Article Abstract
Tardive dyskinesia (TD) is an involuntary movement disorder associated with antipsychotic treatment. Because of the serious and potentially irreversible nature of TD, accurate diagnosis is crucial. However, diagnosing TD can be challenging, since the subtle and often fluctuating symptoms can be easily mistaken for symptoms of mental illness or other side effects. Although the risk of developing TD in relation to treatment with second-generation antipsychotics is lower than that associated with first-generation antipsychotics, the risk still exists and may be greater than once believed. Clinicians prescribe antipsychotics for a variety of illnesses and may underestimate the possibility of a patient developing TD, thus missing early signs of the disorder. In this ACADEMIC HIGHLIGHTS, experts review the prevalence, phenomenology, risk factors, and impact of TD, illustrated by case examples, and provide valuable clinical information to guide early recognition and accurate diagnosis.
From the Department of Psychiatry, Texas Tech University Health Sciences Center Medical School, Midland (Dr Jain); the Department of Psychiatry and Molecular Medicine, Hofstra Northwell School of Medicine, Hempstead, and the Department of Psychiatry, The Zucker Hillside Hospital, Glen Oaks, New York, and the Department of Child and Adolescent Psychiatry, Charitש Universitפtsmedizin, Berlin, Germany (Dr Correll)
‘ ‹’ ‹’ ‹’ ‹’ ‹’ ‹’ ‹

This Academic Highlights section of The Journal of Clinical Psychiatry presents the highlights of the planning teleconference series “Tardive Dyskinesia: Recognition, Patient Assessment, and Differential Diagnosis,” which was held in September and October 2017. This report was prepared and independently developed by the CME Institute of Physicians Postgraduate Press, Inc., and was supported by an educational grant from Neurocrine Biosciences, Inc.
The teleconference was chaired by Rakesh Jain, MD, MPH, from the Department of Psychiatry, Texas Tech University Health Sciences Center Medical School, Midland, and private practice, Austin. The faculty was Christoph U. Correll, MD, from the Department of Psychiatry and Molecular Medicine, Hofstra Northwell School of Medicine, Hempstead, and the Department of Psychiatry, The Zucker Hillside Hospital, Glen Oaks, New York, and the Department of Child and Adolescent Psychiatry, Charité Universitפtsmedizin, Berlin, Germany.
Financial Disclosure
Dr Jain is a consultant for Addrenex, Allergan, Avanir, Janssen, Eli Lilly, Lundbeck, Merck, Neos Therapeutics, Neurocrine Biosciences, Otsuka, Pamlab, Pfizer, Shionogi, Shire, Sunovion, Supernus, Takeda, and Teva; has received grant/research support from AstraZeneca, Allergan, Eli Lilly, Lundbeck, Otsuka, Pfizer, Shire, and Takeda; and is a member of the speakers/advisory boards for Addrenex, Alkermes, Allergan, Avanir, Eli Lilly, Forum, Janssen, Lundbeck, Merck, Neos, Neurocrine, Otsuka, Pamlab, Pfizer, Rhodes, Shionogi, Shire, Sunovion, Supernus, Takeda, Teva, and Tris. Dr Correll is a consultant for and has received honoraria from Alkermes, Allergan, Angelini, Gerson Lehrman Group, IntraCellular Therapies, Janssen/Johnson & Johnson, LB Pharma, Lundbeck, Medavante, Medscape, Neurocrine, Otsuka, Pfizer, Rovi, Sunovion, Takeda, and Teva; has received grant/research support from Janssen and Takeda; and has given expert testimony for Bristol-Myers Squibb, Janssen, and Otsuka.
The opinions expressed herein are those of the faculty and do not necessarily reflect the opinions of the CME provider and publisher or the commercial supporter.
J Clin Psychiatry 2018;79(2):nu17034ah1c
To cite: Jain R, Correll CU. Tardive dyskinesia: recognition, patient assessment, and differential diagnosis. J Clin Psychiatry. 2018;79(2):nu17034ah1c.
To share: https://doi.org/10.4088/JCP.nu17034ah1c
© Copyright 2018 Physicians Postgraduate Press, Inc.
Tardive dyskinesia (TD), which literally means “late-appearing distortion” and “impairment of bodily movement,” is a dyskinetic disorder associated with long-term use of antipsychotic medications. Tardive dyskinesia can have serious physical and psychological implications, including increased medical morbidity and poor quality of life.1 One of the most troubling aspects of TD is that this condition may be irreversible and persist, even after the antipsychotic treatment has been discontinued. In this Academic Highlights, Drs Rakesh Jain and Christoph U. Correll discuss the prevalence and phenomenology of TD, providing case examples to illuminate the true burden of this condition, and offer valuable clinical information to guide early recognition and accurate diagnosis.
