Because this piece does not have an abstract, we have provided for your benefit the first 3 sentences of the full text.
As of March 2019, in the United States, “medical marijuana” has been approved in 33 states, the District of Columbia, Guam, and Puerto Rico. Marijuana, however, remains a schedule I drug per the US Drug Enforcement Administration (DEA) based on its findings that marijuana (a) has a high potential for abuse, (b) has no currently accepted medical use in treatment in the United States, and (c) lacks accepted safety for use under medical supervision. Nevertheless, today the marketplace is flooded with over 2,500 “strains” of the marijuana plant (named variously as “Mango Kush,” “Skywalker,” “Purple Haze,” “Obama Kush,” and so-called cannabidiol [CBD] strains such as “Harlequin,” “Ringo’s Gift,” “ACDC,” and “Cannatonic”) and marijuana-infused products (including edibles, concentrates, dabs, waxes, oils, and vaping fluids), each claiming unique, yet unproven, “medical” benefit.
Department of Psychiatry, Yale University School of Medicine, New Haven, Connecticut
*Corresponding author: Rajiv Radhakrishnan, MBBS, MD, 300 George St, Suite 901, New Haven, CT 06511 ([email protected]).
J Clin Psychiatry 2019;80(5):18ac12537
To cite: Radhakrishnan R, Ranganathan M, D’Souza DC. Medical marijuana: what physicians need to know. J Clin Psychiatry. 2019;80(5):18ac12537.
To share: https://doi.org/10.4088/JCP.18ac12537
© Copyright 2019 Physicians Postgraduate Press, Inc.
Medical Marijuana: What Physicians Need to Know
As of March 2019, in the United States, “medical marijuana” has been approved in 33 states, the District of Columbia, Guam, and Puerto Rico.1 Marijuana, however, remains a schedule I drug per the US Drug Enforcement Administration (DEA) based on its findings that marijuana (a) has a high potential for abuse, (b) has no currently accepted medical use in treatment in the United States, and (c) lacks accepted safety for use under medical supervision. Nevertheless, today the marketplace is flooded with over 2,500 “strains” of the marijuana plant (named variously as “Mango Kush,” “Skywalker,” “Purple Haze,” “Obama Kush,” and so-called cannabidiol [CBD] strains such as “Harlequin,” “Ringo’s Gift,” “ACDC,” and “Cannatonic”) and marijuana-infused products (including edibles, concentrates, dabs, waxes, oils, and vaping fluids), each claiming unique, yet unproven, “medical” benefit. These products are not regulated by the US Food and Drug Administration (FDA) and do not meet the FDA standards for approval of other medications, although the product labels accompanying these products may appear similar to those seen with pharmaceutical medications.
This presents a challenge for physicians treating patients who use “medical marijuana” and tout the benefits of the products based on manufacturers’ product labels. Furthermore, psychiatrists may encounter patients who are using medical marijuana for a psychiatric indication or may comanage patients who are receiving medical marijuana for a nonpsychiatric (medical) condition. This article is intended to provide physicians with an overview of “medical marijuana” and cannabinoids, the current state of the evidence (or lack thereof) for marijuana in psychiatric indications, and the potential risks, side effects, and drug interactions that may be encountered during psychiatric treatment of individuals who use “medical marijuana.”
Overview of Marijuana and the Endocannabinoid System
Marijuana, or cannabis, typically refers to the dried, flowering tops of 3 main species of the cannabis plant, namely, Cannabis indica, Cannabis sativa, and Cannabis ruderalis. The flowering tops contain over 565 chemical constituents including > 120 phytocannabinoids, terpenoids, and flavonoids.2 The most well-studied phytocannabinoids are Δ9-tetrahydrocannabinol (THC) and CBD. THC is the main psychoactive constituent of marijuana. Unlike THC, CBD, another important constituent cannabinoid, is not known to have rewarding/reinforcing effects or perception altering effects. CBD is thought to have some effects with therapeutic potential, such as antiepileptic and anxiolytic effects.3 While the effects of THC have been extensively studied and are well known, the reasons for individual differences in response and interactive effects of some of the other cannabinoids on the net effect of marijuana are not clear.
“Medical marijuana” strains are derived by cross-breeding Cannabis indica and sativa plant species to produce hybrids that vary in their contents of THC and CBD. Strains with THC content > 20% are considered “high THC,” but strains need only have CBD > 4% to be considered “high CBD.”
