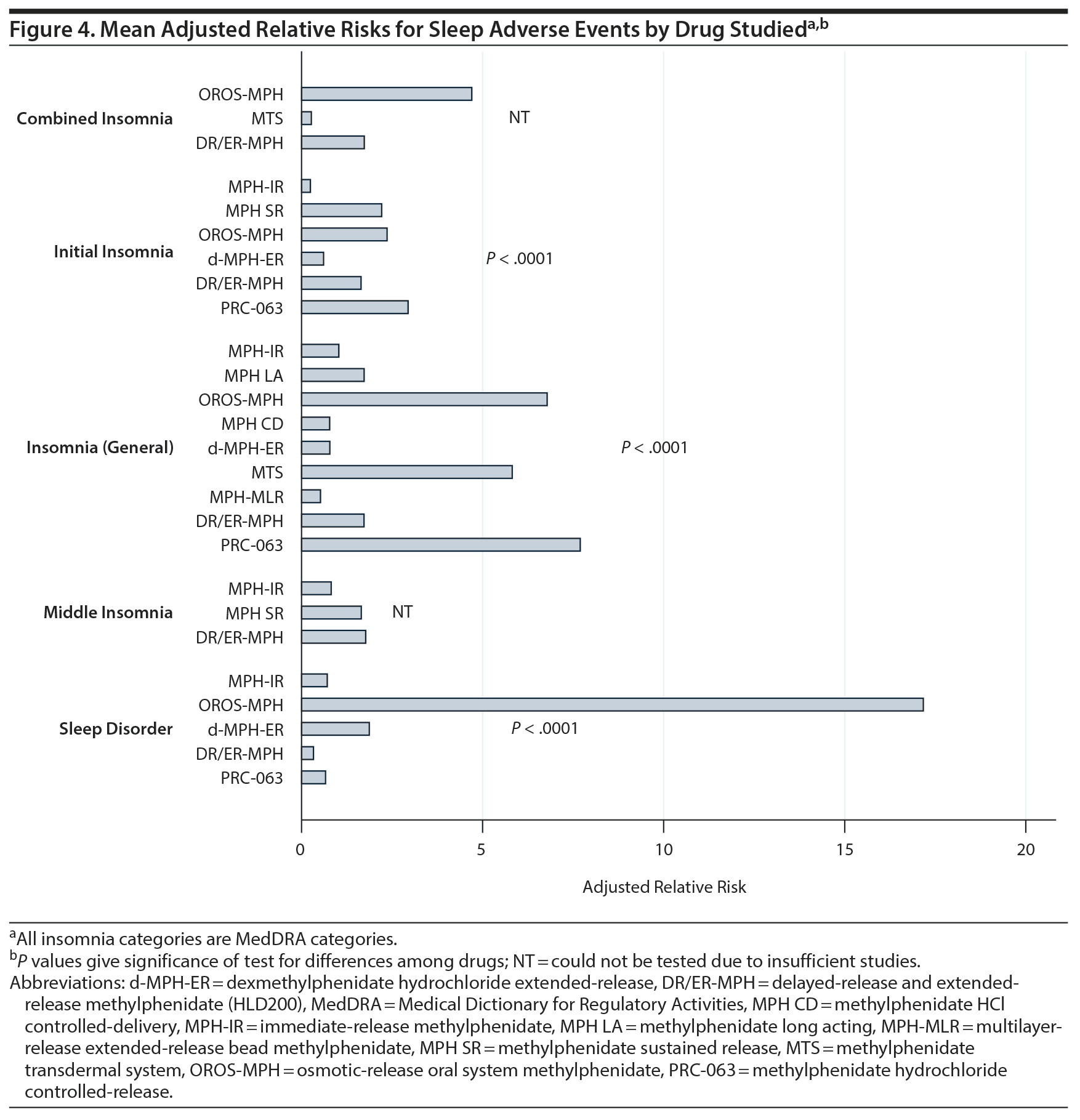
Figure 4 compares the mean adjusted RRs for all drugs and types of sleep-related AEs for AE categories studied by more than 1 drug. As shown in Figure 4, after correcting for confounding variables, we found significant differences among drugs for initial insomnia, insomnia, and sleep disorder. For initial insomnia and insomnia, the greatest RRs were seen for osmotic-release oral system methylphenidate (OROS-MPH), methylphenidate transdermal system (MTS), and methylphenidate hydrochloride controlled-release (PRC-063). For sleep disorder, OROS-MPH had a much larger RR compared with other drugs. There were too few studies to test drug differences for middle or combined insomnia.
