Long-Term Effects of ADHD Medication on Adult Height: Results From the NESARC
To the Editor: Methylphenidate is the most commonly prescribed medication to treat attention-deficit/hyperactivity disorder (ADHD) in children and adolescents. The long-term impact of methylphenidate on adult height remains debated1 because a significant negative impact on children’s growth has been suggested. The Multimodal Treatment Study of ADHD showed that children receiving methylphenidate had heights of 0.17 standard deviations below the population mean at the age of 9 years.2 It has been suggested that height velocity is negatively affected by methylphenidate early in the treatment period, with a normalized growth later on.3 As evidence, Biederman et al4 found no significant association between adult height and methylphenidate in a longitudinal study of 124 patients treated for ADHD during childhood. To our knowledge, this association has never been examined in a larger sample of adult individuals with a lifetime diagnosis of ADHD.
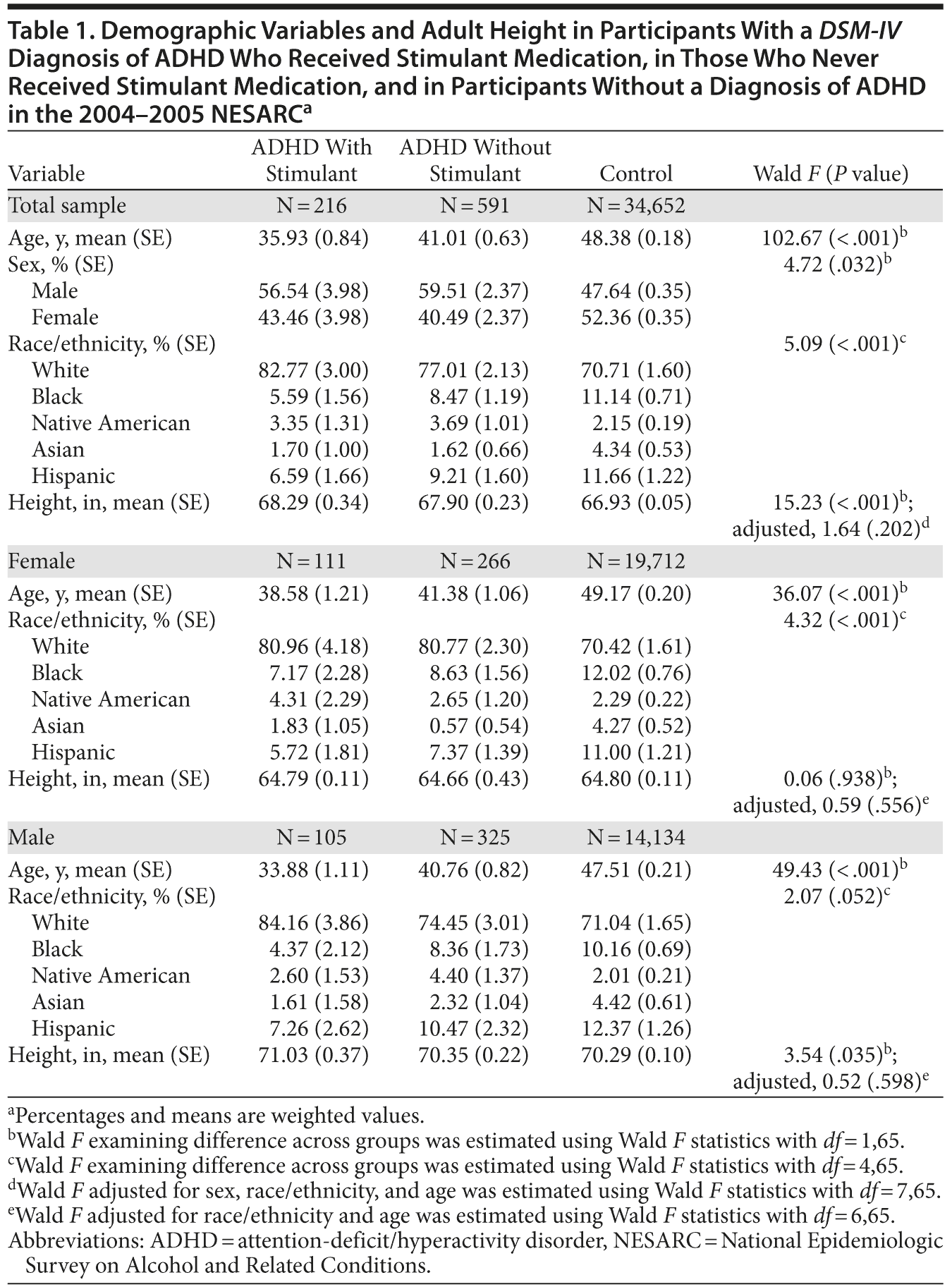
Method. Using data from the 2004-2005 National Epidemiologic Survey on Alcohol and Related Conditions (NESARC), a nationally representative survey,5 we investigated the long-term impact of ADHD medications on adult height in naturalistic conditions. In a recent study using data from the NESARC, Bernardi et al6 identified 807 subjects with a DSM-IV diagnosis of ADHD among whom 27.6% reported having received specific medication for ADHD. We compared adult height between 3 mutually independent groups: (1) participants with a lifetime DSM-IV diagnosis of ADHD who received medication, (2) participants with a lifetime DSM-IV diagnosis of ADHD who never received medication, and (3) participants without such a condition. Multinomial logistic regressions were performed following adjustments for confounders known to affect height, ie, sex, age, and ethnicity. Analyses were also performed stratified by gender. Because of the weighting and clustering used in the NESARC design, all statistical analyses were performed using the Taylor series linearization method, a designed-based method implemented using SUDAAN, version 10 (RTI International, Research Triangle Park, North Carolina).
