This work may not be copied, distributed, displayed, published, reproduced, transmitted, modified, posted, sold, licensed, or used for commercial purposes. By downloading this file, you are agreeing to the publisher’s Terms & Conditions.
Objective: Restless legs syndrome (RLS), also known as Willis-Ekbom disease, is a sensorimotor disorder that can result in considerable sleep disruption. This narrative review provides an overview of RLS diagnosis and reports epidemiologic evidence for an association between RLS and mood disorders. Possible links between RLS, sleep disturbances, and mood disorders are considered, and theoretical pathophysiologic pathways are discussed. Finally, pharmacologic therapies for RLS are summarized.
Data Sources: A PubMed search was performed using the search term restless legs syndrome in combination with affective/anxiety, antidepressants, anxiety/anxiety disorder, attention deficit hyperactivity disorder, depression/depressive disorder, mood/mood disorder, neuropsychiatric, panic/panic disorder, psychiatric disorder, and psychosis. English-language articles published between January 1993 and May 2013 were retrieved. Additional studies were identified from the reference lists of relevant publications.
Study Selection: 173 publications were retrieved. Articles related to the association between idiopathic RLS and depression, anxiety, and mood disorders were reviewed. In total, 32 epidemiologic studies were identified. These studies were reviewed in detail and ranked according to quality.
Data Extraction: Data were extracted on the basis of relevance to the topic. Epidemiologic studies were assessed using 3 parameters: methodology, data quality, and generalizability of the results. Each factor was scored from 1 (high quality) to 4 (low quality), giving a total score of between 3 and 12 for each study.
Results and Conclusions: RLS and mood disorders are frequently comorbid. Recognition and appropriate treatment of comorbid RLS are particularly important in patients with psychiatric disorders, as RLS is a common medical reason for insomnia, and antidepressant use may exacerbate sensory symptoms.
Mood Disorders in Restless Legs Syndrome (Willis-Ekbom Disease)

ABSTRACT
Objective: Restless legs syndrome (RLS), also known as Willis-Ekbom disease, is a sensorimotor disorder that can result in considerable sleep disruption. This narrative review provides an overview of RLS diagnosis and reports epidemiologic evidence for an association between RLS and mood disorders. Possible links between RLS, sleep disturbances, and mood disorders are considered, and theoretical pathophysiologic pathways are discussed. Finally, pharmacologic therapies for RLS are summarized.
Data Sources: A PubMed search was performed using the search term restless legs syndrome in combination with affective/anxiety, antidepressants, anxiety/anxiety disorder, attention deficit hyperactivity disorder, depression/depressive disorder, mood/mood disorder, neuropsychiatric, panic/panic disorder, psychiatric disorder, and psychosis. English-language articles published between January 1993 and May 2013 were retrieved. Additional studies were identified from the reference lists of relevant publications.
Study Selection: 173 publications were retrieved. Articles related to the association between idiopathic RLS and depression, anxiety, and mood disorders were reviewed. In total, 32 epidemiologic studies were identified. These studies were reviewed in detail and ranked according to quality.
Data Extraction: Data were extracted on the basis of relevance to the topic. Epidemiologic studies were assessed using 3 parameters: methodology, data quality, and generalizability of the results. Each factor was scored from 1 (high quality) to 4 (low quality), giving a total score of between 3 and 12 for each study.
Results and Conclusions: RLS and mood disorders are frequently comorbid. Recognition and appropriate treatment of comorbid RLS are particularly important in patients with psychiatric disorders, as RLS is a common medical reason for insomnia, and antidepressant use may exacerbate sensory symptoms.
J Clin Psychiatry 2014;75(7):e679-e694
© Copyright 2014 Physicians Postgraduate Press, Inc.
Submitted: July 18, 2013; accepted February 24, 2014 (doi:10.4088/JCP.13r08692).
Corresponding author: Philip M. Becker, MD, Sleep Medicine Associates of Texas, PA, 5477 Glen Lakes Dr, Ste 100, Dallas, TX 75321 ([email protected]).
Restless legs syndrome (RLS), also known as Willis-Ekbom disease, is a common, chronic sensorimotor disorder with a prevalence of between 3%-29% of the general population in Western industrialized countries.1-4 Around 2.7% of individuals experience at least moderately distressing symptoms likely to warrant treatment, of whom 55.5% experience disturbance of daytime functioning.1 In a US survey of RLS prevalence and burden, 1.5% of respondents were identified as RLS sufferers (symptoms ≥ 2 times per week with moderate-to-severe distress), with a mean work productivity loss of 1 day per week.5 The disorder is characterized by an overwhelming urge to move the legs (or other body parts), often accompanied by paresthesias or dysesthesias. Symptoms worsen in the evening and at night around the peak of core body temperature and can result in significant sleep disruption.6 RLS is thought to be a central nervous system disorder related to dopaminergic dysfunction and impaired iron homeostasis in the brain.7 Proteomic analysis of the cerebrospinal fluid of patients with RLS reveals a protein profile that is consistent with clinical findings of disruptive sleep, cardiovascular dysfunction, and painful symptoms.8 Around 60% of patients with idiopathic/primary RLS have a first-degree relative who also suffers from the disorder,6,9 and genome-wide association studies have identified genetic risk factors.10-12 Secondary RLS is associated with iron deficiency, pregnancy, and end-stage renal disease; all 3 conditions are characterized by reduced central iron availability.13
RLS has a significant impact on patient well-being and has been associated with reduced general health, poor mental health status, and depressed mood.14-18 Although the association between RLS and cognitive impairment needs further evaluation, recent evidence suggests that the disorder may also affect cognitive function19-21; impaired executive functioning, as indicated by impaired performance in the Wisconsin Card Sorting Test, has been observed in 10 adults with frequent RLS symptoms (≥ 2 per week).21 Common comorbidities of RLS include insomnia, anxiety, and depression.22 A bidirectional relationship between clinically relevant depressive symptoms and RLS has been suggested.23 As RLS has a significant impact on sleep and is associated with mood disorders, it may present in psychiatric practice. A large US cross-sectional study (N = 1,024) reported a strong association between RLS and a diagnosis of major depressive disorder (12-month prevalence: 9.5% [odds ratio (OR) = 4.7 (95% CI, 1.2-18.3)]).24 Among 99 elderly patients attending a Spanish psychiatric clinic, the prevalence of confirmed RLS was 11%.25 An additional 10% of patients had “possible RLS”; the diagnosis could not be confirmed due to doubts regarding the accuracy and reliability of the information provided by the patient. None of the patients in the study had previously been diagnosed with RLS. In many cases, patients commented either that they had never reported their symptoms to a clinician or that their symptoms had never been investigated. These results suggest that, although relatively common among psychiatric patients, RLS is frequently underdiagnosed. The potential presence of RLS should be considered in all patients presenting with major sleep onset insomnia and mood disorders.
Periodic leg movements during sleep (PLMS) are rhythmic movements of the toes, ankles, and/or legs and are observed in 85%-95% of patients with RLS.26 The presence of PLMS is important because these movements can lead to partial or complete arousals from sleep, which may lead to other comorbid complications. Selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), and other types of antidepressants can increase the occurrence of PLMS. Treatment with antidepressants, such as amitriptyline, mirtazapine, and multiple SSRIs (fluoxetine, paroxetine, sertraline), has been shown to evoke or increase the frequency of PLMS compared to placebo.27-31 The effect of these agents on PLMS has been observed in healthy subjects as well as in subjects with depression. In patients with depression, Yang et al30 reported that patients given venlafaxine or SSRIs had a more than 5 times greater risk of PLMS compared to control subjects. In healthy subjects, antidepressants, such as amitriptyline and mirtazapine, also induce or increase PLMS.27,28
This narrative review first discusses the diagnosis of RLS and then examines the epidemiologic evidence for an association between RLS and mood disorders. The relationships between RLS, sleep disturbances, and mood disorders and the theoretical pathophysiologic pathways linking RLS and mood disorders are discussed. Finally, potential associations between RLS and antidepressant use and current pharmacologic therapies for RLS are summarized.
METHOD

- Epidemiologic studies report a higher risk of depression and/or anxiety or a higher severity of anxiety and depression in patients with restless legs syndrome (RLS).
- Although the etiology of RLS remains unclear, other factors, such as pharmacologic agents affecting dopamine neurotransmission, may influence development of the disease, thus underscoring the importance of recognizing RLS in patients with psychiatric disorders.
Relevant publications were identified via a comprehensive PubMed search using the term restless legs syndrome in combination with affective/anxiety, antidepressants, anxiety/anxiety disorder, attention deficit hyperactivity disorder, depression/depressive disorder, mood/mood disorder, neuropsychiatric, panic/panic disorder, psychiatric disorder, and psychosis. English-language articles published between January 1993 and May 2013 were retrieved. Additional studies were identified by careful review of the reference lists of identified publications. Although a true systematic approach to study selection was not implemented, data were extracted on the basis of their relevance to the potential link between mood disorders and idiopathic RLS.
A total of 173 publications were retrieved, of which 32 studies (25 studies in adults and 7 in children) reported epidemiologic evidence for the relationship between idiopathic RLS and depression, anxiety, and mood disorders. These studies were reviewed in detail and ranked according to their quality. Study quality was assessed by the authors using 3 parameters: the study methodology, the quality of data, and the generalizability of the results. Each parameter was scored from 1 (high quality) to 4 (low quality), resulting in a total score of between 3 and 12. Therefore, the highest-quality studies received the lowest scores (ie, those studies that used a face-to-face interview and the help of valid, replicable instruments to collect data from the general population). Each of the authors provided their ratings independently, and an average total score for each study was calculated. These ratings are presented to provide context and as a means for comparison of the currently available evidence resulting from the above searches.
Diagnosis of RLS
As RLS is diagnosed clinically, physicians must rely on patient history rather than specific physical findings. Diagnosis can be difficult and time-consuming if a patient’s urge to move is not elicited. Many patients with RLS are initially referred to specialist care with a diagnosis of insomnia and/or depression. In the RLS Epidemiology, Symptoms and Treatment (REST) primary care study, only 13.6% of RLS sufferers (patients with at least twice-weekly symptoms likely to warrant treatment) had a previous diagnosis of the disorder; conditions commonly diagnosed by the primary care physician included back pain (34.8%), depressed mood/depression (26.9%), hypertension (26.1%), insomnia/sleeping disorder (26.0%), and anxiety (23.2%).32 A retrospective database review, which examined diagnoses received by patients in the 5 years prior to a sleep laboratory evaluation, found that a prior diagnosis of a mood disorder was significantly more common among both male (43.7% vs 10.4%) and female (46.1% vs 22.8%) patients with RLS than among matched controls.33
The observed associations between RLS and depression and anxiety disorders may make differential diagnosis challenging. Many patients with RLS, as well as those with depression and anxiety, complain of sleep disturbance.32 Sleep disruption due to RLS symptoms may result in 4 of the symptoms of depression: insomnia, loss of energy, diminished concentration, and psychomotor retardation/agitation.24 In many cases, it can be difficult to determine whether these symptoms occur as the result of depression or are related to RLS. Differential diagnosis may be difficult in cognitively impaired patients with affective disorders, who may have difficulty explaining their array of sensory complaints and distinguishing them from psychiatric complaints.25 Recognition of RLS is particularly important in patients receiving treatment for mood disorders and anxiety, as some of the medications commonly used to treat insomnia and depression may result in exacerbation of RLS symptoms.
There are no physical markers for RLS; therefore, the disorder is a clinical diagnosis based on the essential criteria defined by the International Restless Legs Syndrome Study Group (IRLSSG).6,34 These criteria can be easily remembered using the acronym URGE: U = urge to move the legs often accompanied by unpleasant/uncomfortable sensations; patients may describe their symptoms using terms such as creeping, crawling, burning, electric-current, or bugs underneath the skin; R = rest or being still worsens the urge to move; G = gyration or movement provides partial or total relief from symptoms (at least temporarily); E = evening or nighttime worsening/symptoms only occur in the evening or at night. A fifth criterion was added in 2011,35 changing the acronym to “URGED”: D = denial of other explanations of urge—occurrence of features 1-4 are not solely accounted for as symptoms primary to another medical or behavioral condition. Several other conditions can cause motor restlessness and/or leg pain and discomfort and may mimic the essential diagnostic criteria of RLS.36 For example, akathisia is characterized by a feeling of motor restlessness; in contrast to RLS, this restlessness is not circadian, generally affects the entire body, may not be relieved by movement, and is not accompanied by unpleasant sensations.36 Dysesthesias occur in patients with claudication in addition to those with RLS; however, the symptoms of claudication are not associated with an urge to move, generally increase in intensity with walking, and are relieved by rest.36 Peripheral neuropathy is characterized by painful sensations in the extremities, but, unlike RLS symptoms, these sensations are not accompanied by an urge to move, do not improve with movement, and do not show a prominent circadian pattern.36 In the majority of cases, these mimic disorders can be ruled out by physical examination and patient history; however, clinicians may find themselves occasionally perplexed as to which sensory disorder best explains a patient’s particular complaint.
