Background: Extrapyramidal side effects (EPS) have been identified as a complication of antipsychotic treatment. Previous meta-analyses have investigated EPS prevalence and risk factors in randomized clinical trials with highly selected patients, but studies in real-world schizophrenia are missing.
Objective: To examine the prevalence and clinical correlates associated with EPS in a nonselected national multicenter sample of stabilized patients with schizophrenia.
Methods: Between 2010 and 2016, patients suffering from schizophrenia (DSM-IV-TR criteria) were recruited through the FondaMental Academic Centers of Expertise for Schizophrenia (FACE-SZ) network and data were collected during a comprehensive 1-day-long standardized evaluation. The Simpson-Angus Scale and the Abnormal Involuntary Movement Scale were used to assess drug-induced parkinsonism (DIP) and tardive dyskinesia, respectively.
Results: The overall prevalence of DIP and tardive dyskinesia was 13.2% and 8.3%, respectively, in this community-dwelling sample of 674 patients. DIP was associated with negative symptoms (Positive and Negative Syndrome Scale [PANSS] subscore) (adjusted odds ratio [aOR] = 1.102, P < .001), first-generation antipsychotic prescription (aOR = 2.038, P = .047), and anticholinergic drug administration (aOR = 2.103, P = .017) independently of sex, age, disorganization (PANSS disorganized factor), and antipsychotic polytherapy. Tardive dyskinesia was associated with PANSS disorganized factor (aOR = 1.103, P = .049) independently of sex, age, negative symptoms, excitation, first-generation antipsychotic prescription, and benzodiazepine and anticholinergic drug administration.
Conclusions: Our results indicate the high prevalence of EPS in a nonselected community-dwelling clinically stable sample of outpatients with schizophrenia. In the monitoring of antipsychotic treatment, EPS should be systematically evaluated, especially when negative symptoms and disorganization or cognitive alteration are present. Monotherapy with a second-generation antipsychotic should be preferentially initiated for patients with these side effects.

CME Background
Articles are selected for credit designation based on an assessment of the educational needs of CME participants, with the purpose of providing readers with a curriculum of CME articles on a variety of topics throughout each volume. Activities are planned using a process that links identified needs with desired results.
To obtain credit, read the article, correctly answer the questions in the Posttest, and complete the Evaluation. A $10 processing fee will apply.
CME Objective
After studying this article, you should be able to:
- Identify risk factors for extrapyramidal side effects in your patients taking antipsychotic agents
Accreditation Statement
The CME Institute of Physicians Postgraduate Press, Inc., is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Credit Designation
The CME Institute of Physicians Postgraduate Press, Inc., designates this journal-based CME activity for a maximum of 1 AMA PRA Category 1 Creditâ„¢. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Note: The American Academy of Physician Assistants (AAPA) accepts certificates of participation for educational activities certified for AMA PRA Category 1 Creditâ„¢ from organizations accredited by ACCME or a recognized state medical society. Physician assistants may receive a maximum of 1 hour of Category I credit for completing this program.
Release, Expiration, and Review Dates
This educational activity was published in January 2019 and is eligible for AMA PRA Category 1 Creditâ„¢ through February 28, 2021. The latest review of this material was December 2018.
Financial Disclosure
All individuals in a position to influence the content of this activity were asked to complete a statement regarding all relevant personal financial relationships between themselves or their spouse/partner and any commercial interest. The CME Institute has resolved any conflicts of interest that were identified. In the past year, Marlene P. Freeman, MD, Editor in Chief, has received research funding from JayMac and Sage; has been a member of the advisory boards for Otsuka, Alkermes, and Sunovion; has been a member of the Independent Data Safety and Monitoring Committee for Janssen; and, as a Massachusetts General Hospital (MGH) employee, works with the MGH National Pregnancy Registry, which is sponsored by Teva, Alkermes, Otsuka, Actavis, and Sunovion, and works with the MGH Clinical Trials Network and Institute, which receives research funding from multiple pharmaceutical companies and the National Institute of Mental Health. No member of the CME Institute staff reported any relevant personal financial relationships. Faculty financial disclosure appears at the end of the article.