Results. Participants with ADHD who received medication (N = 216; mean treatment duration = 7.4 years [SE = 0.22]; median age at first prescription was 15.9 years; 36.2% [SE = 0.81%] of these participants with an actual medication usage) had a mean height of 68.29 inches [SE = 0.34]. Participants with ADHD who never received medication (N = 591) had a mean height of 67.90 inches (SE = 0.23), and those without ADHD (N = 34,652) had a mean height of 66.93 inches (SE = 0.05). Following adjustments, no significant difference in height between groups was found (Wald F = 1.64, P = .202) (Table 1). Similar results were obtained when comparisons were stratified by sex. Also, no significant association between treatment duration and adult height was observed (total sample: Wald F7,65 = 0.04, P = .834; female: Wald F6,65 = 0.21, P = .652; male: Wald F6,65 = 0.08, P = .781).
The strengths of this study lie in the naturalistic conditions and the large and nationally representative sample. Based on pharmacoepidemiologic studies,7,8 we can reasonably consider that most of the participants who received specific medication for ADHD were treated with methylphenidate. Data on dose reduction, treatment discontinuation (eg, weekend breaks and summer breaks), and treatment interruption were not available in this study and have been positively associated with growth velocity.3,9,10 Such interventions are commonly prescribed in case of significant slowdown in growth of children treated with methylphenidate and must therefore be taken into consideration in the interpretation of our results.
As did Biederman et al,4 we found no significant association between height into adulthood and either ADHD or medication treatment for ADHD. The slowdown in the growth observed in certain children treated with methylphenidate2 for ADHD appears to be reversible.