THE REALITY OF TARDIVE DYSKINESIA
The incidence of TD has decreased since atypical antipsychotics became available, but the reduction of its prevalence may not be as much as was hoped or believed.1 A 4-year prospective community-based study2 involving 352 psychiatric outpatients compared the incidence of TD with first-generation antipsychotics (FGAs) versus second-generation antipsychotics (SGAs). The authors found a greater risk of developing TD with FGAs, but the risk was still present with SGAs (rate-ratio = 0.68; 95% CI, 0.29-1.64), and the incidence of TD remained relatively unchanged since the 1980s. A review3 of 12 studies that reported on TD prevalence and incidence associated with FGAs versus SGAs confirmed that TD occurs significantly less frequently in patients treated with SGAs. The TD prevalence rate for SGAs was 13.1% compared with a prevalence rate of 32.4% for FGAs (P < .0001). The annualized incidence rate of TD across all studies was 3.9% for SGAs and 5.5% for FGAs. The annualized incidence rate of TD across the 6 direct comparison studies was 4.2% for SGAs and 5.5% for FGAs. Dr Correll pointed out that methodological reasons may explain why the incidence rates appear drastically lower than the prevalence rates. These data were obtained from long-term studies whereas the prevalence data were obtained from cross-sectional studies, for which patients tend to have had longer medication exposure. Due to the lower requirements for inclusion in these studies, participants often are more severely ill, have more comorbidities, and have greater chronicity, duration, and treatment refractoriness.3
More recently, Carbon and colleagues4 conducted a meta-analysis that evaluated the prevalence of TD in 41 studies that investigated TD rates during treatment with FGAs and/or SGAs. The total study population included 11,493 patients with a mean age of 42.8 years; 66.4% were male and 77.1% were diagnosed with a schizophrenia spectrum disorder. Across all studies, which used standardized rating scales to assess for TD, the global mean TD prevalence was 25.3% (95% CI, 22.7%-28.1%). Tardive dyskinesia rates were significantly lower with current SGA treatment (20.7%; 95% CI, 16.6%-25.4%) compared with current FGA treatment (30%; 95% CI, 26.4%-33.8%; P = .002).
Although the evidence from these studies2-4 seems to establish that SGAs carry a reduced risk of TD compared with FGAs, Dr Correll warned that prevalence studies such as these are often flawed by methodological problems. He explained that the current treatment may or may not be causally related to the presenting syndrome, but patients could also possibly have developed TD during prior treatment and been switched to an SGA to reduce the severity or risk of TD. Thus, the results of these studies need to be interpreted carefully. Furthermore, as these studies illustrate, although the rates of TD may be lower in patients treated with SGAs, TD is still a serious risk during treatment with these medications. For example, the meta-analysis conducted by Carbon et al4 found a TD prevalence of 7.2% in the 4 studies that restricted the sample to schizophrenia patients without a lifetime history of FGA exposure. This means that approximately 1 out of every 14 individuals taking an SGA is likely to develop TD. Although TD rates are less clear in patients diagnosed with disorders other than schizophrenia, this finding is troubling, according to Dr Correll, because newer antipsychotic drugs are increasingly being used to treat conditions other than psychosis, such as mood disorders, autism, and attention-deficit/hyperactivity disorder.5 Clinicians may have developed a false sense of security and become less vigilant about necessary safeguards against TD, such as educating patients about the risk of TD, obtaining informed consent prior to initiating treatment, and monitoring for early signs of abnormal movement.6

- Remember that tardive dyskinesia (TD) is a risk with both first-generation and second-generation antipsychotics.
- Screen all patients receiving antipsychotic treatment for abnormal movements at baseline and at regular intervals throughout treatment.
- Avoid high doses of or unnecessary treatment with antipsychotic agents in patients with known risk factors for TD.
- Use objective, quantitative rating scales such as the Abnormal Involuntary Movement Scale (AIMS) in conjunction with established diagnostic criteria to accurately identify patients with TD as early as possible.
Case presentation. Ms A is a 47-year-old married woman who was diagnosed with bipolar disorder over 20 years ago. She experiences several mood episodes a year. Ms A’s condition stabilized over a decade ago after treatment with lithium, divalproex, sertraline, and quetiapine. However, about 7 years ago, she began to gain weight, and after gaining 10 pounds, quetiapine was discontinued and she was started on aripiprazole. Her symptoms were responding well to the switch. However, after about a year on this therapy, one night she experienced a new sensation as if her “legs were jumping” and both of her big toes “wanted to stretch out” for no apparent reason. She thought nothing of it and went to sleep, but this problem persisted, so she went to see her psychiatrist who provisionally diagnosed her with restless legs syndrome (RLS) and referred her to a neurologist.
The neurologist agreed with the diagnosis and started her on a prodopaminergic treatment specific for RLS; unfortunately, this made her twitching symptoms worse. At that time, she started noticing that her “tongue darts in and out” and her eyebrows “jump up and down for no reason.” Ms A consulted another neurologist, who diagnosed her with TD and discontinued the aripiprazole without consulting the psychiatrist. The movement difficulties initially worsened significantly, her mood destabilized, and a severe mixed episode emerged.
Ms A’s jaw clenching and snapping became so severe that, over time, she needed 4 major surgeries to repair dental problems secondary to cracked teeth. She was extremely frightened by this experience and feared being permanently disabled because of her movement disorder and her worsening mood disorder. Her patient chart had no documented informed consent about the risk of TD, and there was no documentation that any formal screening for involuntary movement was ever conducted.