Background: Extrapyramidal side effects (EPS) have been identified as a complication of antipsychotic treatment. Previous meta-analyses have investigated EPS prevalence and risk factors in randomized clinical trials with highly selected patients, but studies in real-world schizophrenia are missing.
Objective: To examine the prevalence and clinical correlates associated with EPS in a nonselected national multicenter sample of stabilized patients with schizophrenia.
Methods: Between 2010 and 2016, patients suffering from schizophrenia (DSM-IV-TR criteria) were recruited through the FondaMental Academic Centers of Expertise for Schizophrenia (FACE-SZ) network and data were collected during a comprehensive 1-day-long standardized evaluation. The Simpson-Angus Scale and the Abnormal Involuntary Movement Scale were used to assess drug-induced parkinsonism (DIP) and tardive dyskinesia, respectively.
Results: The overall prevalence of DIP and tardive dyskinesia was 13.2% and 8.3%, respectively, in this community-dwelling sample of 674 patients. DIP was associated with negative symptoms (Positive and Negative Syndrome Scale [PANSS] subscore) (adjusted odds ratio [aOR] = 1.102, P < .001), first-generation antipsychotic prescription (aOR = 2.038, P = .047), and anticholinergic drug administration (aOR = 2.103, P = .017) independently of sex, age, disorganization (PANSS disorganized factor), and antipsychotic polytherapy. Tardive dyskinesia was associated with PANSS disorganized factor (aOR = 1.103, P = .049) independently of sex, age, negative symptoms, excitation, first-generation antipsychotic prescription, and benzodiazepine and anticholinergic drug administration.
Conclusions: Our results indicate the high prevalence of EPS in a nonselected community-dwelling clinically stable sample of outpatients with schizophrenia. In the monitoring of antipsychotic treatment, EPS should be systematically evaluated, especially when negative symptoms and disorganization or cognitive alteration are present. Monotherapy with a second-generation antipsychotic should be preferentially initiated for patients with these side effects.
J Clin Psychiatry 2019;80(1):18m12246
To cite: Misdrahi D, Tessier A, Daubigney A, et al. Prevalence of and risk factors for extrapyramidal side effects of antipsychotics: results from the national FACE-SZ cohort. J Clin Psychiatry. 2019;80(1):18m12246.
To share: https://doi.org/10.4088/JCP.18m12246
© Copyright 2019 Physicians Postgraduate Press, Inc.
aFondaMental Foundation, Créteil, France
bDepartment of Adult Psychiatry, Charles Perrens Hospital, University of Bordeaux, CNRS UMR 5287-INCIA, Bordeaux, France
cNeurology Department, Pellegrin Hospital, Bordeaux University Hospital; and Institute of Neurodegenerative Diseases, University of Bordeaux, UMR 5293, Bordeaux, France
dDepartment of Adult Psychiatry, Charles Perrens Hospital; and Laboratory of Nutrition and Integrated Neurobiology (UMR INRA 1286), University of Bordeaux, Bordeaux, France
eStrasbourg University Hospital, University of Strasbourg, INSERM U1114, Federation of Translational Psychiatry, Strasbourg, France
fAcademic Department of Adult Psychiatry, La Colombiרre Hospital, CHRU Montpellier, University of Montpellier, Inserm 1061, Montpellier, France
gCMP B, CHU, EA 7280 Faculty of Medicine, University of Auvergne, Clermont-Ferrand, France
hAP-HP, Department of Psychiatry, Louis Mourier Hospital, Colombes, Inserm U894 Paris Diderot University, Sorbonne Paris Cité, Faculty of Medicine, Paris, France
iINSERM U1028, CNRS UMR5292, Neurosciences Research Center of Lyon, Claude Bernard University, PSYR2 team, Le Vinatier Hospital, Bron, France
jPsychosocial Rehabilitation Reference Centre, Alpes Isרre Hospital, Grenoble, France
kAP-HM, Academic Department of Psychiatry, Marseille, France
lDepartment of Adult Psychiatry, Versailles Hospital, Le Chesnay, EA 4047 HANDIReSP, Versailles Saint-Quentin en Yvelines University, Versailles, France
mINSERM U955, Translational Psychiatry Team, Créteil, France, Paris-Est Créteil University, DHU Pe-PSY, Psychiatry and Addictions Department, Henri Mondor Hospital, Créteil, France
nCarémeau Hospital, N×®mes, France
oAix-Marseille University, School of Medicine, La Timone, EA 3279: CEReSS—Study and Research Center on Health Services and Quality of Life, Marseille, France
pSorbonne University, UPMC University of Paris, UMR_S 1136, Pierre Louis institute of Epidemiology and Public Health, Paris, France
qMembers of the FACE-SZ Group are listed at the end of the article.