Supportive features for an RLS diagnosis include a history of the disorder in first-degree relatives, a good response to dopaminergic therapy, and the presence of PLMS.6 It is important to recognize that RLS and PLMS are not synonymous. Although not unique to RLS, PLMS are highly suggestive of the disorder and are observed in up to 95% of patients with RLS.26 Polysomnography is required to diagnose PLMS. PLMS are defined as repetitive leg movements that occur in 5- to 90-second intervals and are 0.5 to 10 seconds in duration.37,38 PLMS can be quantified either by recording the activity of the anterior tibialis muscles with surface electrodes during polysomnography (electromyography) or by using accelerometers attached to the big toe, ankle, or foot to capture leg movements (actigraphy). This latter procedure can be done in the patient’s home.26 Ascertaining the presence and frequency of PLMS in patients with RLS is valuable, as these stereotypical movements can result in arousals from sleep, with consequent autonomic activation and excessive daytime sleepiness.37,39 The suggested immobilization test (SIT) was also developed to quantify subjective leg discomfort and objective leg movement and has a high sensitivity and specificity for diagnosing RLS.40 The SIT is performed during a 1-hour period prior to bedtime.41 Leg movements are quantified via surface electromyography from anterior tibialis muscles, and leg discomfort is measured via a visual analog scale. A SIT index of > 40 leg movements per hour can be used to identify patients with RLS in approximately 80% of cases.42
Epidemiologic Evidence for an Association Between RLS and Mood Disorders in Adults
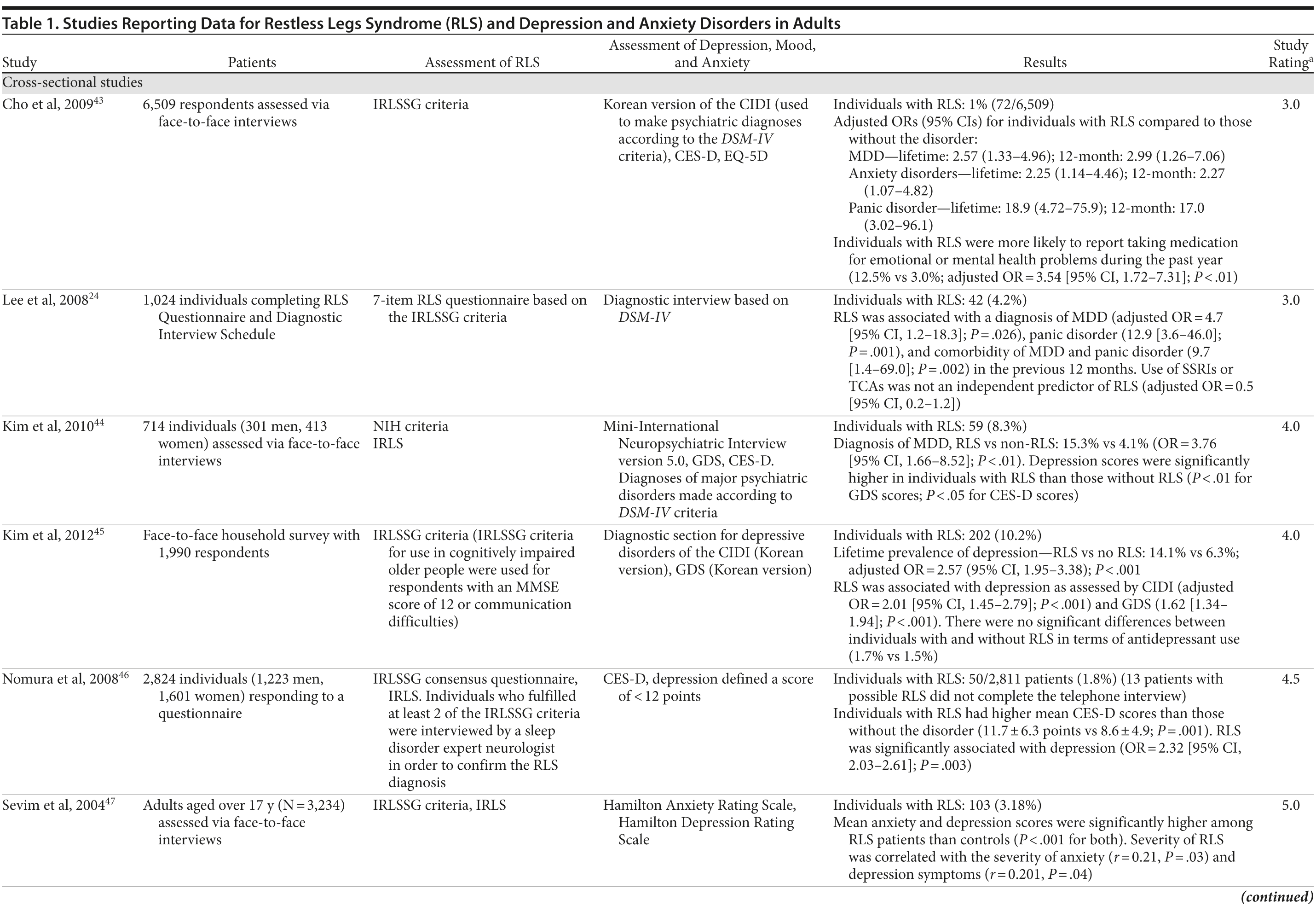
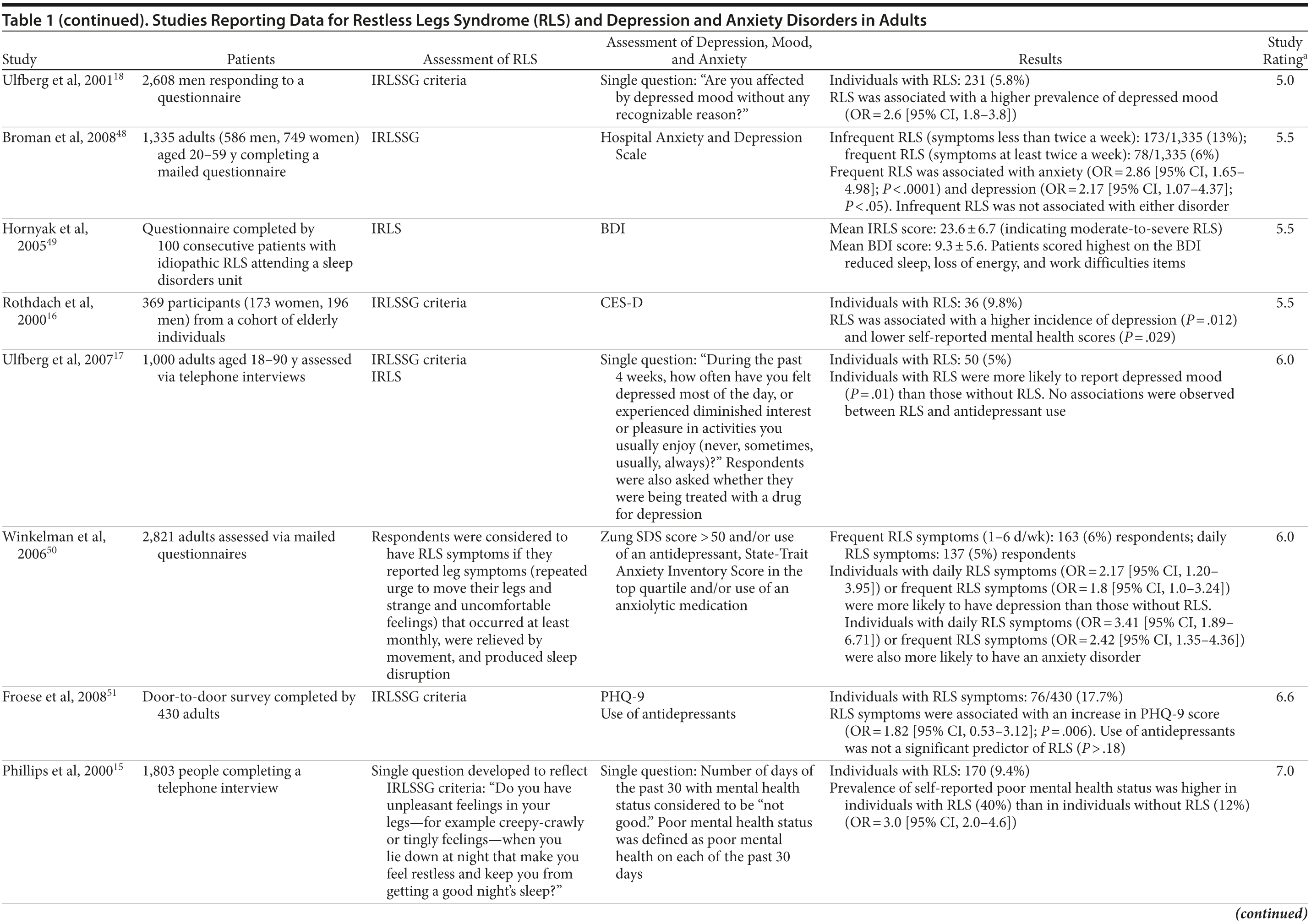
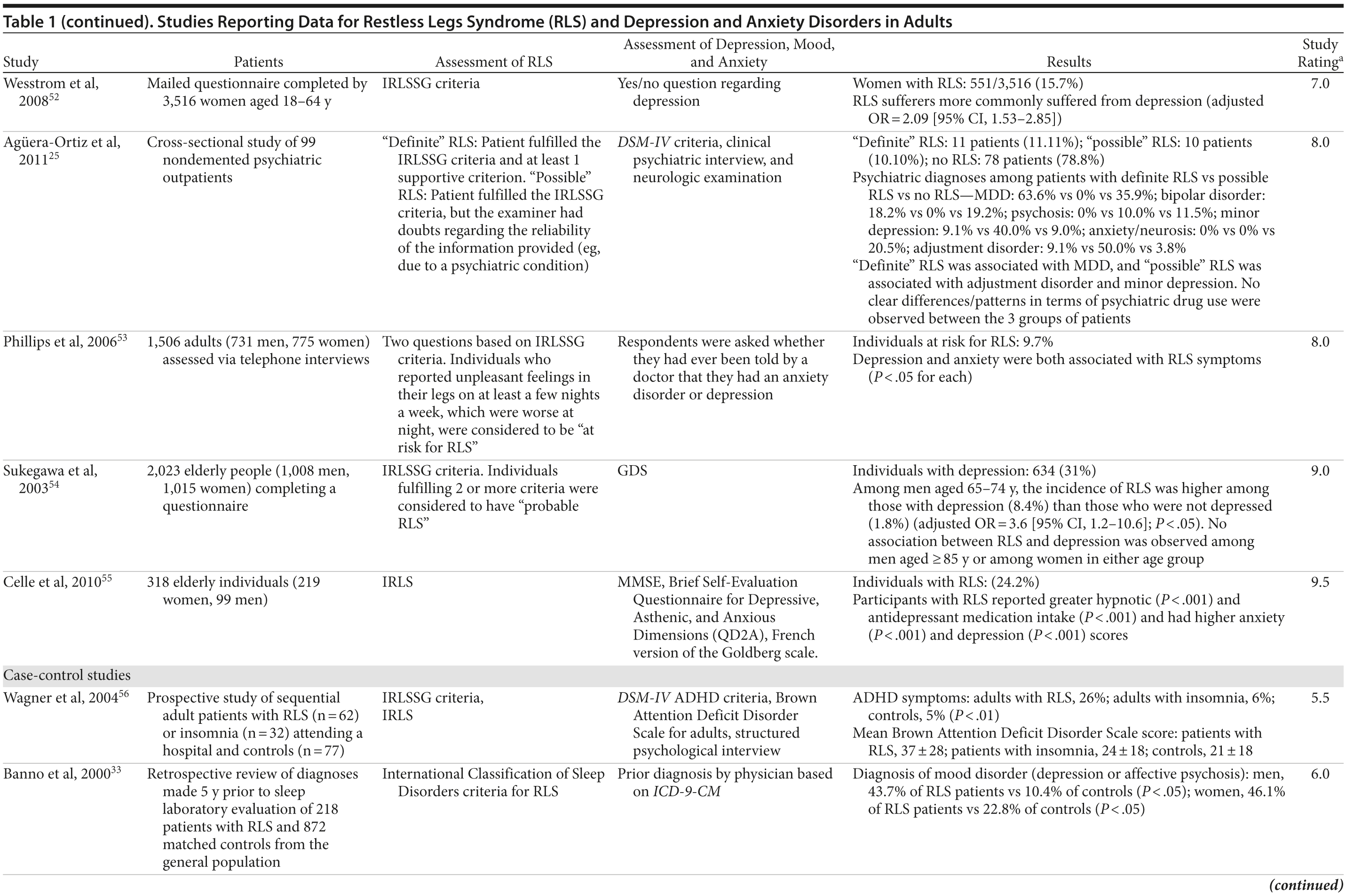
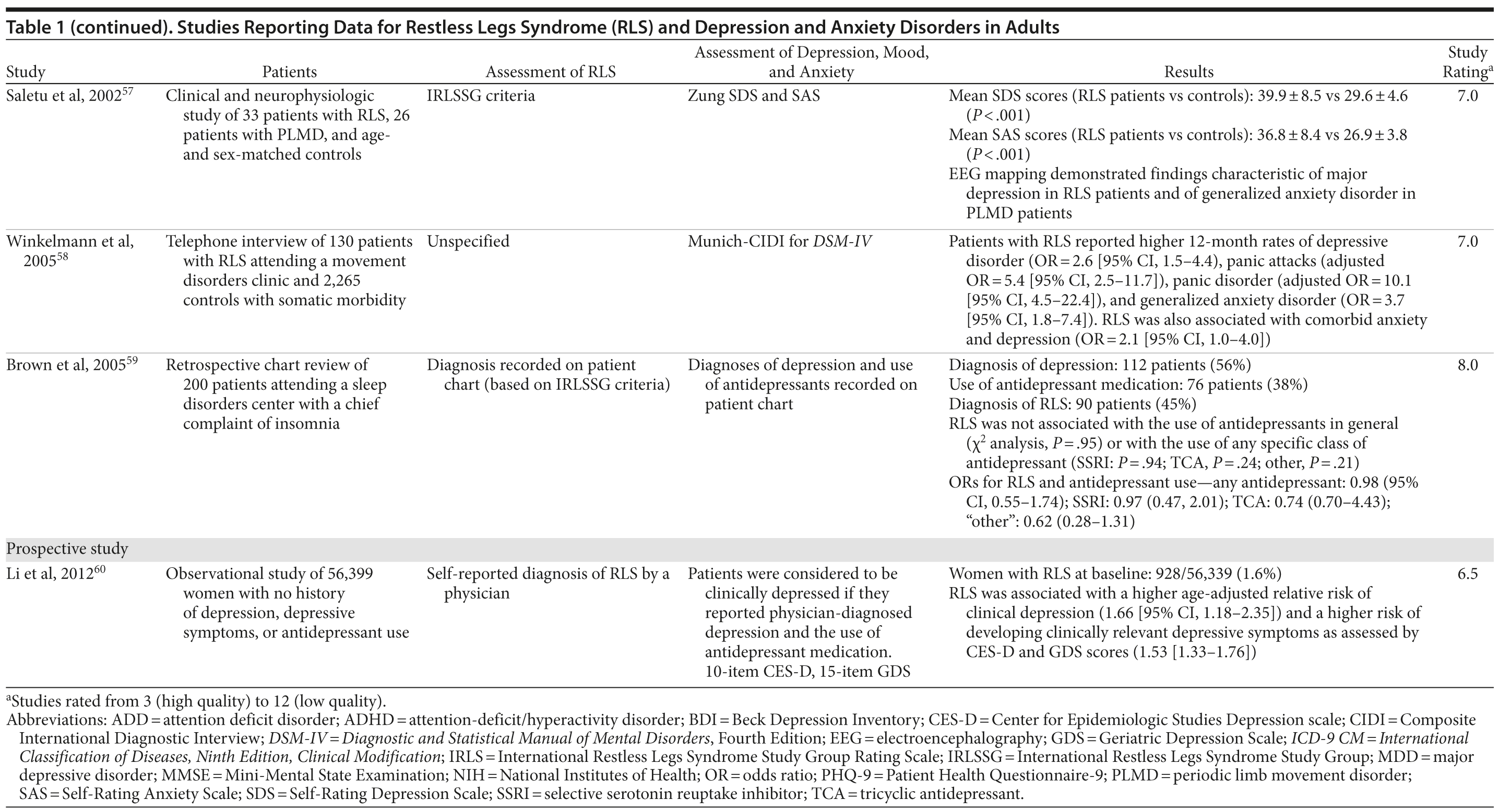
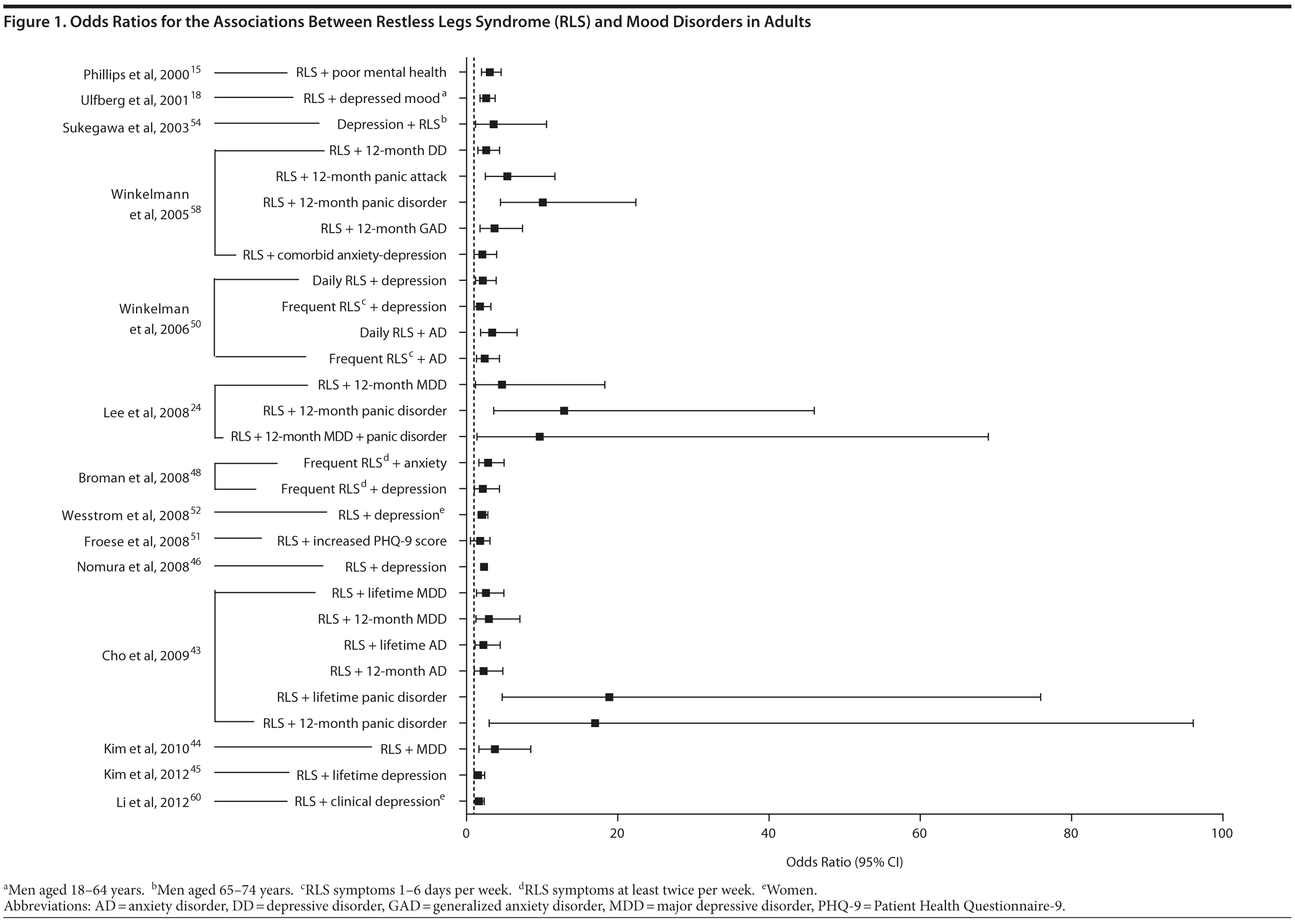
The association between RLS and mood disorders has been investigated in several epidemiologic studies (Table 1, Figure 1). Most used the IRLSSG criteria to identify patients with RLS and assessed depression and/or anxiety according to the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV)/International Classification of Diseases, Ninth Edition, Clinical Modification (ICD-9-CM) criteria or via a validated scale. The highest-quality studies applied the IRLSSG diagnostic criteria, performed elevated measures of RLS and psychiatric disorders, excluded disorders that might complicate diagnosis, and had well-defined populations of patients and controls.
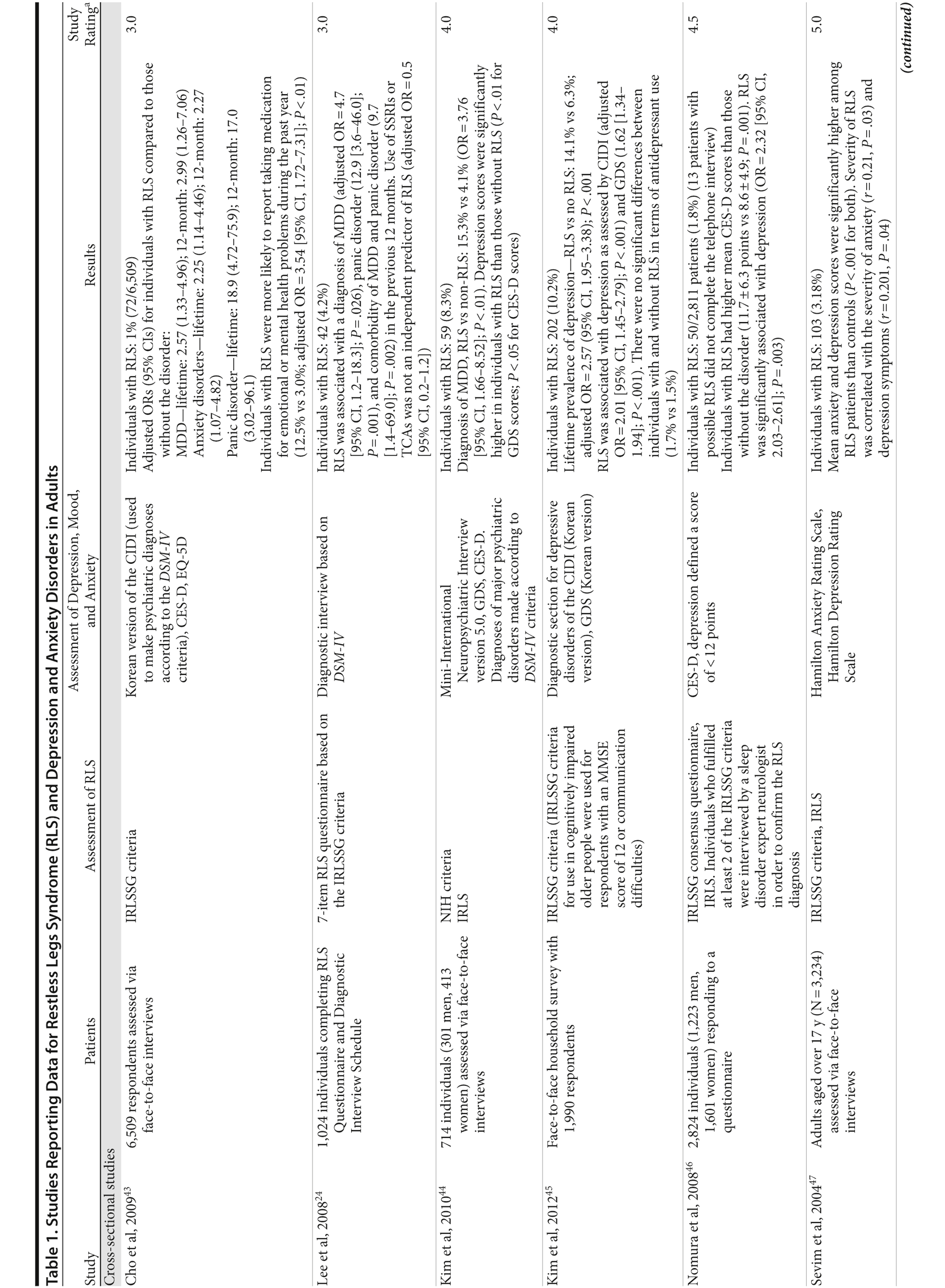
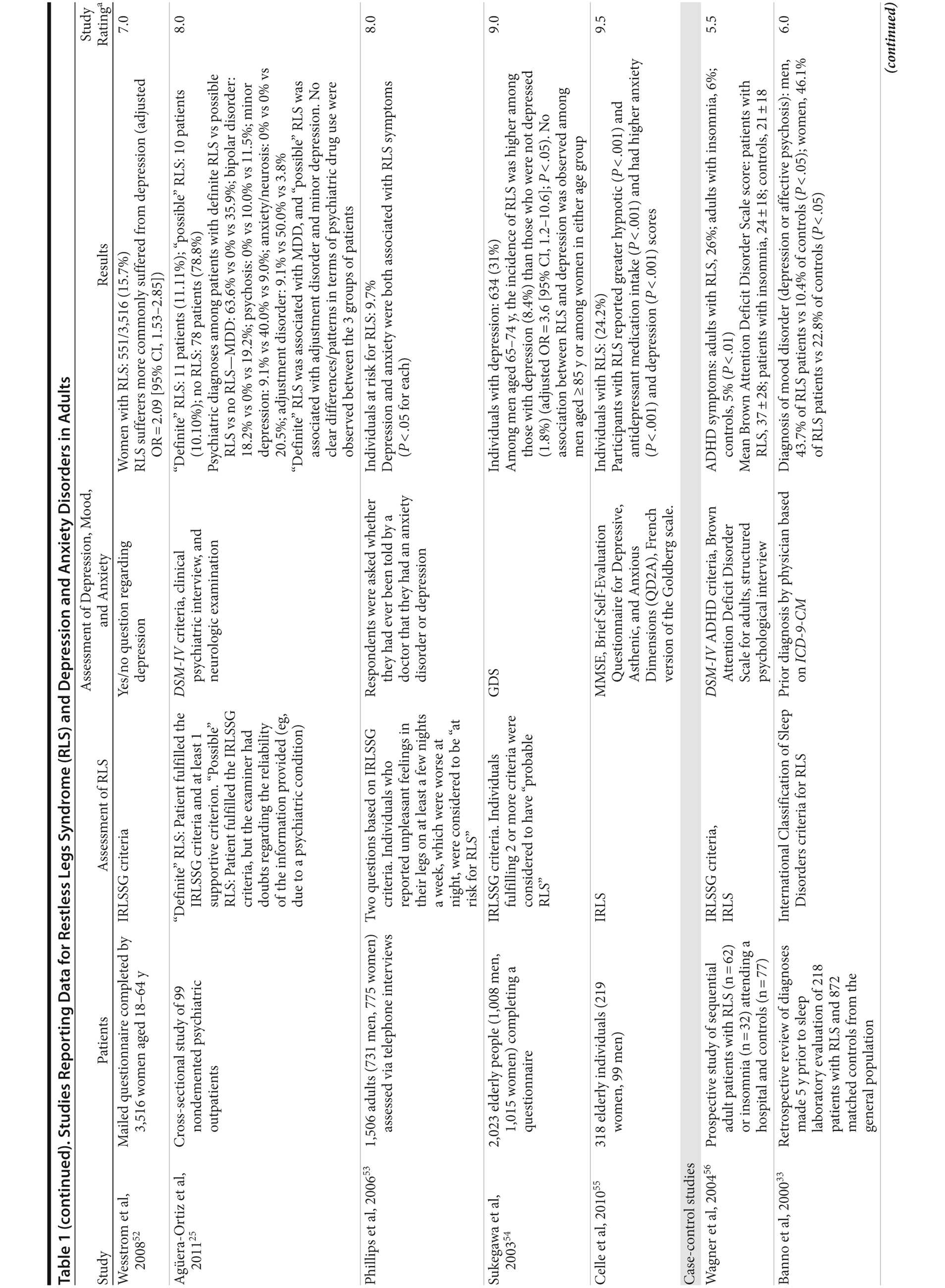

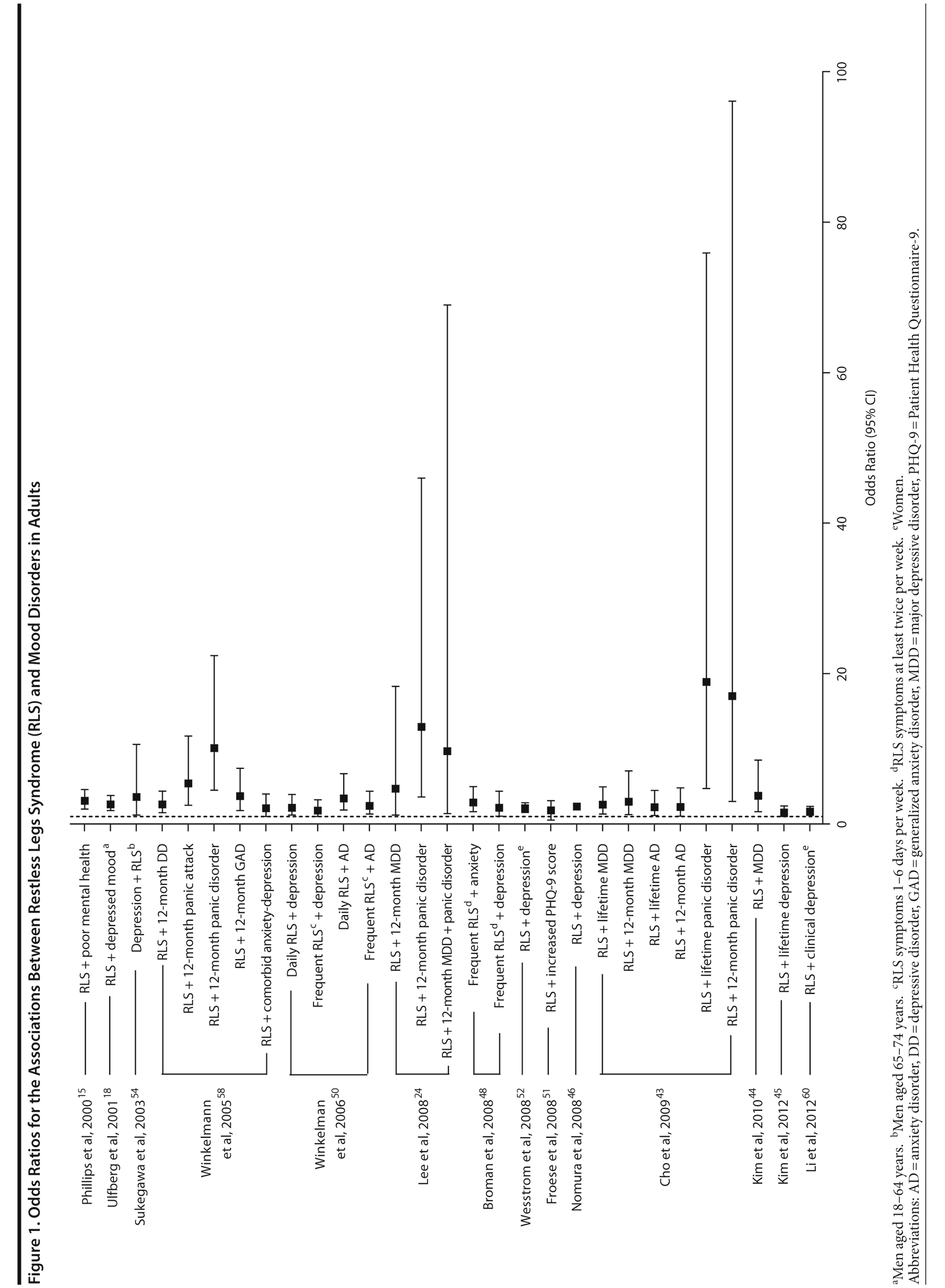
In each study, RLS was associated with a higher risk of depression and/or anxiety or a higher severity of anxiety/depression scores (Table 1). In a cross-sectional study performed in the United States using the RLS diagnostic interview, lifetime prevalence of any psychiatric disorder was found to be higher among individuals with RLS (36.8% [n = 14]) than among those without RLS symptoms (14.6% [n = 129]).24 The most common psychiatric diagnosis was DSM-IV major depressive disorder, with a lifetime prevalence of 19% (adjusted OR = 2.7 [95% CI, 1.1-6.7]; P = .031) and a 12-month prevalence of 9.5% (OR = 4.7 [95% CI, 1.2-18.3]; P = .026) among patients with RLS, in comparison to lifetime and 12-month prevalences