Abstract
Approximately 1 in 5 US adults have at least one episode of major depression in their lifetime. Of those who do, the majority will relapse over the long term and many will have psychosocial disabilities and reduced functioning. Over the past century, a range of medications have been developed to treat depression, although some effective medications have been superseded by newer treatments with an improved safety and tolerability profile. A reconsideration of some older medications is warranted until agents with truly novel mechanisms of action and good safety and tolerability profiles are available.
(J Clin Psychiatry 2012;73[10]:e29)
From the Department of Psychiatry and Behavioral Sciences, University of Texas Medical Branch, Galveston.
doi:10.4088/JCP.11096tx3c
This CME activity is expired. For more CME activities, visit cme.psychiatrist.com.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
Abstract
Approximately 1 in 5 US adults have at least one episode of major depression in their lifetime. Of those who do, the majority will relapse over the long term and many will have psychosocial disabilities and reduced functioning. Over the past century, a range of medications have been developed to treat depression, although some effective medications have been superseded by newer treatments with an improved safety and tolerability profile. A reconsideration of some older medications is warranted until agents with truly novel mechanisms of action and good safety and tolerability profiles are available.
(J Clin Psychiatry 2012;73[10]:e29)
Robert M. A. Hirschfeld, MD: From the Department of Psychiatry and Behavioral Sciences, University of Texas Medical Branch, Galveston.
From the Series: A Fresh Look at Monoamine Oxidase Inhibitors for Depression
Supported by an educational grant from Dey Pharmaceuticals.
doi:10.4088/JCP.11096tx3c
Epidemiology of Depression
Depression is a prevalent and pernicious illness with a mean age at onset of 26 years. Depression is considerably more frequent in women than in men (AV 1).1 In the United States, 8% of adults experience a major depressive episode in any 12-month period and about 20% have at least one episode in their lifetime.1
AV 1. Lifetime Prevalence of Depression in US Adults By Age and Gender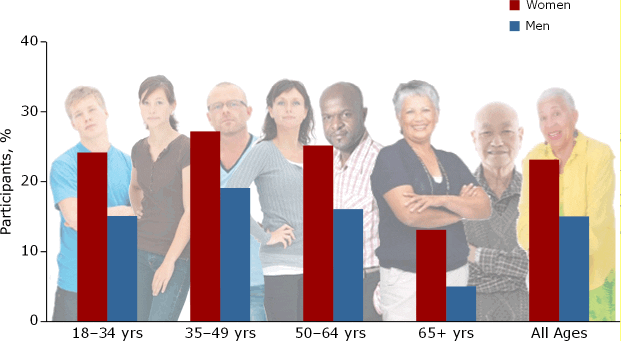
Data from Kessler et al1
The rate of depression is fairly evenly distributed across age groups, but women clearly experience more depression than men over their lifetime. Researchers noted that the risk for major depressive episode is elevated for those who are female, poor, disabled, unemployed, or unmarried.
With regard to clinical course, nearly one-half of all patients experiencing an MDE recovered within 6 months; by 1 year more than two-thirds; and by 2 years, 80%.2 This left 1 in 5 patients continuously depressed for 2 years, most of whom stayed depressed for at least 5 years (12%). Of those who recovered from a depressive episode, most had a recurrence (85% within 2 years). Over the long term (15-year follow-up), only 1 in 9 recovered from their episode of depression and stayed well.3
Depression exacts a heavy psychosocial toll. People with major depression have substantial long-term psychosocial disability and functional impairment4 and have at least some depressive symptoms more than half of the time.5 In fact, the WHO places depression as the third-highest in terms of disease burden in the world.6
A crucial issue to address when evaluating a patient who presents with depression is whether the patient has unipolar depression or bipolar depression. The clinical course and treatment differs substantially for these 2 disorders. The presence of bipolar disorder is easy to miss in depressed patients because many depressed bipolar patients fail to mention prior manic or hypomanic episodes. For example, one study7 of patients diagnosed with major depression found that 20% of participants had experienced mania or hypomania (8% bipolar I and 12% bipolar II).