The Impact of Placebo AE Rates on the RR Statistic
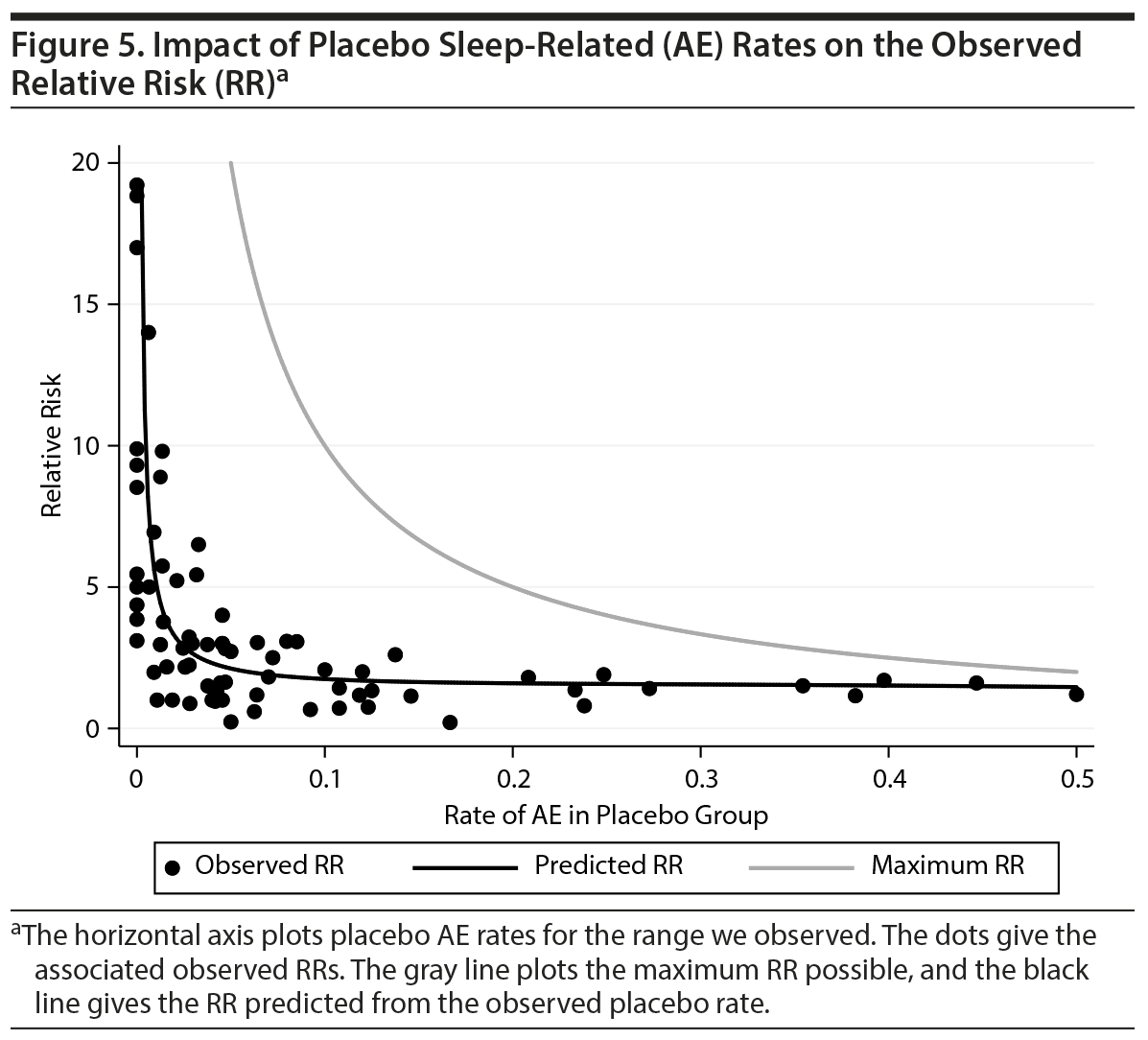
We conducted analyses to further investigate the relationship between the rate of AEs in the placebo group and the RR for these AEs. An obvious, yet vital, point is that the maximum possible RR is bounded by the fact that the rate of AEs in the drug group cannot exceed 1.0. So, for example, if the rate of placebo AEs is 0.50, the maximum possible RR is only 2.0. Less obvious is that drug AE rates and placebo AE rates are highly correlated (r = 0.89, P < .0001). This high correlation is relevant because it allows us to predict the RR by using the denominator for the observed rate of AEs in the placebo group and the numerator for the rate of AEs in the drug group predicted by the AE rate in the placebo group. Figure 5 shows that calculating RR is almost futile with methodologies that estimate high AE rates because their RRs maximize at a point close to the value predicted by the placebo AE rate. The figure also shows that all of the observed RRs are close to what is predicted by simply knowing the placebo AE rate.
DISCUSSION
This meta-analysis is the first to focus on the rates of sleep-related AEs in RCTs of methylphenidate. Our findings, based on literature comprising 35 randomized controlled trials yielding 75 different observations of sleep-related AEs, have important clinical and research implications. We found a significant impact of methylphenidate on sleep-related AEs. Another important finding was that several study design and sample features predicted these RRs and confounded comparisons of methylphenidate formulations.
In terms of the impact of methylphenidate on sleep, we found that its use was significantly associated with reports of several types of insomnia (initial, middle, terminal, combined). Thus, our findings suggest that prescribers focus the clinical interview not only on delays in sleep onset but also on middle and terminal insomnia and, more generally, sleep quality. Furthermore, our findings give pooled estimates of reported sleep-related AEs for each drug. When we compared the RR among drug types after adjusting for confounding variables, we found significant differences among drugs. As shown in Figure 4, the significant differences are due to relatively high RRs for OROS-MPH, MTS, and PRC-063 compared with other methylphenidate compounds. Sleep-related AEs are, however, only one of the several considerations physicians should keep in mind when choosing a methylphenidate formulation.