of 8.4% and 2.2%, respectively, among controls. Individuals with RLS also had a higher lifetime prevalence of panic disorder than controls (16.7% vs 4.1%), and lifetime (adjusted OR = 5.3 [95% CI, 2.0-14.0]; P = .001) and 12-month (adjusted OR = 12.9 [95% CI, 3.6-46.0]; P = .001) odds ratios showed a strong association between panic disorder and RLS. Similar results were observed in a Korean study.43 Lifetime prevalence of any DSM-IV psychiatric disorder was higher in individuals with RLS than in controls (40.3% vs 27.7%; adjusted OR = 2.26 [95% CI, 1.39-3.67]); major depressive disorder was the most common psychiatric diagnosis. RLS was also strongly associated with lifetime (RLS vs controls: 13.9% vs 6.7%; adjusted OR = 2.25 [95% CI, 1.14-4.46]) and 12-month (11.1% vs 5.3%; adjusted OR = 2.27 [95% CI, 1.07-4.82]) prevalence of DSM-IV anxiety disorders, particularly panic disorder (lifetime: 4.2% vs 0.1%; adjusted OR = 18.9 [95% CI, 4.72-75.9]; 12-month: 2.8% vs 0.1%; adjusted OR = 17.0 [95% CI, 3.02-96.1]). Collectively, the results of these studies indicate that RLS and depression, and potentially other mood disorders, have significant comorbidity.
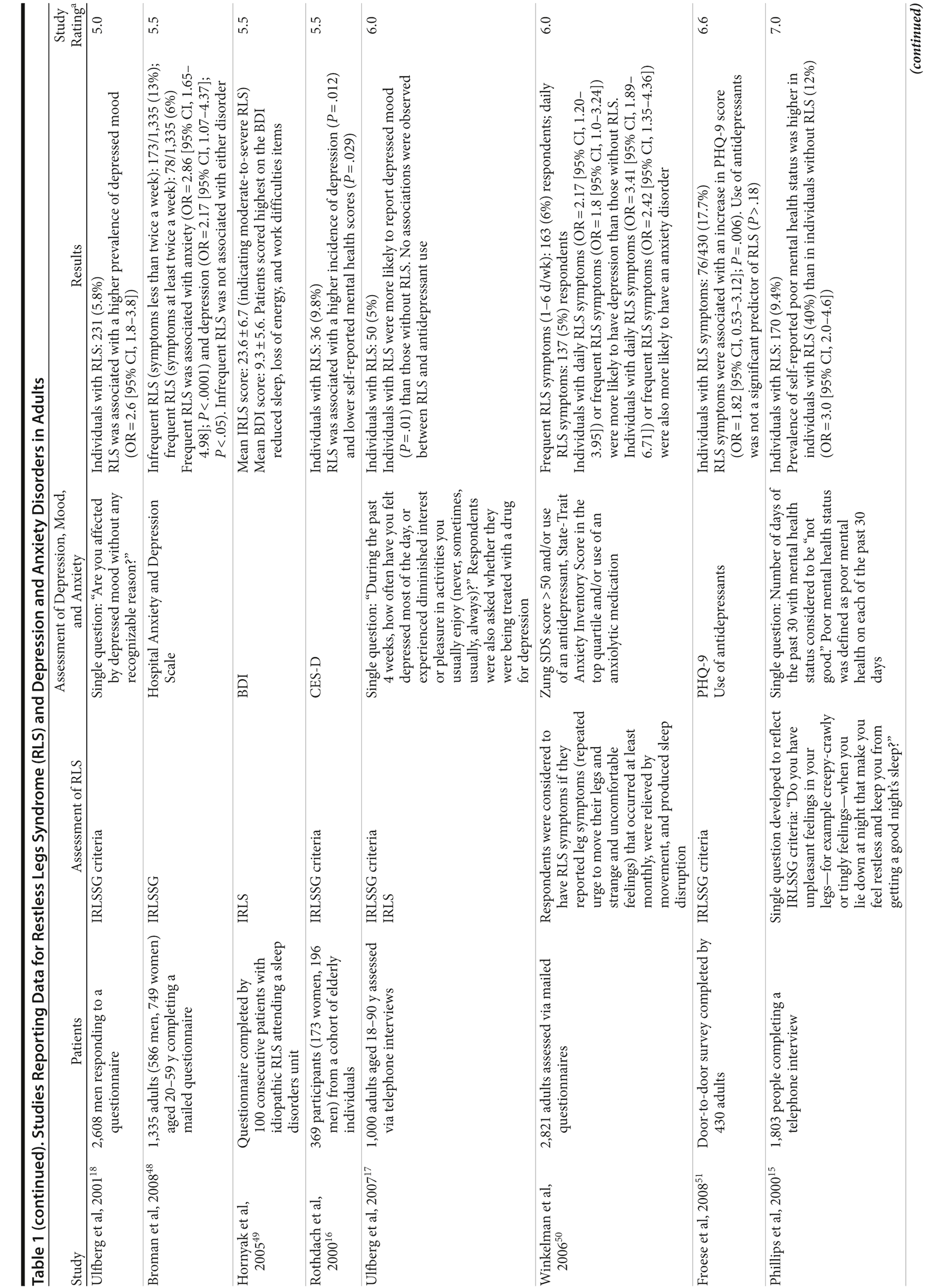
The correlation between RLS and mood disorders may be related to the frequency and severity of RLS symptoms (Table 1). The results from the Wisconsin Sleep Cohort Study indicated an increased risk of anxiety and depression among individuals with RLS.50 These associations were stronger among those with daily RLS symptoms than among those whose RLS symptoms occurred between 1 and 6 times per week. Similarly, a Swedish questionnaire study48 reported associations between RLS and anxiety and depression in individuals whose RLS symptoms occurred at least twice a week; no such associations were observed among individuals with infrequent symptoms (occurring less than twice a week). A Turkish study47 showed a correlation between the severity of RLS (as assessed by the International Restless Legs Syndrome Study Group Rating Scale [IRLS]) and the severity of anxiety and depression symptoms. As severity and frequency of RLS symptoms generally increase over time, further studies are needed to determine whether progression of RLS is associated with an increasing risk of developing depression and mood disorders.
Gender differences have been reported in the prevalence of depression among patients with RLS. In 2 studies,16,54 an increased risk of depression was observed only in male patients; female patients did not show an elevated depression risk (Table 1). In the Memory and Morbidity in Augsburg Elderly (MEMO) study, high scores on the Center for Epidemiologic Studies Depression scale (CES-D) were associated with RLS in men (OR = 13.06 [95% CI, 2.32-73.26]), but no such association was observed in women (OR = 0.90 [95% CI, 0.24-3.34]).16 In a study of elderly Japanese patients, RLS was associated with depression in men aged 65-74 years.54 However, no associations were observed among men aged ≥ 75 years or among women in either age group. Other studies, however, have shown stronger associations between depression and RLS in women (Table 1). In a Swedish study,52 women with RLS (n = 551) were more likely to suffer from depression than individuals without the disorder (n = 2,950) (adjusted OR = 2.09 [95% CI, 1.53-2.85]). A very large prospective study60 