It is estimated that between 15% and 26% of the US labor force works night, evening, or rotating shifts1 and that approximately 10% of these individuals suffer from circadian rhythm sleep disorder, shift work type, usually referred to as shift work disorder (SWD). Shift work disorder involves insomnia and/or excessive sleepiness that impair the ability to function.2 The goal of this article, based on a series of teleconferences held in the summer of 2010, is to help clinicians better understand the pathophysiology, prevalence, and consequences of SWD and how to diagnose this problem when they encounter it in clinical practice.
OVERVIEW OF THE CIRCADIAN RHYTHM
Andrew D. Krystal, MD, MS, presented an overview of the circadian rhythm and its effects on sleep, wakefulness, and overall health.
Case Vignette
Mr B, a 45-year-old police officer, was referred for evaluation for “probable narcolepsy.” He had been experiencing sleepiness, irritability, problems with job performance, social limitations, and trouble falling asleep. He had had 2 car accidents due to falling asleep while driving. He reported frequent upper respiratory infections, but no other health or psychiatric problems. Mr B first began having problems with insomnia and sleepiness 4 years earlier when he started his new job, which requires at least 1 to 3 night shifts per week. He has no history of other sleep problems, including narcolepsy or breathing-related sleep disorder. A diagnosis of SWD was made, which was confirmed when Mr B stopped shift work for a time at the clinician’s suggestion; his difficulties with insomnia and excessive sleepiness completely resolved, his job performance and social life improved, and he had more energy. His problems with upper respiratory infections also improved.
Properties of the Circadian Rhythm
Circadian rhythms were “discovered” in an experiment published in 1729,3 which found that plants whose leaves open and close over 24 hours continue this pattern of leaf movement even in constant darkness. The finding that plants undergo physiologic changes not driven by environmental light but as if by some kind of internal clock launched the field of circadian rhythm research.
The circadian rhythm is a self-sustained rhythm of biological processes that entrains the organism’s various functions to the environmental cycle of light and dark. This rhythm is observed in nearly all species and plays an important role in synchronizing organ systems to optimal phase relationships with each other. Variations in many biological processes occur over roughly a 24-hour period. For example, core body temperature oscillates over a range of about 2 degrees Centigrade on a cycle of about 24 hours,4 which is a fairly significant variation. Such endogenous rhythmicity is seen in many other biological measures, including levels of plasma melatonin, which increase in the evening and early part of the night, and plasma cortisol, which increase over the course of the night, peak at waking, and diminish throughout the day.4
A number of factors, especially light, can modify the endogenous circadian rhythm. For example, when traveling to a new time zone, the human body is temporarily on a different schedule from the new environment, because the circadian rhythm developed in the previous location persists for a time. The longer a person stays in the new environment, the more the body aligns with its new environmental clock, a process driven by cues, especially appropriate exposure to light, that tell us when it is daytime or nighttime.
Light affects the circadian rhythm differently depending on timing.3 If we are exposed to light late during the night, it shifts our rhythm so that we tend to go to bed and wake up earlier. If we are exposed to light in the early part of the night, it shifts our rhythm so that we tend to stay up and sleep later. During the habitual wake period, light has no effect at all.
Other inputs affect our internal clock including time of meals, activity level, and caffeine intake.5,6 We often experience gastrointestinal upset in a new time zone due to eating when the body does not expect to eat.7 The release of insulin and other digestive hormones is still on the previous time zone’s clock and out of synchrony with meal times in the new time zone.8 The circadian rhythm pacemaker also affects brain functioning (eg, alertness or sleepiness) and the autonomic nervous system.9,10
Anatomy and Genetics of the Circadian Rhythm
The master clock that synchronizes all periodic behavior in our body is located in the brain in the suprachiasmatic nucleus (SCN) of the hypothalamus. The SCN plays a key role in regulating timing of activity in a wide range of cells throughout the body via outputs to different brain regions that regulate temperature, sleep, hormone release, and eating behavior,11 as confirmed by the following findings. Melatonin, which promotes sleep, is produced by the pineal gland under the control of the SCN. Exposure to light suppresses melatonin output via the retinohypothalamic tract of neurons that runs from the retina to the SCN. If the SCN is lesioned, circadian rhythmicity goes away.12 When cells are removed from the SCN, they continue to show self-sustaining circadian rhythmicity in vitro.13 Finally, if the SCN is transplanted from one animal to another, the recipient manifests the circadian rhythm of the donor, showing that the SCN can entrain biological activity and drive a circadian process on its own.14
The SCN’s chemical clock is under genetic control. Although researchers had previously been able to breed for changes such as different eye or hair color, Benzer15 was the first to show that behavior could be modified genetically by breeding circadian behavioral patterns into fruit flies. A relatively small number of genes and proteins regulate this biological clock. These include a series of peptides that are synthesized in the nucleus, diffuse into the cytoplasm where they bond with each other, and then diffuse back into the nucleus where they turn off their own synthesis, a process that takes about 24 hours and then starts over again. The critical genes responsible for the process (eg, period, clock, and Cry genes) can be manipulated to alter the circadian cycle.16
The role of genetic factors in the circadian rhythm is supported by the observation that preferred sleep/wake schedules (eg, being a “night owl” or a “morning lark”) tend to run in families. The tendency to go to bed and get up very early (sleep phase advance) is linked to a change from serine to glycine, a mutation in the human period-2 (hPer2) gene that is an autosomal dominant trait.17 The tendency to stay up late and sleep late (sleep phase delay) is associated with several genes, including the human period-3 (hPer3) gene.18-20
Effects on Sleep/Wake Function
The SCN affects sleep/wake function via its effect on 3 brain regions. The ventrolateral preoptic area, which releases γ-aminobutyric acid (GABA) and is the most important for sleep functioning, plays an important role in promoting sleep.11 The lateral hypothalamic area releases the transmitter hypocretin/orexin, a peptide that promotes wakefulness.11 The paraventricular hypothalamus is involved in the release of melatonin.11