Another novel finding is that many study design and sample features, including type of formulation, number of doses per day, age, sex, percentage of stimulant responders enrolled, year of study, number of sites, type of rater, and use of rating scale, predicted both the RR for sleep AEs and the type of drug studied. Interpretation of the effects of confounders (Supplementary Table 2) warrants caution because it is likely that many of these confounders are correlated with one another. Our goal was not to understand the nature of these confounders but to adjust for them in our primary analyses.
Among these confounders, the one that is most relevant from a clinical standpoint is the finding that long-acting formulations affected sleep more than immediate-release formulations. This finding is probably due to patients’ taking their dose too late in the morning or to individual differences in pharmacokinetics, which extend the duration of action into the bedtime period. The high RR for once-a-day dosing reflects that long-acting formulations are typically dosed once per day.
Except for age, all of the potential confounders that we assessed stayed significant when we added drug type to the multivariate confounder model. This latter finding shows that these covariates add independent information to predicting RR above and beyond what can be predicted by type of drug. This observation has important implications for the interpretation of differences among methylphenidate formulations in terms of their impact on sleep. Indeed, one should not compare rates of sleep-related AEs among drugs across studies without taking such features into account. Studies that enroll a larger fraction of stimulant responders have lower RRs than other studies. That difference makes sense to the degree that the definition of “response” includes tolerability. Regarding other sample features, higher RRs were seen for very young samples, and lower RRs were seen for samples that were nearly all male.