*Corresponding author: David Misdrahi, MD, Centre Hospitalier Charles Perrens, CNRS UMR 5287-INCIA, 121 Rue de la Béchade, 33076 Bordeaux, France ([email protected]).
Extrapyramidal side effects including drug-induced parkinsonism (DIP) and tardive dyskinesia (TD) have been identified as frequent side effects of antipsychotics and have been associated with impaired quality of life1 and depression2 in patients with schizophrenia.
More specifically, parkinsonism (bradykinesia, rigidity, and tremor) occurs after a relatively short period of antipsychotic treatment3 and has been mainly related to the intrinsic antidopaminergic potency of the antipsychotic treatment.4-7 DIP symptoms usually remit within a few months after medication withdrawal,8 while they may unmask neurodegenerative dopamine denervation in some patients.9,10 Clinical studies have underlined that parkinsonism was positively correlated with the intensity of negative symptoms.11,12
TD is a drug-induced movement disorder, mainly related to antipsychotic treatment and defined by involuntary, repetitive orofacial movements, often accompanied by choreiform movements of the upper extremities. The term tardive means delayed after months of antipsychotic treatment.13,14 TD can be difficult to treat and may be permanent in some people. Age, duration of treatment with antipsychotics, first-generation antipsychotic (FGA) treatment, treatment with anticholinergics, substance abuse, and negative symptoms have been suggested to be associated with TD.15
EPS has been recently investigated in 2 comprehensive meta-analyses including clinical trials to compare second-generation antipsychotics (SGAs) with FGAs.16,17 Overall, SGAs including clozapine, olanzapine, and risperidone have been found to be associated with fewer EPS than haloperidol (FGA). However, clinical trials are not representative of “real world” schizophrenia, as many patients are administered antipsychotic polytherapy combined with other psychotropic drugs (antidepressant, anxiolytic, anticholinergic), with various degrees of adherence and substance use disorder comorbidities, especially daily tobacco smoking, that may impact blood antipsychotic levels.11,18-20
The objective of the present study was to determine the prevalence and clinical correlates associated with antipsychotic extrapyramidal side effects in a nonselected national sample of stabilized community-dwelling outpatients with schizophrenia.

- The prevalence of drug-induced parkinsonism and tardive dyskinesia (TD) was, respectively, 13.2% and 8.3% in a large multicenter cohort of patients with schizophrenia (674 patients).
- Drug-induced parkinsonism was associated with higher negative symptoms level, first-generation antipsychotics, and anticholinergic drugs, and TD was associated with higher disorganization/cognitive symptoms level.
- The choice of monotherapy with second-generation antipsychotics should be recommended as soon as possible to prevent onset of extrapyramidal side effects in patients with schizophrenia.
METHODS
Study Participants
The FondaMental Academic Centers of Expertise for Schizophrenia (FACE-SZ) cohort is based on a French national network of 10 Schizophrenia Expert Centers (Bordeaux, Clermont-Ferrand, Colombes, Créteil, Grenoble, Lyon, Marseille, Montpellier, Strasbourg, and Versailles) set up by a scientific cooperation foundation in France, the FondaMental Foundation (www.fondation-fondamental.org), and formed by the French Ministry of Research to create a platform that links thorough and systematic assessment to research. Clinically stable patients aged above 16 years are referred by their general practitioner or psychiatrist, who subsequently receives a detailed evaluation report with suggestions for personalized interventions. Patients diagnosed with schizophrenia or schizoaffective disorders according to DSM-IV-TR criteria were enrolled in the FACE-SZ cohort. This study includes patients recruited between March 2010 and January 2016.21
The assessment protocol was approved by the relevant ethical review board (CPP-Ile de France IX, January 18, 2010) and was therefore performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. All subjects gave informed consent prior to their inclusion in the study.