Phytocannabinoids are thought to exert their effects primarily via the endocannabinoid system and downstream effects on other neurotransmitter systems such as serotonin, GABA, and other G-protein–coupled receptors.4 Endocannabinoids, primarily anandamide (AEA) and 2-arachidonoylglycerol (2-AG), are endogenous lipid neurotransmitters that inhibit the release of both GABA and glutamate. They hence maintain the balance of excitatory/inhibitory neurotransmitters in the brain.5 They are synthesized and released on demand, activate cannabinoid receptor subtypes 1 and 2 (CB1R and CB2R), and are rapidly deactivated thereafter. In contrast, THC and other CB1R agonists bind to cannabinoid receptors and activate them for a longer duration. THC is a weak partial agonist of both CB1Rs and CB2Rs. In contrast, the mechanism underlying CBD’s effects is not fully understood as yet. Among the proposed mechanisms are negative allosteric modulation of THC and 2-AG,6 inhibition of AEA reuptake, 5-HT1A agonism, 5-HT2A weak partial agonism, and a noncompetitive 5-HT3A antagonism.4
Labeling
Unlike the standard FDA-approved labeling of commercially available medications in the United States, each state in the United States has its own requirements with regard to the product labeling of medical marijuana. While the majority of states require that the product label state the quantity of THC and CBD, some, such as Connecticut and Illinois, require that the level of tetrahydrocannabinolic acid (THCA) is also included, while others, such as Washington and Nevada, require that “total THC” content is listed. Most products labeled as “CBD-alone” typically also contain THC or THCA since they are derived from the whole plant. The potential benefits of CBD-alone products hence need to be evaluated in the context of potential risk of psychosis and other adverse psychiatric outcomes related to the presence of other cannabinoids in the product. Doses of THC as low as 2.5–5 mg have been shown to worsen psychosis despite adequate antipsychotic treatment.7
An additional concern is that the concentrations noted on the product labels are sometimes inaccurate. The FDA has issued warning letters regarding 45 such commercially available products since 2015, which either contain no cannabinoids despite claiming to do so or contain variable concentrations of THC, CBD, and THCA (https://www.fda.gov/newsevents/publichealthfocus/ucm484109.htm). In a study of 75 edible medical marijuana products from San Francisco and Los Angeles, California, and Seattle, Washington, 17% were accurately labeled, 23% were underlabeled, and 60% were overlabeled with respect to THC content, and 41% had undetectable levels of CBD.8
Current Evidence for the Use of “Medical Marijuana” in Psychiatric Indications
Medical marijuana has been approved for a number of psychiatric disorders in many states in the United States including posttraumatic stress disorder (PTSD), agitation in Alzheimer’s disease, and Tourette’s disorder (Table 1). A recent systematic review of the evidence found that, at the present time, the strength of evidence for the use of medical marijuana for these conditions was “very low.”9 The Veterans Administration Evidence-based Synthesis Program conducted a systematic review of marijuana in PTSD and concluded that further evidence is necessary to support claims of beneficial effects.10 The 2013 American Psychiatric Association “Position Statement on Marijuana as Medicine” notes that “there is no current scientific evidence that marijuana is in any way beneficial for the treatment of any psychiatric disorder. In contrast, current evidence supports, at minimum, a strong association of cannabis use with the onset of psychiatric disorders. Adolescents are particularly vulnerable to harm, given the effects of cannabis on neurodevelopment.”11 There are no randomized controlled trials (RCTs) of adequate power that have examined the efficacy of marijuana for Tourette’s disorder, PTSD, or Alzheimer’s disease. The existing evidence is from studies of THC and nabilone that have been extrapolated to “medical marijuana.” As discussed earlier, THC is one but not the only constituent of marijuana.
