ABSTRACT
Importance: “Psychotropic” drugs have widespread reach and impact throughout the brain and body. Thus, many of these drugs could be repurposed for non-psychiatric indications of high public health impact.
Observations: The selective serotonin reuptake inhibitor (SSRI) fluvoxamine was shown efficacious as a COVID-19 treatment based on randomized controlled trials (RCTs), and a benefit of other antidepressants has been posited based on observational and preclinical studies. In this review, we illuminate features of SSRIs and other psychiatric drugs that make them candidates to repurpose for non-psychiatric indications. We summarize research that led to fluvoxamine’s use in COVID-19 and provide guidance on how to use it safely. We summarize studies suggestive of benefit of other antidepressants versus COVID-19 and long COVID. We also describe putative mechanisms of psychiatric drugs in treating long COVID, Alzheimer’s disease, cancer, and other conditions.
Conclusion and Relevance: There is a potentially great clinical and public health impact of psychotropic drug repurposing. Challenges exist to such repurposing efforts, but solutions exist for researchers, regulators, and funders that overcome these challenges.
J Clin Psychiatry 2023;84(3):22r14494
To cite: Lenze EJ, Reiersen AM, Zorumski CF, et al. Beyond “psychotropic”: repurposing psychiatric drugs for COVID-19, Alzheimer’s disease, and cancer. J Clin Psychiatry. 2023;84(3):22r14494.
To share: https://doi.org/10.4088/JCP.22r14494
© 2023 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry, Washington University School of Medicine, St Louis, Missouri.
bDepartment of Child and Adolescent Psychiatry, King’s College London, London, United Kingdom
cDepartment of Child and Adolescent Mental Health, South London and Maudsley NHS Foundation Trust, London, United Kingdompu
*Corresponding author: Eric Lenze, MD, 660 S Euclid Box 8134, St Louis, MO 63110 ([email protected]).
“PSYCHOTROPIC” DRUGS AROSE FROM NON-PSYCHIATRIC MEDICINE—TIME TO RETURN THE FAVOR
Repurposing refers to using a drug for a reason other than its originally designed purpose. The birth of modern psychopharmacology was a serendipitous repurposing effort that began with, among other things, the observation that the antituberculosis drug iproniazid, a monoamine oxidase (MAO) inhibitor, had antidepressant properties.1 More recent advances have also been repurposing efforts. The 50-plus–year-old anesthetic agent ketamine is now being used at subanesthetic doses for depression.2 Similarly, allopregnanolone was isolated from adrenal glands more than 80 years ago, but it was the discovery of its neurosteroid properties and actions that led to its testing in depression.3
“Psychotropic” drugs consistently arise from broader fields of inquiry, so it is not surprising that many of them may be suitable molecules to repurpose for non-psychiatric indications. Most psychiatric drugs have molecular properties that make them good candidates for repurposing. To penetrate the central nervous system (CNS), they tend to be highly lipophilic weak bases,4 with rapid substantial intracellular uptake in brain and other tissues such as lung.5 Many also have pragmatic advantages of safety, widespread availability, low cost (if generic versions are available), and ease of use.
MANY REPURPOSING OPPORTUNITIES: THE EXAMPLE OF SEROTONIN REUPTAKE INHIBITORS
Psychiatrists are aware that serotonin reuptake inhibitors (SRIs) have been repurposed for other CNS indications. They are used to treat neuropathic pain6 and fibromyalgia7 and are also used to prevent migraines.8,9 SRIs are also used for vasomotor symptoms of menopause,10 premature ejaculation,11 stress urinary incontinence, and neurocardiogenic syncope.12
But there are broader opportunities, as SRIs have anti-inflammatory, antiproliferative, anti-infectious, and neuroprotective and cardioprotective effects.13 These effects, demonstrated in vitro or in animal models, would need to be translated into human studies—clinical trials—to determine their relevance for patients with diseases. Herein, we first describe a successful journey from discovery to translation in COVID-19. Then, we describe additional possibilities for long COVID, Alzheimer’s disease and other neurodegenerative conditions, and cancer. We conclude by discussing challenges to drug repurposing and solutions to those challenges.
Repurposing Fluvoxamine for COVID-19
Fluvoxamine recently vaulted from a little-known (to non-psychiatrists) medication to a COVID-19 treatment. How this happened is a case report for future repurposing efforts. Early in the COVID-19 pandemic, it was recognized that developing new drugs would be a lengthy process and that repurposing existing drugs may be the best approach for reducing the morbidity and mortality risk of those who were infected. The United Kingdom RECOVERY trial,14 a large platform trial to test existing medications, was a huge success in this regard, demonstrating the benefits of dexamethasone for treatment of COVID-induced severe respiratory illness, which led to widespread adoption of this medication by June 2020.
In April 2020, a team of researchers at Washington University School of Medicine (St Louis, Missouri) initiated a randomized controlled trial (RCT) to test fluvoxamine for the treatment of early COVID-19. The motivation for testing fluvoxamine was its strong activation of the sigma-1 receptor (S1R), leading to anti-inflammatory effects.15 Specifically, preclinical research found that S1R activation dampens cellular stress responses by inhibiting activity of the endoplasmic reticulum stress sensor, IRE1. This S1R-IRE1 pathway appears to be an essential mechanism by which organisms regulate inflammation in response to proinflammatory signals such as infection. Preclinical rodent models demonstrated that absence of S1R causes animals to succumb quickly to hyperinflammation induced by inflammatory challenges.16 In contrast, by modulating the S1R-IRE1 pathway, fluvoxamine restricts cytokine expression in models of sepsis, resulting in reduced morbidity and mortality.16 Moreover, fluvoxamine does so without inhibiting classical inflammatory signaling pathways that may be necessary for a successful host immune response to the virus.16 Fluvoxamine is the strongest activator of S1R among the selective serotonin reuptake inhibitors (SSRIs), with a Ki = 36 nM, compared to 120 nM for fluoxetine and 292 nM for citalopram.17
The S1R activation path is thought to be the main mechanism of fluvoxamine’s effects versus COVID-19. However, other potential mechanisms have been suggested for the effects of SSRIs, including fluvoxamine and fluoxetine in particular, versus COVID-19.18 A mechanism for several antidepressants, including fluvoxamine and fluoxetine, is via functional inhibition of acid sphingomyelinase (FIASMA), leading to inhibition of viral entry and propagation of SARS-CoV-2 into cells. Two observational studies19,20 showed that antidepressants support this hypothesis, finding that antidepressants with high FIASMA were associated with a reduced incidence of emergency department admission, hospital admission, and mortality among SARS-CoV-2 patients in a dose-dependent manner. As well, all SSRIs reduce serotonin uptake by platelets, thus depleting platelets of serotonin and reducing their function in both coagulation and the immune response to illness. Severe COVID-19 is often marked by a hypercoagulation and hyperserotonergic state due to florid platelet degranulation of serotonin into plasma. Platelet inhibition by SSRIs may counterbalance this deleterious effect and activate macroautophagy, an intracellular process that helps to dampen cellular stress.