Clinicians generated higher RRs compared with other raters. Because all studies that did not use rating scales had AEs evaluated by clinicians and all studies with nonclinical raters used rating scales, we cannot separate the effect of type of rater and whether a rating scale was used. That said, these results suggest that, compared with other raters using rating scales, clinician raters using a clinical interview without a rating scale produce higher RR for sleep-related AEs. This finding may simply be due to clinicians’ defining AEs conservatively, which would lead to overall low rates and a high relative risk. It is intriguing that RRs for sleep AEs have increased over time. Further work should assess if this increase is due to corresponding change in study design or sample characteristics. It is difficult to interpret the effects of forced dose titration and whether patients were stabilized or optimized prior to treatment. The reason is that these features were correlated and had to be analyzed jointly for confounder analyses. The finding of higher RRs after forced dose titration for patients that are neither stabilized nor optimized makes intuitive sense, but the results for other categories in Supplementary Table 1 suggest that the effects are more complex.
Our work is the first to assess the impact of the placebo AE rate on the RR statistic. The placebo AE rate was associated with both type of drug studied and the RR. It thereby confounds the comparison of RRs among drugs. We have also shown that the placebo AE rate places constraints on the RR. These constraints should be considered when comparing 2 drugs or 2 studies of the same drug. As Figure 5 shows, high placebo AE rates cannot lead to high RRs due to how the RR is defined. Of equal importance, the high correlation between placebo and drug AE rates places further constraints on the RR. As shown in Figure 5, the placebo response rate alone is a good predictor of the RRs seen in the studies we reviewed.
Taken together, Figure 5 cautions readers not to interpret RR reported in clinical trials without considering both the absolute risks and the constraints placed on the RR by that absolute risk. For example, a low RR may seem comforting, but as is clear from Figure 5, low RRs are associated with high absolute risks. Similarly, although a high RR may seem concerning, it is very likely due to a low absolute risk, which diminishes the degree of clinical concern. Also, of note from Figure 5, as the absolute risks approach 0.5, the size of the RR becomes less and less meaningful due to the low value of the maximum RR. In fact, in Figure 5, any study with a placebo AE rate greater than 0.4 has an RR very close to the largest possible RR. Because of these issues, comparisons of RRs between studies are not straightforward. In theory, the RR for sleep-related AEs should not be sensitive to study design and sample features in a blinded, randomized controlled design. The above considerations show that this theoretical position does not hold up to empirical scrutiny.
This conclusion is also corroborated by the work of Wernicke et al,45 who showed that eliciting AEs leads to higher absolute rates in both the drug and placebo groups versus spontaneous reporting of AEs, whereas spontaneous reporting results in larger drug-placebo RRs more often than solicitation. In our multivariate analyses, we showed that the drug studied, the placebo AE rate, and several covariates provided independent predictive information about the RR statistic. This finding means that the RR can be parsed into a part due to drug differences, a part due to specific covariates, a part due to unmeasured covariates for which the placebo AE rate is a proxy, and a part due to the constraints that the placebo AE rate places on the RR due to the mathematical definition of the RR and the empirical association between drug and placebo AE rates. Thus, it is essential for cross-study comparisons to focus on the RR adjusted for these components.