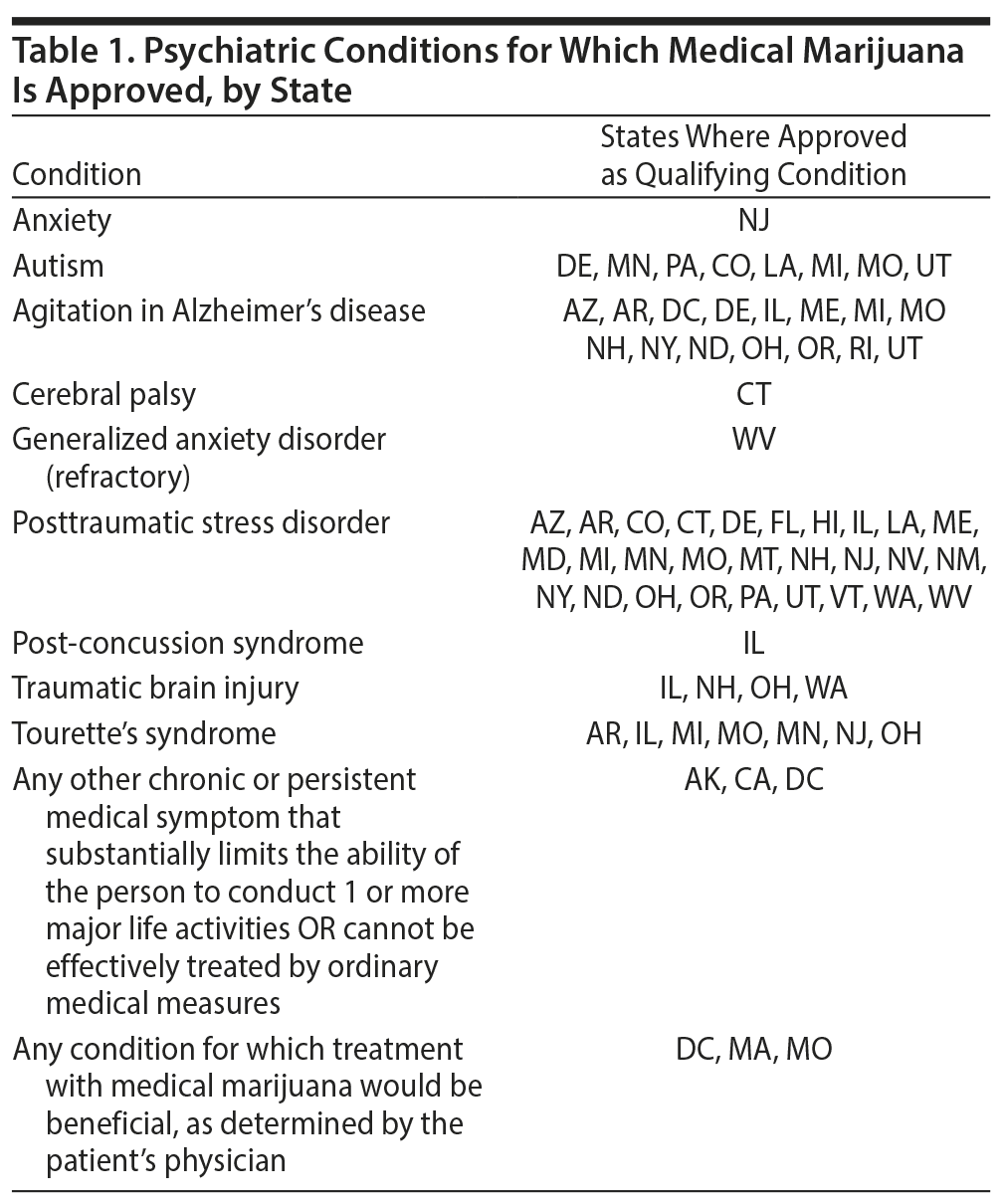
Other psychiatric disorders for which “medical marijuana” is touted to be beneficial include autism, attention-deficit/hyperactivity disorder, sleep disorders, and psychosis, with inadequate evidence to support these claims.