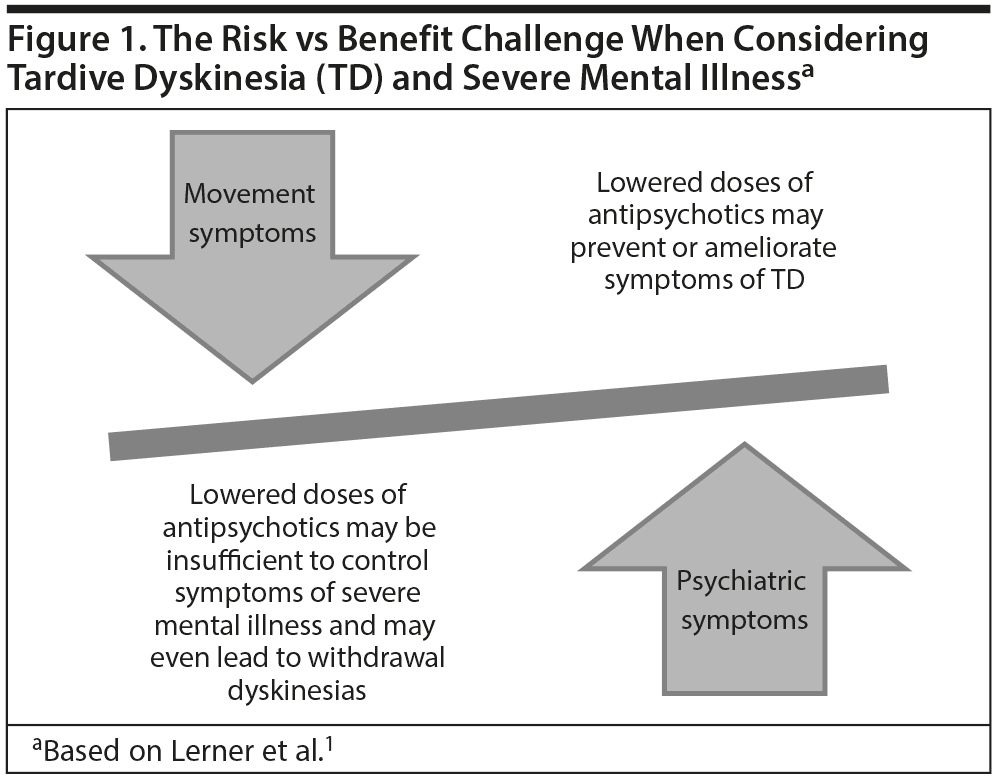
Case lesson. Dr Jain commented that Ms A’s case illustrates the manner in which TD may emerge in a patient and the impact it may have on a patient’s life, including considerable psychosocial impairment and distress. Her case also highlights the difficult position clinicians often confront—having to weigh the risks of withdrawing an antipsychotic treatment, which could potentially improve TD but worsen the psychiatric disorder, or continue the antipsychotic treatment to maintain mental health but potentially maintaining or worsening TD (Figure 1).1 Yet, as in Ms A’s case, the decision can become even more complicated, because, in some cases, withdrawing the antipsychotic actually leads to (at least temporarily) worsening dyskinesia. Dr Jain concluded that Ms A’s case can be seen as a cautionary tale, reminding clinicians that every antipsychotic carries some risk of TD. Thus, informed consent should always be obtained and documented in the charts of all patients on antipsychotic therapy, and patients should be assessed for potential TD at the start of treatment and at regular time intervals, but at least at the earliest sign of abnormal or involuntary movements.
Phenomenology
Dr Correll explained that TD presents clinically as involuntary, repetitive, but irregular choreiform movements that mostly appear in the oral, lingual, and buccal regions.1 Movements commonly include tongue protruding, puckering, chewing, and grimacing. Less often, movements in the hands, legs, feet, and trunk are also present. Tardive motor disturbances resulting from antipsychotic treatment can also manifest as tardive dystonia or tardive akathisia, which are less common than TD but can occur simultaneously with TD. Movement disorders, added Dr Correll, can occur in patients with psychiatric disorders who have not been treated with antipsychotic agents. In fact, spontaneous, involuntary movements, such as those that characterize TD, have been observed in patients with schizophrenia since long before the introduction of antipsychotics.1 However, he pointed out that treatment with dopamine-blocking agents, such as antipsychotics and metoclopramide, increases the risk that TD will develop.