References
1. Poulton A. Growth on stimulant medication; clarifying the confusion: a review. Arch Dis Child. 2005;90(8):801-806. PubMed doi:10.1136/adc.2004.056952
2. Swanson JM, Elliott GR, Greenhill LL, et al. Effects of stimulant medication on growth rates across 3 years in the MTA follow-up. J Am Acad Child Adolesc Psychiatry. 2007;46(8):1015-1027. PubMed doi:10.1097/chi.0b013e3180686d7e
3. Faraone SV, Biederman J, Morley CP, et al. Effect of stimulants on height and weight: a review of the literature. J Am Acad Child Adolesc Psychiatry. 2008;47(9):994-1009. PubMed
4. Biederman J, Spencer TJ, Monuteaux MC, et al. A naturalistic 10-year prospective study of height and weight in children with attention-deficit hyperactivity disorder grown up: sex and treatment effects. J Pediatr. 2010;157(4):635-640, 640. e1. PubMed doi:10.1016/j.jpeds.2010.04.025
5. Grant BF, Goldstein RB, Chou SP, et al. Sociodemographic and psychopathologic predictors of first incidence of DSM-IV substance use, mood and anxiety disorders: results from the Wave 2 National Epidemiologic Survey on Alcohol and Related Conditions. Mol Psychiatry. 2009;14(11):1051-1066. PubMed doi:10.1038/mp.2008.41
6. Bernardi S, Faraone SV, Cortese S, et al. The lifetime impact of attention deficit hyperactivity disorder: results from the National Epidemiologic Survey on Alcohol and Related Conditions (NESARC). Psychol Med. 2012;42(4):875-887. PubMed doi:10.1017/S003329171100153X
7. Safer DJ, Zito JM, Fine EM. Increased methylphenidate usage for attention deficit disorder in the 1990s. Pediatrics. 1996;98(6, pt 1):1084-1088. PubMed
8. Chai G, Governale L, McMahon AW, et al. Trends of outpatient prescription drug utilization in US children, 2002-2010. Pediatrics. 2012;130(1):23-31. PubMed doi:10.1542/peds.2011-2879
9. Klein RG, Landa B, Mattes JA, et al. Methylphenidate and growth in hyperactive children: a controlled withdrawal study. Arch Gen Psychiatry. 1988;45(12):1127-1130. PubMed doi:10.1001/archpsyc.1988.01800360075011
10. Marcus SCWG, Wan GJ, Kemner JE, et al. Continuity of methylphenidate treatment for attention-deficit/hyperactivity disorder. Arch Pediatr Adolesc Med. 2005;159(6):572-578. PubMed doi:10.1001/archpedi.159.6.572
Author affiliations: Assistance Publique-H×´pitaux de Paris (APHP), Robert Debré Hospital, Child and Adolescent Psychiatry Department, Paris (Drs Peyre, Acquaviva, and Delorme); Cognitive Sciences and Psycholinguistic Laboratory, Ecole Normale Supérieure, Paris (Dr Peyre); Assistance Publique-H×´pitaux de Paris (APHP), Corentin Celton Hospital, Department of Psychiatry, 92130 Issy-les-Moulineaux; Paris Descartes University, PRES Sorbonne Paris Cité, Paris (Drs Hoertel and Limosin); INSERM UMR 894, Psychiatry and Neurosciences Center; Paris Descartes University, PRES Sorbonne Paris Cité, Paris (Drs Hoertel and Limosin); INSERM, UMR 669, Universities of Paris Sud and Paris Descartes, Paris (Drs Hoertel and Acquaviva), France; Phyllis Green and Randolph Cowen Institute for Pediatric Neuroscience, Child Study Center of the NYU Langone Medical Center, New York, New York; and Child Neuropsychiatry Unit, Department of Life Sciences and Reproduction, Verona University, Verona, Italy (Dr Cortese); and Human Genetics and Cognitive Functions, Pasteur Institute, Paris, France (Dr Delorme).
Potential conflicts of interest: Dr Cortese was a consultant for Shire Pharmaceuticals in 2009 and 2010. Dr Limosin has been a consultant for Lundbeck and Eutherapie. Drs Peyre, Hoertel, Acquaviva, and Delorme report no potential conflicts of interest relevant to the subject of this letter.
Funding/support: None reported.
Additional information: The National Epidemiologic Survey on Alcohol and Related Conditions was conducted by the National Institute on Alcohol Abuse and Alcoholism; requests for data can be made to http://www.niaaa.nih.gov/foia.
J Clin Psychiatry 2013;74(11):1123-1124 (doi:10.4088/JCP.13l08580).
© Copyright 2013 Physicians Postgraduate Press, Inc.