The most common model of our sleep/wake cycle is based on the interface between the circadian rhythm system’s drive for wakefulness, through the effects of the SCN, and the homeostatic drive for sleep (ie, the longer awake, and the shorter the previous sleep periods, the more a person feels the drive for sleep).21 This interaction produces a consolidated period of wakefulness, driven by the circadian rhythm, and a consolidated period of sleep that occurs when the homeostatic sleep drive has built up and the wake-promoting systems have shut down.22 The circadian rhythm system enables us to stay awake for extended periods, despite a growing homeostatic drive for sleep, by modulating the release of neurotransmitters, in particular hypocretin/orexin, that maintain wakefulness. If not for this interaction, we would have great difficulty functioning, since we would fall asleep as soon as the drive to sleep built up sufficiently. This is what happens in narcolepsy, which involves abnormalities in the hypocretin/orexin system (studies have found that lesioning or loss of the neurons that release hypocretin/orexin seems to lead to narcolepsy).23
Problems can arise when the 2 systems are not synchronized appropriately. Trying to sleep when the body doesn’ t normally sleep will result in less and poor quality sleep because the circadian processes are fighting the sleep drive. Individuals with circadian rhythm sleep disorders are likely to experience at least partial sleep loss on a long-term basis because they are trying to sleep at an unfavorable time for extended periods. Both animal and human research studies indicate that even modest prolonged sleep deprivation can produce 4 types of serious physiologic abnormalities24-29:
- Metabolic dysfunction (increased appetite, metabolism, or oxygen consumption; sympathetic nervous system activation; decreased cerebral glucose utilization in certain subcortical structures)
- Neuroendocrine abnormalities (low thyroid-stimulating hormone; decreased levels of growth hormone, prolactin, or leptin)
- Decreased resistance to infectious disease
- Oxidative stress
Humans who experience prolonged sleep deprivation also demonstrate higher rates of obesity and type 2 diabetes as well as neurobehavioral impairment, including a shortening of voluntary and involuntary sleep latency resulting in daytime sleepiness, microsleeps (intrusion of sleep into wakefulness), and errors of omission and commission on cognitive testing.30,31
Role in Health and Disease
By synchronizing the body’s biological clocks, the SCN has extensive influence on peripheral tissues through the autonomic nervous system,32 including the heart (blood circulation), brown adipose tissue (temperature regulation), the kidney (electrolyte balance), the liver (glucose metabolism), the pancreas (glucose stability), the adrenal cortex (corticosteroid release), and white adipose tissue (leptin release and energy storage). For example, glucose is released in a gradual, oscillating, sinusoidal-like pattern over a 24-hour period. If animals in an experimental paradigm are fed at times other than their natural feeding times, the original cycle continues. However, if the SCN is cut out, glucose release becomes entrained to feeding times33 and is no longer linked to other physiologic processes related to eating and digestion.
Phase dyssynchrony occurs when the rhythms of organs are out of synchrony with the SCN. Research in animals and humans has shown that such disruptions can have negative effects on health. For example, disrupting the normal circadian rhythmicity of hamsters with cardiomyopathy significantly reduced median life span by 11%.34 The SCN also appears to regulate the circadian rhythm of cellular growth and division. Mice with osteosarcoma randomized to either a “jet lag” model or lesioning of the SCN had a significant increase in tumor weight and decrease in survival rates compared with controls.35,36
Similar types of negative effects are also seen in humans who experience phase dyssynchrony, as Dr Roth discusses in the next section.
Summary
The circadian rhythm, a self-sustained rhythm of biological processes observed in nearly all species, is determined by both genetic and behavioral factors. It plays an important role in coordinating and modulating sleep/wake function and in many other biological processes. Disturbances of the circadian rhythm cause misalignment among biological and behavioral processes that can lead to disturbances in sleep/wake function and other types of impaired functioning and may affect our capacity to fight off disease.
SHIFT WORK DISORDER
Circadian Rhythm Sleep Disorders
Thomas Roth, PhD, presented an overview of circadian rhythm sleep disorders. with a focus on SWD. According to the second edition of the American Academy of Sleep Medicine’s International Classification of Sleep Disorders (ICSD-2),37 the major feature of circadian rhythm sleep disorders is “a misalignment between the patient’s sleep pattern and the sleep pattern that is desired or regarded as the societal norm.” In addition to SWD, the ICSD-2 lists 8 other types of circadian rhythm sleep disorders, including time zone change (jet lag) syndrome and delayed and advanced sleep phase syndromes. Many people have experienced jet lag syndrome, caused by a lack of synchrony between the body’s internal clock and a new time zone in which the person is trying to function. Circadian rhythm disturbances can also involve delayed or advanced sleep phases. In delayed sleep phase syndrome, the person has difficulty falling asleep and difficulty getting up in the morning (ie, the person’s clock is set several hours later than the norm). Delayed sleep phase syndrome occurs very commonly in adolescents, who may not go to sleep until 2 am and then have great difficulty getting up in the morning. In advanced sleep phase syndrome, which occurs most often in the elderly, people tend to go to sleep at 8 or 9 pm and then wake up at 3 or 4 am (ie, the person’s clock is set several hours earlier than the norm). Because delayed and advanced sleep phase syndromes often cause the person to be out of synchrony with the prevailing sleep/wake patterns of society, they can lead to significant morbidity. Recent studies have found that, when high school classes were started an hour or 2 later, the number of car accidents decreased and academic functioning improved.38-41 Problems can also arise when there is chronic dyssynchrony between the person’s internal clock and external light and dark (ie, when a person is required to stay awake and work when it is dark and sleep when it is light), which can, in some cases, lead to SWD.
Shift Work
It is important to distinguish between shift work and SWD. Shift work is a job description. The vast majority of people who work shifts adjust and do well. However, a subgroup of people have great difficulty adjusting their internal clocks and develop SWD due to a mismatch between the sleep/wake schedule required by their jobs and their own circadian sleep/wake cycles.
Prevalence of shift work. It is estimated that 15%-26% of the US labor force works night, evening, or rotating shifts.1 The 2004 Current Population Survey (CPS)42 reported that almost 15% of full-time wage and salary workers (over 14 million workers) usually worked an alternative shift: 4.7% evening shifts, 3.2% night shifts, 3.1% employer-arranged irregular schedules, and 2.5% rotating shifts. The prevalence of shift work was greatest among workers in protective services (eg, police, firefighters, security guards) (50.6%), food preparation/serving (40.4%), health care support (28%), personal care and service (28%), and transportation and material moving (28%).
