There is a substantial body of literature comprising anecdotal material and descriptions of uncontrolled and randomized controlled trials addressing the use of subanesthetic doses of ketamine for the off-label treatment of major depressive episodes. This article examines diagnostic indications for the off-label use of ketamine as an antidepressant and possible contexts in which ketamine may be trialled. Ketamine is indicated in patients who are in a major depressive episode. Most of the research data have been collected from patients with major depressive disorder, but patients with bipolar depression have also been studied. Ketamine is effective in both diagnostic groups, but its benefits are impermanent, perhaps more so in bipolar depression. There are several contexts within this diagnostic framework when a ketamine trial may be considered. These include severe depression and depression that is refractory to conventional antidepressant medication; this is because there is little purpose in trialling an experimental treatment in patients who are less severely ill and those who are antidepressant responsive. More importantly, ketamine has demonstrated efficacy in the rapid reduction of suicidal symptoms and can therefore be trialled when rapid reduction in suicidality is necessary. Likewise, because of its swift and dramatic antidepressant action, it can be trialled in patients in whom improvement is urgently desired in order to allow the patient to attend to pressing life circumstances. Some data suggest that the use of ketamine early during the course of an antidepressant trial, or as anesthesia during electroconvulsive therapy, can improve early antidepressant outcomes. It is not clear whether the presence of psychotic symptoms is a contraindication for ketamine use. Issues related to these indications and contexts are briefly discussed.
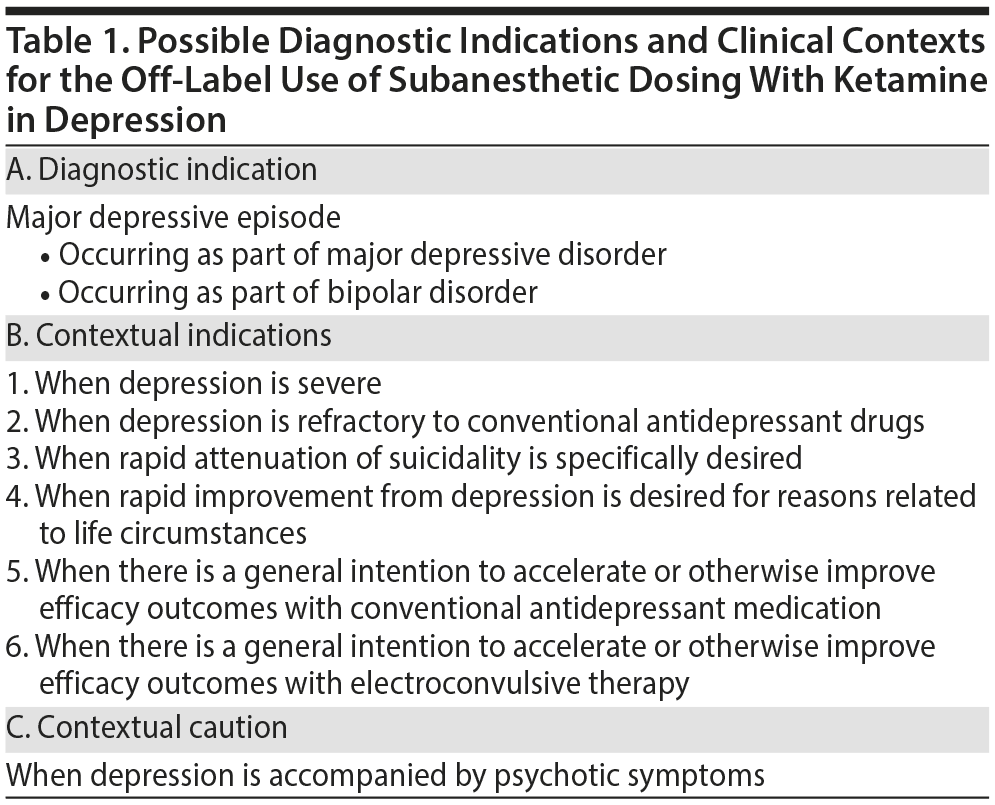
- The off-label use of subanesthetic doses of ketamine to treat depression may be considered in patients with major depressive disorder or bipolar disorder who are in a current major depressive episode.