Pathophysiology
The pathophysiology of TD is not fully understood, but several hypotheses have been explored. Drs Jain and Correll discussed several of the most prominent theories regarding the pathophysiology of TD. The postsynaptic dopamine receptor hypersensitivity theory holds that exposure to antipsychotic drugs, which are known to block dopamine receptors, leads these receptors to become hypersensitive.7 The nigrostriatal dopaminergic pathway is known to be central to the control of motor functions,8 and a significant correlation (P = .024) has been observed between the binding potential of antipsychotic drugs to dopamine receptors in the substantia nigra of patients with schizophrenia and the experience of drug-induced motor symptoms.9
An additional hypothesis suggests that antipsychotic treatment causes damage to neurons containing glutamic acid decarboxylase, which leads to γ-aminobutyric acid (GABA) insufficiency in the anatomic loop that controls motor function. Much of the support7,10 for this hypothesis, however, is based on animal models; evidence from human studies is limited. Degeneration of striatal cholinergic interneurons has also been implicated as a potential mechanism underlying TD. Animal studies have observed reduced cholinergic cells in the striatum after exposure to antipsychotic drugs, particularly in brain regions that are critical in the regulation of oral movements.11 Reduced cholinergic neurons have been observed in the basal ganglia of patients with schizophrenia.7 Another hypothesis proposes that TD is caused by oxidative stress created from chronic antipsychotic use. In addition, noted Dr Correll, a subgroup of patients have a genetic vulnerability that predisposes them to developing TD when receiving antipsychotics. Tardive dyskinesia has been associated with several different polymorphisms of dopamine receptor genes, the dopamine transporter gene, and the manganese superoxide dismutase gene.12
Risk Factors
Dr Correll stated that several risk factors for TD have been identified, and an awareness of known risk factors is important because TD is an often irreversible condition; therefore, the best strategy for combating TD is prevention (Figure 2).13,14 Many of the risk factors for TD are associated with antipsychotic medications themselves. For example, the risk of TD has been found to be greater with FGAs than with newer SGAs. A meta-analysis by O’ Brien15 compared the risk of TD in older adults treated with FGAs versus SGAs. O’ Brien reported that after 1 year of treatment, the risk of TD was 3 times lower in the patients being treated with SGAs compared with those being treated with FGAs. After 3 years of treatment, the risk of TD associated with FGAs was quite high, with 57% of participants exhibiting probable TD (95% CI, 45.3-69.1).15 The risk of TD also appears to increase with higher antipsychotic doses16,17 and to be greater in patients who experience extrapyramidal symptoms as an early side effect of antipsychotic treatment.18,19
Older patients are particularly susceptible to TD,17,20,21 added Dr Correll, although the risk seems to be greater with FGAs than with SGAs.20 Woerner and colleagues21 assessed the risk of TD in 261 antipsychotic-naive patients over the age of 55 from the point at which they began treatment with an FGA. The cumulative rates of TD were 25% after 1 year of antipsychotic treatment, 34% after 2 years of treatment, and 53% after 3 years of treatment. These rates were 3 to 5 times higher than those that had been found in separate studies for younger patients, despite the fact that the older participants had received lower doses and had shorter durations of antipsychotic medication exposure.21 In this elderly patient population, risk factors identified for TD included a history of electroconvulsive therapy, higher mean daily and cumulative antipsychotic doses, and early signs of extrapyramidal symptoms.21 Woerner and colleagues20 conducted a similar study to assess the risk of TD in elderly patients treated with the SGAs risperidone and olanzapine. The cumulative TD rate with risperidone was 5.3% after 1 year and 7.2% after 2 years. With olanzapine, the cumulative TD rate was 6.7% after 1 year and 11.1% after 2 years. Risk for TD was higher in women, African Americans, and patients without past antidepressant treatment or with FGA cotreatment.
Female sex22 and non-white ethnicity19 have also been found to be associated with an increased risk of TD. In addition, Drs Jain and Correll agreed that antipsychotic agents are used to treat a number of conditions, and a patient’s diagnosis and certain comorbid conditions may affect their likelihood of developing TD. Individuals experiencing psychosis, particularly associated with schizophrenia, and individuals with intellectual disabilities have been found to experience elevated rates of TD,22,23 as have individuals with mood disorders and symptoms.24 Individuals with diabetes or human immunodeficiency virus (HIV) may be also be at greater risk for TD.13
Case Practice Question
Case 1. Fred is a 44-year-old African American man with a diagnosis of schizoaffective disorder. He presents with new onset involuntary movements, which emerged after 18 months of treatment with an SGA. ALL of the following of Fred’s characteristics are established risk factors for TD, EXCEPT:
- Male sex
- African American race
- Schizoaffective disorder diagnosis
- Long-term treatment with an SGA
Discussion of Case Practice Question
Preferred response is a. Male sex.
Being male is not an established risk factor for TD. However, women, especially of older age, have an elevated risk of developing TD.
Patient Experience of TD
The experience of TD can have a severe impact on a patient’s everyday functioning and quality of life. Dr Correll explained that the effect of TD on everyday functioning can be categorized into either medical or psychological domains.
