Manic and Psychotic Symptoms Following Subcutaneous Leuprolide in a Male Patient With No Prior Psychiatric History
To the Editor: Leuprolide is a synthetic luteinizing hormone-releasing hormone (LHRH) agonist used in the treatment of advanced prostate cancer. It is available as an immediate-release intramuscular injection, a depot suspension for intramuscular injection, and an extended-release suspension for subcutaneous injection. After a 45-mg dose of subcutaneous leuprolide, 99.1% of patients experience clinical castration by day 28, defined as serum testosterone levels < 50 ng/mL.1 In addition to this decrease in serum testosterone levels, decreases in sex hormone-binding globulin and estradiol are also seen.1,2
Although there have been reports3,4 of emotional lability, depression, and anxiety with LHRH agonists, the data are not consistent. Furthermore, all published reports on psychiatric side effects of LHRH agonists have been exclusively in female patients. The package insert1 of leuprolide depot subcutaneous injection does list anxiety, depression, and delusions as occurring in less than 5% of patients. Here, we describe a case of a 65-year-old man with no prior psychiatric history who developed manic and psychotic symptoms after receiving a 45-mg subcutaneous injection of leuprolide. This is the only case available in the literature that describes a male patient experiencing such severe psychiatric symptoms in association with leuprolide.
Case report. Mr A, a 65-year-old white man, was brought into the emergency department by the police after they were called by his wife. She reported that he was extremely agitated, shouting profanities, threatening her, and accusing her of being schizophrenic. Over the last 3 to 4 weeks, he had been progressively sleeping less, only sleeping 1 to 2 hours per night immediately prior to admission. He had persecutory delusions regarding the Mafia and FBI. He was hyperverbal, had pressured speech, and was repeatedly quoting the Bible. Over the past several weeks, he had been displaying bizarre behavior, such as impulsively buying objects he had no use for and excessively spending money. He had also been irritable with his wife and using profanity, which was unusual behavior for him. At the time of admission, an accurate history could not be obtained from the patient, and therefore the majority of the information was obtained from his spouse.
Laboratory test results on admission, including thyroid panel, basic metabolic panel, complete blood count, and urinalysis, were all within normal limits, except the patient’s red blood cell count (RBC) was 3.88 M/mm3 (normal: 4.00-5.30 M/mm3) and his hematocrit was 35.5% (normal: 38.0%-46.0%). The rapid plasma reagin test result was negative. A toxicology screen was negative for illegal substances. The patient’s blood alcohol level was undetectable, and neurologic examination results were unremarkable.
When Mr A arrived in the emergency department, he was agitated and combative. He struck a nurse and threatened to hurt other staff members. At this time, the patient had to be placed in 4-point restraints. He also received haloperidol 5 mg intramuscularly. He was then involuntarily committed to the psychiatric inpatient unit.
The patient’s past medical history was significant for prostate cancer, and he had received radiation seed implants 4 years before. However, 3 months prior to this admission, his prostate-specific antigen levels became elevated. Upon the recommendation of his physician, the patient received leuprolide 45 mg subcutaneously approximately 2 months prior to this admission; this dose is intended to be administered once every 6 months. His past medical history was otherwise unremarkable. The patient had no history of psychiatric illness. His wife reported that his current behavior was an extreme deviation from his normal behavior. She reported no other recent medication changes. There was no evidence of metastasis.
Mr A was a college graduate who had a successful career and retired about 5 years ago. He denied tobacco or illicit drug use. He did state that he drank rarely. The patient had no family history of psychiatric illness, but one of his sisters did have Alzheimer’s disease. He had been married for 30 years; he had 2 children who lived out of the home and had no psychiatric history of their own. His home medications were loratadine 10 mg/d, aspirin 81 mg/d, multivitamin daily, and fish oil daily. He had no known drug allergies.
Upon admission, he was started on treatment with oral olanzapine 10 mg/d. At day 4 of hospitalization, he began to become less paranoid and guarded. He was still irritable and quick to anger with staff and patients; however, he was not physically assaultive. His speech remained pressured and tangential. A computed tomographic scan and magnetic resonance imaging of the head yielded unremarkable findings. On day 7 of hospitalization, the patient’s olanzapine dose was increased to 20 mg/d. By day 9, he began to participate more appropriately in groups and was easier to talk to. He was exhibiting no paranoid or aggressive behaviors. His speech became regular and less tangential. He no longer exhibited any persecutory delusions. He required no other medications during his hospitalization. A repeat complete blood count did show an increase in both RBC and hematocrit to 4.11 M/mm3 and 38.3%, respectively. He was discharged home with appropriate follow-up after 13 days of inpatient hospital treatment.