Clinical Trials of Fluvoxamine for COVID-19
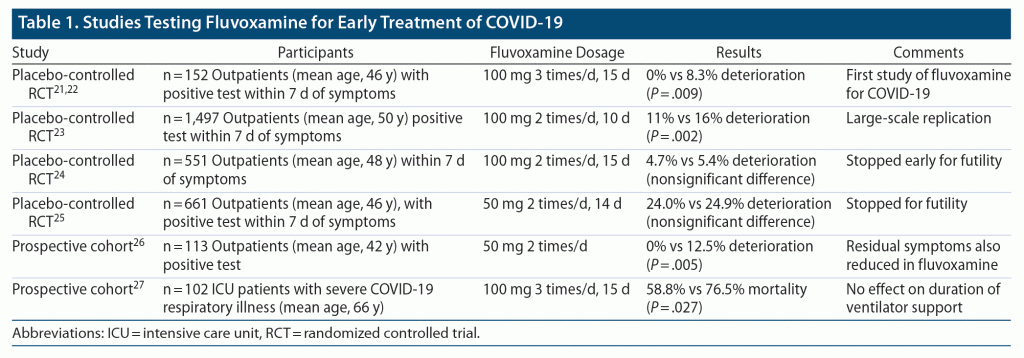
Four clinical trials and two prospective cohort studies have reported results with fluvoxamine for the treatment of COVID-19 (see Table 1). In November 2020, the Washington University team reported results from the first placebo-controlled RCT in outpatients with early COVID-19 (defined as SARS CoV-2 positive and < 7 days since symptom onset).21 The study arms were fluvoxamine, titrated rapidly up to 100 mg 3 times daily as tolerated, or placebo for 15 days. The prespecified primary outcome was clinical deterioration, defined as shortness of breath and/or hospitalization for same, plus hypoxia (SpO2 < 92%). Among 80 patients randomized, none clinically deteriorated, compared to 6 of 72 (8.3%) in the placebo group (P = .009). There were no differences in symptom reduction; the researchers speculated that the highly individually dynamic nature of COVID-19 symptoms would interfere with the ability to discern a treatment effect.22
The second RCT was from the TOGETHER Trial, a large adaptive platform trial led by researchers in Canada and Brazil, which randomized more than 4,000 patients internationally to evaluate drugs for early treatment of COVID-19.23 This study randomized 1,497 patients to fluvoxamine 100 mg twice daily or placebo for 10 days. The prespecified primary outcome was severe disease progression, a composite endpoint of either retention in a COVID-19 emergency setting or transfer to tertiary hospital due to COVID-19 up to 28 days post–random assignment. In August 2021, the fluvoxamine arm was stopped for superiority: the independent Data and Safety Monitoring Board (DSMB) determined that the criteria for proving fluvoxamine’s efficacy had been met. The TOGETHER trial found a 32% reduction in risk for severe disease progression in patients randomized to fluvoxamine compared to placebo. Among patients who were adherent with their treatment regimen, taking at least 80% of their pills, the effect was even more striking: more than 66% reduction in risk for hospitalization with fluvoxamine and only 1 death in the fluvoxamine group compared to 12 in the placebo group.
Of note, the TOGETHER trial was a platform trial that tested multiple other treatments in 2020 and 2021: hydroxychloroquine, ritonavir/lopinavir, metformin, and both low-dose and high-dose ivermectin. All of these were stopped for futility: the trial did not show evidence of efficacy of these drugs. In particular, the high-dose ivermectin arm was stopped for futility at approximately the same time as the fluvoxamine arm was stopped for efficacy.23
The third trial was the STOP COVID 2 trial, a multisite, US- and Canada-based trial that recruited both locally at each study site and nationally throughout the US.24 The study randomized 547 outpatients to fluvoxamine 100 mg twice daily or placebo for 15 days. The study was stopped for futility at the recommendation of the DSMB based on low conditional power plus slowing recruitment attributable to the vaccination campaign. Clinical deterioration, defined in the same way as in the earlier trial, occurred in 13 (4.7%) of the fluvoxamine group versus 15 (5.4%) in the placebo group, a nonsignificant difference. Several issues may have interfered with the trial, especially low event rates in the placebo group but also participants’ starting the medication relatively late in the illness (median = 5 days after symptom onset), and frequent occurrences of poor adherence, early stopping of study medication, and coprescribed medications including steroids. These methodological issues highlight the challenges of carrying out a fully remote trial for an acute illness such as COVID-19.
A meta-analysis of these 3 clinical trials concluded that fluvoxamine showed a high probability of reducing hospitalization from COVID-19.24 Recently, a fourth trial25 testing fluvoxamine at a lower dose of 50 mg twice daily reported negative results. The COVID-OUT trial used a factorial design to test metformin, ivermectin, and fluvoxamine in preventing a composite endpoint (hypoxemia, emergency department visit, hospitalization, or death) and randomized 1,431 total patients. None of the drugs showed efficacy in preventing this composite endpoint; the adjusted odds ratio for fluvoxamine was 0.94.
Two prospective cohort studies have also shown a clinical benefit of fluvoxamine for COVID-19. One was conducted at a horse race track in Berkeley, California, which suffered a major occupational outbreak of COVID-19.26 The track physician offered workers who tested positive a prescription of fluvoxamine 50 mg twice daily; 65 took the drug, and 48 did not take it. None of those who opted to take fluvoxamine were hospitalized, versus 12.5% (6/48) of those who did not take it. At 14 days, none of those who took fluvoxamine had persistent symptoms, versus 60% (29/48) of those who did not. The second trial27 was conducted in COVID-19 intensive care unit (ICU) patients with matched controls. The study reported lower mortality in the fluvoxamine group than the control group (30/51 [58.8%] vs 39/51 [76.5%]; hazard ratio [HR]= 0.58, P = .027). However, there were no significant differences in days on ventilator support, duration of ICU stay, or total hospital stay.
Overall, these studies demonstrate a clinically and statistically significant effect of fluvoxamine on reducing morbidity and possibly mortality in COVID-19 patients. Earlier treatment may be more effective; for example, the Berkeley horse track study,26 which provided fluvoxamine earliest in the course of illness, showed the best outcomes. The studies do not clarify the optimal dose or duration of fluvoxamine. Two trials have tested fluvoxamine at a lower dose of 50 mg twice daily (the ACTIV-6 trial and the COVID OUT trial) but have not found this lower dose to be effective. A recommended strategy is to take an initial dose of 50 mg and then, depending on tolerability, either immediately increase to 100 mg twice daily or keep at 50 mg twice daily. If 100 mg is not well-tolerated, the dose can be decreased to 50 mg. After a 10- to 15-day course, fluvoxamine can be stopped with no need to taper.