Impairments in medical domains. The hallmark motor symptoms of TD can lead to considerable impairment, and Dr Correll emphasized that the topography of the symptoms should be considered in order to determine its impact on functioning. Orolingual TD can adversely affect communication, respiration, and even swallowing and food intake.5 Dysarthria is common in TD and leads to difficulty in articulation, which decreases verbal communication when orolingual TD symptoms are present. Orolingual TD can also cause problems with dentition through attrition or abfraction, as well as to temporomandibular joint pain and myalgia. Furthermore, orolingual TD can be associated with swallowing difficulties and problems with food intake.5
Upper extremity TD can impair an individual’s ability to carry out daily activities required to live independently. Specifically, the reduced functioning in the upper extremities leads to impairments in fine motor skills and complicates simple tasks such as writing, handling money, using technology, preparing food, and practicing self-care. Truncal or lower-extremity TD can interfere with gait and posture or reduce postural stability, which can lead to poor balance and increase the patient’s fall risk. Furthermore, strength, power flexibility, physical capacity, and exercise can be impaired.5
Impairments in psychological domains. The psychological impairments of TD, clarified Dr Correll, include lack of awareness of the TD-associated involuntary movements, cognitive dysfunction, and anxiety resulting from experiencing the stigma associated with this disorder. Individuals with TD have also been found to have lower cognitive capacity.5
Perhaps due to the cognitive impairments associated with TD and severe mental illness in general, many patients lack awareness of their TD.25 In fact, as many as one-half to two-thirds of patients with TD are unaware of their involuntary movements.25-27
The association between cognitive dysfunction and TD was assessed in a study28 involving 64 outpatients with schizophrenia. The authors found a significant association between orofacial TD and cognitive dysfunction as measured by the Trail Making Test B29 (P < .01). In a small study of patients with schizophrenia, Baribeau and colleagues30 found that patients experiencing formal thought disorders presented more severe orofacial dyskinesia and greater attentional/cognitive deficits. In a larger study, Wu et al31 assessed 206 patients with chronic schizophrenia, 102 of whom had TD, and 104 controls with the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS).32 The participants with TD scored significantly lower on RBANS visuospatial/constructional, attentional, and total index scores (P < .001).31
Finally, Dr Correll pointed out that TD has been associated with emergence of worrisome issues, such as suicidality and social isolation when patients feel scrutinized and looked at by others around them.5,33
Case presentation. Mr C is 58 years old and was diagnosed with schizophrenia nearly 40 years ago. He has never married and has been on permanent disability for many decades. Mr C tried numerous medications for his schizophrenia. He started with haloperidol, and his last treatment was with risperidone, which he calls a “gift from heaven.” For the first time in years, he was free of hallucinations and was actually able to live independently in an apartment his parents left him in their will. Mr C improved so much that he became a volunteer peer-guidance counselor at the community mental health center where he receives treatment. As a result, his social life improved significantly.
However, 10 years ago, Mr C noticed something odd while he was shaving. His face was twitching, and the movements were so sudden and unexpected that he accidentally nicked himself. He initially thought that tiredness must be causing his face to have tics, and he assumed they would go away in an hour or so. The abnormal movements, in fact, have not gone away, and Mr C has been affected by these abnormal movements every single day for over a decade. These movements are now affecting his lips, tongue, face, neck, fingers, wrists, shoulder, pelvis, and feet. Mr C has stumbled and nearly fallen on several occasions because his hyperkinetic body movements are so significant. His neck is now in a permanently twisted position. When he goes out in public, he notices that people, particularly children, stare and avoid him. Mr C’s psychiatrist diagnosed him with TD and offered the option of reducing his risperidone dose, but he adamantly refused, stating, “I don’ t want to go crazy again and end up in a hospital.”
Case lesson. Dr Jain explained that Mr C’s case illustrates the enormous physical and emotional burden that TD places on a patient’s life. He emphasized that the psychosocial impact of these movement difficulties, both in social settings as well as in mobility, can be significant and must be proactively assessed and addressed. Additionally, TD can affect multiple areas of the body, including the lips, tongue, jaws, muscles of facial expression, upper extremities, trunk, and lower extremities. Another important lesson to learn from Mr C’s case is that TD can emerge after many years of antipsychotic treatment, so clinicians must remain vigilant. Although the antipsychotic agent may have triggered the TD, reducing and stopping these medications should be considered carefully, as it may destabilize patients’ mental health.
Case Practice Question
Case 2. Zelda is a 62-year-old white woman who works as a receptionist and administrative assistant in a law office. She has been diagnosed with major depressive disorder and received an SGA as augmentation to an antidepressant due to treatment-resistant depression. She developed TD 2 years after starting the SGA, which mainly manifested as involuntary orofacial movements with less severe truncal and upper extremity movements.
What feature best describes the most relevant factor for the functional impact of TD symptoms on Zelda’s instrumental activities?
- Severity of TD movements
- Self-awareness of TD movements
- Awareness of the TD symptoms by others around the patient
- Movement location relative to the instrumental use of the affected muscle groups
Discussion of Case Practice Question
Preferred response is d. Movement location relative to the instrumental use of the affected muscle groups.
Although a-c are also potentially relevant factors for the determination of the functional impact of TD on Zelda’s instrumental activities, the location of the TD symptoms, and how much these muscles are relevant for the instrumental activities that she undertakes are likely most relevant. Because Zelda’s job requires her to answer the telephone and speak with clients, her orofacial TD may cause considerable impairment. The TD symptoms in her extremities may make it difficult for her to place calls, use a computer, or even write notes or messages.