Treatment Evolution
Treatments for MDD, particularly pharmacologic treatments, have advanced considerably over the last 100 years (AV 2).
AV 2. Timeline: Somatic Treatments for Depression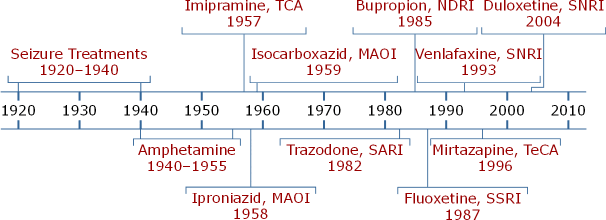
Treatments for depression have evolved from seizure and convulsion treatments, to amphetamines, to the modern era of psychotropics, beginning with imipramine, iproniazid, and isocarboxazid. Next came medications that specifically targeted serotonin, norepinephrine, or dopamine like trazadone, bupropion, fluoxetine, venlafaxine, mirtazapine, and duloxetine. The newer medications have become more popular than TCAs and older MAOIs because of their improved safety and tolerability profiles.
Seizure or convulsive treatments. The first somatic treatments for depression that emerged in the 20th century evolved from serendipitous observations of patients undergoing seizures. The Hungarian psychiatrist Meduna induced seizures in the 1920s and 1930s with Metrazol in patients with schizophrenia. In the 1930s, insulin-induced hypoglycemic coma and seizures were found to calm patients with schizophrenia. In the late 1930s, Cerletti and Bini reported that they had induced seizures in Italy by passing electrical current through the brain—a technique that was much more controllable and safer than previous methods, beginning the first ECT treatments.
Amphetamines. During the 1930s came the first antidepressant medicine of the 20th century—amphetamine. Originally developed for the treatment of asthma and hay fever and subsequently found ineffective for schizophrenia and anxiety, amphetamine was shown to relieve mild depression, particularly anhedonia.8 Positive clinical trials comparing amphetamine with placebo led to inclusion in AMA treatment guidelines for mood elevation in depression in 1937. By the mid 1940s, more than a million amphetamine tablets were being consumed daily in the United States, for not only mood elevation but also weight loss. However, nonprescription abuse of amphetamine spread quickly, and safety concerns, coupled with the introduction of new drugs in the 1960s, led to the decline of amphetamine to treat depression.
Tricyclic antidepressants. During World War II, a search for antimalarial drugs ushered in modern psychopharmacology with the creation of new agents. Modification of the phenothiazine methylene blue led to the synthesis of chlorpromazine in 1950.9 Although ineffective for malaria, chlorpromazine had powerful antihistaminic qualities, particularly sedation and anxiety reduction, and was beneficial in patients with mania and schizophrenia.10 By 1957, a compound closely related chemically to chlorpromazine—imipramine—was found to improve mood in patients with depression.11 These discoveries led to the development of several other TCAs that were widely used. The mechanism of action of the TCAs was to block the reuptake of specific neurotransmitters in the synapse—serotonin and norepinephrine—which produced an antidepressant effect.
Monoamine oxidase inhibitors. In the 1950s, Crane observed that patients with tuberculosis who were treated with the MAOI iproniazid had substantially improved mood.12 Use of iproniazid as a mood elevator was discontinued because of substantial liver toxicity. Similar drugs that were less toxic, such as tranylcypromine and phenelzine, were developed. The mechanism of action of the MAOIs differs from that of the TCAs. The MAOIs increase synaptic catecholamines by inhibiting the enzyme monoamine oxidase, which metabolizes them. Currently, MAOIs may be the most effective medications available to treat depression, but their widespread use has been limited by dietary restrictions and the potential for serious adverse events through drug-drug interactions.