Fluvoxamine has a high safety margin. It is not fatal even in overdose, and it is not prone to increase the cardiac corrected QT interval (QTc) unlike some other SSRIs.28 Thus, no laboratory monitoring is needed for its use. Published trials of fluvoxamine in COVID-19 have reported its characteristic side effects, particularly mild nausea, and no adverse events suggestive of drug-drug interactions. However, fluvoxamine potently inhibits cytochrome P450 (CYP) 1A2 and 2C19.29 Caffeine is a substrate of CYP1A2, so its use should be eliminated or greatly reduced during fluvoxamine treatment. Otherwise, there are few narrow–therapeutic index drugs for which these enzymes are the primary metabolic pathways. For patients taking drugs that are primarily metabolized by CYP1A2 or 2C19 and have a narrow therapeutic index (namely, theophylline, clozapine, olanzapine, and tizanidine, which are CYP1A2 substrates), they should either not take fluvoxamine or should be evaluated to determine if the drug can be safely given. Similarly, many COVID-19 patients are already taking SRIs and should probably avoid adding fluvoxamine, which risks serotonin syndrome, or switching from an existing medication, which risks worsening mental illness if that medication was helpful.
Other Evidence of the Benefits of SSRIs for COVID-19
The potential for other SSRIs, particularly fluoxetine, to reduce morbidity or even infection from COVID-19 has been raised by in vitro studies. One study30 screening existing drugs versus SARS-CoV-2 infection found that fluoxetine significantly inhibited it at a concentration of 0.8 µg/mL. Another line of research31 proposed that functional inhibition of acid sphingomyelinase, leading to inhibition of viral entry and propagation of SARS-CoV-2 into cells, was a common pathway for several psychiatric drugs (including all SSRIs) and non-psychiatric drugs. These researchers showed that amitriptyline prevented ex vivo infection of nasal epithelial cells with pseudoviral SARS CoV-2 by this route.
Several observational studies have shown a reduced incidence of morbidity, or COVID-19 illness, in individuals taking SSRIs. A study in France32 of 2,846 adults hospitalized for severe COVID-19 found that patients who were taking a medication that was a functional inhibitor of acid sphingomyelinase (including all SSRIs) were less likely to be intubated or die (HR = 0.58); the same study found that patients with severe COVID-19 who were taking one of these medications tended to be older and have more medical comorbidities, suggesting that the medications prevented severe COVID-19 in the first place. Another observational study by the same group33 followed 7,230 adults hospitalized for COVID-19 and found that patients who received an antidepressant within 48 hours of admission had lower risk for intubation or death (HR = 0.56). A study of psychiatric inpatients in New York State during the first wave of the pandemic in 202034 found a significant protective association between antidepressant use and COVID-19 infection (odds ratio [OR] = 0.33) and found that SSRIs as well as serotonin-norepinephrine reuptake inhibitors drove this protective effect, with fluoxetine specifically showing a protective effect. A large electronic health record database study of 83,584 patients35 found that those who were taking SSRIs, and in particular those who were taking fluoxetine or fluvoxamine, had a reduced mortality (relative risk [RR] = 0.74). Finally, a case-control study of 264 patients hospitalized for severe COVID-19 pneumonia36 found that fluoxetine use was associated with improved survival (OR = 0.30); this study is notable in that participants were receiving fluoxetine specifically as adjunctive treatment for COVID-19, not chronically for psychiatric purposes.
Overall, these observational studies show a consistent association of SSRI use—especially fluoxetine—with more favorable outcomes in COVID-19, including reduced risk of infection and, in particular, reduced intubation or death in those who have serious COVID-19 illness. It is important to highlight that none of these studies was a randomized controlled trial, and, as described subsequently in this report, there are limitations in estimating treatment effects from observational studies.
Repurposing Psychotropics for Long-Term Neuropsychiatric Manifestations of COVID-19
It has also been posited that SSRIs could help with the longer-term neuropsychiatric manifestations of COVID-19.37 Understanding this benefit is important: due to high worldwide rates of COVID-19, we could face an onslaught of new-onset and recurrent neuropsychiatric problems. Long COVID is a patient-generated term referring to the burden of symptoms that persist beyond acute illness. In this article, we use the term neuropsychiatric long COVID, given that long COVID commonly and prominently manifests as neuropsychiatric problems. The Patient-Led Research Collaborative assessed the prevalence of symptoms in 3,762 persons over 7 months post-COVID; the authors state,
Memory and cognitive dysfunction, experienced by over 85% of respondents, were the most pervasive and persisting neurologic symptoms in this cohort, equally common across all ages, and with substantial impact on work. Headaches, insomnia, vertigo, neuralgia, neuropsychiatric changes, tremors, sensitivity to noise and light, hallucinations (olfactory and other), tinnitus, and other sensorimotor symptoms were also all common among respondents and may point to larger neurological issues involving both the central and peripheral nervous system. The reduced work capacity because of cognitive dysfunction, in addition to other debilitating symptoms, translated into the loss of hours, jobs, and ability to work relative to pre-illness levels.38(p14)
More studies have been published adding to this conclusion that long COVID is prominently neuropsychiatric. A meta-analysis39 of 51 studies of persistent COVID-19 neuropsychiatric symptoms, comprising 18,917 patients, found that the most common symptoms were sleep disturbance (pooled prevalence = 27.4%), fatigue (24.4%), cognitive impairment (20.2%), anxiety (19.1%), posttraumatic stress (15.7%), and depression (12.9%).
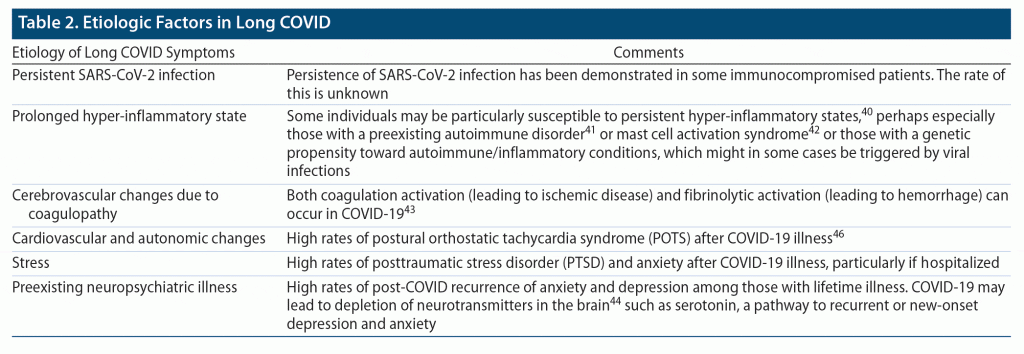
What remains unknown is how persistent these symptoms will be. There are several proposed etiologies for neuropsychiatric sequelae of COVID-19 (Table 2); many of these etiologies are suggestive of long-term problems that could last many years without treatment. Further, large numbers of new infections continue.