There is great interest in CBD-alone products. The utility of CBD in psychiatric disorders including psychosis is, however, controversial.12 While one RCT13 showed improvement in Positive and Negative Syndrome Scale positive symptoms (CBD dose = 1,000 mg/d), another14 (CBD dose = 800 mg/d) was negative. In the former study, the CBD group had lower levels of positive psychotic symptoms at the 6 week time point that were statistically significant, but the differences may be of small clinical significance. Two additional RCTs, judged to have a high risk of bias (71 total participants with schizophrenia or schizophreniform psychosis), found CBD (maximum dose 800 mg/d) to be comparable to amisulpride (800 mg/d) in chronic schizophrenia14 and CBD (600 mg/d) not to be significantly different from placebo in first-episode schizophrenia.15–17 Of note, the studies of CBD with adjunctive antipsychotics did not examine serum antipsychotic levels. This would be important to rule out a pharmacokinetic effect since CBD potentially inhibits cytochrome P450 (CYP)2D6.18
Risks Associated With Acute Marijuana Use
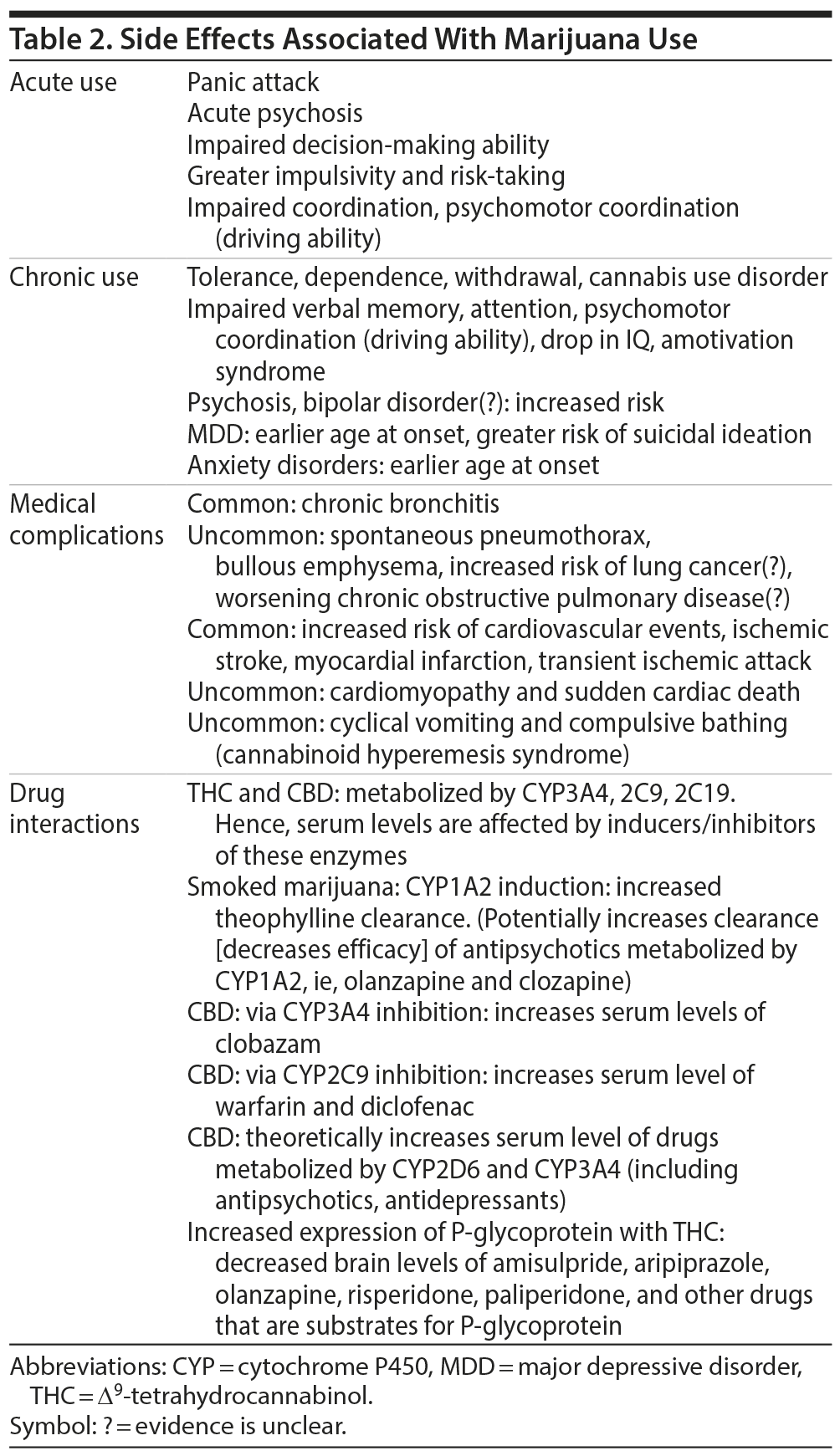
Acute marijuana intoxication can present with increased anxiety, panic attacks, acute psychosis, impaired decision-making, and greater risk-taking and impulsivity (Table 2).19 THC acutely produces deficits in attention, verbal learning, working memory, and electrophysiologic indices of information processing.7,20
Risks Related to Chronic Use of Marijuana
The consequences of chronic marijuana use include development of tolerance, dependence, withdrawal, and cannabis use disorder; cognitive deficits such as impairment in attention, memory, IQ, and driving ability; and amotivation syndrome.21–24 The effects of marijuana use on adolescents are of particular concern, since adolescence is increasingly well recognized as a critical period of neurodevelopment.