RECOGNITION AND DIAGNOSIS
The symptoms of TD can be difficult to recognize and diagnose because they can be subtle and fluctuating, often have an insidious onset, and can easily be mistaken for symptoms of the patient’s mental disorder. Dr Jain commented that TD symptoms can emerge after 3 months of antipsychotic treatment (and occasionally even with much shorter exposure), after several years, or after the medication is discontinued. For the majority of patients, once symptoms develop and evolve into TD, they will stabilize but maintain a chronic yet fluctuating course.34 The American Psychiatric Association (APA) has recommended that all patients being treated with an antipsychotic be monitored for TD,35 with assessments for abnormal involuntary movements conducted at baseline, at least every 6 months for patients taking FGAs, and at least every 12 months for patients taking SGAs. Objective, quantitative rating scales should be used in conjunction with established diagnostic criteria to accurately identify patients with TD as early as possible.
Dr Jain remarked that the Abnormal Involuntary Movement Scale (AIMS)36 is a well-recognized and important screening instrument for identifying the severity and location of abnormal involuntary movements, but it cannot be used alone to diagnose TD. Rather, this tool must be used in conjunction with diagnostic criteria for TD to make a diagnosis. The AIMS consists of 7 items, each of which measures the severity of involuntary movements in a different anatomic area, such as the face, extremities, or trunk. Multiple diagnostic criteria have been put forth for TD. The Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5),37 contains an entire chapter on medication-induced movement disorders, which states that the diagnosis should be based on the presence of involuntary athetoid or choreiform movements that have lasted at least a few weeks and that have developed in association with the use of antipsychotic treatment of at least a few months’ duration.
According to Dr Jain, one set of diagnostic criteria that is still very important in the field is the Schooler-Kane criteria,38 which were published in 1982. These criteria were originally developed to be used in research settings in an attempt to reduce diagnostic heterogeneity that was common in clinical trials, thus making results more consistent and easily comparable.38 However, these criteria are easily used in routine practice and consist of 3 prerequisites that must be met in order to render a probable diagnosis of TD. The first prerequisite is that the patient must have at least 3 months of cumulative exposure to antipsychotic medication, but these 3 months can be broken up and do not have to be continuous. The next prerequisite is that the patient must exhibit abnormal, involuntary movements that meet either of the following criteria: (a) the movements are present in one or more body area and are of at least moderate severity (an AIMS score ≥ 3), or (b) the movements are mild (an AIMS score ≥ 2) but are present in 2 or more body areas. The final prerequisite is that the patient must be free of other conditions that might produce abnormal involuntary movements. However, Dr Jain emphasized that even if some or all of these criteria are met, sometimes the diagnosis is not as clear-cut whether a patient absolutely does or does not have TD. The Schooler-Kane criteria are helpful for these situations, because they break the diagnosis down into the following 6 categories to reflect the progressive nature of TD: (1) probable TD, (2) masked probable TD, (3) transient TD, (4) withdrawal TD, (5) persistent TD, and (6) masked persistent TD (Table 1).38
Glazer et al39 also proposed diagnostic criteria for TD. Their criteria, which were published in 1993, state that a diagnosis of TD should be based on the results of 2 assessments with the AIMS administered at the beginning and end of a clinical visit. For patients to be diagnosed with TD, they must receive a total AIMS score of 3 or greater, with at least one body area rated as 2 or greater, indicating at least mild severity. They must receive these scores on at least 2 successive visits.
Because TD is a diagnosis of exclusion, other conditions associated with abnormal movement must be ruled out.40 Dr Correll explained that clinicians should highly suspect TD if a patient exhibits tongue protrusions or “fly-catcher tongue,” smacking or puckering of the lips, opening or closing of the mouth or jaw, or lateral movements of the jaw.41 However, he warned that clinicians should think twice about the presence of TD if they observe any rapidly evolving movements, vivid “piano player” movements, isolated dyskinesia of the lower body, or a localized, fixed dystonia only. The symptoms of TD must be distinguished from other involuntary movement syndromes, such as dystonia, tics, tremor, stereotypies, and myoclonus, that can occur alone or coexist with TD.41 Other conditions that may resemble TD that must be ruled out to make an accurate diagnosis include Huntington disease, Sydenham chorea, neurologic complications of metabolic disorders, and drug-induced dyskinesias.40,41
Case Practice Question
Case 3. Sally is a 62-year-old white woman who has been taking adjunctive SGA treatment for refractory depression for 8 weeks. She reports jerky movements in her face, muscles, and tongue, and you suspect signs of TD.
Which of the following steps is most appropriate at this stage of assessment?
- Immediately stop the SGA.
- Immediately increase the dose of the SGA to suppress the abnormal movements.
- Conduct an examination for abnormal movements using the AIMS.
- Schedule a follow-up for 3 months after the antipsychotic was initiated to evaluate for abnormal movements, at which point a diagnosis of TD can be reliably made.
Discussion of Case Practice Question
Preferred response is c. Conduct an examination for abnormal movements using the AIMS.