What Is the Best Strategy for Testing Treatments for Neuropsychiatric Long COVID?
A preliminary study45 found SSRIs may be helpful for post-COVID depression, prompting the need for clinical trials to test their role in neuropsychiatric long COVID. Arguably, then, we are in the same place now with long COVID as 3 years ago with the start of the pandemic: How do we rigorously test drugs that show promise based on theorized mechanisms or preliminary studies? This is a key question right now not just for researchers but for funders, policy-makers, patients, and health care providers.
One research strategy is to carry out studies similar to the projects that have been most successful in assessing acute COVID-19 therapeutics: large platform trials that assess at least one treatment (as well as combinations), similar to the REMAP-CAP,46 RECOVERY, TOGETHER, and ACTIV trials. Such trials could add treatments as they show promise in smaller trials, while removing treatments that are found to be futile. Just as the critical illness and infectious diseases communities conducted large trials for acute COVID-19, the psychiatric community, working together with long COVID clinics that have emerged throughout the US and world, can mount a similar effort.
There are major challenges to this strategy, outlined in Table 3: identifying valid outcomes given the heterogeneity of COVID-19 symptoms both across individuals and within individuals over time22; the need to determine optimal treatment combinations, as monotherapies may have small effect sizes; and personalizing treatment to the individual patient, given the likely heterogeneity of etiologies and presentations in neuropsychiatric long COVID. The table suggests solutions to these challenges; the psychiatric research field should be central to these solutions given that the challenges are similar to what we have faced for testing treatment in all of the major psychiatric disorders. Table 3 also describes generic drug repurposing challenges and solutions, discussed subsequently in this article.
Repurposing SRIs for Alzheimer’s’ Disease
Alzheimer’s disease, originally characterized by the psychiatrist Alois Alzheimer in 1901, is a progressive neurodegenerative disease that is the cause of most cases of dementia and for which there is still no preventative or disease-modifying treatment. At the time of this writing, there is positive news from a large study of lecanemab, a monoclonal antibody thought to reduce amyloid-β deposition and thereby reduce the first pathogenic step of Alzheimer’s disease.52
It is likely that interventions to prevent Alzheimer’s disease or its progression will need to be “multi-hit,” affecting multiple steps along the pathway of the disease. If treatments will soon be available that increase clearance of amyloid plaques, combining these with other mechanisms is a promising next step.
As already noted, some SRIs have anti-inflammatory functions that could improve outcome in Alzheimer’s (and other) neurodegenerative illness. For example, fluvoxamine and fluoxetine appear to inhibit the NLRP3 inflammasome, an immunomodulatory effect that could be repurposed for macular degeneration and other illnesses whose pathophysiology involves neuroinflammation or peripheral inflammation.53 Beyond these anti-inflammatory effects, several serotonergic drugs appear to have neuroprotective effects in the context of CNS amyloid deposition (a precursor to Alzheimer’s dementia), including trazodone54,55 vortioxetine,56 and fluvoxamine.57 Many may be therapeutic in neuropsychiatric diseases because of sigma-1 receptor agonist activity.58,59 Finally, SRIs appear to reduce amyloid production and aggregation60; they could provide a potent combination together with medications that increase amyloid plaque clearance.
Repurposing Psychotropic Drugs for Cancer Treatment
Many psychotropics have potential to be repurposed for anticancer drug discovery. SSRIs and other psychotropic drugs are particularly promising because of their high safety and tolerability, given that toxicity is a key reason for the very high failure rate of new compounds for cancer treatment.61
Antidepressants can modulate antitumor activity through different mechanisms including (1) disrupting mitochondrial and lysosomal function,62 (2) inhibiting cell proliferation, (3) reducing metastasis, (4) disrupting the cell cycle, (5) inducing apoptosis and autophagy, (6) affecting oxidative-associated pathways, and (7) modulating tumor immunology.63 Some antidepressants have advanced into clinical trials for cancer therapy. Repurposing tranylcypromine has led to the discovery of several tranylcypromine-based histone lysine-specific demethylase 1 (LSD1) inhibitors for cancer therapy.63,64 The success of tranylcypromine-based LSD1 inhibitors in clinical trials suggests that both monotherapy and combinational therapy are promising therapeutic strategies for cancer treatment. Repurposing approved antidepressants also provides hope for patients with both depression and cancer.63
Antipsychotic drugs are also being investigated for anticancer properties, and, lately, there has been an increasing interest in their role in cancer treatment.65 Haloperidol has been found to induce autophagy, apoptosis, and cell cycle arrest.66 Penfluridol induces apoptosis by activation of protein phosphatase 2A (PP2A) and exerts antiproliferative effects potentially through the inhibition of the integrin α6β4 signaling axis and is considered a candidate for repurposing in triple-negative breast cancer.67 Chlorpromazine has antitumor effects in white blood cell malignancies. Chlorpromazine appears to inhibit mitochondrial DNA polymerase and induce apoptosis selectively in several leukemia types, including acute T and acute B lymphoblastic leukemia and Burkitt lymphoma cell lines but not in normal lymphocytes.68 Human clinical trials include a phase 2 study of chlorpromazine in glioblastoma, a phase 2 trial of pimozide in metastatic melanoma, and a phase 1 trial of thioridazine in acute myeloid leukemia. Finally, the estrogen- and prolactin-sparing69 drug aripiprazole has in vitro activity against estrogen receptor–positive breast cancer cells comparable to that of standard anticancer drugs.
Lithium, a mood stabilizer, has long helped manage aplastic anemia and congenital or chronic neutropenia.70,71 There are multiple clinical trials registered in ClinicalTrials.gov (as of January 29, 2022) in which lithium is being tested in various cancers, as well as those in which it is being tested as a neuroprotective agent when patients are undergoing radiotherapy.
Propranolol, a β-blocker anxiolytic, interferes with angiogenesis and cell proliferation and modulates the expression and activation of angiopoietin/TIE2, vascular endothelial growth factor (VEGF), and hypoxia-inducible factor signaling.72,73 Propranolol use is associated with lower cancer risk (HR = 0.75)74 and lower prostate cancer–specific mortality75,76 and can modify the post-surgical metastatic process77; multiple clinical trials with propranolol as a chemotherapy adjunct are ongoing (per ClinicalTrials.gov, as of January 29, 2022). Overall, this section shows that there are many avenues toward better cancer treatment outcomes with psychotropic drugs, which require testing in clinical trials.