Effects of shift work on the sleep/wake cycle. Shift work affects the 2-process sleep/wake cycle in a number of ways. No matter how many hours a person has slept during the day, trying to work during the down side of the circadian rhythm (eg, between 12 am and 6 am) is very difficult unless the internal clock shifts. Studies have found that, over a 24-hour cycle, subjective alertness and cognitive functioning both decline between 2 am and 4 am.43 Also, because a person is not sleeping at night, the homeostatic pressure to sleep is not relieved, producing ever increasing pressure to sleep.44 However, only a subset of individuals who work night or rotating shifts develop SWD because circadian rhythms are modulated not only by light and dark, but also by other factors such as “clock genes,” melatonin, and environmental cues (eg, noise).45,46
Shift Work Disorder: An Overview
Prevalence. Drake et al found that 28% of those who work night or rotating shifts, compared with 18% of day workers, experienced insomnia and/or excessive sleepiness and estimated the “true prevalence” of SWD to be approximately 10% of those who work night or rotating shifts.2 A study of 103 shift workers on a North Sea oil rig (working 2 weeks on 7 nights/7 days, 12-hour shifts, 4 weeks off) by Waage et al found a relatively high prevalence of SWD.47 They reported that 24 (23.3%) of the shift workers were suffering from SWD, and that, during their 4-week period off work, the workers with SWD reported significantly poorer sleep quality, more subjective health complaints, and greater problems in coping than individuals who did not have SWD. Shift workers without SWD reported results similar to day workers on the rig with regard to sleep, sleepiness, subjective health complaints, and coping.
Diagnosis. The ICSD-2 criteria for SWD are listed in Table 1. The disorder is diagnosed primarily based on sleep history. In assessing for SWD, the clinician should ask, “When do you go to work? When do you sleep? How sleepy are you now?” It is also important to rule out other sleep and psychiatric disorders that can lead to insomnia and/or excessive sleepiness.37
Differential diagnosis. The differential diagnosis of SWD includes excessive sleepiness due to obstructive sleep apnea, narcolepsy, restless legs syndrome, and chronic insufficient sleep due to daytime conflicts (eg, child care, environmental factors, “moonlighting” at a second job). Comorbid conditions (eg, prevalence of sleep apnea is increased in shift workers) can complicate the diagnosis of SWD. Clinicians also need to rule out comorbid disorders that can cause insomnia and excessive sleepiness (eg, primary insomnia, insomnia associated with psychiatric disorders such as major depression), as well as consider whether the person may be taking medications or abusing drugs or alcohol to help with sleep, which may be causing impairment at work.
Consequences of Shift Work Disorder
Just as animal studies have found that disruptions in circadian rhythm can affect health outcomes, studies in humans have produced similar findings.
Gastrointestinal problems (eg, ulcers, functional bowel disorders) are significantly increased in individuals who work night or rotating shifts. However, the increased prevalence of ulcers is associated not just with shift work, but with SWD. In a study comparing 360 workers on rotating shifts, 174 on night shifts, and 2,036 who worked days, Drake et al found that, among those who reported excessive sleepiness and/or insomnia, the prevalence of ulcers was higher among rotating shift workers (12.5%) and night shift workers (15.4%) than day workers (6%).2 This effect was not seen to any marked degree in those who worked rotating or night shifts but did not have excessive sleepiness and/or insomnia. Zhen Lu et al found that the prevalence of functional bowel disorders was higher in a sample of nurses who worked rotating shifts (38%) than those who worked day shifts (20%) and that symptoms of functional bowel disorders were positively correlated with level of sleep disturbance.48
Cancer. Shift work (whether or not the person has SWD) has been found to be a risk factor for cancer. Increased odds ratios for breast cancer have been found in large samples of women who worked night shifts, particularly with increasing duration of nighttime employment.49-53 A study of 14,052 working men in Japan also found a significantly increased risk of prostate cancer in men who worked rotating shifts.54 The World Health Organization International Agency for Research on Cancer has concluded that “shift work that involves circadian disruption is probably carcinogenic to humans.”55
Depression. The prevalence of depression is significantly higher in those who work rotating and night shifts than in day workers. In addition, while insomnia or daytime sleepiness is a risk factor for depression for all individuals, it is a much greater risk factor for rotating or night shift workers.2
Cardiovascular effects. While insomnia is a risk factor for hypertension in all individuals, it is a significantly higher risk factor for shift workers with insomnia.2 In contrast, although shift work is associated with a significantly increased risk of heart disease compared with nonshift work, this increased risk is not associated with SWD.2
Excessive sleepiness and accidents. Insomnia is associated with excessive sleepiness in rotating shift workers compared with day workers,2 which can impair functioning, Studies have found a 12% frequency of drowsy driving and an increased risk of driving accidents related to sleepiness in rotating shift workers with SWD compared with those without the disorder. Relative risk of injuries and accidents increases with each successive night shift worked.56 The effects of shift work on patient and employee safety are an important consideration in the health care field, in which many workers have extended shifts.57
Productivity. Similarly, it is the combination of night or rotating shift work and the daytime sleepiness or insomnia that decreases productivity, not each factor alone.2 Rotating shift workers with insomnia and/or excessive sleepiness (SWD) missed significantly more days of work (an average of 3 days/mo over 3 months, a 10% decrease in productivity) than day workers with these symptoms, who missed approximately half a day of work/mo over the 3-month period. This effect was not seen in shift workers who did not have insomnia or excessive sleepiness: both they and the day workers without these symptoms missed a half day or less of work over 3 months.2 Rotating shift workers who experience both insomnia and excessive sleepiness are at the greatest risk for lost productivity. See Keller (2009) for a review of potential productivity problems in health care workers on extended shifts.57
Summary
Shift work is very prevalent in our society. However, only a subset of shift workers meet criteria for SWD and need treatment. Potential targets for treatment are (1) the person’s work schedule; (2) difficulty sleeping during the day; and, (3) most important, given the accident data discussed above, difficulty functioning during the day because of excessive sleepiness (eg, commuting home safely).
SHIFT WORK DISORDER: CLINICAL RECOGNITION AND ASSESSMENT
Richard D. Simon, Jr, MD, provided guidance for clinicians on how to recognize and assess shift work disorder in clinical settings.