Data Collection
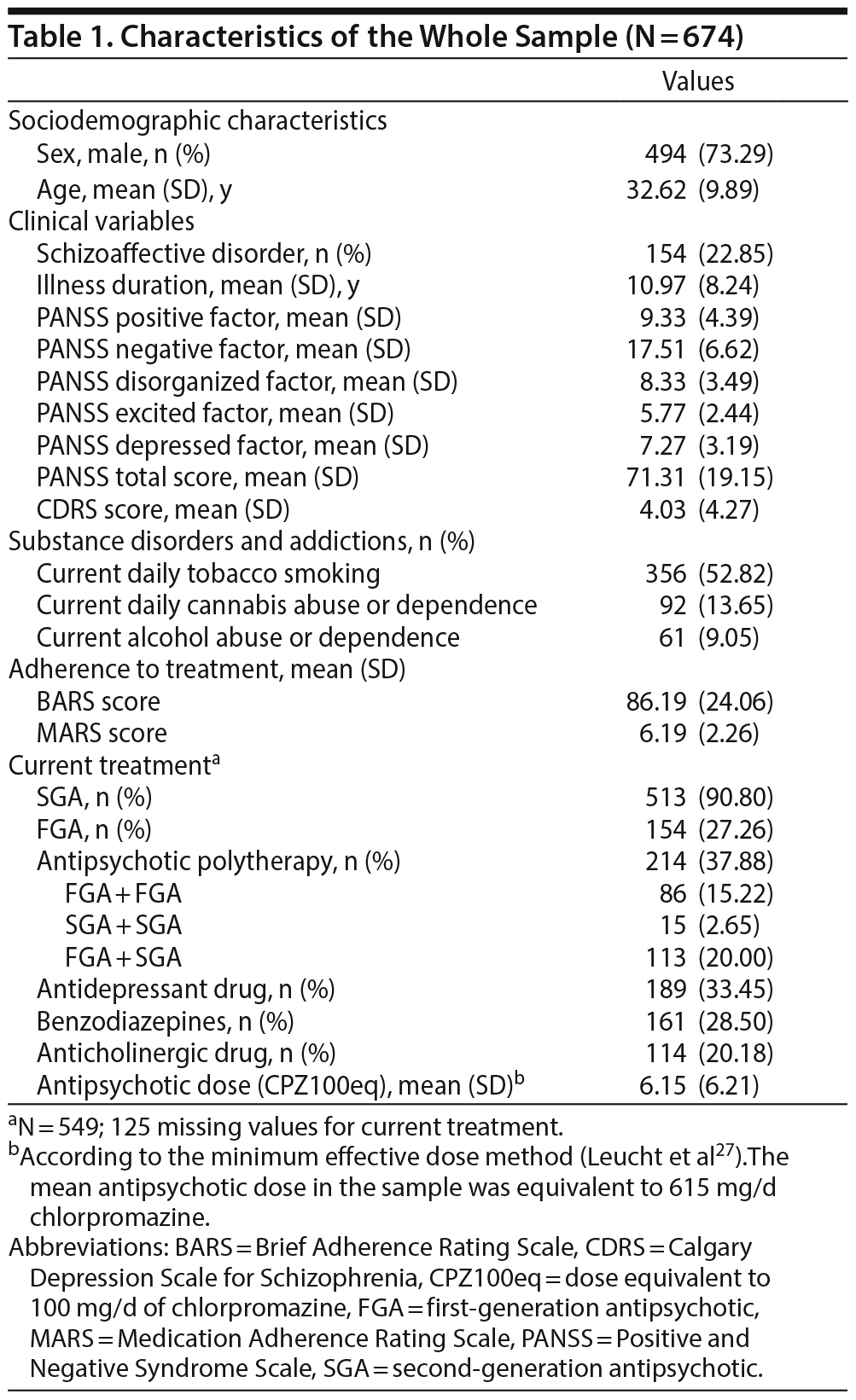
Sociodemographic and clinical variables. Clinical and sociodemographic factors were collected during an extensive evaluation. Standardized assessments were used to assess psychotic and negative symptoms and general psychopathology with the Positive and Negative Syndrome Scale (PANSS) subscores.22 For the purposes of this study and to explore specifically depressive and cognitive symptoms, we used the validated 5-factor model of the PANSS23 described as follows: “positive symptoms,” “negative symptoms,” “disorganized/concrete” (ie, the factor found to account for the largest share of the PANSS association with cognition [IQ]), “excitement,” and “depression.” Depressive symptoms were also assessed with the French-validated Calgary Depression Rating Scale for Schizophrenia (CDRS).24 A cutoff of 6 was considered as a current major depressive episode. Treatment adherence was evaluated using the Brief Adherence Rating Scale25 and the Medication Adherence Rating Scale.26 Daily tobacco smoking and alcohol and cannabis use disorders were defined according to the Structured Clinical Interview for DSM-IV Axis I Disorders. Type and number of ongoing psychotropic treatments were recorded (antipsychotics, antidepressants, benzodiazepines, and anticholinergics). Chlorpromazine equivalent doses (CPZ100eq) were calculated according to the minimum effective dose method.27 All patients were on stable medication for more than 4 weeks.