In clinical practice, clinicians should systematically explore sleep issues before starting a treatment with methylphenidate and at each follow-up visit. Our findings highlight that, rather than querying nonspecifically for “sleep problems,” clinicians should specifically explore initial, middle, terminal, and combined insomnia as well as sleep quality and the restorative value of sleep. These issues can be explored with simple unstructured questions or with user-friendly questionnaires such as the Sleep Disturbance Scale for Children46 or the Children’s Sleep Habits Questionnaire.47
Our work has implications for future clinical trial designs. Given that insomnia occurs with all methylphenidate products, studies would be more informative if they reported results for different classes of insomnia rather than lumping all types of insomnia under one nonspecific term. Insomnia and other sleep AEs should be routinely reported, and study reports should provide sufficient data for inclusion in future meta-analyses. Given the impact of prevalence on the RR (Figure 5), studies should report whether AEs were elicited or were based on spontaneous reports. No report should make cross-study comparisons of AE rates or RRs without taking into account sample and study design features. If cross-study comparisons are needed, the best current way to do so (lacking a formal meta-analysis) is to refer to Figure 5. Because the placebo AE rate summarizes the effects of study design and sample features, Figure 5 can be used to determine the expected RR from future studies.
Our conclusions are limited by methodological issues. Like all meta-analyses, our analyses of covariates were limited by the information provided in the reports we reviewed. There were also many studies screened from the initial pool reviewed that did not have sufficient data for inclusion (Figure 1). For studies that were included, data were not uniformly available for some covariates. Although we addressed this issue by categorizing continuous variables and estimating parameters for “missing” data, the issue may have limited our ability to find significant effects. It is, however, unlikely to have created spurious results. Another limitation is that we restricted the search to reports published in English, given practical constraints with the translations of reports in other languages.
Despite these limitations, our meta-analytic estimates of the association between sleep AEs and methylphenidate highlight that several types of insomnia and sleep problems, and not just initial insomnia, are reported as possible AEs of methylphenidate. We also provide new data about study design and sample features that influence the RR statistic. By showing that the rate of placebo AEs impacts the RR, we provide the field with a useful and typically nonmissing covariate for adjusting RR statistics. Although our work suggests that different methylphenidate formulations differ in their RR for sleep AEs, such information is only one of many considerations for selecting a methylphenidate formulation.
Submitted: February 22, 2018; accepted November 19, 2018.
Published online: April 30, 2019.
Potential conflicts of interest: In the past year, Dr Faraone received income, travel expenses, continuing education support, and/or research support and had the potential to receive income from Otsuka, Lundbeck, KenPharm, Rhodes, Arbor, Ironshore, Shire, Akili Interactive Laboratories, Alcobra, Enzymotec, Sunovion, Genomind. and NeuroLifeSciences. With his institution, he has US patent US20130217707 A1 for the use of sodium-hydrogen exchange inhibitors in the treatment of attention-deficit/hyperactivity disorder (ADHD). Dr Cortese reports receiving reimbursement for travel and accommodation expenses from the Association for Child and Adolescent Mental Health (ACAMH), a non-profit organization, in relation to lectures that he delivered for ACAMH. Drs Komolova and Po are employees of Highland Therapeutics Inc.
Funding/support: Dr Faraone is supported by the K. G. Jebsen Centre for Research on Neuropsychiatric Disorders, University of Bergen, Bergen, Norway; the European Union’s Seventh Framework Programme for Research, Technological Development, and Demonstration under grant agreement No. 602805; the European Union’s Horizon 2020 Research and Innovation Programme under grant agreements Nos. 667302 and 728018; and National Institute of Mental Health grants 5R01MH101519 and U01 MH109536-01. Funding for the development of this manuscript was provided by Ironshore Pharmaceuticals & Development, Inc, a subsidiary of Highland Therapeutics Inc.
Role of the sponsor: Stacey Curtiss, PharmD, and Zachary Henney, PharmD, from Ironshore Pharmaceuticals & Development, Inc, only assisted with data extraction so that all data extraction was done in duplicate. The funders had no other role in this study.
Supplementary material: Available at PSYCHIATRIST.COM.