followed 56,399 women over a 6-year period. At baseline, the women were free from depressive symptoms, and 928 reported a diagnosis of RLS. The presence of RLS at baseline was associated with a higher risk of developing clinical depression (multivariate adjusted relative risk [RR]: 1.5 [95% CI, 1.1-2.1]; P = .02) and clinically relevant depressive symptoms as assessed by the 10-item CES-D and the 15-item Geriatric Depression Scale. The authors also performed a meta-analysis including data from 19 case-control and cross-sectional studies; the pooled odds ratio for developing clinical depression and/or depressive symptoms was 2.25 (95% CI, 1.94-2.60) (P for heterogeneity = .02) for women with RLS in comparison to women without the disorder. Thus, the results from these 2 studies indicate that the presence of RLS in women and in men may contribute to an increased risk of developing depression.
RLS, Sleep Disturbances, and Mood Disorders
Sleep disruption is thought to be a risk factor for both panic disorder and depression58,61; therefore, sleep disturbances and impaired quality of life due to RLS are likely to play a role in the development of these disorders. The results of a controlled study have suggested that, in most cases, RLS occurs as the primary condition (Table 1).58 The majority of patients with major depression (76.7%), dysthymia (83.3%), generalized anxiety disorder (63.6%), and panic disorder (60%) reported that the onset of their RLS symptoms preceded that of the psychiatric disorder. In comparison to patients with somatic morbidities, patients with RLS were more likely to consider sleep disturbances (47.8% vs 5.4%, OR = 12.7 [95% CI, 4.0-40.7]), depressed mood (43.5% vs 3.1%, OR = 24.9 [95% CI, 7.5-82.3]), reduced interest (34.8% vs 2.8%, OR = 19.7 [95% CI, 5.2-74.2]), and suicidal thoughts (34.8% vs 2.6%, OR = 24.1 [95% CI, 6.4-90.5]) as being entirely due to their underlying medical condition.
Epidemiologic data have suggested that RLS may be more strongly associated with sleep-related physical features of depression (eg, reduced sleep, loss of energy) than with nonsomatic depressive symptoms. In a questionnaire study,49 untreated patients with RLS generally scored higher on the Beck Depression Inventory (BDI) items related to sleep, such as reduced sleep, fatigue, and irritability, than on items related to cognitive symptoms, such as feelings of failure, guilt, and self-reproach. In addition, comparable levels of depressive symptoms unrelated to sleep impairment such as depressed mood, anhedonia, and guilt have been seen in individuals with and without RLS.24 However, the association between RLS and depression and anxiety disorders cannot be fully explained on the basis of sleep disruption. In a female-only prospective study,60 the association between RLS and depression (multivariate-adjusted RR: 1.51 [95% CI, 1.07-2.14; P = .02]) was only slightly weaker after adjusting for sleep duration and persistent snoring (multivariate-adjusted RR: 1.49 [95% CI, 1.06-2.10]; P = .02). Similarly, in a cross-sectional study62 of patients with chronic kidney disease, the association between RLS symptoms and depression (OR = 3.96 [95% CI, 2.12-7.1], P < .001) remained significant after adjusting for insomnia (OR = 2.9 [95% CI, 1.55-5.43]; P < .001), suggesting that this association may be independent of the sleep disorder.