Epidemiologic studies point to an association between marijuana use and increased risk of psychiatric disorders such as schizophrenia,20,25 bipolar disorder,23,26 major depressive disorder, and suicidal ideation.27 Marijuana use has been associated with a worse course of illness, greater number of hospitalizations, and overall poorer outcomes in both schizophrenia and bipolar disorder28–30 (also reviewed in Satre et al31).
Medical complications associated with chronic, heavy marijuana use include increased risk of pulmonary complications (chronic pulmonary obstructive disease, spontaneous pneumothorax, bullous emphysema, lung cancer)32 and cardiovascular complications (myocardial infarction, cardiomyopathy, stroke, transient ischemic attack, and sudden cardiac death).33,34 Chronic marijuana use is also associated with a syndrome of cyclic vomiting and compulsive bathing (cannabinoid hyperemesis syndrome), the diagnosis of which requires a high index of suspicion.35
Drug Interactions
THC and CBD are both substrates for cytochrome P450 enzymes. THC and CBD are also both metabolized by CYP2C9 and 3A4, while CBD is additionally metabolized by CYP2C19.36 Serum levels of THC and CBD are hence altered by concomitant CYP3A4 inhibitors and inducers.
Clinically significant drug interactions include increased serum levels of clobazam (possibly via CYP3A inhibition), warfarin, and diclofenac (via CYP2C9 inhibition) with CBD37 and with marijuana.38 CBD is a potent inhibitor of CYP2D6, CYP2C19, and CYP3A439,40 in in vitro studies. Theoretically, CBD could increase serum levels of antipsychotics and antidepressants that are metabolized by these enzymes.17,40 THC, on the other hand, has been shown to decrease brain concentrations of risperidone and its active metabolite, 9-hydroxyrisperidone, via induction of P-glycoprotein expression.41
Like cigarette smoking, smoking marijuana may induce CYP1A2. Consequently, frequent marijuana smokers have been shown to have a 42%–48% higher clearance of theophylline.42 Also of clinical relevance is the finding that alcohol in combination with marijuana impairs driving ability at a lower blood alcohol level.
In summary, the use of medical marijuana presents a challenge to physicians, in the absence of good evidence of its efficacy. It is important for physicians to note that “medical marijuana” and THC are schedule I substances per the DEA in the United States. Therefore, even when US states permit their use under the guise of medical marijuana law, “prescribing” these compounds is not without liability. Additionally, as noted earlier, the contents in the product do not match the product label in many cases. It would be important for physicians to document a discussion of risks versus benefits, including acute and chronic side effects, drug interactions, and alternative treatment options.
Published online: May 7, 2019.
Potential conflicts of interest: None.
Funding/support: Dr Radhakrishnan is supported by the Dana Foundation David Mahoney grant program and Clinical and Translational Science Awards Grant Number UL1 TR001863 from the National Center for Advancing Translational Sciences, a component of the National Institutes of Health (NIH) and NIH Roadmap for Medical Research.
Disclaimer: The contents of this publication are solely the responsibility of the authors and do not necessarily represent the official view of NIH.