Identifying Circadian Rhythm Disturbances
A number of clinical clues suggest a possible circadian rhythm sleep disorder (Table 2), with the most important being an irregular sleep/wake schedule. It is not possible to change one’s circadian rhythm by more than 2-4 hours in any given day,58,59 so that a sleep/wake schedule that varies by more than that between work days and days off suggests possible circadian issues. One of the best ways to identify circadian sleep problems is to ask if the person experiences a combination of insomnia (difficulty falling asleep at bedtime) and hypersomnia (difficulty waking up at the desired time), indicating a delayed sleep phase syndrome (ie, a “night owl”). Individuals with this sleep pattern often abuse the snooze button, hitting it repeatedly. This pattern is frequently seen in teenagers. People may also have hypersomnia at bedtime (eg, falling asleep at 8 pm) and insomnia long before the desired wake time (eg, waking at 3 am), indicating that the person has advanced sleep phase syndrome, a pattern frequently seen in the elderly.
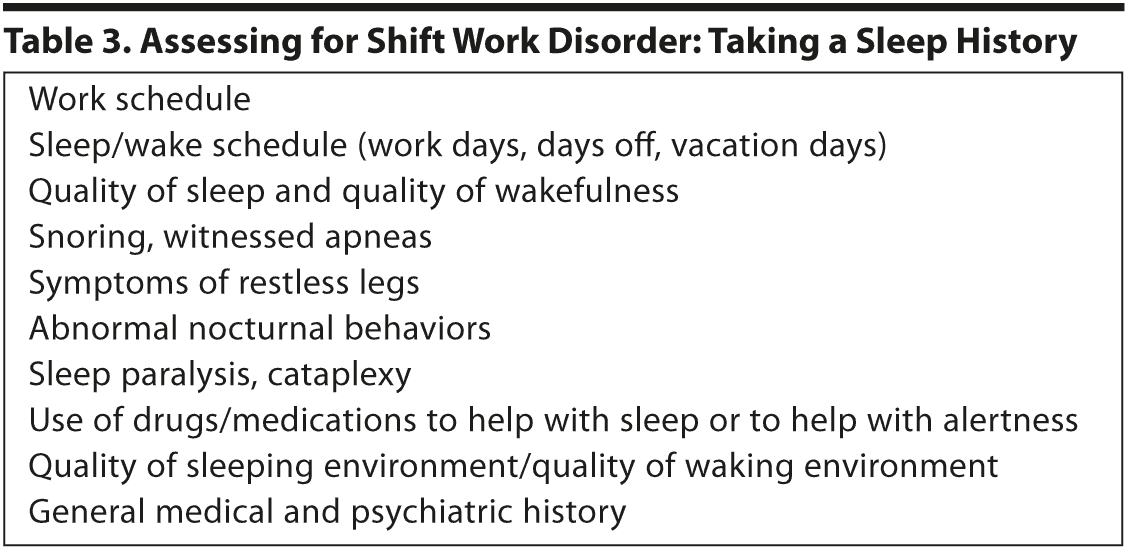
Taking a Sleep History
Items to assess in taking a sleep history are summarized in Table 3. Most important is to see how the person’s sleep/wake schedule differs on work days, days off, and vacation days (which can be particularly helpful in identifying the person’s intrinsic sleep/wake schedule). Quality of sleep and wakefulness should be assessed by asking question such as:
- Do you sleep all night? Do you feel refreshed in the morning? Or do you have a fragmented sleep?
- Do you find it easy to maintain alertness throughout the day? Or do you find yourself getting fatigued and sleepy during the day?
- Do you snore? Has anyone you live with witnessed any episodes of apnea?
Clinicians should assess for restless legs syndrome, characterized by an uncomfortable, creeping, crawling, restless feeling in the legs that can make it very difficult to fall asleep. If the person reports snoring or witnessed episodes of apnea, abnormal nocturnal behaviors (eg, injuring self or others by acting out dreams), or symptoms suggesting narcolepsy, a sleep study is required. The clinician should ask about use of drugs or medications to help with sleep or alertness (eg, caffeine in the daytime, pills or alcohol to promote sleep) and the quality and safety of the sleeping and waking environments, as well as taking a medical and psychiatric history to identify conditions that might be contributing to the sleep problems (eg, respiratory problems, pain, depression, anxiety).
Assessment Tools
The simplest and most important assessment tool for day-to-day clinical use by primary care physicians and general psychiatrists is a sleep diary. An easy-to-use diary has 25 columns (the first for recording the date and then one column for each hour of the day beginning with 10 pm) and multiple rows. Each day, the patient records the date and marks an X in the box for each hour when he or she was sleeping. While there is likely to be little day-to-day variation in the diary of someone with a regular sleep/wake schedule, the diary of someone who works night or rotating shifts may show variations of much more than 2-4 hours from day to day, suggesting the need to evaluate for possible circadian misalignment.
Several easy-to-use scales are also commonly used in sleep assessments. The Stanford Sleepiness Scale (SSS)60 and the Epworth Sleepiness Scale (ESS)61 measure level of excessive sleepiness. The ESS asks the person to rate the likelihood of dozing in 8 different situations on a 4-point scale (0 = would never doze to 3 = high chance of dozing), with a score of 10 or greater suggesting the need for further evaluation. The Insomnia Severity Index (ISI)62 assesses severity of current sleep problems and their effect on daytime functioning. Dr Simon indicated that scales such as the SSS, ESS, and ISI are particularly useful for tracking the effectiveness of an intervention over time. In some situations, depression or anxiety scales or a general outcome scale such as the Short-Form 36-Item Health Survey, Version 2 (SF-36v2) may also be useful.63
Actigraphy, which uses a device worn on the wrist to record motion (ie, suggesting the person is awake) is not generally necessary in assessing for SWD, since an accurate history and a sleep diary will usually supply all the necessary information. Referral for overnight sleep studies or polysomnography is also not indicated to diagnose SWD, but is indicated if one suspects the patient may have obstructive sleep apnea, parasomnias leading to injurious nocturnal behaviors during sleep, or narcolepsy. If narcolepsy is suspected in a shift worker, it is usually necessary to have the worker discontinue shift work for 2-4 weeks, because shift work itself and the associated circadian misalignment can confuse testing for narcolepsy. Narcolepsy is suggested by a history of excessive sleepiness that predates the patient’s shift work and often started in the teenage years.
Differential Diagnosis and Comorbid Conditions
Conditions that frequently occur in conjunction with SWD include obstructive sleep apnea and restless legs syndrome. Signs suggesting sleep apnea include large neck size, crowded oropharynx, and reports of witnessed apneas. The poor sleep habits of shift workers can also cause them to develop learned insomnia behaviors, referred to as psychophysiologic insomnia. Other comorbid conditions include depressive and/or anxiety disorders, and chronic fatigue, which can be difficult to distinguish in a person with chronic circadian dyssynchrony.