Theoretical Pathophysiologic Pathways of RLS and Mood Disorders
RLS and mood disorders, in particular depression, may have similar neurobiological pathways. The results of a neurophysiologic study add support to the association between RLS and depression.57 Daytime electroencephalography (EEG) mapping demonstrated significant differences in electrophysiologic brain function in 33 patients with RLS in comparison to age- and sex-matched controls. The greatest differences between patients with RLS and controls were seen in EEG measures that have shown the highest correlation with the Hamilton Depression Rating Scale (HDRS) score in depressed patients: alpha and delta power, dominant frequency, and the relationship of different EEG bands across the hemispheres. These findings suggest dissociated vigilance in patients with RLS, similar to the changes described in major depression.
Although the neurobiological pathways involved in RLS have not yet been established, the effect of dopamine agonists on RLS symptom alleviation suggests that dopaminergic dysfunction may be involved.13,63-65 A dopaminergic deficiency has also been implicated in the pathophysiology of depression.66-68 Dopamine-containing circuits in the central nervous system are thought to be involved in the regulation of motivation, concentration, and the ability to experience pleasure, all 3 of which are impaired in patients with depression.69 Several dopamine receptor agonists such as pramipexole and bromocriptine have been shown to be effective in reducing depressive symptoms in otherwise healthy patients.66,70-72 Antidepressant effects of dopamine agonists may be related to their receptor binding profiles. Positron emission tomography has shown that pramipexole binds to D2/D3 receptors in extrastriatal dopaminergic regions in the human brain, including the mesolimbic dopamine system, which is involved in mood and behavior.73 The antidepressant effect of pramipexole may be related to its activation of the D2 receptor subfamily, particularly D3 receptors, which have been implicated in the pathogenesis of depression due to their pharmacology and distribution in the brain.73
The potential role of dopaminergic dysfunction in the pathogenesis of depression is supported by the high prevalence of depressive symptoms among patients with Parkinson’s disease, as this disorder is associated with loss of nigrostriatal dopaminergic neurons.69 Improvements in depressive symptoms have been observed in patients with Parkinson’s disease receiving dopamine agonists.74 Two double-blind studies75,76 evaluated the effects of pramipexole in patients with Parkinson’s disease without motor complications. In a 14-week double-blind trial comparing the antidepressant effects of pramipexole to the SSRI sertraline, both pramipexole (n = 67) and sertraline (n = 34) improved depressive symptoms as measured by the HDRS total score (-10.76 ± 5.74 for pramipexole vs -9.03 ± 7.28 for sertraline; P = .055).75 In a 12-week, placebo-controlled trial, pramipexole (n = 139) reduced depressive symptoms as measured by the BDI (treatment difference vs placebo [n = 148]: -1.9 [95% CI, -3.4 to −0.5]; P = .01) and Geriatric Depression Scale (-0.8 [95% CI, -1.5 to -0.1]; P = .035).76 Further analysis revealed that 80% of the total treatment effect could be accounted for by the direct antidepressant effect of pramipexole, with the remaining 20% mediated through alleviation of motor symptoms.76 These results suggest that the antidepressant effects of pramipexole in Parkinson’s disease are not simply the result of a treatment-mediated improvement in motor symptoms.
Dopamine dysregulation cannot fully explain the relationship between RLS and depression. Improvements in depressive symptoms have been seen in some patients with RLS receiving dopamine agonists, but not in others. In a placebo-controlled crossover trial,77 a significant improvement in mean Zung Self-Rating Depression Scale scores was observed in 11 patients with RLS following 4 weeks of therapy with the dopamine agonist pramipexole (pretreatment: 45.6 ± 11.7; posttreatment: 37.0 ± 11.6; P = .006). However, a similar study78 in 18 patients with RLS receiving levodopa showed no difference between pretreatment and posttreatment Zung Self-Rating Depression Scale scores following 4 weeks of therapy. In a larger-scale 12-week double-blind study79 of patients with moderate-to-severe idiopathic RLS and at least mild depressive symptoms, Montgomery-Asberg Depression Rating Scale (adjusted mean treatment difference: -3.6 [95% CI, -5.6 to -1.6]; P < .001), HDRS (adjusted mean treatment difference: -2.7 [95% CI, -4.4, -1.1]; P < .001), and BDI-II (adjusted mean treatment difference: -2.6 [95% CI, -4.6, -0.7]; P = .009) scores all improved with ropinirole (n = 171) vs placebo (n = 60). More research is needed to elucidate fully the potential role of dopamine and dopaminergic therapy in the relationship between RLS and mood disorders.
RLS and Mood Disorders in Children
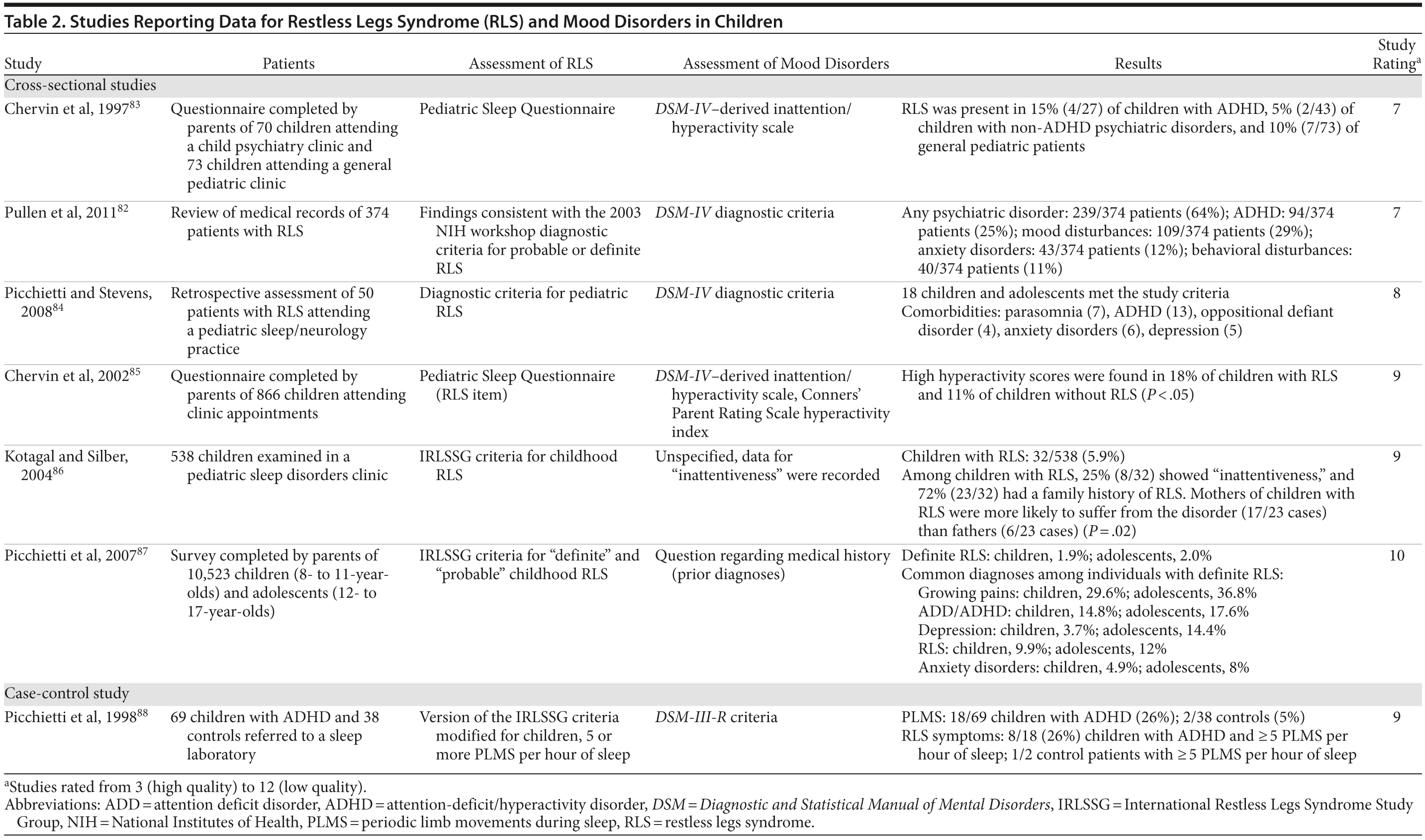
Many adults with RLS report that their symptoms began during childhood or adolescence, with around 25% reporting symptom onset between the ages of 10 and 20 years.80 Children with RLS may exhibit mood and behavioral problems such as inattentiveness, daytime somnolence, hyperactivity, and aggression, which are often attributed to disrupted sleep.81 The IRLSSG criteria have been adjusted for the diagnosis of pediatric RLS, to account for possible difficulties children may have in describing their symptoms.6 Diagnosis may be complicated by the presence of comorbid mood and anxiety disorders. In a study that examined psychiatric disorders among 347 children with RLS, 64% were found to have 1 or more psychiatric comorbidities.82 Common comorbidities included mood disturbances (29.1%), attention-deficit/hyperactivity disorder (ADHD) (25%), anxiety disorders (11.5%), and behavioral disturbances (10.9%). Girls with RLS were more likely to experience mood disturbances (OR = 1.6) and anxiety disorders (OR = 1.26), whereas ADHD and disruptive behavior disorders were more common in boys (OR = 1.94).
Evidence for the possible association between childhood RLS and ADHD has been provided by several clinical and epidemiologic studies (Table 2). Lack of sleep can lead to inattentiveness and hyperactivity in children89; however, adults with RLS have been reported to have higher Brown Attention Deficit Disorder Scale scores than adults with insomnia, suggesting that the association between RLS and ADHD is not purely due to sleep disruption.56 There may be a pathophysiologic link between the 2 disorders; deficits in both dopamine and iron have been associated with ADHD,89 and dopamine agonists may be beneficial in reducing ADHD symptoms in patients with RLS.56,90,91 ADHD and RLS may share some genetic components, and a positive family history is a feature of both disorders. The results of a large-scale cross-sectional study (N = 65,554) indicated that mothers of children with ADHD have an increased risk of having RLS (multivariate adjusted OR = 1.27 [95% CI, 1.15-1.41]; P < .0001); these results suggest that an association between the 2 disorders may exist across generations.92 Among the parents of children with ADHD, the presence of RLS may indicate an increased vulnerability to psychiatric disorders such as ADHD and anxiety disorders.93 There is also the possibility that children with RLS may be misdiagnosed with ADHD, as their leg discomfort may lead to hyperactivity, restlessness, and difficulty concentrating.88,89 As is the case with RLS, the symptoms of childhood-onset ADHD may persist into adulthood. In a prospective study, a higher prevalence of ADHD symptoms was observed among adult patients with RLS (26%) than among patients with insomnia (6%) or controls (5%).56

RLS and Antidepressants
Anecdotal evidence and case reports have suggested a possible association between RLS and the use of antidepressants.59,80 Given the purported involvement of dopamine deficiency in the etiology of RLS, it is logical to presume that medications that decrease or modify the availability of dopamine may lead to or exacerbate RLS symptomatology. Elevated PLMS indexes have been observed in patients receiving the SNRI venlafaxine and the SSRIs citalopram, fluoxetine, paroxetine, and sertraline.30 The effects of these medications on PLMS may be the result of serotonergically mediated dopamine inhibition.30 The results of a large cross-sectional study94 indicated that the use of SSRIs (OR = 3.11 [95% CI, 1.66-5.79]; P < .001) is more prevalent in those with questionnaire-defined symptoms of RLS. No significant associations were observed between RLS and the use of tricyclic antidepressants (TCAs) (OR = 0.56 [95% CI, 0.22-1.45]) or “other” types of antidepressants (OR = 0.70 [95% CI, 0.17-2.98]). Patients taking hypnotics were at greater risk of periodic limb movement disorder (OR = 2.35 [95% CI, 1.68-3.27]; P < .001) but were not significantly more likely to have RLS (OR = 1.17 [95% CI, 0.89-1.55]). In an observational study95 of 271 patients with depression receiving antidepressants for the first time, 9% experienced the appearance or worsening of RLS symptoms. The frequency of drug-induced RLS varied between the antidepressants. RLS was particularly common with mirtazapine (28%), occurred occasionally (frequency of 2%-10%) with the SSRIs (paroxetine, sertraline, escitalopram, fluoxetine, and citalopram) and SNRIs (venlafaxine and duloxetine), and did not occur with the norepinephrine reuptake inhibitor reboxetine.
Other studies have not shown a link between RLS and antidepressants. A retrospective chart review in patients with sleep disorders found no significant associations between RLS and use of antidepressants in general (OR = 0.98 [95% CI, 0.55-1.74]) or between RLS and the use of any specific class of antidepressant (SSRI: OR = 0.97 [95% CI, 0.47-2.01]; TCA: 0.74 [95% CI, 0.70-4.43]; “other”: OR = 0.62 [95% CI, 0.28-1.31]) (Table 2).59 In a study of 243 patients with affective and anxiety disorders, antidepressants and neuroleptics were not shown to be a major risk factor for RLS; nonopioid analgesics did appear to increase the risk.96 A case-control study24 indicated that the use of SSRIs and TCAs was not an independent predictor of RLS (adjusted OR = 0.5 [95% CI, 0.2-1.2]; P = NS) (Table 1). Further studies are needed to examine fully the relationship between RLS and antidepressants.