REFERENCES
1. van Os J, Reininghaus U. Psychosis as a transdiagnostic and extended phenotype in the general population. World Psychiatry. 2016;15(2):118–124. PubMed CrossRef
2. Turner SE, Williams CM, Iversen L, et al. Molecular pharmacology of phytocannabinoids. Prog Chem Org Nat Prod. 2017;103:61–101. PubMed CrossRef
3. Devinsky O, Cilio MR, Cross H, et al. Cannabidiol: pharmacology and potential therapeutic role in epilepsy and other neuropsychiatric disorders. Epilepsia. 2014;55(6):791–802. PubMed CrossRef
4. Morales P, Hurst DP, Reggio PH. Molecular targets of the phytocannabinoids: a complex picture. Prog Chem Org Nat Prod. 2017;103:103–131. PubMed CrossRef
5. Radhakrishnan R, Ross DA. From “azalla” to anandamide: distilling the therapeutic potential of cannabinoids. Biol Psychiatry. 2018;83(2):e27–e29. PubMed CrossRef
6. Laprairie RB, Bagher AM, Kelly ME, et al. Cannabidiol is a negative allosteric modulator of the cannabinoid CB1 receptor. Br J Pharmacol. 2015;172(20):4790–4805. PubMed CrossRef
7. Radhakrishnan R, Wilkinson ST, D’Souza DC. Gone to pot: a review of the association between cannabis and psychosis. Front Psychiatry. 2014;5:54. PubMed CrossRef
8. Vandrey R, Raber JC, Raber ME, et al. Cannabinoid dose and label accuracy in edible medical cannabis products. JAMA. 2015;313(24):2491–2493. PubMed CrossRef
9. Wilkinson ST, Radhakrishnan R, D’Souza DC. A systematic review of the evidence for medical marijuana in psychiatric indications. J Clin Psychiatry. 2016;77(8):1050–1064. PubMed CrossRef
10. Kansagara D, O’Neil M, Nugent S, et al. Benefits and Harms of Cannabis in Chronic Pain or Post-traumatic Stress Disorder: A Systematic Review. Washington, DC: US Department of Veterans Affairs; 2017.
11. Zaman T, Rosenthal RN, Renner JA, et al. Position Statement on Marijuana as Medicine. American Psychiatric Association. November 10, 2013. https://www.psychiatry.org/file%20library/about-apa/organization-documents-policies/policies/position-2013-marijuana-as-medicine.pdf.
12. Mandolini GM, Lazzaretti M, Pigoni A, et al. Pharmacological properties of cannabidiol in the treatment of psychiatric disorders: a critical overview. Epidemiol Psychiatr Sci. 2018;27(4):327–335. PubMed CrossRef
13. McGuire P, Robson P, Cubala WJ, et al. Cannabidiol (CBD) as an adjunctive therapy in schizophrenia: a multicenter randomized controlled trial. Am J Psychiatry. 2018;175(3):225–231. PubMed CrossRef
14. Boggs DL, Surti T, Gupta A, et al. The effects of cannabidiol (CBD) on cognition and symptoms in outpatients with chronic schizophrenia: a randomized placebo controlled trial. Psychopharmacology (Berl). 2018;235(7):1923–1932. PubMed CrossRef
15. Whiting PF, Wolff RF, Deshpande S, et al. Cannabinoids for medical use: a systematic review and meta-analysis. JAMA. 2015;313(24):2456–2473. PubMed CrossRef
16. Leweke FM, Piomelli D, Pahlisch F, et al. Cannabidiol enhances anandamide signaling and alleviates psychotic symptoms of schizophrenia. Transl Psychiatry. 2012;2(3):e94. PubMed CrossRef
17. Leweke M. The endocannabinoid system in schizophrenia: a mechanistically new approach to its pathophysiology and treatment. Schizophr Bull. 2013;39(1):S341.
18. Iffland K, Grotenhermen F. An update on safety and side effects of cannabidiol: a review of clinical data and relevant animal studies. Cannabis Cannabinoid Res. 2017;2(1):139–154. PubMed CrossRef
19. Crean RD, Crane NA, Mason BJ. An evidence based review of acute and long-term effects of cannabis use on executive cognitive functions. J Addict Med. 2011;5(1):1–8. PubMed CrossRef
20. Sherif M, Radhakrishnan R, D’Souza DC, et al. Human laboratory studies on cannabinoids and psychosis. Biol Psychiatry. 2016;79(7):526–538. PubMed CrossRef
21. Ranganathan M, Skosnik PD, D’Souza DC. Marijuana and madness: associations between cannabinoids and psychosis. Biol Psychiatry. 2016;79(7):511–513. PubMed CrossRef
22. Chadi N, Levy S. Radhakrishnan R; et al. Introduction. In: Sabet KA, Winters KC, eds. Contemporary Health Issues on Marijuana. New York, NY: Oxford University Press; 2018.