Treatment Goals
The primary goal of treatment for SWD is to reduce the degree of circadian misalignment, by fostering better sleep when desired and improved alertness and functioning when appropriate. Other goals are to identify and appropriately treat any intrinsic sleep disorders (eg, apnea) and any medical or psychiatric disorders that are present. The strategies discussed below should be tried before considering use of medications to promote sleep and/or alertness.
Zeitgebers: Strategies for Shifting the Biological Clock
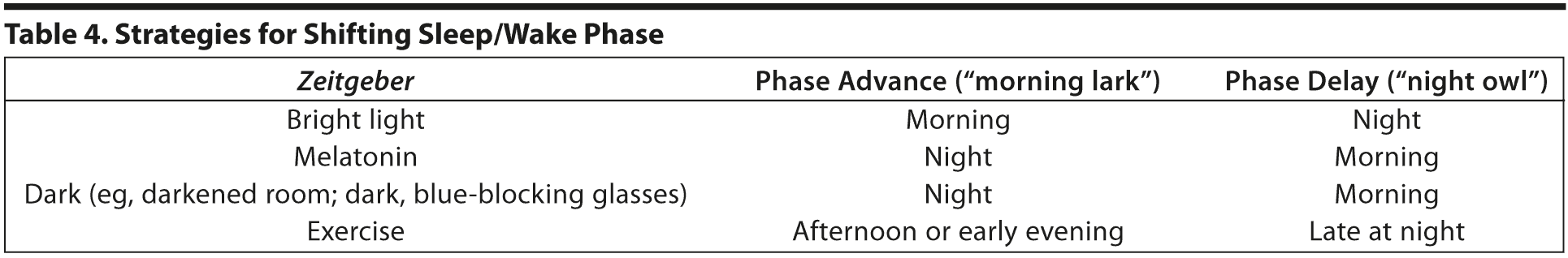
The term Zeitgeber (German for time giver) describes an external cue that helps synchronize a plant or animal’s internal clock to the earth’s 24-hour light/dark cycle.64 The most powerful zeitgebers in humans are light, exogenous melatonin, dark, and exercise.
Very bright light has extremely powerful effects if timed properly,58,59 with individuals most sensitive to the effects of light approximately 2 hours before or an hour or 2 after their spontaneous wake time. If one gives a pulse of very bright light 2-4 hours before a person’s spontaneous wake time (eg, 3 am for someone with a usual wake time of 6 am), the person is likely to wake up 2-4 hours later (ie, to move toward a delayed sleep phase, becoming more of a night owl). On the other hand, if exposed to very bright light at the spontaneous wake time or in the hour or so after, the person is likely to wake up 2-4 hours earlier (ie, to move toward an advanced sleep phase, becoming more of a morning lark).
Melatonin acts in the opposite way.58,59 When administered in the evening, it tends to make the person fall asleep and wake up earlier (ie, to advance the sleep phase). When administered in the morning, it tends to make the person stay up later and wake up later (ie, to delay the sleep phase).
Dark also has powerful effects on sleep phase.58,59 Thus, taking naps in a darkened environment acts in much the same way as melatonin. Greatly limiting exposure to light in the evening will help going to sleep earlier. Conversely, absence of light in the morning will help sleeping later. Because primarily the shorter wave lengths (eg, blue light) lead to phase shifts, one strategy for exposing the biological clock to dark is wearing dark or blue-blocking sunglasses.
Similar phase response curves have been found for exercise.58,59 Exercise in the early evening tends to promote phase advance to make more of a morning person. Exercise after midnight generally does the opposite. However, exercise is not often used to adjust sleep phase in humans.
Combinations of light, dark, and melatonin can be used to help shift the biological clock (Table 4). Melatonin can be thought of as a pill form of dark. Taking melatonin or using dark blue-blocking sunglasses near bedtime (eg, 10 hours before normal wake time) and bright morning light at the normal waking time (with exercise in the afternoon or early evening) will phase advance the cycle, making the person more of a morning lark. Conversely, bright light in the evening (and exercise late in the evening) and then melatonin (or use of dark glasses) at wake time will phase delay the cycle, making the person more of a night owl.
Practical Strategies for Sleep Problems Associated With Shift Work
To minimize problems associated with shift work, workers should have as predictable a work schedule as possible. It is also helpful if employers provide sufficient breaks at work, allow shift workers to take a short nap at work, avoid schedules that involve working multiple days in a row, and provide sufficient time off between work days. These strategies are important because the vast majority of shift workers do not fully entrain (ie, their biological clocks never fully synchronize with their required work and sleep schedules). It is also useful to try to limit commuting time and overtime.
Another key strategy is to minimize circadian misalignment between work days and days off, which involves educating and enlisting the support of significant others in the shift worker’s family and immediate social circle. For most shift workers, this means producing a phase delay in their biological clocks (ie, to make them more “night owls”). This is done by changing the person’s environment so that he or she gets as much light as possible during the scheduled “day” and as little light as possible during the scheduled “night” and by minimizing the difference in sleep/wake patterns between work days and days off. Practically, this means having bright light at work, wearing dark glasses during the drive home when likely to be exposed to light, and keeping the bedroom, bathroom, and other rooms the person will use at home as dark as possible during the desired sleep period.
Shift workers who achieve complete or even partial entrainment (ie, biological clock realigned with a new sleep/wake schedule) show marked improvements in psychomotor vigilance, memory, reaction time, night work performance, and mood and reductions in fatigue, excessive sleepiness, and mental exhaustion compared with those who do not.65,66
Case Vignette
The patient, a 44-year-old man who complains of insomnia and daytime fatigue, currently works the evening shift (4 pm to midnight) and has a 60-minute drive to work. On work days, he goes to bed about 4 am. He would like to sleep until noon, but his wife, who goes to work at 7 am, wakes him at 6:30 am to get his 3 children ready for school. He gets home at 8 am after taking the children to school and sleeps until noon when he gets up to do household chores. On weekends, he tries to go to bed with his wife at 10:00 pm and wake up with her at 6:00 am, but he tosses and turns for several hours before he can sleep so that he can’ t get up with her. He consumes 6-12 cups of coffee in the morning on weekends to try to wake up and function with his family but is still exhausted. He doesn’ t snore or have symptoms of restless legs syndrome. The patient’s job requires him to work rotating shifts: 3 months on evening shifts, 3 months on graveyard shifts (midnight to 7 am), and 3 months on day shifts. He has little difficulty maintaining wakefulness during the evening shift. However, when he works the graveyard shift, he has great difficulty maintaining alertness on the drive home, and when he gets home, has trouble sleeping past noon. On day shift, he has incredible difficulty maintaining alertness on the drive to work and during the first half of his shift. The patient says his wife is unhappy that he seems to be unwilling and unable to go to bed at 10 pm and wake up at 6 am and participate in family events on his days off.