Drug-induced parkinsonism. The Simpson-Angus Scale (SAS) is a 10-item scale used in clinical and research practices to assess DIP. One item measures gait, 6 items measure stiffness, and 3 items measure tremor, salivation, and palpebral reflex. This scale is validated and is among those most used in individuals with schizophrenia in naturalistic conditions.28 We used a cutoff of 0.65 to define presence of DIP. This threshold was previously used and validated.29
Tardive dyskinesia. The Abnormal Involuntary Movement Scale (AIMS), a 10-item scale designed to record the occurrence of dyskinetic movements, was used to assess the incidence of TD.30 Items evaluate dyskinetic movements in 3 body regions (facial and oral, extremity, trunk) on a 5-point scale (with 0 indicating no dyskinetic movements and 4 indicating severe dyskinetic movements) for a total score ranging from 0 to 40. Each item on the AIMS ranges from 0 to 4, and the total AIMS score was calculated by adding items 1-7. In accordance with a recent meta-analysis,20 we used the widely recognized Schooler-Kane criteria to define patients with TD. Thus, dyskinesia was classified as present when an AIMS score of 2 was noted in at least 2 items or when a score of > 2 was noted in at least 1 item.20
Statistical Analysis
The sociodemographic and clinical characteristics, presence of substance use disorder, medications, and the scores for each scale were compared between the 2 groups for each side effect studied (“DIP” vs “No DIP” and “TD” vs “No TD”) using the Student t test for continuous variables and χ2 test for categorical variables. A logistic regression analysis was used to estimate the odds ratio (OR) for risk factors associated with DIP and TD, after adjusting for confounding factors. Variables relevant to the model were selected based on their clinical interest and/or a threshold P value ≤ 0.20 in univariate analyses (exclusion of collinear variables). The final model incorporated the adjusted odds ratios (aORs) with 95% confidence interval. An aOR > 1 was considered as an increased risk factor for the presence of studied side effects, and an aOR < 1, as a protective risk factor for studied side effects. The statistical significance level was set at P = .05 in a 2-sided test. Data were analyzed using SPSS 20.0 software.
RESULTS
Sample Characteristics
Altogether, 674 outpatients suffering from schizophrenia were included in this study. Clinical and demographic characteristics are presented in Table 1. Patients were mostly men (73.3%), the mean age at inclusion was 32.6 years (SD = 9.9), and the mean illness duration was 11.0 years (SD = 8.2). Criteria for current tobacco smoking were met by 52.8% of the patients, and current alcohol and cannabis use disorders were present in, respectively, 9.1% and 13.7%. SGAs were prescribed in 90.8% of patients, 114 (20.2%) patients were administered an anticholinergic drug, and 214 patients (37.9%) were treated with antipsychotic combination therapy.