Changing the atypical antipsychotic dose is not a safe choice because it may destabilize Sally’s psychiatric symptoms. Although some diagnostic criteria38 for TD require 3 months of antipsychotic exposure, TD can emerge sooner in some patients, and Sally has several risk factors, including older age, female sex, and the presence of mood symptoms. Conducting an AIMS examination to better understand and quantify the movement abnormalities is indicated at this time.
CONCLUSION
Despite optimism that the prevalence of TD would improve with the introduction of atypical antipsychotics, the reality has been very different. Tardive dyskinesia remains a very real problem, and clinicians must be proactive to enable their patients to both recover from their psychiatric illnesses and remain free from this serious and potentially irreversible movement disorder. These goals can be accomplished by assessing patients for signs of movement disorders at baseline and throughout treatment, screening patients for TD risk factors, and using reliable rating scales and diagnostic criteria to identify this disorder as early as possible.
Published online: March 20, 2018.
Disclosure of off-label usage: Drs Jain and Correll have determined that, to the best of their knowledge, no investigational information about pharmaceutical agents that is outside US Food and Drug Administration-approved labeling has been presented in this activity.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
REFERENCES
1. Lerner PP, Miodownik C, Lerner V. Tardive dyskinesia (syndrome): current concept and modern approaches to its management. Psychiatry Clin Neurosci. 2015;69(6):321-334. PubMed CrossRef
2. Woods SW, Morgenstern H, Saksa JR, et al. Incidence of tardive dyskinesia with atypical versus conventional antipsychotic medications: a prospective cohort study. J Clin Psychiatry. 2010;71(4):463-474. PubMed CrossRef
3. Correll CU, Schenk EM. Tardive dyskinesia and new antipsychotics. Curr Opin Psychiatry. 2008;21(2):151-156. PubMed CrossRef
4. Carbon M, Hsieh C-H, Kane JM, et al. Tardive dyskinesia prevalence in the period of second-generation antipsychotic use: a meta-analysis. J Clin Psychiatry. 2017;78(3):e264-e278. PubMed CrossRef
5. Strassnig M, Rosenfeld A, Harvey PD. Tardive dyskinesia: motor system impairments, cognition and everyday functioning [published online ahead of print September 7, 2017]. CNS Spectr. 2017:1-8. PubMed CrossRef
6. Kim J, MacMaster E, Schwartz TL. Tardive dyskinesia in patients treated with atypical antipsychotics: case series and brief review of etiologic and treatment considerations. Drugs Context. 2014;3:212259. PubMed CrossRef
7. Margolese HC, Chouinard G, Kolivakis TT, et al. Tardive dyskinesia in the era of typical and atypical antipsychotics, part 1: pathophysiology and mechanisms of induction. Can J Psychiatry. 2005;50(9):541-547. PubMed CrossRef
8. Crocker AD. The regulation of motor control: an evaluation of the role of dopamine receptors in the substantia nigra. Rev Neurosci. 1997;8(1):55-76. PubMed CrossRef
9. Tuppurainen H, Kuikka JT, Viinamפki H, et al. Extrapyramidal side-effects and dopamine D(2/3) receptor binding in substantia nigra. Nord J Psychiatry. 2010;64(4):233-238. PubMed CrossRef
10. Casey DE. Tardive dyskinesia: pathophysiology and animal models. J Clin Psychiatry. 2000;61(suppl 4):5-9. PubMed
11. Grimm JW, Chapman MA, Zahm DS, et al. Decreased choline acetyltransferase immunoreactivity in discrete striatal subregions following chronic haloperidol in rats. Synapse. 2001;39(1):51-57. PubMed CrossRef
12. Chang FC, Fung VS. Clinical significance of pharmacogenomic studies in tardive dyskinesia associated with patients with psychiatric disorders. Pharmgenomics Pers Med. 2014;7:317-328. PubMed CrossRef
13. Jankelowitz SK. Treatment of neurolept-induced tardive dyskinesia. Neuropsychiatr Dis Treat. 2013;9:1371-1380. PubMed CrossRef
14. Solmi M, Pigato G, Kane J, et al. Clinical risk factors for the development of tardive dyskinesia [published online ahead of print February 5, 2018]. J Neurol Sci. PubMed CrossRef
15. O’ Brien A. Comparing the risk of tardive dyskinesia in older adults with first-generation and second-generation antipsychotics: a systematic review and meta-analysis. Int J Geriatr Psychiatry. 2016;31(7):683-693. PubMed CrossRef
16. Tarsy D, Lungu C, Baldessarini RJ. Epidemiology of tardive dyskinesia before and during the era of modern antipsychotic drugs. Handb Clin Neurol. 2011;100:601-616. PubMed CrossRef
17. Achalia RM, Chaturvedi SK, Desai G, et al. Prevalence and risk factors associated with tardive dyskinesia among Indian patients with schizophrenia. Asian J Psychiatr. 2014;9:31-35. PubMed CrossRef