The catecholamine hypothesis. The presumed mechanism of action of the tricyclics and of the MAOIs led scientists to develop a comprehensive theory of the pathophysiology of depression—the catecholamine hypothesis. Simply put, this hypothesis states that depression results from a deficiency of catecholamines (particularly serotonin and norepinephrine) in synapses in the brain. Elation results from an excess of such amines.
Mixed-action antidepressants. In the 1980s and 1990s, medications with other mechanisms of action were developed.13 Trazodone and nefazodone inhibit norepinephrine and serotonin reuptake and act as an antagonist of serotonin. Bupropion has dual inhibition of norepinephrine and dopamine reuptake, and mirtazapine acts as an antagonist of noradrenergic and serotonergic neurotransmission.
Selective serotonin reuptake inhibitors. A major change in the treatment of depression occurred with the development of SSRIs. These medications were specific inhibitors of serotonin reuptake into afferent neurons.14 The FDA approved the first SSRI, fluoxetine in 1987, and the development of several other SSRIs quickly followed.
The SSRIs do not necessarily offer increased efficacy over the TCAs or the MAOIs; they do, however, have attractive properties. Unlike TCAs, SSRIs are not lethal in overdose and do not cause cardiac arrhythmia problems.15 In general, the SSRIs cause less sedation and weight gain and have fewer anticholinergic side effects than earlier antidepressants15 and do not require dietary restrictions. Dose titration is generally simpler. The improved safety and tolerability profiles of SSRIs have led to their widespread use. They surpassed use of all other antidepressant agents by the mid-1990s.
Dual-reuptake inhibitors. In the 1990s, several dual-reuptake inhibitors were developed to inhibit both norepinephrine and serotonin reuptake. The intent was to provide greater efficacy than SSRIs and cause fewer side effects than other agents that affect both serotonin and norepinephrine (eg, MAOIs, TCAs, mixed-action agents) by avoiding additional action at other receptors.16 The best known SNRIs are venlafaxine, which was approved by the FDA in 1994, and duloxetine, approved in 2004.
Conclusion
Depression continues to be a pernicious, debilitating illness. Clinicians now have a wide variety of treatment options for patients who present with depression. Since the TCAs and MAOIs were developed, however, no truly novel mechanisms of action have arisen in the antidepressant armamentarium. Although many treatments were discovered serendipitously, the search for improved safety and tolerability has driven the recent evolution of treatment in an effort to increase patients’ adherence. Some of the current treatment options are considerably underused and clinicians should continue to be alert for new and better treatments.
For Clinical Use
- Monitor patients with depression for any sign of manic or hypomanic symptoms
- Address residual symptoms of depression
- Consider treatments other than SSRIs for patients with depression
Drug Names
bupropion (Wellbutrin, Aplenzin, and others), duloxetine (Cymbalta), fluoxetine (Prozac and others), imipramine (Tofranil and others), isocarboxazid (Marplan), mirtazapine (Remeron and others), phenelzine (Nardil), trazodone (Oleptro and others), tranylcypromine (Parnate and others), venlafaxine (Effexor and others)
Abbreviations
AMA = American Medical Association, ECT = electroconvulsive therapy, FDA = US Food and Drug Administration, MAOI = monoamine oxidase inhibitor, MDD = major depressive disorder, MDE = major depressive episode, NDRI = norepinephrine-dopamine reuptake inhibitor, SARI = serotonin agonist and reuptake inhibitor, SNRI = serotonin-norepinephrine reuptake inhibitor, SSRI = selective serotonin reuptake inhibitor, TCA = tricyclic antidepressant, TeCA = tetracyclic antidepressant, WHO = World Health Organization
References