- A trial of ketamine may be appropriate when depression is severe or refractory to conventional antidepressant drugs; when rapid reduction in suicidal symptoms is necessary; and when urgent improvement in depression is desired to allow patients to attend to pressing life circumstances.
- Some antidepressant outcomes may be improved by the use of ketamine early during an antidepressant course or as anesthesia during electroconvulsive therapy.

ABSTRACT
There is a substantial body of literature comprising anecdotal material and descriptions of uncontrolled and randomized controlled trials addressing the use of subanesthetic doses of ketamine for the off-label treatment of major depressive episodes. This article examines diagnostic indications for the off-label use of ketamine as an antidepressant and possible contexts in which ketamine may be trialled. Ketamine is indicated in patients who are in a major depressive episode. Most of the research data have been collected from patients with major depressive disorder, but patients with bipolar depression have also been studied. Ketamine is effective in both diagnostic groups, but its benefits are impermanent, perhaps more so in bipolar depression. There are several contexts within this diagnostic framework when a ketamine trial may be considered. These include severe depression and depression that is refractory to conventional antidepressant medication; this is because there is little purpose in trialling an experimental treatment in patients who are less severely ill and those who are antidepressant responsive. More importantly, ketamine has demonstrated efficacy in the rapid reduction of suicidal symptoms and can therefore be trialled when rapid reduction in suicidality is necessary. Likewise, because of its swift and dramatic antidepressant action, it can be trialled in patients in whom improvement is urgently desired in order to allow the patient to attend to pressing life circumstances. Some data suggest that the use of ketamine early during the course of an antidepressant trial, or as anesthesia during electroconvulsive therapy, can improve early antidepressant outcomes. It is not clear whether the presence of psychotic symptoms is a contraindication for ketamine use. Issues related to these indications and contexts are briefly discussed.
J Clin Psychiatry 2017;78(5):e555-e558
https://doi.org/10.4088/JCP.17f11629
© Copyright 2017 Physicians Postgraduate Press, Inc.
Introduction
A previous article in this column1 summarized issues related to the efficacy, adverse effects, and possible mechanism(s) of action of subanesthetic dosing with ketamine in the treatment of depression. The present article examines possible diagnostic indications and clinical contexts for the use of ketamine as an off-label antidepressant (Table 1). Readers may note here that the distinction between diagnostic indications and clinical contexts was made previously with regard to electroconvulsive therapy (ECT). In explanation, depression, mania, and schizophrenia are examples of indications for ECT, and severe illness, medication-refractory illness, presence of catatonic symptoms, presence of psychotic symptoms, presence of suicidality, and the need to discharge the patient early are examples of clinical and administrative contexts in which the use of ECT may be preferred.2
Diagnostic Indications
The antidepressant efficacy of subanesthetic doses of ketamine has been specifically studied in patients who were in a major depressive episode. Antidepressant benefits were observed to achieve statistical and clinical significance within 1-4 hours of ketamine administration. Such benefits peak after a day and are progressively lost 3-12 days later.1
Samples recruited in ketamine trials include subjects with major depressive disorder and those with bipolar depression; although the latter are a minority in terms of numbers of subjects and trials,3 there have been no signals suggesting poorer response or greater risks, such as that of manic switch, or other critical issues, except, perhaps, for an earlier offset of antidepressant action.4,5 Nonrefractory,6 mixed,7 and antidepressant-refractory8,9 samples have all been studied, with depression being at least moderately severe, and, more usually, severe, in all samples.
Depression with psychotic features has generally been an exclusionary condition in clinical trials of ketamine for depression. However, there have been anecdotal reports of the safe and effective use of ketamine in patients with psychotic depression, suggesting the possibility that this subgroup of patients may also benefit from a ketamine trial.10,11 At present, however, depression with psychotic features is best considered as a cautionary context for the use of ketamine.