REFERENCES
1. Faraone SV, Asherson P, Banaschewski T, et al. Attention-deficit/hyperactivity disorder. Nat Rev Dis Primers. 2015;1:15020. PubMed CrossRef
2. Faraone SV, Buitelaar J. Comparing the efficacy of stimulants for ADHD in children and adolescents using meta-analysis. Eur Child Adolesc Psychiatry. 2010;19(4):353-364. PubMed CrossRef
3. Cortese S, Faraone SV, Konofal E, et al. Sleep in children with attention-deficit/hyperactivity disorder: meta-analysis of subjective and objective studies. J Am Acad Child Adolesc Psychiatry. 2009;48(9):894-908. PubMed
4. Stein MA, Weiss M, Hlavaty L. ADHD treatments, sleep, and sleep problems: complex associations. Neurotherapeutics. 2012;9(3):509-517. PubMed CrossRef
5. Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ. 2009;339:b2700. PubMed CrossRef
6. DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7(3):177-188. PubMed CrossRef
7. Egger M, Davey Smith G, Schneider M, et al. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629-634. PubMed CrossRef
8. Huber PJ. The behavior of maximum likelihood estimates under non-standard conditions. Proceedings of the Fifth Berkeley Symposium on Mathematical Statistics and Probability, Vol 1. Berkeley, CA: University of California Press; 1967:221-233.
9. Stata User’s Guide: Release 9 [computer program]. College Station, TX: Stata Corp LP; 2005.
10. Pliszka SR, Wilens TE, Bostrom S, et al. Efficacy and safety of HLD200, delayed-release and extended-release methylphenidate, in children with attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2017;27(6):474-482. PubMed CrossRef
11. Greenhill LL, Findling RL, Swanson JM; ADHD Study Group. A double-blind, placebo-controlled study of modified-release methylphenidate in children with attention-deficit/hyperactivity disorder. Pediatrics. 2002;109(3):e39. PubMed CrossRef
12. Findling RL, Quinn D, Hatch SJ, et al. Comparison of the clinical efficacy of twice-daily Ritalin and once-daily Equasym XL with placebo in children with attention deficit/hyperactivity disorder. Eur Child Adolesc Psychiatry. 2006;15(8):450-459. PubMed CrossRef
13. Pearson DA, Santos CW, Aman MG, et al. Effects of extended release methylphenidate treatment on ratings of attention-deficit/hyperactivity disorder (ADHD) and associated behavior in children with autism spectrum disorders and ADHD symptoms. J Child Adolesc Psychopharmacol. 2013;23(5):337-351. PubMed CrossRef
14. Biederman J, Quinn D, Weiss M, et al. Efficacy and safety of Ritalin LA, a new, once-daily, extended-release dosage form of methylphenidate hydrochloride, in children with attention deficit hyperactivity disorder. Paediatr Drugs. 2003;5(12):833-841. PubMed CrossRef
15. Simonoff E, Taylor E, Baird G, et al. Randomized controlled double-blind trial of optimal dose methylphenidate in children and adolescents with severe attention deficit hyperactivity disorder and intellectual disability. J Child Psychol Psychiatry. 2013;54(5):527-535. PubMed CrossRef
16. Lee SH, Kim SW, Lee MG, et al. Lack of association between response of OROS-methylphenidate and norepinephrine transporter (SLC6A2) polymorphism in Korean ADHD. Psychiatry Res. 2011;186(2-3):338-344. PubMed CrossRef
17. Rapport MD, Kofler MJ, Coiro MM, et al. Unexpected effects of methylphenidate in attention-deficit/hyperactivity disorder reflect decreases in core/secondary symptoms and physical complaints common to all children. J Child Adolesc Psychopharmacol. 2008;18(3):237-247. PubMed CrossRef
18. Daviss WB, Patel NC, Robb AS, et al. Clonidine for attention-deficit/hyperactivity disorder, II: ECG changes and adverse events analysis. J Am Acad Child Adolesc Psychiatry. 2008;47(2):189-198. PubMed CrossRef