- Kessler RC, Birnbaum H, Bromet E, et al. Age differences in major depression: results from the National Comorbidity Survey Replication (NCS-R). Psychol Med. 2010;40(2):225–237. PubMed
- Keller MB, Lavori PW, Mueller TI, et al. Time to recovery, chronicity, and levels of psychopathology in major depression: a 5-year prospective follow-up of 431 subjects. Arch Gen Psychiatry. 1992;49(10):809–816. PubMed
- Mueller TI, Leon AC, Keller MB, et al. Recurrence after recovery from major depressive disorder during 15 years of observational follow-up. Am J Psychiatry. 1999;156(7):1000–1006. PubMed
- Fichter MM, Kohlboeck G, Quadflieg N. The Upper Bavarian longitudinal community study 1975-2004, pt 2: long-term course and outcome of depression: a controlled study. Eur Arch Psychiatry Clin Neurosci. 2008;258(8):476–488. PubMed
- Judd LL, Akiskal HS, Maser JD, et al. A prospective 12-year study of subsyndromal and syndromal depressive symptoms in unipolar major depressive disorders. Arch Gen Psychiatry. 1998;55(8):694–700. PubMed
- World Health Organization. The Global Burden of Disease: 2004 Update. Geneva, Switzerland: WHO Press; 2008. http://www.who.int/healthinfo/global_burden_disease/GBD_report_2004update_full.pdf. Accessed February 20, 2012.
- Fiedorowicz JG, Endicott J, Leon AC, et al. Subthreshold hypomanic symptoms in progression from unipolar major depression to bipolar disorder. Am J Psychiatry. 2011;168(1):40–48. PubMed
- Rasmussen N. Making the first anti-depressant: amphetamine in American medicine 1929–1950. J Hist Med Allied Sci. 2006;61(3):288–323. PubMed
- Nassralah HA, Tandon R. Classic Antipsychotic Medications. In: Schatzberg AF, Nemeroff CB, eds. The American Psychiatric Publishing Textbook of Psychopharmacology. 4th ed. Arlington, VA: American Psychiatric Publishing; 2009:533–554. http://psychiatryonline.org/content.aspx?bookid=29§ionid=1356426. Accessed March 2, 2012.
- Frankenburg FR, Baldessarini RJ. Neurosyphilis, malaria, and the discovery of antipsychotic agents. Harv Rev Psychiatry. 2008;16(5):299–307. PubMed
- Ban TA. The role of serendipity in drug discovery. Dialogues Clin Neurosci. 2006;8(3):335–344. PubMed
- Crane GE. The psychiatric side-effects of iproniazid. Am J Psychiatry. 1956;112(7):494–501. PubMed
- Feighner JP. Mechanism of action of antidepressant medications. J Clin Psychiatry. 1999;60(suppl 4):4–11. Full Text
- Zahajszky J, Rosenbaum JF, Tollefson GD. Fluoxetine. In: Schatzberg AF, Nemeroff CB, eds. The American Psychiatric Publishing Textbook of Psychopharmacology. 4th ed. Arlington, VA: American Psychiatric Publishing; 2009:289–306. http://psychiatryonline.org/content.aspx?bookid=29§ionid=1353194. Accessed February 20, 2012
- Ferguson JM. SSRI antidepressant medications: adverse effects and tolerability. Prim Care Companion J Clin Psychiatry. 2001;3(1):22–27. Full Text
- Jain R. Single-action versus dual action antidepressants. Prim Care Companion J Clin Psychiatry. 2004;6(suppl 1):7–11. Full Text
This CME activity is expired. For more CME activities, visit cme.psychiatrist.com.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
CME Background Information
Supported by an educational grant from Dey Pharmaceuticals.
Objective
After completing this educational activity, you should be able to:
- Identify patients who would benefit from treatment with an MAOI and develop a treatment and monitoring plan
Financial Disclosure
The faculty for this CME activity and the CME Institute staff were asked to complete a statement regarding all relevant personal and financial relationships between themselves or their spouse/partner and any commercial interest. The CME Institute has resolved any conflicts of interest that were identified. No member of the CME Institute staff reported any relevant personal financial relationships. Faculty financial disclosures are as follows:
Dr Hirschfeld has received royalties from Jones and Bartlett, has received honoraria from Merck Manual Editorial Board, and has received other financial support from CME Outfitters and Nevada Psychiatric Association.