Refractory Depression as a Clinical Context
Conventional antidepressant drugs are well established with regard to safety, efficacy, and convenience of administration for depressed patients in everyday clinical care. Ketamine may therefore be appropriate only in depressed patients who do not respond to conventional antidepressant drugs. In fact, most of the research on the off-label use of ketamine in depression has been conducted in patients in a (usually) severe antidepressant-refractory major depressive episode.1 There is, however, no guidance presently available that addresses where in the hierarchy of treatments an experimental treatment such as ketamine belongs in managing medication-refractory depression; patient selection, presently, is therefore a matter of judgment.12
Severe Suicidal Ideation as a Clinical Context
Suicidal ideation with strong intent is a clinical emergency; depressed patients at high risk of suicide are usually managed as inpatients, with ECT commonly offered as a treatment option.13 ECT, however, is associated with a risk of cognitive and other adverse effects.14 Ketamine has demonstrated efficacy against measures of suicidality in uncontrolled studies and in randomized controlled trials (RCTs); the effect size is large,15 and the benefits are apparent within a day.16-18 Small studies conducted in emergency department settings have demonstrated that the antisuicidal effect of ketamine may even be apparent within 40 minutes of treatment.19,20 The anecdotal use of ketamine as an emergency treatment for intense and persistent suicidal ideation in a young, depressed woman has also been described.21
The antisuicidal effect of ketamine is probably mediated by its antidepressant effect17 and wears off within days to a week18 but may be sustained for as long as 10 days.19 The time thus bought could be valuable because it would allow the initiation of other antidepressant and psychosocial intervention measures, as appropriate.
Life Circumstances as a Clinical Context
In 2009, Kellner et al22 reported the use of ECT for an "urgent social indication." The patient was a woman with recurrent depression who presented to the ECT services days before her daughter’s wedding. A single ECT administered with bifrontal electrode placement resulted in sufficient improvement for the woman to participate in and enjoy the family celebrations. There have been other reports, too, of the use of ECT to attenuate depression in the face of pressing life circumstances such as an examination or the patient’s own wedding.2
Given the dramatic benefits associated with the off-label use of ketamine in depression, it seems logical that ketamine could be more helpful than ECT in social emergencies. There are at least 2 reasons for this assertion. One is that ketamine but not ECT is commonly associated with marked antidepressant effect after a single treatment. The other is that ECT but not ketamine is associated with cognitive adverse effects that can compromise the ability of the patient to deal with the pressing life circumstances.14 If the administration of ketamine by intranasal,23,24 intramuscular,25 or subcutaneous25 routes can be established to be as effective as administration by the intravenous route, then the treatment procedure also becomes far more convenient than ECT.
Given the transience of the antidepressant benefits of ketamine,26 the treatment may need to be repeated if the social context is still present at the time the benefits wear off. The value of ketamine to allow depressed patients to recover sufficiently to deal with pressing life circumstances has not been formally studied, so far, and certainly merits attention.
Antidepressant Drug Treatment Initiation as a Clinical Context
If ketamine has an acute antidepressant effect, can it accelerate recovery or otherwise improve antidepressant outcomes in nonrefractory depressed patients started on conventional antidepressant drugs? This is an important question because if the answer is yes, then ketamine augmentation could become a useful strategy to reduce the duration of suffering and disability and to hasten functional recovery in depression.
In what is possibly the only RCT on the subject published so far, Hu et al7 administered a single dose of intravenous ketamine or saline placebo to 27 patients with severe nonpsychotic major depressive disorder, all of whom were also prescribed escitalopram (10 mg/d). At the end of the 4-week trial, response rates (92% vs 57%) and remission rates (77% vs 14%) were significantly higher in the ketamine group relative to the placebo group. Ketamine was also associated with shorter mean time to response (6.4 vs 26.5 days) and remission (14 vs 27 days), and the effect sizes, favoring ketamine, were large, on various rating instruments.
All cause discontinuation was numerically higher with ketamine than with placebo (20% vs 7%). Whereas ketamine was associated with more adverse events, no patient dropped out because of adverse events.7
In this study,7 56% of the sample was classified as treatment-refractory; analyses were presented for the superiority of ketamine over placebo in the whole sample and in the refractory subgroup but not in the nonrefractory patients. Therefore, a limitation of the study is that it is uncertain to what extent the findings can be generalized to nonrefractory samples in routine clinical care. This, therefore, remains an important area for future investigation.