Pharmacologic Treatment for RLS
In milder cases, RLS symptoms may be improved by changes in lifestyle. Improvements in sleep hygiene and the avoidance of caffeine and excessive alcohol can be beneficial.97 Patients may find that activities such as a pre-bedtime hot bath, massage, moderate exercise, and stretching can help to relieve their symptoms.97 These measures may not be sufficient for patients with moderate-to-severe symptoms, for whom pharmacologic treatment may be indicated.98
Dopamine agonists are the agent of choice for the signs and symptoms of RLS.37,99 Three non-ergot dopaminergic agonists, ropinirole (0.25-4 mg/d), pramipexole (0.125-0.75 mg/d), and rotigotine transdermal patch (1-3 mg/24 hours), are approved by the US Food and Drug Administration (FDA) for the treatment of moderate-to-severe primary RLS.100 Ropinirole and pramipexole are oral medications administered 2 hours prior to symptom onset. Rotigotine is administered via a patch on the skin. Potential side effects of the dopamine agonists include nausea, vomiting, headaches, insomnia, and somnolence.22,99 In randomized, double-blind, placebo-controlled clinical trials, administration of ropinirole,101,102 pramipexole,103-105 and rotigotine106,107 has resulted in significant improvements in RLS symptoms, which are typically measured using the IRLS and the Clinical Global Impressions scale (CGI). The IRLS measures changes in RLS symptom severity (ie, no symptoms to very severe symptoms),108 whereas the CGI measures changes in disease status (ie, not ill at all to extremely ill). In addition, all of these agents have reported response rates (generally determined by a > 50% increase in CGI score) ranging from 46% to 60% in treated patients.101-107 It should be noted, however, that response rates for placebo in clinical trials with dopamine agonists for RLS have been as high as 43%.101-107
The anticonvulsant gabapentin enacarbil (600 mg/d) is also approved for moderate-to-severe primary RLS and should be considered for patients with RLS-related pain or painful comorbid conditions.100 Orally administered at 5:00 pm with a light snack, gabapentin enacarbil has been shown to significantly improve IRLS and CGI scores in randomized clinical trials. Over 77% of patients randomized to gabapentin enacarbil responded to treatment; 45% of patients receiving placebo also responded.109 Common adverse events included somnolence and dizziness.98 Patients should be closely monitored for any indication of suicidal thoughts or depression. In addition to the approved RLS therapies, levodopa/carbidopa (25-100 mg) is commonly used off-label.
The main limitation of dopaminergic therapy is the long-term side effect of augmentation, defined as a treatment-emergent worsening of RLS symptoms. Among 266 patients with RLS receiving dopaminergic treatment over an average of 2.7 ± 2.4 years, 54 (20%) reported definite or highly suggestive clinical indicators of augmentation.110 Augmentation rates with different drugs cannot be directly compared, as the available studies differ in terms of their duration and the methodology used to assess augmentation. However, the risk of developing augmentation does appear to be far lower with dopamine agonists than with levodopa. In 6-month studies that used established diagnostic criteria,6,111 incidences of augmentation were 60% with levodopa,112 9.2% with pramipexole (placebo: 6%),104 3.5% with ropinirole (placebo: < 1%),113 and 1.5% with rotigotine (placebo: 0.5%).114 Augmentation has not been reported in clinical studies of gabapentin enacarbil, which has no dopaminergic activity.98
The potential complication of augmentation should be considered and monitored in all patients with RLS who are receiving dopaminergic medications, particularly those who are taking doses at or above the FDA-approved limits. Ferritin levels should be assessed, as low ferritin may represent a potential biomarker of augmentation.115 Characteristic features of augmentation are an earlier onset of symptoms during the day, more rapid onset of symptoms when at rest, an increase in symptom intensity, a shorter period of relief following administration of medication, and the spread of symptoms to previously unaffected parts of the body.111 Augmentation can be distinguished from tolerance and from the natural course of disease progression, as it involves an initial response to dopaminergic therapy and then a paradoxical response (increase of symptoms with increasing dosages/decrease of symptoms with lower dosages) with a worsening of symptoms beyond baseline levels.111 Augmentation may be addressed by switching the patient to a longer-acting dopamine agonist or by reducing or stopping agonist therapy113,116; symptom worsening due to disease progression or tolerance would not respond similarly to a reduction in dosage.111 It is sometimes necessary to change to an entirely different class of medication such as alpha 2-delta compounds, benzodiazepines, or opioids.
As previously mentioned, dopaminergic treatment may have beneficial effects on comorbid mood disorders in addition to improving symptoms of RLS.80 While treatment of the underlying RLS may improve mood in patients with mild mood disorders, those with more severe symptoms may require an antidepressant.80 The choice of antidepressant should be carefully considered, as SSRIs and SNRIs may exacerbate RLS symptoms. Atypical antidepressants such as bupropion,30,117,118 trazodone,119 and nefazodone120 do not appear to exacerbate PLMS and may be suitable therapies. Other drugs that may worsen or induce RLS symptoms include the atypical antipsychotics quetiapine,121-123 risperidone,124 and olanzapine.125-127 There are case reports of improvement of RLS symptoms in patients taking the atypical antipsychotic aripiprazole, which has partial agonist and antagonist properties.128-130 At present, there is no literature on RLS and electroconvulsive therapy for depression.
Practical Points for the Diagnosis and Treatment of RLS
Some practical points regarding the diagnosis and treatment of RLS are worth considering. First, it is important to establish whether the symptoms of RLS are of an idiopathic nature or triggered by an underlying cause. As discussed, potential secondary causes include iron deficiency, pregnancy, and end-stage renal failure with dialysis.37 Iron deficiency can be addressed with iron supplementation in both patients with anemia and those receiving dialysis.131 In women who develop RLS during pregnancy, RLS symptoms may resolve within the first postpartum weeks.37 Since patients with mood disorders, including depression, may be prescribed agents that may affect central dopamine activity, it is important to recognize that these drugs are potential triggers of RLS symptoms. Exacerbating agents include dopamine antagonists (including antiemetics), centrally active antihistamines, and many antidepressants (except bupropion).37
Diagnosis of RLS can be achieved following the 5 criteria described by the IRLSSG.35 One of the key diagnostic factors of RLS is that the urge to move the legs due to the uncomfortable sensations has a very definite circadian rhythmicity. There are conditions that mimic and may fulfill some of the diagnostic criteria for RLS, such as akathisia and peripheral neuropathy; however, these syndromes lack the circadian rhythmicity that defines RLS.36 Sleep disruption is a major complaint of patients with RLS.32,49 Therefore, patients reporting sleep disturbances also should be evaluated for the presence of RLS.
The symptoms of RLS are chronic and progressively worsen over time in a majority of patients; therefore, long-term pharmacologic treatment of symptoms is quite likely necessary. In more mild or moderate cases of RLS, symptoms may be alleviated by eliminating alcohol or caffeine later in the day or into the early evening, practicing good sleep hygiene, and considering brief activity before bedtime.37 The decision to pharmacologically treat RLS is based on the frequency and severity of symptoms and the extent to which these symptoms negatively affect the patient’s sleep, quality of life, and overall daytime and social functioning.37 In many cases, a suspicion of RLS can be confirmed by a positive response to a trial course of dopaminergic therapy.6 Dopamine agonists are thought to be the first-line pharmacologic therapy for symptoms of RLS.65 Treatment complications, such as augmentation, can develop with dopaminergic agonists over time. Other medications, including the alpha 2-delta compounds, benzodiazepines, sleep-promoting compounds, and opioids, can be added to the pharmacologic regimen if sleep problems persist, if the response to dopamine agonists begins to wane, if the disease progresses to more severe levels, or if augmentation develops.
CONCLUSIONS
As RLS and mood disorders are frequently comorbid, RLS may present in the psychiatry practice. RLS is a sensorimotor neurologic disorder with a significant impact on sleep and quality of life. Sleep disruption due to RLS symptoms may have a negative impact on mood and may contribute to higher rates of depression and anxiety disorders in RLS patients. In addition, RLS, depression, and mood disorders may share similar pathophysiologic networks in the central nervous system. Dopaminergic dysfunction has been implicated in the pathophysiology of both RLS and depression. Recognition of comorbid RLS is particularly important in patients with psychiatric disorders, as many commonly used antidepressants and atypical antipsychotics may exacerbate the sensory symptoms of RLS, worsening sleep disruption and the underlying psychiatric disorder. Dopamine agonists are generally effective in improving RLS symptoms and may have a beneficial effect on comorbid mood disorders. Given the likely associations between RLS and mood disorders, clinicians should consider the potential presence of RLS in patients who are prescribed medications that may alter dopaminergic neurotransmission, who complain of sleep disturbances (especially in the first half of the night), or who have psychiatric comorbidities that may be related to RLS.
Drug names: aripiprazole (Abilify), bromocriptine (Parlodel, Cycloset, and others), bupropion (Wellbutrin, Aplenzin, and others), citalopram (Celexa and others), duloxetine (Cymbalta), escitalopram (Lexapro and others), fluoxetine (Prozac and others), gabapentin enacarbil (Horizant), levodopa and carbidopa (Sinemet CR, Parcopa, and others), mirtazapine (Remeron and others), olanzapine (Zyprexa), paroxetine (Paxil, Pexeva, and others), pramipexole (Mirapex and others), quetiapine (Seroquel), risperidone (Risperdal and others), ropinirole (Requip and others), rotigotine (Neupro), sertraline (Zoloft and others), trazodone (Oleptro and others), venlafaxine (Effexor and others). Author affiliations: Sleep Medicine Associates of Texas, PA, and Department of Psychiatry, University of Texas Southwestern Medical Center, Dallas (Dr Becker); and Advanced Sleep Center, Metairie, and Tulane University School of Medicine, New Orleans, Louisiana (Dr Sharon).
Potential conflicts of interest: Dr Becker has received financial support for participation as a consultant/medical advisor for UCB Pharma and Xenoport, has received grant/research support as a principal investigator from UCB Pharma and Aerial Biopharma, and has received financial support for participation in speakers/advisory boards for UCB Pharma. Dr Sharon has received grant/research support as a principal investigator from UCB Pharma and has received financial support for participation in speakers/advisory boards for UCB Pharma.
Funding/support: Writing and editorial assistance was funded by UCB Pharma, Smyrna, Georgia.
Role of the sponsor: The sponsor provided funding for writing and editorial assistance, which included completing literature searches. Although the sponsor was provided an opportunity to review and comment on a draft of the manuscript, the authors are responsible for the content of this article.
Acknowledgments: The authors acknowledge Hannah Carney, PhD, Evidence Scientific Solutions, London, England, for writing and editorial assistance, which was funded by UCB Pharma, Smyrna, Georgia.
REFERENCES
1. Allen RP, Walters AS, Montplaisir J, et al. Restless legs syndrome prevalence and impact: REST general population study. Arch Intern Med. 2005;165(11):1286-1292. PubMed doi:10.1001/archinte.165.11.1286
2. Berger K, Luedemann J, Trenkwalder C, et al. Sex and the risk of restless legs syndrome in the general population. Arch Intern Med. 2004;164(2):196-202. PubMed doi:10.1001/archinte.164.2.196
3. Innes KE, Selfe TK, Agarwal P. Prevalence of restless legs syndrome in North American and Western European populations: a systematic review. Sleep Med. 2011;12(7):623-634. PubMed doi:10.1016/j.sleep.2010.12.018
4. Ohayon MM, O’ Hara R, Vitiello MV. Epidemiology of restless legs syndrome: a synthesis of the literature. Sleep Med Rev. 2012;16(4):283-295. PubMed doi:10.1016/j.smrv.2011.05.002
5. Allen RP, Bharmal M, Calloway M. Prevalence and disease burden of primary restless legs syndrome: results of a general population survey in the United States. Mov Disord. 2011;26(1):114-120. PubMed doi:10.1002/mds.23430
6. Allen RP, Picchietti D, Hening WA, et al; International Restless Legs Syndrome Study Group. Restless legs syndrome: diagnostic criteria, special considerations, and epidemiology: a report from the restless legs syndrome diagnosis and epidemiology workshop at the National Institutes of Health. Sleep Med. 2003;4(2):101-119. PubMed doi:10.1016/S1389-9457(03)00010-8
7. Winkelman JW. Considering the causes of RLS. Eur J Neurol. 2006;13(suppl 3):8-14. PubMed
8. Patton SM, Cho YW, Clardy TW, et al. Proteomic analysis of the cerebrospinal fluid of patients with restless legs syndrome/Willis-Ekbom disease. Fluids Barriers CNS. 2013;10(1):20. PubMed doi:10.1186/2045-8118-10-20
9. Ferini-Strambi L. RLS-like symptoms: differential diagnosis by history and clinical assessment. Sleep Med. 2007;8(suppl 2):S3-S6. PubMed doi:10.1016/j.sleep.2007.04.009
10. Stefansson H, Rye DB, Hicks A, et al. A genetic risk factor for periodic limb movements in sleep. N Engl J Med. 2007;357(7):639-647. PubMed doi:10.1056/NEJMoa072743
11. Winkelmann J, Czamara D, Schormair B, et al. Genome-wide association study identifies novel restless legs syndrome susceptibility loci on 2p14 and 16q12.1. PLoS Genet. 2011;7(7):e1002171. PubMed doi:10.1371/journal.pgen.1002171
12. Winkelmann J, Schormair B, Lichtner P, et al. Genome-wide association study of restless legs syndrome identifies common variants in three genomic regions. Nat Genet. 2007;39(8):1000-1006. PubMed doi:10.1038/ng2099
13. Allen RP, Earley CJ. The role of iron in restless legs syndrome. Mov Disord. 2007;22(suppl 18):S440-S448. PubMed doi:10.1002/mds.21607
14. Becker PM. The biopsychosocial effects of restless legs syndrome (RLS). Neuropsychiatr Dis Treat. 2006;2(4):505-512. PubMed doi:10.2147/nedt.2006.2.4.505
15. Phillips B, Young T, Finn L, et al. Epidemiology of restless legs symptoms in adults. Arch Intern Med. 2000;160(14):2137-2141. PubMed doi:10.1001/archinte.160.14.2137
16. Rothdach AJ, Trenkwalder C, Haberstock J, et al. Prevalence and risk factors of RLS in an elderly population: the MEMO study. Memory and Morbidity in Augsburg Elderly. Neurology. 2000;54(5):1064-1068. PubMed doi:10.1212/WNL.54.5.1064
17. Ulfberg J, Bjorvatn B, Leissner L, et al; Nordic RLS Study Group. Comorbidity in restless legs syndrome among a sample of Swedish adults. Sleep Med. 2007;8(7-8):768-772. PubMed doi:10.1016/j.sleep.2006.11.015
18. Ulfberg J, Nyström B, Carter N, et al. Prevalence of restless legs syndrome among men aged 18 to 64 years: an association with somatic disease and neuropsychiatric symptoms. Mov Disord. 2001;16(6):1159-1163. PubMed doi:10.1002/mds.1209
19. Choi JW, Ko D, Lee GT, et al. Reduced neural synchrony in patients with restless legs syndrome during a visual oddball task. PLoS ONE. 2012;7(7):e42312. PubMed doi:10.1371/journal.pone.0042312
20. Fulda S, Beitinger ME, Reppermund S, et al. Short-term attention and verbal fluency is decreased in restless legs syndrome patients. Mov Disord. 2010;25(15):2641-2648. PubMed doi:10.1002/mds.23353
21. Fulda S, Szesny N, Ising M, et al. Further evidence for executive dysfunction in subjects with RLS from a non-clinical sample. Sleep Med. 2011;12(10):1003-1007. PubMed doi:10.1016/j.sleep.2011.04.013
22. Earley CJ, Silber MH. Restless legs syndrome: understanding its consequences and the need for better treatment. Sleep Med. 2010;11(9):807-815. PubMed doi:10.1016/j.sleep.2010.07.007
23. Szentkiralyi A, Völzke H, Hoffmann W, et al. The relationship between depressive symptoms and restless legs syndrome in two prospective cohort studies. Psychosom Med. 2013;75(4):359-365. PubMed doi:10.1097/PSY.0b013e31828bbbf1
24. Lee HB, Hening WA, Allen RP, et al. Restless legs syndrome is associated with DSM-IV major depressive disorder and panic disorder in the community. J Neuropsychiatry Clin Neurosci. 2008;20(1):101-105. PubMed doi:10.1176/appi.neuropsych.20.1.101
25. Agüera-Ortiz L, Perez MI, Osorio RS, et al. Prevalence and clinical correlates of restless legs syndrome among psychogeriatric patients. Int J Geriatr Psychiatry. 2011;26(12):1252-1259. PubMed doi:10.1002/gps.2674
26. Rye DB, Trotti LM. Restless legs syndrome and periodic leg movements of sleep. Neurol Clin. 2012;30(4):1137-1166. PubMed doi:10.1016/j.ncl.2012.08.004
27. Fulda S, Kloiber S, Dose T, et al. Mirtazapine provokes periodic leg movements during sleep in young healthy men. Sleep. 2013;36(5):661-669. PubMed
28. Goerke M, Rodenbeck A, Cohrs S, et al. The influence of the tricyclic antidepressant amitriptyline on periodic limb movements during sleep. Pharmacopsychiatry. 2013;46(3):108-113. PubMed doi:10.1055/s-0032-1331702
29. Hoque R, Chesson AL Jr. Pharmacologically induced/exacerbated restless legs syndrome, periodic limb movements of sleep, and REM behavior disorder/REM sleep without atonia: literature review, qualitative scoring, and comparative analysis. J Clin Sleep Med. 2010;6(1):79-83. PubMed
30. Yang C, White DP, Winkelman JW. Antidepressants and periodic leg movements of sleep. Biol Psychiatry. 2005;58(6):510-514. PubMed doi:10.1016/j.biopsych.2005.04.022
31. Zhang B, Hao Y, Jia F, et al. Sertraline and periodic limb movements during sleep: an 8-week open-label study in depressed patients with insomnia. Sleep Med. 2013;14(12):1405-1412. PubMed doi:10.1016/j.sleep.2013.07.019
32. Hening W, Walters AS, Allen RP, et al. Impact, diagnosis and treatment of restless legs syndrome (RLS) in a primary care population: the REST (RLS epidemiology, symptoms, and treatment) primary care study. Sleep Med. 2004;5(3):237-246. PubMed doi:10.1016/j.sleep.2004.03.006
33. Banno K, Delaive K, Walld R, et al. Restless legs syndrome in 218 patients: associated disorders. Sleep Med. 2000;1(3):221-229. PubMed doi:10.1016/S1389-9457(00)00023-X
34. Walters AS; The International Restless Legs Syndrome Study Group. Toward a better definition of the restless legs syndrome. Mov Disord. 1995;10(5):634-642. PubMed doi:10.1002/mds.870100517
35. International Restless Legs Syndrome Study Group. Revised IRLSSG Diagnostic Criteria for RLS. http://irlssg.org/diagnostic-criteria/. Accessed August 6, 2012.