The Co-Chair for this activity, Lawrence J. Cohen, PharmD, BCPP, FASHP, FCCP, is a consultant for Dey and has received honoraria from and is a member of the speakers/advisory boards for Sunovion and Merck.
The Co-Chair for this activity, David A. Sclar, BPharm, PhD, is a consultant for and has received grant/research support and honoraria from Eli Lilly, Pfizer, GlaxoSmithKline, Forest, Dey, and Bristol-Myers Squibb and is a member of the speakers/advisory board for Eli Lilly.
Accreditation Statement
The CME Institute of Physicians Postgraduate Press, Inc., is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Credit Designation
The CME Institute of Physicians Postgraduate Press, Inc., designates this enduring material for a maximum of 0.5 AMA PRA Category 1 Credit™. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
The American Academy of Physician Assistants (AAPA) accepts certificates of participation for educational activities certified for AMA PRA Category 1 Credit™ from organizations accredited by ACCME or a recognized state medical society. Physician assistants may receive a maximum of 0.5 hours of Category I credit for completing this program.
To obtain credit for this activity, study the material and complete the CME Posttest and Evaluation.
Release, Review, and Expiration Dates
This Psychlopedia activity was published in March 2012 and is eligible for AMA PRA Category 1 Credit™ through March 30, 2015. The latest review of this material was March 2012.
Statement of Need and Purpose
Depression is a leading cause of disability, affecting 121 million people. Due to its heterogeneous nature, depression varies in symptom presentation, course, history, and treatment response. In some cases, the subtype of depression can guide treatment selection to help establish a better response and prognosis. For example, atypical depression, a subtype characterized by mood reactivity with increased appetite or weight gain, hypersomnia, leaden paralysis, or sensitivity to interpersonal rejection, typically has an early onset and chronic course and may be over-represented among patients classified as suffering from treatment-resistant depression. Monoamine oxidase inhibitors (MAOIs) are often effective for patients with atypical depression, but, due to side effects, dietary restrictions, and drug-drug interactions, they are typically recommended only after patients fail to respond to at least several other classes of antidepressants. However, the efficacy of these agents has led to the development of newer formulations and delivery methods, such as the transdermal system, that retain the antidepressant effects but can circumvent some of the adverse events. This unique administration method can also improve patients’ adherence rates, which are typically low with antidepressants. Clinicians need education on the available antidepressant options for depression, including their mechanism of action, and strategies for administering antidepressant treatments to patients with atypical depression to maximize response, minimize side effects, and promote adherence. This activity was designed to meet the needs of participants in CME activities provided by the CME Institute of Physicians Postgraduate Press, Inc., who have requested information on depression.
Disclosure of Off-Label Usage
Dr Hirschfeld has determined that, to the best of his knowledge, chlorpromazine is not approved by the US Food and Drug Administration for the treatment of major depressive disorder, and amphetamine sulfate, iproniazid, and pentylenetetrazol are no longer available in the United States.
Review Process
The entire faculty of the series discussed the content at a peer-reviewed planning session, the Co-Chairs reviewed the activity for accuracy and fair balance, and a member of the External Advisory CME Board who is without conflict of interest reviewed the activity to determine whether the material is evidence-based and objective.
Acknowledgment
This Psychlopedia activity is derived from the planning teleconference series “A Fresh Look at Monoamine Oxidase Inhibitors for Depression,” which was held in December 2011 through February 2012 and is supported by an educational grant from Dey Pharmaceuticals. The opinions expressed herein are those of the faculty and do not necessarily reflect the opinions of the CME provider and publisher or the commercial supporter.