Repurposing Psychotropics for Other Conditions
Another area of interest has been exploring antipsychotics for antirheumatic effects, as several cytokines modulated by them are like existing antirheumatic drugs. Data mining of real-world data and bioinformatics databases have identified haloperidol, which also targets the S1R78 and dampens proinflammatory responses,79 as a potential antirheumatic drug candidate. Haloperidol may exert antirheumatic effects by modulating cytokine and chemokine signaling, major histocompatibility complex class-II antigen presentation, and toll-like receptor cascade pathways.80
A recent study81 reported results from a follow-up of 50,000 patients with bipolar disorder from the Swedish Patient Register investigating the impact of lithium or sodium valproate on respiratory infections. During follow-up, 5,760 respiratory infections were documented, and the incidence rate was 28% lower during lithium treatment and 35% higher during valproate treatment compared with periods off treatment. This study provides real-world evidence that lithium protects against respiratory infections and suggests that the repurposing potential of lithium for antiviral effects is worthy of investigation. Finally, in an example of drug repurposing possibilities for rare diseases, buspirone, an anxiolytic, has been shown to improve autonomic dysregulation in Rett syndrome.82
Challenges in Drug Repurposing: Research and Practice
There is more opportunity than ever to identify repurposable drugs for new indications based on the development of modern strategies, including signature matching, omics sequencing, artificial intelligence, and phenotypic screening.83 There are many pitfalls in translating in vitro and preclinical research into human studies. Therefore, promising candidate drugs must be tested in RCTs to gain sufficient evidence for widespread adoption, including regulators’ approving these drugs for new uses. Therein lie several challenges that, to date, have limited repurposing for psychotropic (and other) drugs (Table 3).84
First, difficulties with enforcing patent rights on a new indication are a double-edged sword: it means that a repurposed generic drug could be widely available and inexpensive because of the lack of patent protection, but it also removes the financial incentive for carrying out costly studies to demonstrate efficacy for new indications.85 Another hurdle is in regulatory approval: the US Food and Drug Administration (FDA) and European Medicines Agency (EMA) drug approval processes are expensive and laborious, which again provides an incentive for approving only drugs that have patent protection, thereby providing a financial incentive for this route. Without such official approvals, doctors may be reluctant to prescribe a medication “off-label” or may even be barred from doing so, as has been observed during the COVID-19 pandemic with non-approved medications.
Given the time, costs, and other limitations of conducting large RCTs of drugs, physicians may be tempted to turn to observational studies to guide treatment. Yet, observational studies suffer from biases, including confounding—namely, the association may be due to the reason for which the drug is prescribed (eg, depression) rather than due to the drug itself. Such confounds in observational studies of psychotropic drugs (especially antidepressants) tend to show adverse associations, such as more falls.86 Similar confounds, including healthy user bias, have been proposed to underlie seeming benefits of drugs in observational studies; a famous example of this is the discrepancy between observational and RCT data for neuropsychiatric benefits of treatments such as improved cognitive function with estrogen.87 Therefore it is, at best, controversial to assert a drug’s benefit based purely on observational data. The value of rigorous clinical trials continues to be demonstrated during the COVID-19 pandemic, in disproving ineffective treatments and supporting effective ones. One opportunity to be explored would be interinstitutional collaborations within the National Institiutes of Health, such as the National Institute of Mental Health (NIMH) and the National Institute of Allergy and Infectious Diseases for psychotropic drugs that have anti-infectious or anti-inflammatory properties, or the NIMH and National Institute on Aging for similar drugs with properties versus Alzheimer’s disease.
CONCLUSION: PSYCHOTROPIC DRUGS NEED REPURPOSING EFFORTS IN A RANGE OF ILLNESSES
With the pandemic continuing to evolve, key questions remain unanswered about the role of SSRIs in the treatment of acute COVID-19. In particular, what is the best dose and timing of starting fluvoxamine, and how effective is it in combination with other treatments for COVID-19 (such as monoclonal antibodies)? Is fluoxetine, which has shown promise in preclinical and observational studies, also an effective treatment given that it is more widely available and easier to use? And what are the best treatments for long-term neuropsychiatric manifestations of COVID-19 and in which patients? Given that many psychotropics are known to have widespread molecular, cellular, and physiologic effects, including anti-inflammatory, neuroprotective, cardioprotective, and antiproliferative mechanisms, lessons learned in testing psychotropic medications versus COVID-19 will be important for other drug repurposing efforts such as for Alzheimer’s disease and other neurodegenerative illnesses and for cancer.13 Many challenges, including stigma, exist in repurposing these drugs, but their low cost, high tolerability, safety, and widespread availability make them potentially high-impact, low-cost solutions for these common and devastating public health problems.
Submitted: April 22, 2022; accepted January 11, 2023.
Published online: March 20, 2023.
Relevant financial relationships: Drs Lenze and Reiersen are co-inventors on a patent for sigma-1 agonists in COVID-19 treatment. Dr Lenze reports consulting with Prodeo, IngenioRx, Boehringer-Ingelheim, and Pritikin ICR and research support from Janssen. Dr Zorumski reports consulting with Sage Pharmaceuticals. Dr Santosh reports no conflicts.
Funding/support: The manuscript was supported by Mercatus FastGrants, Taylor Family Institute for Innovative Psychiatric Research at Washington University School of Medicine, and grant UL1TR002345 from the National Institutes of Health.
Role of the sponsor: The funders had no role in the conduct of the study; design, management, analysis, or interpretation of the data; or preparation, review, or approval of the manuscript.
CLINICAL POINTS
- Selective serotonin reuptake inhibitors (SSRIs)—and other “psychotropic” drugs—have mechanisms that may be beneficial for other diseases, including COVID-19, long COVID, Alzheimer’s disease, and cancer.
- A series of drug repurposing studies in acute COVID-19 showed that fluvoxamine at a target dose of 100 mg twice daily is an effective early treatment strategy.
- To successfully repurpose psychiatric drugs requires clinical trials using innovative precision medicine solutions.