A month after discharge, Mr A continued to do well and decided to discontinue olanzapine on his own. After another month off olanzapine treatment, he continued to do well with no psychiatric symptoms. He never received a second shot of leuprolide.
This is the first case report of a male patient with no prior psychiatric history developing manic and psychotic symptoms after receiving a leuprolide injection.
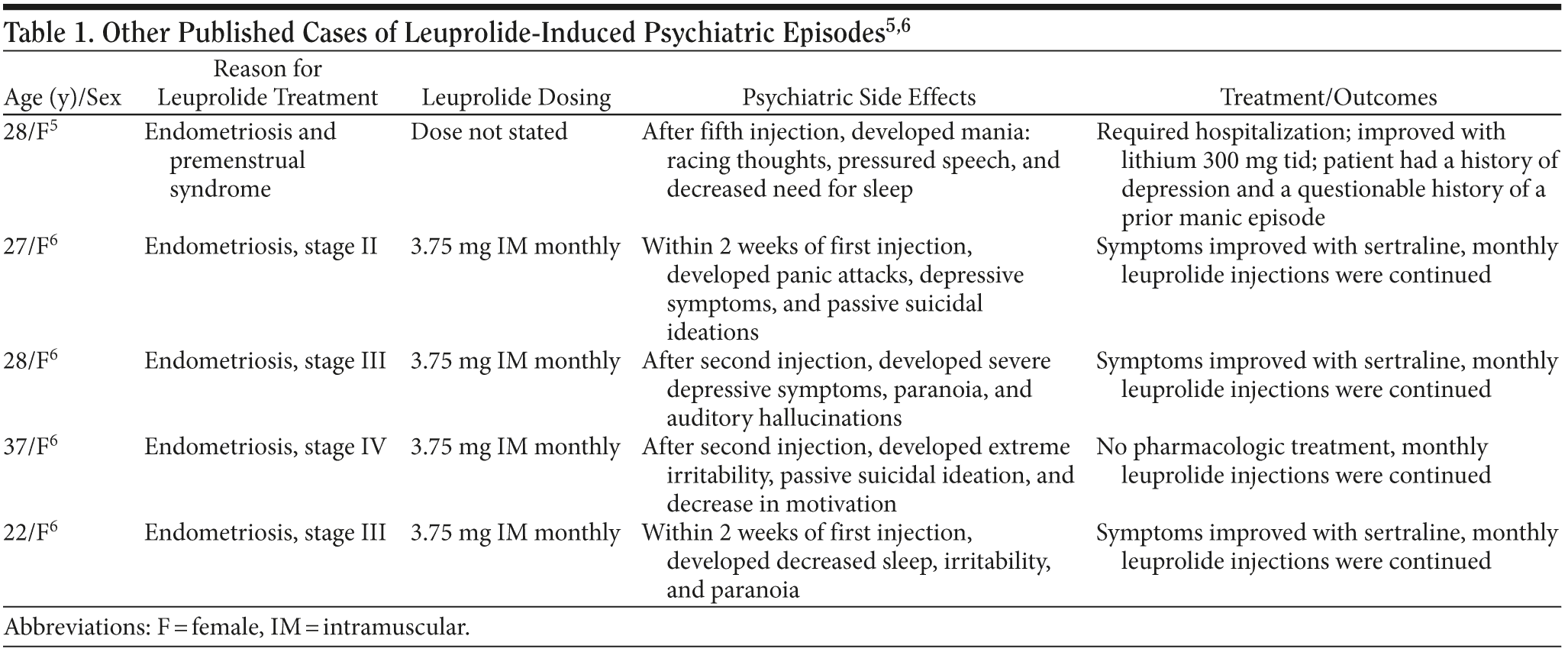
A PubMed/MEDLINE search of publications from 1980-2010 using the terms leuprolide, goserelin, mania, psychosis, mood disorder, and bipolar disorder revealed 2 publications,5,6 for a total of 5 cases, describing leuprolide-induced mood disturbances, and only 2 of those cases involved mania and/or psychosis. The rest of the cases described only depressive symptoms. Previous case reports5,6 have described psychotic symptoms in women after receiving leuprolide, but none have demonstrated this in men (Table 1). In these reports, it was theorized that estrogen’s rapid withdrawal following leuprolide therapy was a cause for the psychiatric symptoms.