ECT Initiation as a Clinical Context
ECT is administered under anesthesia, and ketamine is an approved anesthetic agent. Might the use of ketamine in ECT anesthesia improve ECT outcomes? This question has been examined in several RCTs as well as in meta-analyses.
In one systematic review and meta-analysis, McGirr et al27 identified 10 parallel-group RCTs conducted in patients with unipolar or bipolar depression. These patients had received ECT in ketamine (0.3 to 2.0 mg/kg; n = 333) or control (n = 269) arms. Ketamine had been administered either as standard (anesthesia) dose monotherapy or as low dose augmentation of sodium thiopental or propofol anesthesia. Analyses conducted at the ECT treatment endpoint found no difference between ketamine and control groups in response rates (53.9% vs 53.4%; 7 RCTs; n = 339) and remission rates (16.8% vs 18.9%; 7 RCTs; n = 339), nor did the groups differ significantly in the magnitude of antidepressant effect in either main or sensitivity analyses. Ketamine was associated with an increased risk of confusion.
In a second systematic review and meta-analysis published at approximately the same time as the previously described meta-analysis,27 Li et al28 identified 16 RCTs with 346 and 329 patients in ketamine and control arms, respectively. The use of ketamine in the ECT anesthesia regimen was associated with significantly greater antidepressant improvement at 1-2 as well as at 3-4 week assessments. The advantage for ketamine persisted in different subgroup and sensitivity analyses.
How does one reconcile these disparate conclusions? One explanation is that Li et al28 included a broader range of studies; another is that Li et al28 examined early (1-2 week) outcomes whereas McGirr et al27 studied only endpoint outcomes. It is possible that ketamine can improve early outcomes before the antidepressant effect of ECT manifests, and that at the end of the course, because ECT is a gold standard treatment for depression and because it is hard to improve a gold standard, a ceiling effect prevents the emergence of further benefit with ketamine.
Therefore, it appears that, at best, the use of ketamine in ECT anesthesia may carry benefits in the early part of an ECT course, but this will require further verification because the larger RCTs in the field were mostly poor in quality. Of note, a very recent RCT, not included in either meta-analysis, also did not find an advantage for the inclusion of ketamine in the ECT anesthesia.29 Readers who desire more information on the subject are referred to a recently published, extensive qualitative review.30
Parting Notes: Use of Concurrent Medications During a Ketamine Trial
Patients treated with ketamine were medication-free in some studies6,31; ongoing medications were continued unchanged in others.8,32 In this context, a suggestion that psychiatrists can consider is to continue necessary antidepressant and other medications during a ketamine trial, especially because maintenance antidepressant treatment will be required should the patient respond or remit. With specific reference to diagnostic subgroups, patients could need mood stabilizer and/or antipsychotic medication when the illness being treated is bipolar depression or depression with psychotic symptoms. This is to contain the hypothetical risk of a switch into mania or a ketamine-induced worsening of psychosis. Such precautions have been reported in literature.11,32
 Each month in his online column, Dr Andrade considers theoretical and practical ideas in clinical psychopharmacology with a view to update the knowledge and skills of medical practitioners who treat patients with psychiatric conditions.
Each month in his online column, Dr Andrade considers theoretical and practical ideas in clinical psychopharmacology with a view to update the knowledge and skills of medical practitioners who treat patients with psychiatric conditions.
Department of Clinical Psychopharmacology and Neurotoxicology, National Institute of Mental Health and Neurosciences, Bangalore, India ([email protected]).
Financial disclosure and more about Dr Andrade.
REFERENCES
1. Andrade C. Ketamine for depression, 1: clinical summary of issues related to efficacy, adverse effects, and mechanism of action. J Clin Psychiatry. 2017;78(4):e415-e419.