19. Pelham WE, Gnagy EM, Chronis AM, et al. A comparison of morning-only and morning/late afternoon Adderall to morning-only, twice-daily, and three times-daily methylphenidate in children with attention-deficit/hyperactivity disorder. Pediatrics. 1999;104(6):1300-1311. PubMed CrossRef
20. Stein MA, Blondis TA, Schnitzler ER, et al. Methylphenidate dosing: twice daily versus three times daily. Pediatrics. 1996;98(4 Pt 1):748-756. PubMed
21. Buitelaar JK, van der Gaag RJ, Swaab-Barneveld H, et al. Pindolol and methylphenidate in children with attention-deficit hyperactivity disorder. Clinical efficacy and side-effects. J Child Psychol Psychiatry. 1996;37(5):587-595. PubMed CrossRef
22. Ahmann PA, Waltonen SJ, Olson KA, et al. Placebo-controlled evaluation of Ritalin side effects. Pediatrics. 1993;91(6):1101-1106. PubMed
23. Barkley RA, McMurray MB, Edelbrock CS, et al. Side effects of methylphenidate in children with attention deficit hyperactivity disorder: a systemic, placebo-controlled evaluation. Pediatrics. 1990;86(2):184-192. PubMed
24. Ramtvedt BE, Aabech HS, Sundet K. Minimizing adverse events while maintaining clinical improvement in a pediatric attention-deficit/hyperactivity disorder crossover trial with dextroamphetamine and methylphenidate. J Child Adolesc Psychopharmacol. 2014;24(3):130-139. PubMed CrossRef
25. Arnold LE, Lindsay RL, Conners CK, et al. A double-blind, placebo-controlled withdrawal trial of dexmethylphenidate hydrochloride in children with attention deficit hyperactivity disorder. J Child Adolesc Psychopharmacol. 2004;14(4):542-554. PubMed CrossRef
26. Klorman R, Brumaghim JT, Fitzpatrick PA, et al. Clinical effects of a controlled trial of methylphenidate on adolescents with attention deficit disorder. J Am Acad Child Adolesc Psychiatry. 1990;29(5):702-709. PubMed CrossRef
27. Florida International University. Behavioral treatment, drug treatment, and combined treatment for attention deficit hyperactivity disorder (ADHD). Clinicaltrials.gov website. https://clinicaltrials.gov/ct2/show/NCT00050622. 2016.
28. Wigal SB, Nordbrock E, Adjei AL, et al. Efficacy of methylphenidate hydrochloride extended-release capsules (Aptensio XR) in children and adolescents with attention-deficit/hyperactivity disorder: a phase III, randomized, double-blind study. CNS Drugs. 2015;29(4):331-340. PubMed CrossRef
29. Findling RL, Turnbow J, Burnside J, et al. A randomized, double-blind, multicenter, parallel-group, placebo-controlled, dose-optimization study of the methylphenidate transdermal system for the treatment of ADHD in adolescents. CNS Spectr. 2010;15(7):419-430. PubMed CrossRef
30. Wilens TE, Hammerness P, Martelon M, et al. A controlled trial of the methylphenidate transdermal system on before-school functioning in children with attention-deficit/hyperactivity disorder. J Clin Psychiatry. 2010;71(5):548-556. PubMed CrossRef
31. Findling RL, Bukstein OG, Melmed RD, et al. A randomized, double-blind, placebo-controlled, parallel-group study of methylphenidate transdermal system in pediatric patients with attention-deficit/hyperactivity disorder. J Clin Psychiatry. 2008;69(1):149-159. PubMed CrossRef
32. Becker SP, Froehlich TE, Epstein JN. Effects of methylphenidate on sleep functioning in children with attention-deficit/hyperactivity disorder. J Dev Behav Pediatr. 2016;37(5):395-404. PubMed CrossRef
33. Coghill D, Banaschewski T, Zuddas A, et al. Long-acting methylphenidate formulations in the treatment of attention-deficit/hyperactivity disorder: a systematic review of head-to-head studies. BMC Psychiatry. 2013;13(1):237. PubMed CrossRef