Discussion. The patient is a “night owl” whose optimal sleep time appears to be 4 am to noon. When on the evening shift, he maintains good alertness and typically socializes for 1 to 2 hours after work before driving home. He could probably sleep until noon, but he must get up to drive his children to school. On weekends, he can’ t fall asleep at 10 pm with his family, who lead a “regular” diurnal life, because that is when his biological clock is very active. On the graveyard shift, he is very sleepy during the last few hours of work, which is when he normally goes to bed (4 am) and has a great deal of trouble driving home because his biological clock has turned off. Although tired, he also has trouble sleeping past noon, because his intrinsic biological clock begins to turn on about noon. When the patient works days, he has great difficulty getting up at 5 or 6 am, driving to work, and staying awake during the first half of his shift, because his biological clock is basically off. During the second half of his shift, when his clock is beginning to turn on, he is better able to stay awake.
During a joint visit, the patient and his wife were educated about circadian principles and the physiology of sleep and wakefulness. This helped the patient’s wife understand that he does not have as much choice about when he sleeps as she thought and allowed her to “buy in” to the idea that the patient must have protected sleep time, the timing of which will vary depending on the shift he is working. The patient acknowledged he would need to make some changes too. The patient and his wife agreed to the following strategies.
When on the evening shift, rather than socializing after work, the patient will go home immediately, keep the environment as dark as possible between midnight and 2 am, and try to sleep from 2 am to 10 am, with his bedroom as dark as possible. At 10 am, the patient will get as much bright light as possible, to alert his brain that the day starts at 10 am. His wife agreed to go to work an hour later so that she can get the children off to school.
When on the graveyard shift, the patient will wear very dark glasses on the drive home, will try to sleep from 9 am to 5 pm, get bright light after 5 pm, and consider taking melatonin as soon as he gets home. He will also consider having a nap and then a cup of coffee before going to work. On weekends, the patient will follow a compromise routine of sleeping from 4 am to noon.
When working the day shift, his most difficult shift, the patient will try to wake up at 5:30 am, have some coffee, and get as much light as possible, and perhaps take some melatonin in the evening. On weekends, rather than going to bed at 10 pm with his wife, he will stay up until midnight and sleep until 8 am
This 3-part system involves the patient making some compromises in his intrinsic biological sleep/wake schedule and the family accepting that, depending on what shift he is working, the patient’s sleep/wake schedule will be different. The clinician also asked the patient and his wife to consider whether he needed to continue working shifts as well as whether it might be possible to move closer to work and reduce his commute time. The clinician also discussed the possible long-term effects of shift work and explained that few data are available concerning whether a compromise re-entrainment, such as proposed here, will reduce the long-term negative health effects of chronic circadian misalignment.
When to Refer for Specialized Care
In the case of the patient described above, the clinician proposed a number of fairly simple strategies to try to resolve his sleep problems. If these strategies did not help and the patient continued to have difficulties with insomnia, or excessive sleepiness when he wishes to be awake, the clinician should consider other interventions, such as use of medications to promote sleep or alertness and possible referral for a specialized sleep consultation. If a clinician is uncomfortable or does not have the time to manage shift work/circadian issues, a referral would also be indicated. Other situations in which a referral for a specialized sleep assessment and use of more aggressive treatment strategies are likely to be needed include patients with a high likelihood of having comorbid intrinsic sleep disorders, in particular sleep apnea; patients at considerable risk of falling asleep on the drive to or from work; and patients involved in dangerous work who are likely to fall asleep at work. Table 5 shows what to expect when a specialized sleep consultation is requested.
Summary
Shift work disorder needs to be considered in all patients who have a sleep/wake schedule that differs by more than 2-4 hours on work days compared with days off and who exhibit symptoms of sleepiness at work and difficulty sleeping during the desired sleep time. Asking about snoring and restless legs symptoms can lead to comorbid diagnoses that, if treated, can improve the shift worker’s sleep. By enlisting the support of family, appropriately identifying and treating comorbid sleep disorders, and appropriately timing light and dark exposure (supplemented by melatonin), many shift workers can improve their ability to sleep and maintain wakefulness and possibly decrease the metabolic and other adverse effects of shift work. More aggressive treatment strategies and referral to a sleep specialist should be considered in patients in whom these simple measures are not as effective as desired.
REFERENCES
1. Beers TM. Flexible schedules and shift work: replacing the “9 to 5” workday? Mon Labor Rev. 2000;(June):33-40.
2. Drake CL, Roehrs T, Richardson G, et al. Shift work sleep disorder: prevalence and consequences beyond that of symptomatic day workers. Sleep. 2004;27(8):1453-1462. PubMed
3. Moore-Ede MC, Sulszman FM, Fuller CA. The Clocks That Time Us: Physiology of the Circadian Timing System. Cambridge, MA: Harvard University Press; 1982.