References (87)

- Leon AC. Evolution of psychopharmacology trial design and analysis: six decades in the making. J Clin Psychiatry. 2011;72(3):331–340. PubMed CrossRef
- Subramanian S, Haroutounian S, Palanca BJA, et al. Ketamine as a therapeutic agent for depression and pain: mechanisms and evidence. J Neurol Sci. 2022;434:120152. PubMed CrossRef
- Zorumski CF, Paul SM, Izumi Y, et al. Neurosteroids, stress and depression: potential therapeutic opportunities. Neurosci Biobehav Rev. 2013;37(1):109–122. PubMed CrossRef
- Lester HA, Miwa JM, Srinivasan R. Psychiatric drugs bind to classical targets within early exocytotic pathways: therapeutic effects. Biol Psychiatry. 2012;72(11):907–915. PubMed CrossRef
- Hallifax D, Houston JB. Saturable uptake of lipophilic amine drugs into isolated hepatocytes: mechanisms and consequences for quantitative clearance prediction. Drug Metab Dispos. 2007;35(8):1325–1332. PubMed CrossRef
- Qureshi Z, Ali MN, Khalid M. An Insight into potential pharmacotherapeutic agents for painful diabetic neuropathy. J Diabetes Res. 2022;2022:9989272. PubMed CrossRef
- Welsch P, Üçeyler N, Klose P, et al. Serotonin and noradrenaline reuptake inhibitors (SNRIs) for fibromyalgia. Cochrane Database Syst Rev. 2018;2(2):CD010292. PubMed CrossRef
- Banzi R, Cusi C, Randazzo C, et al. Selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) for the prevention of migraine in adults. Cochrane Database Syst Rev. 2015;4(4):CD002919. PubMed CrossRef
- d’Amato CC, Pizza V, Marmolo T, et al. Fluoxetine for migraine prophylaxis: a double-blind trial. Headache. 1999;39(10):716–719. PubMed CrossRef
- Joffe H, Guthrie KA, LaCroix AZ, et al. Low-dose estradiol and the serotonin-norepinephrine reuptake inhibitor venlafaxine for vasomotor symptoms: a randomized clinical trial. JAMA Intern Med. 2014;174(7):1058–1066. PubMed CrossRef
- Waldinger MD, Hengeveld MW, Zwinderman AH, et al. Effect of SSRI antidepressants on ejaculation: a double-blind, randomized, placebo-controlled study with fluoxetine, fluvoxamine, paroxetine, and sertraline. J Clin Psychopharmacol. 1998;18(4):274–281. PubMed CrossRef
- Calkins H. Pharmacologic approaches to therapy for vasovagal syncope. Am J Cardiol. 1999;84(8):20–25. PubMed CrossRef
- Nykamp MJ, Zorumski CF, Reiersen AM, et al. Opportunities for drug repurposing of serotonin reuptake inhibitors: potential uses in inflammation, infection, cancer, neuroprotection, and alzheimer’s disease prevention. Pharmacopsychiatry. 2022;55(1):24–29. PubMed CrossRef
- Horby P, Lim WS, Emberson JR, et al; RECOVERY Collaborative Group. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med. 2021;384(8):693–704. PubMed CrossRef
- Nicol GE, Karp JF, Reiersen AM, et al. “What were you before the war?” repurposing psychiatry during the COVID-19 pandemic. J Clin Psychiatry. 2020;81(3):20com13373. PubMed CrossRef
- Rosen DA, Seki SM, Fernández-Castañeda A, et al. Modulation of the sigma-1 receptor-IRE1 pathway is beneficial in preclinical models of inflammation and sepsis. Sci Transl Med. 2019;11(478):eaau5266. PubMed CrossRef
- Narita N, Hashimoto K, Tomitaka S, et al. Interactions of selective serotonin reuptake inhibitors with subtypes of sigma receptors in rat brain. Eur J Pharmacol. 1996;307(1):117–119. PubMed CrossRef
- Sukhatme VP, Reiersen AM, Vayttaden SJ, et al. Fluvoxamine: a review of its mechanism of action and its role in COVID-19. Front Pharmacol. 2021;12:652688. PubMed CrossRef
- Fritz BA, Hoertel N, Lenze EJ, et al. Association between antidepressant use and ED or hospital visits in outpatients with SARS-CoV-2. Transl Psychiatry. 2022;12(1):341. PubMed CrossRef
- Hoertel N, Sánchez-Rico M, Kornhuber J, et al; On Behalf Of Ap-Hp/Université Paris Cité/Inserm Covid-Research Collaboration Ap-Hp Covid Cdr Initiative And Entrepôt de Données de Santé Ap-Hp Consortium. Antidepressant use and its association with 28-day mortality in inpatients with SARS-CoV-2: support for the FIASMA Model against COVID-19. J Clin Med. 2022;11(19):5882. PubMed CrossRef
- Lenze EJ, Mattar C, Zorumski CF, et al. Fluvoxamine vs placebo and clinical deterioration in outpatients with symptomatic COVID-19: a randomized clinical trial. JAMA. 2020;324(22):2292–2300. PubMed CrossRef
- Rodebaugh TL, Frumkin MR, Reiersen AM, et al. Acute symptoms of mild to moderate COVID-19 are highly heterogeneous across individuals and over time. Open Forum Infect Dis. 2021;8(3):b090. PubMed CrossRef
- Reis G, Silva EASM, Silva DCM, et al; TOGETHER Investigators. Effect of early treatment with ivermectin among patients with Covid-19. N Engl J Med. 2022;386(18):1721–1731. PubMed CrossRef
- Lee TC, Vigod S, Bortolussi-Courval É, et al. Fluvoxamine for outpatient management of COVID-19 to prevent hospitalization: a systematic review and meta-analysis. JAMA Netw Open. 2022;5(4):e226269. PubMed CrossRef
- Bramante CT, Huling JD, Tignanelli CJ, et al; COVID-OUT Trial Team. Randomized trial of metformin, ivermectin, and fluvoxamine for Covid-19. N Engl J Med. 2022;387(7):599–610. PubMed CrossRef
- Seftel D, Boulware DR. Prospective cohort of fluvoxamine for early treatment of COVID-19. Open Forum Infect Dis. 2021;8(2):b050. PubMed CrossRef
- Calusic M, Marcec R, Luksa L, et al. Safety and efficacy of fluvoxamine in COVID-19 ICU patients: an open label, prospective cohort trial with matched controls. Br J Clin Pharmacol. 2022;88(5):2065–2073. PubMed CrossRef
- Funk KA, Bostwick JR. A comparison of the risk of QT prolongation among SSRIs. Ann Pharmacother. 2013;47(10):1330–1341. PubMed CrossRef
- Christensen M, Tybring G, Mihara K, et al. Low daily 10-mg and 20-mg doses of fluvoxamine inhibit the metabolism of both caffeine (cytochrome P4501A2) and omeprazole (cytochrome P4502C19). Clin Pharmacol Ther. 2002;71(3):141–152. PubMed CrossRef
- Zimniak M, Kirschner L, Hilpert H, et al. The serotonin reuptake inhibitor fluoxetine inhibits SARS-CoV-2 in human lung tissue. Sci Rep. 2021;11(1):5890. PubMed CrossRef
- Carpinteiro A, Edwards MJ, Hoffmann M, et al. Pharmacological inhibition of acid sphingomyelinase prevents uptake of SARS-CoV-2 by epithelial cells. Cell Rep Med. 2020;1(8):100142. PubMed CrossRef
- Hoertel N, Sánchez-Rico M, Gulbins E, et al; AP-HP / Université de Paris / INSERM COVID-19 research collaboration, AP-HP COVID CDR Initiative, “Entrepôt de Données de Santé” AP-HP Consortium. Association between FIASMAs and reduced risk of intubation or death in individuals hospitalized for Severe COVID-19: an observational multicenter study. Clin Pharmacol Ther. 2021;110(6):1498–1511. PubMed CrossRef