34. Stein MA, Sarampote CS, Waldman ID, et al. A dose-response study of OROS methylphenidate in children with attention-deficit/hyperactivity disorder. Pediatrics. 2003;112(5):e404. PubMed CrossRef
35. Wilens TE, McBurnett K, Bukstein O, et al. Multisite controlled study of OROS methylphenidate in the treatment of adolescents with attention-deficit/hyperactivity disorder. Arch Pediatr Adolesc Med. 2006;160(1):82-90. PubMed CrossRef
36. Lin DY, Kratochvil CJ, Xu W, et al. A randomized trial of edivoxetine in pediatric patients with attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2014;24(4):190-200. PubMed CrossRef
37. Newcorn JH, Kratochvil CJ, Allen AJ, et al; Atomoxetine/Methylphenidate Comparative Study Group. Atomoxetine and osmotically released methylphenidate for the treatment of attention deficit hyperactivity disorder: acute comparison and differential response. Am J Psychiatry. 2008;165(6):721-730. PubMed CrossRef
38. Riggs PD, Winhusen T, Davies RD, et al. Randomized controlled trial of osmotic-release methylphenidate with cognitive-behavioral therapy in adolescents with attention-deficit/hyperactivity disorder and substance use disorders. J Am Acad Child Adolesc Psychiatry. 2011;50(9):903-914. PubMed CrossRef
39. Kollins SHCA, Khattak S, Weiss MD, et al. A randomized double-blind placebo-controlled multicenter study measuring the efficacy and safety of a novel, extended-release formulation of methylphenidate (PRC-063) in adolescents with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2016;55(10 suppl):s217-s218. CrossRef
40. Childress AC, Spencer T, Lopez F, et al. Efficacy and safety of dexmethylphenidate extended-release capsules administered once daily to children with attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2009;19(4):351-361. PubMed CrossRef
41. Greenhill L, Kollins S, Abikoff H, et al. Efficacy and safety of immediate-release methylphenidate treatment for preschoolers with ADHD. J Am Acad Child Adolesc Psychiatry. 2006;45(11):1284-1293. PubMed CrossRef
42. Children’s Hospital Medical Center Cincinnati. Response variability in children with attention deficit hyperactivity disorder. Clinicaltrials.gov website. https://clinicaltrials.gov/ct2/show/NCT01238822. 2012.
43. Stein MA, Waldman ID, Charney E, et al. Dose effects and comparative effectiveness of extended release dexmethylphenidate and mixed amphetamine salts. J Child Adolesc Psychopharmacol. 2011;21(6):581-588. PubMed CrossRef
44. Pelham WE Jr, Greenslade KE, Vodde-Hamilton M, et al. Relative efficacy of long-acting stimulants on children with attention deficit-hyperactivity disorder: a comparison of standard methylphenidate, sustained-release methylphenidate, sustained-release dextroamphetamine, and pemoline. Pediatrics. 1990;86(2):226-237. PubMed
45. Wernicke JF, Faries D, Milton D, et al. Detecting treatment emergent adverse events in clinical trials: a comparison of spontaneously reported and solicited collection methods. Drug Saf. 2005;28(11):1057-1063. PubMed CrossRef
46. Bruni O, Ottaviano S, Guidetti V, et al. The Sleep Disturbance Scale for Children (SDSC): construction and validation of an instrument to evaluate sleep disturbances in childhood and adolescence. J Sleep Res. 1996;5(4):251-261. PubMed CrossRef
47. Owens JA, Spirito A, McGuinn M. The Children’s Sleep Habits Questionnaire (CSHQ): psychometric properties of a survey instrument for school-aged children. Sleep. 2000;23(8):1043-1051. PubMed CrossRef
Editor’s Note: We encourage authors to submit papers for consideration as a part of our Focus on Childhood and Adolescent Mental Health section. Please contact Karen D. Wagner, MD, PhD, at [email protected].
This PDF is free for all visitors!