4. Hastings M. The brain, circadian rhythms, and clock genes. BMJ. 1998;317(7174):1704-1707. PubMed
5. Kobayashi H, Oishi K, Hanai S, et al. Effect of feeding on peripheral circadian rhythms and behaviour in mammals. Genes Cells. 2004;9(9):857-864. PubMed doi:10.1111/j.1365-2443.2004.00769.x
6. Antle MC, Steen NM, Mistlberger RE. Adenosine and caffeine modulate circadian rhythms in the Syrian hamster. Neuroreport. 2001;12(13):2901-2905. PubMed doi:10.1097/00001756-200109170-00029
7. Waterhouse J, Nevill A, Finnegan J, et al. Further assessments of the relationship between jet lag and some of its symptoms. Chronobiol Int. 2005;22(1):121-136. PubMed doi:10.1081/CBI-200036909
8. Scheer FA, Hilton MF, Mantzoros CS, et al. Adverse metabolic and cardiovascular consequences of circadian misalignment. Proc Natl Acad Sci U S A. 2009;106(11):4453-4458. PubMed doi:10.1073/pnas.0808180106
9. Chou TC, Scammell TE, Gooley JJ, et al. Critical role of dorsomedial hypothalamic nucleus in a wide range of behavioral circadian rhythms. J Neurosci. 2003;23(33):10691-10702. PubMed
10. Buijs RM, Scheer FA, Kreier F, et al. Organization of circadian functions: interaction with the body. Prog Brain Res. 2006;153:341-360. PubMed doi:10.1016/S0079-6123(06)53020-1
11. Saper CB, Scammell TE, Lu J. Hypothalamic regulation of sleep and circadian rhythms. Nature. 2005;437(7063):1257-1263. PubMed doi:10.1038/nature04284
12. Stetson MH, Watson-Whitmyre M. Nucleus suprachiasmaticus: the biological clock in the hamster? Science. 1976;191(4223):197-199. PubMed doi:10.1126/science.942799
13. Groos G, Hendriks J. Circadian rhythms in electrical discharge of rat suprachiasmatic neurones recorded in vitro. Neurosci Lett. 1982;34(3):283-288. PubMed doi:10.1016/0304-3940(82)90189-6
14. Matsumoto S, Basil J, Jetton AE, et al. Regulation of the phase and period of circadian rhythms restored by suprachiasmatic transplants. J Biol Rhythms. 1996;11(2):145-162. PubMed doi:10.1177/074873049601100207
15. Benzer S. Behavioral mutants of Drosophila isolated by countercurrent distribution. Proc Natl Acad Sci USA. 1967;58(3):1112-1119. PubMed doi:10.1073/pnas.58.3.1112
16. Ko CH, Takahashi JS. Molecular components of the mammalian circadian clock. Hum Mol Genet. 2006;15(spec no 2):R271-R277. PubMed doi:10.1093/hmg/ddl207
17. Toh KL, Jones CR, He Y, et al. An hPer2 phosphorylation site mutation in familial advanced sleep phase syndrome. Science. 2001;291(5506):1040-1043. PubMed doi:10.1126/science.1057499
18. Lowrey PL, Takahashi JS. Mammalian circadian biology: elucidating genome-wide levels of temporal organization. Annu Rev Genomics Hum Genet. 2004;5(1):407-441. PubMed doi:10.1146/annurev.genom.5.061903.175925
19. Taheri S. The genetics of sleep disorders. Minerva Med. 2004;95(3):203-212. PubMed
20. Kimura M, Winkelmann J. Genetics of sleep and sleep disorders. Cell Mol Life Sci. 2007;64(10):1216-1226. PubMed doi:10.1007/s00018-007-6532-1
21. Borbély AA. A two process model of sleep regulation. Hum Neurobiol. 1982;1(3):195-204. PubMed
22. Dijk DJ, Czeisler CA. Paradoxical timing of the circadian rhythm of sleep propensity serves to consolidate sleep and wakefulness in humans. Neurosci Lett. 1994;166(1):63-68. PubMed doi:10.1016/0304-3940(94)90841-9
23. Dauvilliers Y, Arnulf I, Mignot E. Narcolepsy with cataplexy. Lancet. 2007;369(9560):499-511. PubMed doi:10.1016/S0140-6736(07)60237-2
24. Everson CA. Clinical assessment of blood leukocytes, serum cytokines, and serum immunoglobulins as responses to sleep deprivation in laboratory rats. Am J Physiol Regul Integr Comp Physiol. 2005;289(4):R1054-R1063. PubMed
25. Everson CA, Crowley WR. Reductions in circulating anabolic hormones induced by sustained sleep deprivation in rats. Am J Physiol Endocrinol Metab. 2004;286(6):E1060-E1070. PubMed doi:10.1152/ajpendo.00553.2003
26. Everson CA, Nowak TS Jr. Hypothalamic thyrotropin-releasing hormone mRNA responses to hypothyroxinemia induced by sleep deprivation. Am J Physiol Endocrinol Metab. 2002;283(1):E85-E93. PubMed
27. Everson CA, Szabo A. Recurrent restriction of sleep and inadequate recuperation induce both adaptive changes and pathological outcomes. Am J Physiol Regul Integr Comp Physiol. 2009;297(5):R1430-R1440. PubMed
28. Everson CA, Laatsch CD, Hogg N. Antioxidant defense responses to sleep loss and sleep recovery. Am J Physiol Regul Integr Comp Physiol. 2005;288(2):R374-R383. PubMed
29. Everson CA, Thalacker CD, Hogg N. Phagocyte migration and cellular stress induced in liver, lung, and intestine during sleep loss and sleep recovery. Am J Physiol Regul Integr Comp Physiol. 2008;295(6):R2067-R2074. PubMed
30. Goel N, Rao H, Durmer JS, et al. Neurocognitive consequences of sleep deprivation. Semin Neurol. 2009;29(4):320-339. PubMed doi:10.1055/s-0029-1237117
31. Banks S, Dinges DF. Behavioral and physiological consequences of sleep restriction. J Clin Sleep Med. 2007;3(5):519-528. PubMed
32. Buijs RM, van Eden CG, Goncharuk VD, et al. The biological clock tunes the organs of the body: timing by hormones and the autonomic nervous system. J Endocrinol. 2003;177(1):17-26. PubMed doi:10.1677/joe.0.1770017
33. La Fleur SE. Daily rhythms in glucose metabolism: suprachiasmatic nucleus output to peripheral tissue. J Neuroendocrinol. 2003;15(3):315-322. PubMed doi:10.1046/j.1365-2826.2003.01019.x
34. Penev PD, Kolker DE, Zee PC, et al. Chronic circadian desynchronization decreases the survival of animals with cardiomyopathic heart disease. Am J Physiol. 1998;275(6 Pt 2):H2334-H2337. PubMed
35. Filipski E, King VM, Li X, et al. Host circadian clock as a control point in tumor progression. J Natl Cancer Inst. 2002;94(9):690-697. PubMed
36. Filipski E, Delaunay F, King VM, et al. Effects of chronic jet lag on tumor progression in mice. Cancer Res. 2004;64(21):7879-7885. PubMed doi:10.1158/0008-5472.CAN-04-0674
37. American Academy of Sleep Medicine (AASM). International Classification of Sleep Disorders Diagnostic and Coding Manual, 2nd ed (ICSD-2). Westchester, IL: American Academy of Sleep Medicine; 2005
38. Wolfson AR, Carskadon MA. Sleep schedules and daytime functioning in adolescents. Child Dev. 1998;69(4):875-887. PubMed
39. Wolfson AR, Carskadon MA. Understanding adolescents’ sleep patterns and school performance: a critical appraisal. Sleep Med Rev. 2003;7(6):491-506. PubMed doi:10.1016/S1087-0792(03)90003-7
40. Owens JA, Belon K, Moss P. Impact of delaying school start time on adolescent sleep, mood, and behavior. Arch Pediatr Adolesc Med. 2010;164(7):608-614. PubMed doi:10.1001/archpediatrics.2010.96
41. Pizza F, Contardi S, Antognini AB, et al. Sleep quality and motor vehicle crashes in adolescents. J Clin Sleep Med. 2010;6(1):41-45. PubMed
42. Bureau of Labor Statistics. Workers on flexible and shift schedules in May 2004 (data from the May 2004 Current Population Survey). Washington, DC: United States Department of Labor; www.bls.gov/news.release/pdf/flex.pdf. Updated July 1, 2005. Accessed August 30, 2010.