36. Merlino G, Serafini A, Robiony F, et al. Restless legs syndrome: differential diagnosis and management with rotigotine. Neuropsychiatr Dis Treat. 2009;5:67-80. PubMed
37. Earley CJ, Allen RP, Hening W. Restless legs syndrome and periodic leg movements in sleep. Handb Clin Neurol. 2011;99:913-948. PubMed doi:10.1016/B978-0-444-52007-4.00015-1
38. Walters AS, Rye DB. Review of the relationship of restless legs syndrome and periodic limb movements in sleep to hypertension, heart disease, and stroke. Sleep. 2009;32(5):589-597. PubMed
39. Ferini-Strambi L, Walters AS, Sica D. The relationship among restless legs syndrome (Willis-Ekbom Disease), hypertension, cardiovascular disease, and cerebrovascular disease [published online ahead of print August 21, 2013]. J Neurol. 2013. PubMed doi:10.1007/s00415-013-7065-1
40. Michaud M, Paquet J, Lavigne G, et al. Sleep laboratory diagnosis of restless legs syndrome. Eur Neurol. 2002;48(2):108-113. PubMed doi:10.1159/000062996
41. Michaud M, Lavigne G, Desautels A, et al. Effects of immobility on sensory and motor symptoms of restless legs syndrome. Mov Disord. 2002;17(1):112-115. PubMed doi:10.1002/mds.10004
42. Montplaisir J, Boucher S, Nicolas A, et al. Immobilization tests and periodic leg movements in sleep for the diagnosis of restless leg syndrome. Mov Disord. 1998;13(2):324-329. PubMed doi:10.1002/mds.870130220
43. Cho SJ, Hong JP, Hahm BJ, et al. Restless legs syndrome in a community sample of Korean adults: prevalence, impact on quality of life, and association with DSM-IV psychiatric disorders. Sleep. 2009;32(8):1069-1076. PubMed
44. Kim KW, Yoon IY, Chung S, et al. Prevalence, comorbidities and risk factors of restless legs syndrome in the Korean elderly population: results from the Korean Longitudinal Study on Health and Aging. J Sleep Res. 2010;19(1, pt 1):87-92. PubMed doi:10.1111/j.1365-2869.2009.00739.x
45. Kim WH, Kim BS, Kim SK, et al. Restless legs syndrome in older people: a community-based study on its prevalence and association with major depressive disorder in older Korean adults. Int J Geriatr Psychiatry. 2012;27(6):565-572. PubMed doi:10.1002/gps.2754
46. Nomura T, Inoue Y, Kusumi M, et al. Prevalence of restless legs syndrome in a rural community in Japan. Mov Disord. 2008;23(16):2363-2369. PubMed doi:10.1002/mds.22274
47. Sevim S, Dogu O, Kaleagasi H, et al. Correlation of anxiety and depression symptoms in patients with restless legs syndrome: a population based survey. J Neurol Neurosurg Psychiatry. 2004;75(2):226-230. PubMed
48. Broman JE, Mallon L, Hetta J. Restless legs syndrome and its relationship with insomnia symptoms and daytime distress: epidemiological survey in Sweden. Psychiatry Clin Neurosci. 2008;62(4):472-475. PubMed doi:10.1111/j.1440-1819.2008.01825.x
49. Hornyak M, Kopasz M, Berger M, et al. Impact of sleep-related complaints on depressive symptoms in patients with restless legs syndrome. J Clin Psychiatry. 2005;66(9):1139-1145. PubMed doi:10.4088/JCP.v66n0909
50. Winkelman JW, Finn L, Young T. Prevalence and correlates of restless legs syndrome symptoms in the Wisconsin Sleep Cohort. Sleep Med. 2006;7(7):545-552. PubMed doi:10.1016/j.sleep.2006.01.004
51. Froese CL, Butt A, Mulgrew A, et al. Depression and sleep-related symptoms in an adult, indigenous, North American population. J Clin Sleep Med. 2008;4(4):356-361. PubMed
52. Wesstrom J, Nilsson S, Sundstrom-Poromaa I, et al. Restless legs syndrome among women: prevalence, co-morbidity and possible relationship to menopause. Climacteric. 2008;11(5):422-428. PubMed doi:10.1080/13697130802359683
53. Phillips B, Hening W, Britz P, et al. Prevalence and correlates of restless legs syndrome: results from the 2005 National Sleep Foundation Poll. Chest. 2006;129(1):76-80. PubMed doi:10.1378/chest.129.1.76
54. Sukegawa T, Itoga M, Seno H, et al. Sleep disturbances and depression in the elderly in Japan. Psychiatry Clin Neurosci. 2003;57(3):265-270. PubMed doi:10.1046/j.1440-1819.2003.01115.x
55. Celle S, Roche F, Kerleroux J, et al. Prevalence and clinical correlates of restless legs syndrome in an elderly French population: the synapse study. J Gerontol A Biol Sci Med Sci. 2010;65(2):167-173. PubMed doi:10.1093/gerona/glp161
56. Wagner ML, Walters AS, Fisher BC. Symptoms of attention-deficit/hyperactivity disorder in adults with restless legs syndrome. Sleep. 2004;27(8):1499-1504. PubMed
57. Saletu B, Anderer P, Saletu M, et al. EEG mapping, psychometric, and polysomnographic studies in restless legs syndrome (RLS) and periodic limb movement disorder (PLMD) patients as compared with normal controls. Sleep Med. 2002;3(suppl):S35-S42. PubMed doi:10.1016/S1389-9457(02)00147-8
58. Winkelmann J, Prager M, Lieb R, et al. “Anxietas tibiarum”: depression and anxiety disorders in patients with restless legs syndrome. J Neurol. 2005;252(1):67-71. PubMed doi:10.1007/s00415-005-0604-7
59. Brown LK, Dedrick DL, Doggett JW, et al. Antidepressant medication use and restless legs syndrome in patients presenting with insomnia. Sleep Med. 2005;6(5):443-450. PubMed doi:10.1016/j.sleep.2005.03.005
60. Li Y, Mirzaei F, O’ Reilly EJ, et al. Prospective study of restless legs syndrome and risk of depression in women. Am J Epidemiol. 2012;176(4):279-288. PubMed doi:10.1093/aje/kws016
61. Riemann D, Voderholzer U. Primary insomnia: a risk factor to develop depression? J Affect Disord. 2003;76(1-3):255-259. PubMed doi:10.1016/S0165-0327(02)00072-1
62. Szentkiralyi A, Molnar MZ, Czira ME, et al. Association between restless legs syndrome and depression in patients with chronic kidney disease. J Psychosom Res. 2009;67(2):173-180. PubMed doi:10.1016/j.jpsychores.2009.05.004
63. Allen R. Dopamine and iron in the pathophysiology of restless legs syndrome (RLS). Sleep Med. 2004;5(4):385-391. PubMed doi:10.1016/j.sleep.2004.01.012
64. Jones R, Cavanna AE. The neurobiology and treatment of restless legs syndrome. Behav Neurol. 2013;26(4):283-292. PubMed
65. Trenkwalder C, Paulus W. Restless legs syndrome: pathophysiology, clinical presentation and management. Nat Rev Neurol. 2010;6(6):337-346. PubMed doi:10.1038/nrneurol.2010.55
66. Dailly E, Chenu F, Renard CE, et al. Dopamine, depression and antidepressants. Fundam Clin Pharmacol. 2004;18(6):601-607. PubMed doi:10.1111/j.1472-8206.2004.00287.x
67. Nestler EJ, Carlezon WA Jr. The mesolimbic dopamine reward circuit in depression. Biol Psychiatry. 2006;59(12):1151-1159. PubMed doi:10.1016/j.biopsych.2005.09.018
68. Yadid G, Friedman A. Dynamics of the dopaminergic system as a key component to the understanding of depression. Prog Brain Res. 2008;172:265-286. PubMed doi:10.1016/S0079-6123(08)00913-8
69. Dunlop BW, Nemeroff CB. The role of dopamine in the pathophysiology of depression. Arch Gen Psychiatry. 2007;64(3):327-337. PubMed doi:10.1001/archpsyc.64.3.327
70. Aiken CB. Pramipexole in psychiatry: a systematic review of the literature. J Clin Psychiatry. 2007;68(8):1230-1236. PubMed doi:10.4088/JCP.v68n0810
71. Corrigan MH, Denahan AQ, Wright CE, et al. Comparison of pramipexole, fluoxetine, and placebo in patients with major depression. Depress Anxiety. 2000;11(2):58-65. PubMed doi:10.1002/(SICI)1520-6394(2000)11:2<58::AID-DA2>3.0.CO;2-H
72. Waehrens J, Gerlach J. Bromocriptine and imipramine in endogenous depression: a double-blind controlled trial in out-patients. J Affect Disord. 1981;3(2):193-202. PubMed doi:10.1016/0165-0327(81)90044-6
73. Ishibashi K, Ishii K, Oda K, et al. Binding of pramipexole to extrastriatal dopamine D2/D3 receptors in the human brain: a positron emission tomography study using 11C-FLB 457. PLoS ONE. 2011;6(3):e17723. PubMed doi:10.1371/journal.pone.0017723
74. Seppi K, Weintraub D, Coelho M, et al. The Movement Disorder Society Evidence-Based Medicine Review Update: treatments for the non-motor symptoms of Parkinson’s disease. Mov Disord. 2011;26(suppl 3):S42-S80. PubMed doi:10.1002/mds.23884
75. Barone P, Scarzella L, Marconi R, et al; Depression/Parkinson Italian Study Group. Pramipexole versus sertraline in the treatment of depression in Parkinson’s disease: a national multicenter parallel-group randomized study. J Neurol. 2006;253(5):601-607. PubMed doi:10.1007/s00415-006-0067-5
76. Barone P, Poewe W, Albrecht S, et al. Pramipexole for the treatment of depressive symptoms in patients with Parkinson’s disease: a randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2010;9(6):573-580. PubMed doi:10.1016/S1474-4422(10)70106-X
77. Saletu M, Anderer P, Saletu-Zyhlarz G, et al. Acute placebo-controlled sleep laboratory studies and clinical follow-up with pramipexole in restless legs syndrome. Eur Arch Psychiatry Clin Neurosci. 2002;252(4):185-194. PubMed doi:10.1007/s00406-002-0380-7
78. Saletu M, Anderer P, Högl B, et al. Acute double-blind, placebo-controlled sleep laboratory and clinical follow-up studies with a combination treatment of rr-L-dopa and sr-L-dopa in restless legs syndrome. J Neural Transm. 2003;110(6):611-626. PubMed doi:10.1007/s00702-003-0814-z
79. Benes H, Mattern W, Peglau I, et al. Ropinirole improves depressive symptoms and restless legs syndrome severity in RLS patients: a multicentre, randomized, placebo-controlled study. J Neurol. 2011;258(6):1046-1054. PubMed doi:10.1007/s00415-010-5879-7
80. Picchietti D, Winkelman JW. Restless legs syndrome, periodic limb movements in sleep, and depression. Sleep. 2005;28(7):891-898. PubMed
81. Durmer JS, Quraishi GH. Restless legs syndrome, periodic leg movements, and periodic limb movement disorder in children. Pediatr Clin North Am. 2011;58(3):591-620. PubMed doi:10.1016/j.pcl.2011.03.005
82. Pullen SJ, Wall CA, Angstman ER, et al. Psychiatric comorbidity in children and adolescents with restless legs syndrome: a retrospective study. J Clin Sleep Med. 2011;7(6):587-596. PubMed
83. Chervin RD, Dillon JE, Bassetti C, et al. Symptoms of sleep disorders, inattention, and hyperactivity in children. Sleep. 1997;20(12):1185-1192. PubMed
84. Picchietti DL, Stevens HE. Early manifestations of restless legs syndrome in childhood and adolescence. Sleep Med. 2008;9(7):770-781. PubMed doi:10.1016/j.sleep.2007.08.012
85. Chervin RD, Archbold KH, Dillon JE, et al. Associations between symptoms of inattention, hyperactivity, restless legs, and periodic leg movements. Sleep. 2002;25(2):213-218. PubMed
86. Kotagal S, Silber MH. Childhood-onset restless legs syndrome. Ann Neurol. 2004;56(6):803-807. PubMed doi:10.1002/ana.20292