- Hoertel N, Sánchez-Rico M, Vernet R, et al; AP-HP / Universities / INSERM COVID-19 Research Collaboration and AP-HP COVID CDR Initiative. Association between antidepressant use and reduced risk of intubation or death in hospitalized patients with COVID-19: results from an observational study. Mol Psychiatry. 2021;26(9):5199–5212. PubMed CrossRef
- Clelland CL, Ramiah K, Steinberg L, et al. Analysis of the impact of antidepressants and other medications on COVID-19 infection risk in a chronic psychiatric in-patient cohort. BJPsych Open. 2021;8(1):e6. PubMed CrossRef
- Oskotsky T, Maric I, Tang A, et al. Mortality risk among patients with COVID-19 prescribed selective serotonin reuptake inhibitor antidepressants. JAMA Netw Open. 2021;4(11):e2133090. PubMed CrossRef
- Németh ZK, Szûcs A, Vitrai J, et al. Fluoxetine use is associated with improved survival of patients with COVID-19 pneumonia: A retrospective case-control study. Ideggyogy Sz. 2021;74(11–12):389–396. PubMed CrossRef
- Khani E, Entezari-Maleki T. Fluvoxamine and long COVID-19; a new role for sigma-1 receptor (S1R) agonists. Mol Psychiatry. 2022;27(9):3562. PubMed CrossRef
- Davis HE, Assaf GS, McCorkell L, et al. Characterizing long COVID in an international cohort: 7 months of symptoms and their impact. EClinicalMedicine. 2021;38:101019. PubMed CrossRef
- Badenoch JB, Rengasamy ER, Watson C, et al. Persistent neuropsychiatric symptoms after COVID-19: a systematic review and meta-analysis. Brain Commun. 2022;4(1):fcab297. PubMed CrossRef
- Law JC, Girard M, Chao GYC, et al. Persistence of T cell and antibody responses to SARS-CoV-2 up to 9 months after symptom onset. J Immunol. 2022;208(2):429–443. PubMed CrossRef
- Liu Y, Sawalha AH, Lu Q. COVID-19 and autoimmune diseases. Curr Opin Rheumatol. 2021;33(2):155–162. PubMed CrossRef
- Afrin LB, Weinstock LB, Molderings GJ. Covid-19 hyperinflammation and post-Covid-19 illness may be rooted in mast cell activation syndrome. Int J Infect Dis. 2020;100:327–332. PubMed CrossRef
- Yamada S, Asakura H. Coagulopathy and fibrinolytic pathophysiology in COVID-19 and SARS-CoV-2 vaccination. Int J Mol Sci. 2022;23(6):3338. PubMed CrossRef
- Boldrini M, Canoll PD, Klein RS. How COVID-19 affects the brain. JAMA Psychiatry. 2021;78(6):682–683. PubMed CrossRef
- Mazza MG, Zanardi R, Palladini M, et al. Rapid response to selective serotonin reuptake inhibitors in post-COVID depression. Eur Neuropsychopharmacol. 2022;54:1–6. PubMed CrossRef
- Angus DC, Berry S, Lewis RJ, et al. The REMAP-CAP (Randomized Embedded Multifactorial Adaptive Platform for Community-acquired Pneumonia) Study. Rationale and Design. Ann Am Thorac Soc. 2020;17(7):879–891. PubMed CrossRef
- Ridenour TA, Wittenborn AK, Raiff BR, et al. Illustrating idiographic methods for translation research: moderation effects, natural clinical experiments, and complex treatment-by-subgroup interactions. Transl Behav Med. 2016;6(1):125–134. PubMed CrossRef
- Reitz KM, Seymour CW, Vates J, et al. Strategies to Promote ResiliencY (SPRY): a randomised embedded multifactorial adaptative platform (REMAP) clinical trial protocol to study interventions to improve recovery after surgery in high-risk patients. BMJ Open. 2020;10(9):e037690. PubMed CrossRef
- Lenze EJ, Nicol GE, Barbour DL, et al. Precision clinical trials: a framework for getting to precision medicine for neurobehavioural disorders. J Psychiatry Neurosci. 2021;46(1):E97–E110. PubMed CrossRef
- Sachs RE, Ginsburg PB, Goldman DP. Encouraging new uses for old drugs. JAMA. 2017;318(24):2421–2422. PubMed CrossRef
- Sydes MR, Barbachano Y, Bowman L, et al; Data Enabled Trials Group Workshop Group members. Realising the full potential of data-enabled trials in the UK: a call for action. BMJ Open. 2021;11(6):e043906. PubMed CrossRef
- McDade E, Cummings JL, Dhadda S, et al. Lecanemab in patients with early Alzheimer’s disease: detailed results on biomarker, cognitive, and clinical effects from the randomized and open-label extension of the phase 2 proof-of-concept study. Alzheimers Res Ther. 2022;14(1):191. PubMed CrossRef
- Ambati M, Apicella I, Wang SB, et al. Identification of fluoxetine as a direct NLRP3 inhibitor to treat atrophic macular degeneration. Proc Natl Acad Sci U S A. 2021;118(41):e2102975118. PubMed CrossRef
- de Oliveira P, Cella C, Locker N, et al. Improved sleep, memory, and cellular pathological features of tauopathy, including the NLRP3 inflammasome, after chronic administration of trazodone in rTg4510 Mice. J Neurosci. 2022;42(16):3494–3509. PubMed CrossRef
- Halliday M, Radford H, Zents KAM, et al. Repurposed drugs targeting eIF2α-P-mediated translational repression prevent neurodegeneration in mice. Brain. 2017;140(6):1768–1783. PubMed CrossRef
- Jiang LX, Huang GD, Su F, et al. Vortioxetine administration attenuates cognitive and synaptic deficits in 5×FAD mice. Psychopharmacology (Berl). 2020;237(4):1233–1243. PubMed CrossRef
- Bogár F, Fülöp L, Penke B. Novel therapeutic target for prevention of neurodegenerative diseases: modulation of neuroinflammation with Sig-1R ligands. Biomolecules. 2022;12(3):363. PubMed CrossRef
- Niitsu T, Iyo M, Hashimoto K. Sigma-1 receptor agonists as therapeutic drugs for cognitive impairment in neuropsychiatric diseases. Curr Pharm Des. 2012;18(7):875–883. PubMed CrossRef
- Izumi Y, Reiersen AM, Lenze EJ, et al. SSRIs differentially modulate the effects of pro-inflammatory stimulation on hippocampal plasticity and memory via sigma 1 receptors and neurosteroids. Transl Psychiatry. 2023;13(1):39. PubMed CrossRef
- Cirrito JR, Wallace CE, Yan P, et al. Effect of escitalopram on Aβ levels and plaque load in an Alzheimer mouse model. Neurology. 2020;95(19):e2666–e2674. PubMed CrossRef
- Lin A, Giuliano CJ, Palladino A, et al. Off-target toxicity is a common mechanism of action of cancer drugs undergoing clinical trials. Sci Transl Med. 2019;11(509):eaaw8412. PubMed CrossRef
- Varalda M, Antona A, Bettio V, et al. Psychotropic drugs show anticancer activity by disrupting mitochondrial and lysosomal function. Front Oncol. 2020;10:562196. PubMed CrossRef
- Song Y, Yang X, Yu B. Repurposing antidepressants for anticancer drug discovery. Drug Discov Today. 2022;27(7):1924–1935. PubMed CrossRef
- Dai XJ, Liu Y, Xiong XP, et al. Tranylcypromine based lysine-specific demethylase 1 inhibitor: summary and perspective. J Med Chem. 2020;63(23):14197–14215. PubMed CrossRef
- Vlachos N, Lampros M, Voulgaris S, et al. Repurposing antipsychotics for cancer treatment. Biomedicines. 2021;9(12):1785. PubMed CrossRef
- Papadopoulos F, Isihou R, Alexiou GA, et al. Haloperidol induced cell cycle arrest and apoptosis in glioblastoma cells. Biomedicines. 2020;8(12):595. PubMed CrossRef
- Pareek S, Huang Y, Nath A, et al. The success story of drug repurposing in breast cancer. In: To KKW, Cho WCS, eds. Drug Repurposing in Cancer Therapy, Chapter 6. Academic Press; 2020:173–190.