An estrogen protection hypothesis has been postulated in the literature7; according to this hypothesis, estrogen exerts a protective effect on psychotic symptoms in susceptible patients. This hypothesis is supported by an average age of schizophrenia onset that is 5 years later in women than men; an increase in rates of admissions for schizophrenia in women on the days that estrogen levels are lowest in the menstrual cycle; and lower rates of acute psychosis during pregnancy, when estrogen levels are high, and higher rates of acute psychosis in the postpartum period, when estrogen levels suddenly decrease. Further, the incidence of late-onset schizophrenia in women is double that seen in men, and the combination of estrogen and neuroleptics leads to more rapid symptomatic improvement than neuroleptics alone.7
Initial research in men experiencing new-onset and/or acute psychosis has demonstrated decreased levels of total testosterone, free testosterone, estradiol, and estrone in men with schizophrenia compared to age- and weight-matched control patients.8 It is possible that the decreased levels of estradiol following an LHRH agonist injection may have left our patient vulnerable to the psychotic symptoms that he demonstrated, similar to the case reports in women. Unfortunately, measurements of testosterone and estradiol levels were not ordered for this patient. In retrospect, it would have been extremely helpful to have those levels. However, due to the mechanism of action of leuprolide and the timing of the injection, one would expect the patient to be in a state of chemical castration.
Although this level of severity in psychiatric adverse events with leuprolide is rare, clinicians should be aware of the possibility. A shorter-acting leuprolide formulation may be used if the patient has a history of psychiatric illness or if the clinician is concerned about psychiatric side effects.
References
1. Eligard (leuprolide) [package insert]. New York, NY: Sanofi-Synthelabo; 2008.
2. Smith MR. Obesity and sex steroids during gonadotropin-releasing hormone agonist treatment for prostate cancer. Clin Cancer Res. 2007;13(1):241-245. PubMed doi:10.1158/1078-0432.CCR-06-2086
3. Shaw RW. The role of GnRH analogues in the treatment of endometriosis. Br J Obstet Gynaecol. 1992;99(suppl 7):9-12. PubMed
4. Steingold KA, Cedars M, Lu JK, et al. Treatment of endometriosis with a long-acting gonadotropin-releasing hormone agonist. Obstet Gynecol. 1987;69(3 pt 1):403-411. PubMed
5. Rachman M, Garfield DA, Rachman I, et al. Lupron-induced mania. Biol Psychiatry. 1999;45(2):243-244. PubMed doi:10.1016/S0006-3223(98)00206-6
6. Warnock JK, Bundren JC. Anxiety and mood disorders associated with gonadotropin-releasing hormone agonist therapy. Psychopharmacol Bull. 1997;33(2):311-316. PubMed
7. Riecher-Rössler A. Oestrogen effects in schizophrenia and their potential therapeutic implications—review. Arch Women Ment Health. 2002;5(3):111-118. PubMed doi:10.1007/s00737-002-0003-3
8. Huber TJ, Tettenborn C, Leifke E, et al. Sex hormones in psychotic men. Psychoneuroendocrinology. 2005;30(1):111-114. PubMed doi:10.1016/j.psyneuen.2004.05.010
Author affiliations: Rutgers, The State University of New Jersey, Piscataway (both authors). Dr Chavez is now affiliated with the College of Pharmacy, University of Hawai’ i at Hilo. Potential conflicts of interest: Dr Chavez reports no potential conflict of interest. Dr Reilly‘s spouse is an employee and stockholder of Johnson & Johnson. Funding/support: None reported. Previous presentation: Presented as a poster and platform presentation at the College of Psychiatric and Neurologic Pharmacists Annual Meeting; April 18-19, 2010; San Antonio, Texas.
doi:10.4088/JCP.10l06190yel
© Copyright 2010 Physicians Postgraduate Press, Inc.