2. Devanathan S, Andrade C. Electroconvulsive therapy: indications versus contexts. J ECT. 2011;27(3):268-269. PubMed doi:10.1097/YCT.0b013e3181eb30ec
3. Kraus C, Rabl U, Vanicek T, et al. Administration of ketamine for unipolar and bipolar depression. Int J Psychiatry Clin Pract. 2017;21(1):2-12. PubMed doi:10.1080/13651501.2016.1254802
4. Newport DJ, Carpenter LL, McDonald WM, et al; APA Council of Research Task Force on Novel Biomarkers and Treatments. Ketamine and other NMDA antagonists: early clinical trials and possible mechanisms in depression. Am J Psychiatry. 2015;172(10):950-966. PubMed doi:10.1176/appi.ajp.2015.15040465
5. Romeo B, Choucha W, Fossati P, et al. Meta-analysis of short- and mid-term efficacy of ketamine in unipolar and bipolar depression. Psychiatry Res. 2015;230(2):682-688. PubMed doi:10.1016/j.psychres.2015.10.032
6. Berman RM, Cappiello A, Anand A, et al. Antidepressant effects of ketamine in depressed patients. Biol Psychiatry. 2000;47(4):351-354. PubMed doi:10.1016/S0006-3223(99)00230-9
7. Hu YD, Xiang YT, Fang JX, et al. Single i.v. ketamine augmentation of newly initiated escitalopram for major depression: results from a randomized, placebo-controlled 4-week study. Psychol Med. 2016;46(3):623-635. PubMed doi:10.1017/S0033291715002159
8. Cusin C, Ionescu DF, Pavone KJ, et al. Ketamine augmentation for outpatients with treatment-resistant depression: preliminary evidence for two-step intravenous dose escalation. Aust N Z J Psychiatry. 2017;51(1):55-64. PubMed doi:10.1177/0004867416631828
9. Vande Voort JL, Morgan RJ, Kung S, et al. Continuation phase intravenous ketamine in adults with treatment-resistant depression. J Affect Disord. 2016;206:300-304. PubMed doi:10.1016/j.jad.2016.09.008
10. Atigari OV, Healy D. Sustained antidepressant response to ketamine. BMJ Case Rep. 2013;2013:bcr2013200370. PubMed doi:10.1136/bcr-2013-200370
11. Ribeiro CM, Sanacora G, Hoffman R, et al. The use of ketamine for the treatment of depression in the context of psychotic symptoms: to the editor. Biol Psychiatry. 2016;79(9):e65-e66. PubMed doi:10.1016/j.biopsych.2015.05.016
12. Sanacora G, Frye MA, McDonald W, et al; American Psychiatric Association (APA) Council of Research Task Force on Novel Biomarkers and Treatments. A consensus statement on the use of ketamine in the treatment of mood disorders [published online ahead of print March 1, 2017]. JAMA Psychiatry. PubMed doi:10.1001/jamapsychiatry.2017.0080
13. Fink M, Kellner CH, McCall WV. The role of ECT in suicide prevention. J ECT. 2014;30(1):5-9. PubMed doi:10.1097/YCT.0b013e3182a6ad0d
14. Andrade C, Arumugham SS, Thirthalli J. Adverse effects of electroconvulsive therapy. Psychiatr Clin North Am. 2016;39(3):513-530. PubMed doi:10.1016/j.psc.2016.04.004
15. Bartoli F, Riboldi I, Crocamo C, et al. Ketamine as a rapid-acting agent for suicidal ideation: a meta-analysis [published online ahead of print March 22, 2017]. Neurosci Biobehav Rev. 2017;S0149-7634(16)30843-0. PubMed doi:10.1016/j.neubiorev.2017.03.010
16. Price RB, Nock MK, Charney DS, et al. Effects of intravenous ketamine on explicit and implicit measures of suicidality in treatment-resistant depression. Biol Psychiatry. 2009;66(5):522-526. PubMed doi:10.1016/j.biopsych.2009.04.029
17. Price RB, Iosifescu DV, Murrough JW, et al. Effects of ketamine on explicit and implicit suicidal cognition: a randomized controlled trial in treatment-resistant depression. Depress Anxiety. 2014;31(4):335-343. PubMed doi:10.1002/da.22253