- Zhelev Z, Ohba H, Bakalova R, et al. Phenothiazines suppress proliferation and induce apoptosis in cultured leukemic cells without any influence on the viability of normal lymphocytes. Cancer Chemother Pharmacol. 2004;53(3):267–275. PubMed CrossRef
- Rahman T, Patrick C, Ma C, et al. Prolactin and estrogen levels in postmenopausal women receiving aripiprazole augmentation treatment for depression. J Clin Psychopharmacol. 2021;41(1):31–35. PubMed CrossRef
- Barrett AJ. Clinical experience with lithium in aplastic anemia and congenital neutropenia. Adv Exp Med Biol. 1980;127:305–320. PubMed CrossRef
- Chan HS, Freedman MH, Saunders EF. Stimulation of granulopoiesis in vitro and in vivo using lithium in children with chronic neutropenia. Adv Exp Med Biol. 1980;127:293–304. PubMed CrossRef
- Kum JJ, Khan ZA. Mechanisms of propranolol action in infantile hemangioma. Dermatoendocrinol. 2015;6(1):e979699. PubMed CrossRef
- Lamy S, Lachambre MP, Lord-Dufour S, et al. Propranolol suppresses angiogenesis in vitro: inhibition of proliferation, migration, and differentiation of endothelial cells. Vascul Pharmacol. 2010;53(5-6):200–208. PubMed CrossRef
- Chang PY, Huang WY, Lin CL, et al. Propranolol reduces cancer risk: a population-based cohort study. Medicine (Baltimore). 2015;94(27):e1097. PubMed CrossRef
- Archer M, Dogra N, Dovey Z, et al. Role of α- and β-adrenergic signaling in phenotypic targeting: significance in benign and malignant urologic disease. Cell Commun Signal. 2021;19(1):78. PubMed CrossRef
- Lu H, Liu X, Guo F, et al. Impact of beta-blockers on prostate cancer mortality: a meta-analysis of 16,825 patients. OncoTargets Ther. 2015;8:985–990. PubMed CrossRef
- Pantziarka P, Bouche G, Sukhatme V, et al. Repurposing drugs in oncology (ReDO)-propranolol as an anti-cancer agent. Ecancermedicalscience. 2016;10:680. PubMed CrossRef
- Dalwadi DA, Kim S, Schetz JA. Activation of the sigma-1 receptor by haloperidol metabolites facilitates brain-derived neurotrophic factor secretion from human astroglia. Neurochem Int. 2017;105:21–31. PubMed CrossRef
- Patergnani S, Bonora M, Ingusci S, et al. Antipsychotic drugs counteract autophagy and mitophagy in multiple sclerosis. Proc Natl Acad Sci U S A. 2021;118(24):e2020078118. PubMed CrossRef
- Nakagawa C, Yokoyama S, Hosomi K, et al. Repurposing haloperidol for the treatment of rheumatoid arthritis: an integrative approach using data mining techniques. Ther Adv Musculoskelet Dis. 2021;13:X211047057. PubMed CrossRef
- Landén M, Larsson H, Lichtenstein P, et al. Respiratory infections during lithium and valproate medication: a within-individual prospective study of 50,000 patients with bipolar disorder. Int J Bipolar Disord. 2021;9(1):4. PubMed CrossRef
- Singh J, Lanzarini E, Santosh P. Organic features of autonomic dysregulation in paediatric brain injury—clinical and research implications for the management of patients with Rett syndrome. Neurosci Biobehav Rev. 2020;118:809–827. PubMed CrossRef
- Pham T-H, Qiu Y, Zeng J, et al. A deep learning framework for high-throughput mechanism-driven phenotype compound screening and its application to COVID-19 drug repurposing. Nat Mach Intell. 2021;3(3):247–257. PubMed CrossRef
- Austin CP. Repurposing Challenges: Conceptualization of a Research Agenda. Global Forum: Driving Insights to Action. National Center for Advancing Translational Sciences; National Institutes of Health website. https://globalforum.diaglobal.org/issue/june-2020/repurposing-challenges-conceptualization-of-a-research-agenda/. 2020. Accessed February 23, 2023.
- Sahragardjoonegani B, Beall RF, Kesselheim AS, et al. Repurposing existing drugs for new uses: a cohort study of the frequency of FDA-granted new indication exclusivities since 1997. J Pharm Policy Pract. 2021;14(1):3. PubMed CrossRef
- Gebara MA, Lipsey KL, Karp JF, et al. Cause or effect? selective serotonin reuptake inhibitors and falls in older adults: a systematic review. Am J Geriatr Psychiatry. 2015;23(10):1016–1028. PubMed CrossRef
- Gleason CE, Dowling NM, Friedman E, et al. Using predictors of hormone therapy use to model the healthy user bias: how does healthy user status influence cognitive effects of hormone therapy? Menopause. 2012;19(5):524–533. PubMed CrossRef
This PDF is free for all visitors!