23. Marwaha S, Winsper C, Bebbington P, et al. Cannabis use and hypomania in young people: a prospective analysis. Schizophr Bull. 2018;44(6):1267–1274. PubMed CrossRef
24. Duperrouzel J, Hawes SW, Lopez-Quintero C, et al. The association between adolescent cannabis use and anxiety: a parallel process analysis. Addict Behav. 2018;78:107–113. PubMed CrossRef
25. Marconi A, Di Forti M, Lewis CM, et al. Meta-analysis of the association between the level of cannabis use and risk of psychosis. Schizophr Bull. 2016;42(5):1262–1269. PubMed CrossRef
26. Gibbs M, Winsper C, Marwaha S, et al. Cannabis use and mania symptoms: a systematic review and meta-analysis. J Affect Disord. 2015;171:39–47. PubMed CrossRef
27. Agrawal A, Nelson EC, Bucholz KK, et al. Major depressive disorder, suicidal thoughts and behaviours, and cannabis involvement in discordant twins: a retrospective cohort study. Lancet Psychiatry. 2017;4(9):706–714. PubMed CrossRef
28. Ringen PA, Nesvåg R, Helle S, et al. Premorbid cannabis use is associated with more symptoms and poorer functioning in schizophrenia spectrum disorder. Psychol Med. 2016;46(15):3127–3136. PubMed CrossRef
29. Romer Thomsen K, Thylstrup B, Pedersen MM, et al. Drug-related predictors of readmission for schizophrenia among patients admitted to treatment for drug use disorders. Schizophr Res. 2018;195:495–500. PubMed CrossRef
30. Kvitland LR, Melle I, Aminoff SR, et al. Continued cannabis use at one year follow up is associated with elevated mood and lower global functioning in bipolar I disorder. BMC Psychiatry. 2015;15(1):11. PubMed CrossRef
31. Satre DD, Bahorik A, Zaman T, et al. Psychiatric disorders and comorbid cannabis use: how common is it and what is the clinical impact? J Clin Psychiatry. 2018;79(5):18ac12267. PubMed CrossRef
32. Martinasek MP, McGrogan JB, Maysonet A. A systematic review of the respiratory effects of inhalational marijuana. Respir Care. 2016;61(11):1543–1551. PubMed CrossRef
33. Abouk R, Adams S. Examining the relationship between medical cannabis laws and cardiovascular deaths in the US. Int J Drug Policy. 2018;53:1–7. PubMed CrossRef
34. Thomas G, Kloner RA, Rezkalla S. Adverse cardiovascular, cerebrovascular, and peripheral vascular effects of marijuana inhalation: what cardiologists need to know. Am J Cardiol. 2014;113(1):187–190. PubMed CrossRef
35. Patterson DA, Smith E, Monahan M, et al. Cannabinoid hyperemesis and compulsive bathing: a case series and paradoxical pathophysiological explanation. J Am Board Fam Med. 2010;23(6):790–793. PubMed CrossRef
36. Stout SM, Cimino NM. Exogenous cannabinoids as substrates, inhibitors, and inducers of human drug metabolizing enzymes: a systematic review. Drug Metab Rev. 2014;46(1):86–95. PubMed CrossRef
37. Grayson L, Vines B, Nichol K, et al; UAB CBD Program. An interaction between warfarin and cannabidiol, a case report. Epilepsy Behav Case Rep. 2017;9:10–11. PubMed CrossRef
38. Yamreudeewong W, Wong HK, Brausch LM, et al. Probable interaction between warfarin and marijuana smoking. Ann Pharmacother. 2009;43(7):1347–1353. PubMed CrossRef
39. Yamaori S, Ebisawa J, Okushima Y, et al. Potent inhibition of human cytochrome P450 3A isoforms by cannabidiol: role of phenolic hydroxyl groups in the resorcinol moiety. Life Sci. 2011;88(15–16):730–736. PubMed CrossRef
40. Yamaori S, Okamoto Y, Yamamoto I, et al. Cannabidiol, a major phytocannabinoid, as a potent atypical inhibitor for CYP2D6. Drug Metab Dispos. 2011;39(11):2049–2056. PubMed CrossRef
41. Brzozowska NI, de Tonnerre EJ, Li KM, et al. The differential binding of antipsychotic drugs to the ABC transporter P-glycoprotein predicts cannabinoid-antipsychotic drug interactions. Neuropsychopharmacology. 2017;42(11):2222–2231. PubMed CrossRef
42. Jusko WJ, Schentag JJ, Clark JH, et al. Enhanced biotransformation of theophylline in marihuana and tobacco smokers. Clin Pharmacol Ther. 1978;24(4):405–410. PubMed CrossRef
This PDF is free for all visitors!