87. Picchietti D, Allen RP, Walters AS, et al. Restless legs syndrome: prevalence and impact in children and adolescents—the Peds REST study. Pediatrics. 2007;120(2):253-266. PubMed doi:10.1542/peds.2006-2767
88. Picchietti DL, England SJ, Walters AS, et al. Periodic limb movement disorder and restless legs syndrome in children with attention-deficit hyperactivity disorder. J Child Neurol. 1998;13(12):588-594. PubMed doi:10.1177/088307389801301202
89. Cortese S, Konofal E, Lecendreux M, et al. Restless legs syndrome and attention-deficit/hyperactivity disorder: a review of the literature. Sleep. 2005;28(8):1007-1013. PubMed
90. Konofal E, Arnulf I, Lecendreux M, et al. Ropinirole in a child with attention-deficit hyperactivity disorder and restless legs syndrome. Pediatr Neurol. 2005;32(5):350-351. PubMed doi:10.1016/j.pediatrneurol.2004.11.007
91. Walters AS, Mandelbaum DE, Lewin DS, et al; Dopaminergic Therapy Study Group. Dopaminergic therapy in children with restless legs/periodic limb movements in sleep and ADHD. Pediatr Neurol. 2000;22(3):182-186. PubMed doi:10.1016/S0887-8994(99)00152-6
92. Gao X, Lyall K, Palacios N, et al. RLS in middle aged women and attention deficit/hyperactivity disorder in their offspring. Sleep Med. 2011;12(1):89-91. PubMed doi:10.1016/j.sleep.2010.05.006
93. Steinlechner S, Brüggemann N, Sobottka V, et al. Restless legs syndrome as a possible predictor for psychiatric disorders in parents of children with ADHD. Eur Arch Psychiatry Clin Neurosci. 2011;261(4):285-291. PubMed doi:10.1007/s00406-010-0140-z
94. Ohayon MM, Roth T. Prevalence of restless legs syndrome and periodic limb movement disorder in the general population. J Psychosom Res. 2002;53(1):547-554. PubMed doi:10.1016/S0022-3999(02)00443-9
95. Rottach KG, Schaner BM, Kirch MH, et al. Restless legs syndrome as side effect of second generation antidepressants. J Psychiatr Res. 2008;43(1):70-75. PubMed doi:10.1016/j.jpsychires.2008.02.006
96. Leutgeb U, Martus P. Regular intake of non-opioid analgesics is associated with an increased risk of restless legs syndrome in patients maintained on antidepressants. Eur J Med Res. 2002;7(8):368-378. PubMed
97. Byrne R, Sinha S, Chaudhuri KR. Restless legs syndrome: diagnosis and review of management options. Neuropsychiatr Dis Treat. 2006;2(2):155-164. PubMed doi:10.2147/nedt.2006.2.2.155
98. Aurora RN, Kristo DA, Bista SR, et al. The treatment of restless legs syndrome and periodic limb movement disorder in adults-an update for 2012: practice parameters with an evidence-based systematic review and meta-analyses: an American Academy of Sleep Medicine Clinical Practice Guideline. Sleep. 2012;35(8):1039-1062. PubMed
99. Trenkwalder C, Hening WA, Montagna P, et al. Treatment of restless legs syndrome: an evidence-based review and implications for clinical practice. Mov Disord. 2008;23(16):2267-2302. PubMed doi:10.1002/mds.22254
100. Aurora RN, Kristo DA, Bista SR, et al. Update to the AASM Clinical Practice Guideline: “The Treatment of Restless Legs Syndrome and Periodic Limb Movement Disorder in Adults: An Update for 2012: Practice Parameters With an Evidence-Based Systematic Review and Meta-Analyses.” Sleep. 2012;35(8):1037. PubMed
101. Trenkwalder C, Garcia-Borreguero D, Montagna P, et al; Therapy with Ropiunirole; Efficacy and Tolerability in RLS 1 Study Group. Ropinirole in the treatment of restless legs syndrome: results from the TREAT RLS 1 study, a 12 week, randomised, placebo controlled study in 10 European countries. J Neurol Neurosurg Psychiatry. 2004;75(1):92-97. PubMed
102. Walters AS, Ondo WG, Dreykluft T, et al; TREAT RLS 2 (Therapy with Ropinirole: Efficacy And Tolerability in RLS 2) Study Group. Ropinirole is effective in the treatment of restless legs syndrome. TREAT RLS 2: a 12-week, double-blind, randomized, parallel-group, placebo-controlled study. Mov Disord. 2004;19(12):1414-1423. PubMed doi:10.1002/mds.20257
103. Ferini-Strambi L, Aarskog D, Partinen M, et al. Effect of pramipexole on RLS symptoms and sleep: a randomized, double-blind, placebo-controlled trial. Sleep Med. 2008;9(8):874-881. PubMed doi:10.1016/j.sleep.2008.09.001
104. Högl B, Garcia-Borreguero D, Trenkwalder C, et al. Efficacy and augmentation during 6 months of double-blind pramipexole for restless legs syndrome. Sleep Med. 2011;12(4):351-360. PubMed doi:10.1016/j.sleep.2010.12.007
105. Montagna P, Hornyak M, Ulfberg J, et al. Randomized trial of pramipexole for patients with restless legs syndrome (RLS) and RLS-related impairment of mood. Sleep Med. 2011;12(1):34-40. PubMed doi:10.1016/j.sleep.2010.08.005
106. Hening WA, Allen RP, Ondo WG, et al; SP792 Study Group. Rotigotine improves restless legs syndrome: a 6-month randomized, double-blind, placebo-controlled trial in the United States. Mov Disord. 2010;25(11):1675-1683. PubMed doi:10.1002/mds.23157
107. Trenkwalder C, Benes H, Poewe W, et al; SP790 Study Group. Efficacy of rotigotine for treatment of moderate-to-severe restless legs syndrome: a randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2008;7(7):595-604. PubMed doi:10.1016/S1474-4422(08)70112-1
108. Walters AS, LeBrocq C, Dhar A, et al; International Restless Legs Syndrome Study Group. Validation of the International Restless Legs Syndrome Study Group rating scale for restless legs syndrome. Sleep Med. 2003;4(2):121-132. PubMed doi:10.1016/S1389-9457(02)00258-7
109. Lee DO, Ziman RB, Perkins AT, et al; XP053 Study Group. A randomized, double-blind, placebo-controlled study to assess the efficacy and tolerability of gabapentin enacarbil in subjects with restless legs syndrome. J Clin Sleep Med. 2011;7(3):282-292. PubMed
110. Allen RP, Ondo WG, Ball E, et al. Restless legs syndrome (RLS) augmentation associated with dopamine agonist and levodopa usage in a community sample. Sleep Med. 2011;12(5):431-439. PubMed doi:10.1016/j.sleep.2011.03.003
111. Garc×a-Borreguero D, Allen RP, Kohnen R, et al; International Restless Legs Syndrome Study Group. Diagnostic standards for dopaminergic augmentation of restless legs syndrome: report from a World Association of Sleep Medicine-International Restless Legs Syndrome Study Group consensus conference at the Max Planck Institute. Sleep Med. 2007;8(5):520-530. PubMed doi:10.1016/j.sleep.2007.03.022
112. Högl B, Garc×a-Borreguero D, Kohnen R, et al. Progressive development of augmentation during long-term treatment with levodopa in restless legs syndrome: results of a prospective multi-center study. J Neurol. 2010;257(2):230-237. PubMed doi:10.1007/s00415-009-5299-8
113. Garc×a-Borreguero D, Högl B, Ferini-Strambi L, et al. Systematic evaluation of augmentation during treatment with ropinirole in restless legs syndrome (Willis-Ekbom disease): results from a prospective, multicenter study over 66 weeks. Mov Disord. 2012;27(2):277-283. PubMed doi:10.1002/mds.24889
114. BeneÅ¡ H, Garc×a-Borreguero D, Ferini-Strambi L, et al. Augmentation in the treatment of restless legs syndrome with transdermal rotigotine. Sleep Med. 2012;13(6):589-597. PubMed doi:10.1016/j.sleep.2011.09.016
115. Frauscher B, Gschliesser V, Brandauer E, et al. The severity range of restless legs syndrome (RLS) and augmentation in a prospective patient cohort: association with ferritin levels. Sleep Med. 2009;10(6):611-615. PubMed doi:10.1016/j.sleep.2008.09.007
116. Garc×a-Borreguero D, Williams AM. Dopaminergic augmentation of restless legs syndrome. Sleep Med Rev. 2010;14(5):339-346. PubMed doi:10.1016/j.smrv.2009.11.006
117. Bayard M, Bailey B, Acharya D, et al. Bupropion and restless legs syndrome: a randomized controlled trial. J Am Board Fam Med. 2011;24(4):422-428. PubMed doi:10.3122/jabfm.2011.04.100173
118. Nofzinger EA, Fasiczka A, Berman S, et al. Bupropion SR reduces periodic limb movements associated with arousals from sleep in depressed patients with periodic limb movement disorder. J Clin Psychiatry. 2000;61(11):858-862. PubMed doi:10.4088/JCP.v61n1108
119. Saletu-Zyhlarz GM, Abu-Bakr MH, Anderer P, et al. Insomnia in depression: differences in objective and subjective sleep and awakening quality to normal controls and acute effects of trazodone. Prog Neuropsychopharmacol Biol Psychiatry. 2002;26(2):249-260. PubMed doi:10.1016/S0278-5846(01)00262-7
120. Shen J, Kennedy SH, Levitan RD, et al. The effects of nefazodone on women with seasonal affective disorder: clinical and polysomnographic analyses. J Psychiatry Neurosci. 2005;30(1):11-16. PubMed
121. Coe HV, Hong IS. Safety of low doses of quetiapine when used for insomnia. Ann Pharmacother. 2012;46(5):718-722. PubMed doi:10.1345/aph.1Q697
122. Pinninti NR, Mago R, Townsend J, et al. Periodic restless legs syndrome associated with quetiapine use: a case report. J Clin Psychopharmacol. 2005;25(6):617-618. PubMed doi:10.1097/01.jcp.0000186870.75042.25
123. Webb J. Co-occurring akathisia and restless legs syndrome likely induced by quetiapine. J Neuropsychiatry Clin Neurosci. 2012;24(2):E46-E47. PubMed
124. Wetter TC, Brunner J, Bronisch T. Restless legs syndrome probably induced by risperidone treatment. Pharmacopsychiatry. 2002;35(3):109-111. PubMed doi:10.1055/s-2002-31514
125. Aggarwal S, Dodd S, Berk M. Restless leg syndrome associated with olanzapine: a case series. Curr Drug Saf. 2010;5(2):129-131. PubMed doi:10.2174/157488610790936187
126. Kang SG, Lee HJ, Kim L. Restless legs syndrome and periodic limb movements during sleep probably associated with olanzapine. J Psychopharmacol. 2009;23(5):597-601. PubMed doi:10.1177/0269881108091876
127. Khalid I, Rana L, Khalid TJ, et al. Refractory restless legs syndrome likely caused by olanzapine. J Clin Sleep Med. 2009;5(1):68-69. PubMed
128. Kikukawa S. Effectiveness of aripiprazole in treatment of adults with attention deficit disorder and restless legs syndrome. Int J Neuropsychopharmacol. 2008;11(3):439-440. PubMed doi:10.1017/S1461145707008310
129. McLean AJ. The use of the dopamine-receptor partial agonist aripiprazole in the treatment of restless legs syndrome. Sleep. 2004;27(5):1022. PubMed
130. Virit O, Selek S, Savas HA, et al. Improvement of restless legs syndrome and trichotillomania with aripiprazole. J Clin Pharm Ther. 2009;34(6):723-725. PubMed doi:10.1111/j.1365-2710.2009.01051.x
131. Novak M, Mendelssohn D, Shapiro CM, et al. Diagnosis and management of sleep apnea syndrome and restless legs syndrome in dialysis patients. Semin Dial. 2006;19(3):210-216. PubMed doi:10.1111/j.1525-139X.2006.00157.x
This PDF is free for all visitors!