18. Murrough JW, Soleimani L, DeWilde KE, et al. Ketamine for rapid reduction of suicidal ideation: a randomized controlled trial. Psychol Med. 2015;45(16):3571-3580. PubMed doi:10.1017/S0033291715001506
19. Larkin GL, Beautrais AL. A preliminary naturalistic study of low-dose ketamine for depression and suicide ideation in the emergency department. Int J Neuropsychopharmacol. 2011;14(8):1127-1131. PubMed doi:10.1017/S1461145711000629
20. Burger J, Capobianco M, Lovern R, et al. A double-blinded, randomized, placebo-controlled sub-dissociative dose ketamine pilot study in the treatment of acute depression and suicidality in a military emergency department setting. Mil Med. 2016;181(10):1195-1199. PubMed doi:10.7205/MILMED-D-15-00431
21. Zigman D, Blier P. Urgent ketamine infusion rapidly eliminated suicidal ideation for a patient with major depressive disorder: a case report. J Clin Psychopharmacol. 2013;33(2):270-272. PubMed doi:10.1097/JCP.0b013e3182856865
22. Kellner CH, Tobias KG, Jakubowski LM, et al. Electroconvulsive therapy for an urgent social indication. J ECT. 2009;25(4):274-275. PubMed doi:10.1097/YCT.0b013e3181a4878e
23. Lapidus KA, Levitch CF, Perez AM, et al. A randomized controlled trial of intranasal ketamine in major depressive disorder. Biol Psychiatry. 2014;76(12):970-976. PubMed doi:10.1016/j.biopsych.2014.03.026
24. Andrade C. Intranasal drug delivery in neuropsychiatry: focus on intranasal ketamine for refractory depression. J Clin Psychiatry. 2015;76(5):e628-e631. PubMed doi:10.4088/JCP.15f10026
25. Loo CK, Gálvez V, O’ Keefe E, et al. Placebo-controlled pilot trial testing dose titration and intravenous, intramuscular and subcutaneous routes for ketamine in depression. Acta Psychiatr Scand. 2016;134(1):48-56. PubMed doi:10.1111/acps.12572
26. Kishimoto T, Chawla JM, Hagi K, et al. Single-dose infusion ketamine and non-ketamine N-methyl-d-aspartate receptor antagonists for unipolar and bipolar depression: a meta-analysis of efficacy, safety and time trajectories. Psychol Med. 2016;46(7):1459-1472. PubMed doi:10.1017/S0033291716000064
27. McGirr A, Berlim MT, Bond DJ, et al. An updated systematic review and meta-analysis of randomized controlled trials of adjunctive ketamine in electroconvulsive therapy. Br J Psychiatry. 2017; In press.
28. Li DJ, Wang FC, Chu CS, et al. Significant treatment effect of add-on ketamine anesthesia in electroconvulsive therapy in depressive patients: a meta-analysis. Eur Neuropsychopharmacol. 2017;27(1):29-41. PubMed doi:10.1016/j.euroneuro.2016.11.008
29. Fernie G, Currie J, Perrin JS, et al. Ketamine as the anaesthetic for electroconvulsive therapy: the KANECT randomised controlled trial. Br J Psychiatry. 2017;bjp.bp.116.189134. PubMed doi:10.1192/bjp.bp.116.189134
30. Gálvez V, McGuirk L, Loo CK. The use of ketamine in ECT anaesthesia: a systematic review and critical commentary on efficacy, cognitive, safety and seizure outcomes [published online ahead of print November 28, 2016]. World J Biol Psychiatry. 2016:1-21. PubMed doi:10.1080/15622975.2016.1252464
31. Zarate CA Jr, Singh JB, Carlson PJ, et al. A randomized trial of an N-methyl-d-aspartate antagonist in treatment-resistant major depression. Arch Gen Psychiatry. 2006;63(8):856-864. PubMed doi:10.1001/archpsyc.63.8.856
32. Rybakowski JK, Permoda-Osip A, Bartkowska-Sniatkowska A. Ketamine augmentation rapidly improves depression scores in inpatients with treatment-resistant bipolar depression [published online ahead of print March 8, 2017]. Int J Psychiatry Clin Pract. 2017;1-5. PubMed doi:10.1080/13651501.2017.1297834
This PDF is free for all visitors!