18. Novick D, Haro JM, Bertsch J, et al. Incidence of extrapyramidal symptoms and tardive dyskinesia in schizophrenia: thirty-six-month results from the European schizophrenia outpatient health outcomes study. J Clin Psychopharmacol. 2010;30(5):531-540. PubMed CrossRef
19. Tenback DE, van Harten PN, van Os J. Non-therapeutic risk factors for onset of tardive dyskinesia in schizophrenia: a meta-analysis. Mov Disord. 2009;24(16):2309-2315. PubMed CrossRef
20. Woerner MG, Correll CU, Alvir JMJ, et al. Incidence of tardive dyskinesia with risperidone or olanzapine in the elderly: results from a 2-year, prospective study in antipsychotic-naׯve patients. Neuropsychopharmacology. 2011;36(8):1738-1746. PubMed CrossRef
21. Woerner MG, Alvir JM, Saltz BL, et al. Prospective study of tardive dyskinesia in the elderly: rates and risk factors. Am J Psychiatry. 1998;155(11):1521-1528. PubMed CrossRef
22. Merrill RM, Lyon JL, Matiaco PM. Tardive and spontaneous dyskinesia incidence in the general population. BMC Psychiatry. 2013;13(1):152. PubMed CrossRef
23. Matson JL, Fodstad JC, Neal D, et al. Risk factors for tardive dyskinesia in adults with intellectual disability, comorbid psychopathology, and long-term psychotropic use. Res Dev Disabil. 2010;31(1):108-116. PubMed CrossRef
24. Kane JM. Tardive dyskinesia in affective disorders. J Clin Psychiatry. 1999;60(suppl 5):43-49. PubMed
25. Emsley R, Niehaus DJH, Oosthuizen PP, et al. Subjective awareness of tardive dyskinesia and insight in schizophrenia. Eur Psychiatry. 2011;26(5):293-296. PubMed CrossRef
26. Macpherson R, Collis R. Tardive dyskinesia: patients’ lack of awareness of movement disorder. Br J Psychiatry. 1992;160(1):110-112. PubMed CrossRef
27. Chong SA, Remington G, Mahendran R, et al. Awareness of tardive dyskinesia in Asian patients with schizophrenia. J Clin Psychopharmacol. 2001;21(2):235-237. PubMed CrossRef
28. Waddington JL, O’ Callaghan E, Larkin C, et al. Cognitive dysfunction in schizophrenia: organic vulnerability factor or state marker for tardive dyskinesia? Brain Cogn. 1993;23(1):56-70. PubMed CrossRef
29. Reitan R. Validity of the Trail Making Test as an indicator of organic brain damage. Percept Mot Skills. 1958;8(3):271-276. CrossRef
30. Baribeau J, Laurent JP, Décary A. Tardive dyskinesia and associated cognitive disorders: a convergent neuropsychological and neurophysiological approach. Brain Cogn. 1993;23(1):40-55. PubMed CrossRef
31. Wu JQ, Chen DC, Xiu MH, et al. Tardive dyskinesia is associated with greater cognitive impairment in schizophrenia. Prog Neuropsychopharmacol Biol Psychiatry. 2013;46:71-77. PubMed CrossRef
32. Randolph C, Tierney M, Mohr E, et al. The Repeatable Battery for the Assessment of Neuropsychological Status (RBANS): preliminary clinical validity. J Clin Exp Neuropsychol. 1998;20(3):310-319. PubMed CrossRef
33. Yassa R, Jones BD. Complications of tardive dyskinesia: a review. Psychosomatics. 1985;26(4):305-307, 310, 312-313. PubMed CrossRef
34. Waln O, Jankovic J. An update on tardive dyskinesia: from phenomenology to treatment. Tremor Other Hyperkinet Mov (N Y). 2013;3:tre-03-161-4138-1. PubMed CrossRef
35. Lehman AF, Lieberman JA, Dixon LB, et al. Practice guideline for the treatment of patients with schizophrenia, second edition. Am J Psychiatry. 2004;161(2 suppl):1-56. PubMed
36. Guy W. ECDEU Assessment Manual for Psychopharmacology. Washington, DC: US Department of Health, Education, and Welfare; 1976.
37. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. Fifth Edition. Washington, DC: American Psychiatric Publishing; 2013.
38. Schooler NR, Kane JM. Research diagnoses for tardive dyskinesia. Arch Gen Psychiatry. 1982;39(4):486-487. PubMed CrossRef
39. Glazer WM, Morgenstern H, Doucette JT. Predicting the long-term risk of tardive dyskinesia in outpatients maintained on neuroleptic medications. J Clin Psychiatry. 1993;54(4):133-139. PubMed
40. Citrome L, Dufresne R, Dyrud JM. Tardive dyskinesia: minimizing risk and improving outcomes in schizophrenia and other disorders. Am J Manag Care. 2007;12(Decision Maker News-Tardive Dyskinesia: Minimizing Risk and Improving Outcomes in Schizophrenia and Other Disorders 1-Decis). Available at https://ajmc.s3.amazonaws.com/_media/_pdf/A178%20TD.pdf. Accessed February 14, 2018.
41. Tardive dyskinesia: summary of a Task Force Report of the American Psychiatric Association. By the Task Force on Late Neurological Effects of Antipsychotic Drugs. Am J Psychiatry. 1980;137(10):1163-1172. PubMed CrossRef
This PDF is free for all visitors!