43. Czeisler CA, Gooley JJ. Sleep and circadian rhythms in humans. Cold Spring Harb Symp Quant Biol. 2007;72(1):579-597. PubMed doi:10.1101/sqb.2007.72.064
44. Kryger MH, Roth T, Dement WC. Principles and Practice of Sleep Medicine. 4th ed. Philadelphia, PA: Saunders; 2005.
45. Piggins HD. Human clock genes. Ann Med. 2002;34(5):394-400. PubMed doi:10.1080/078538902320772142
46. Herzog ED, Tosini G. The mammalian circadian clock shop. Semin Cell Dev Biol. 2001;12(4):295-303. PubMed doi:10.1006/scdb.2001.0257
47. Waage S, Moen BE, Pallesen S, et al. Shift work disorder among oil rig workers in the North Sea. Sleep. 2009;32(4):558-565. PubMed
48. Zhen Lu W, Ann Gwee K, Yu Ho K. Functional bowel disorders in rotating shift nurses may be related to sleep disturbances. Eur J Gastroenterol Hepatol. 2006;18(6):623-627. PubMed
49. Hansen J. Increased breast cancer risk among women who work predominantly at night. Epidemiology. 2001;12(1):74-77. PubMed doi:10.1097/00001648-200101000-00013
50. Davis S, Mirick DK, Stevens RG. Night shift work, light at night, and risk of breast cancer. J Natl Cancer Inst. 2001;93(20):1557-1562. PubMed doi:10.1093/jnci/93.20.1557
51. Schernhammer ES, Laden F, Speizer FE, et al. Rotating night shifts and risk of breast cancer in women participating in the nurses’ health study. J Natl Cancer Inst. 2001;93(20):1563-1568. PubMed doi:10.1093/jnci/93.20.1563
52. Schernhammer ES, Kroenke CH, Laden F, et al. Night work and risk of breast cancer. Epidemiology. 2006;17(1):108-111. PubMed doi:10.1097/01.ede.0000190539.03500.c1
53. O’ Leary ES, Schoenfeld ER, Stevens RG, et al; Electromagnetic Fields and Breast Cancer on Long Island Study Group. Shift work, light at night, and breast cancer on Long Island, New York. Am J Epidemiol. 2006;164(4):358-366. PubMed doi:10.1093/aje/kwj211
54. Kubo T, Ozasa K, Mikami K, et al. Prospective cohort study of the risk of prostate cancer among rotating-shift workers: findings from the Japan collaborative cohort study. Am J Epidemiol. 2006;164(6):549-555. PubMed doi:10.1093/aje/kwj232
55. Straif K, Baan R, Grosse Y, et al; WHO International Agency for Research on Cancer Monograph Working Group. Carcinogenicity of shift-work, painting, and fire-fighting. Lancet Oncol. 2007;8(12):1065-1066. PubMed doi:10.1016/S1470-2045(07)70373-X
56. Folkard S, Tucker P. Shift work, safety and productivity. Occup Med (Lond). 2003;53(2):95-101. PubMed doi:10.1093/occmed/kqg047
57. Keller SM. Effects of extended work shifts and shift work on patient safety, productivity, and employee health. AAOHN J. 2009;57(12):497-502, quiz 503-504. PubMed doi:10.3928/08910162-20091116-01
58. Pandi-Perumal SR, Smits M, Spence W, et al. Dim light melatonin onset (DLMO): a tool for the analysis of circadian phase in human sleep and chronobiological disorders. Prog Neuropsychopharmacol Biol Psychiatry. 2007;31(1):1-11. PubMed doi:10.1016/j.pnpbp.2006.06.020
59. Buxton OM, L’ Hermite-Balériaux M, Turek FW, et al. Daytime naps in darkness phase shift the human circadian rhythms of melatonin and thyrotropin secretion. Am J Physiol Regul Integr Comp Physiol. 2000;278(2):R373-R382. PubMed
60. Hoddes E, Zarcone V, Smythe H, et al. Quantification of sleepiness: a new approach. Psychophysiology. 1973;10(4):431-436. PubMed doi:10.1111/j.1469-8986.1973.tb00801.x
61. Johns MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep. 1991;14(6):540-545. PubMed
62. Morin CM, Espie CA. Insomnia: A Clinical Guide to Assessment and Treatment. New York, NY: Kluwer Academic/Plenum; 2003.
63. Ware JE. User’s Manual for the SF-36v2 Health Survey, 2nd ed. Lincoln, RI: QualityMetric Health Outcomes Solutions 2007. www.sf-36.org. Updated September 25, 2007. Accessed January 6, 2011.
64. Aschoff J. The phase-angle difference in circadian periodicity. In: Aschoff J, ed. Circadian Clocks. Amsterdam, The Netherlands: North Holland Press; 1965:262-278.
65. Crowley SJ, Lee C, Tseng CY, et al. Complete or partial circadian re-entrainment improves performance, alertness, and mood during night-shift work. Sleep. 2004;27(6):1077-1087. PubMed
66. Smith MR, Fogg LF, Eastman CI. A compromise circadian phase position for permanent night work improves mood, fatigue, and performance. Sleep. 2009;32(11):1481-1489. PubMed