Drug-Induced Parkinsonism
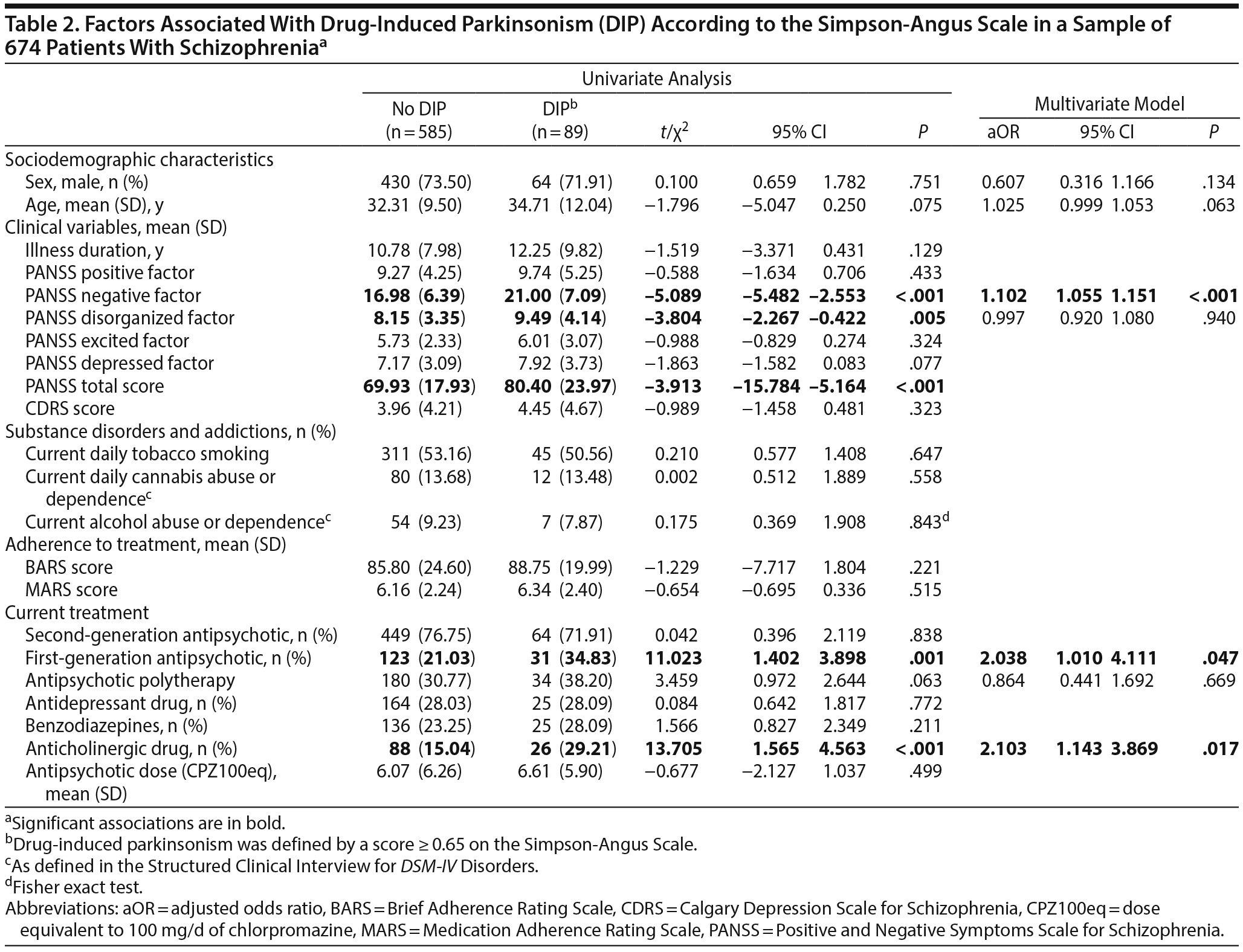
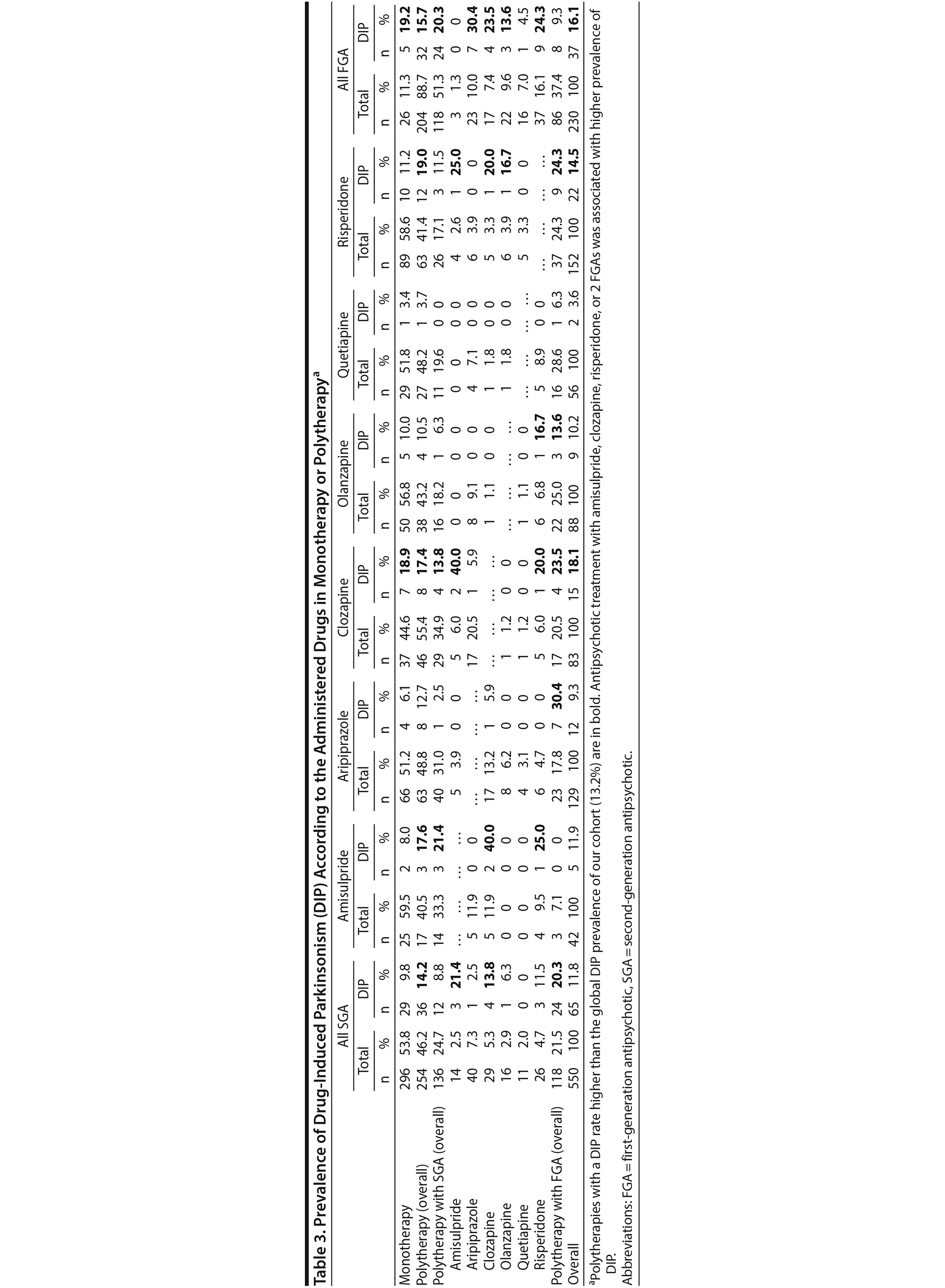
The global prevalence of DIP in our sample was 13.2% (89/674). In the multivariate analysis, after adjustment for sex, age, disorganization, and antipsychotic polytherapy, DIP remained significantly associated with higher negative symptoms (PANSS subscore, aOR = 1.102 [95% CI, 1.055-1.151], P < .001), prescription of FGAs (aOR = 2.038 [1.010-4.111], P = .047), and coprescription of an anticholinergic drug (aOR = 2.103 [1.143-3.869], P = .017) (Table 2). Concerning the PANSS negative factor, scores on all items, which include N1 (Blunted affect), N2 (Emotional withdrawal), N3 (Poor rapport), N4 (Passive/apathetic social withdrawal), N6 (Lack of spontaneity and flow of conversation), and G7 (Motor retardation), were significantly higher in patients with DIP.
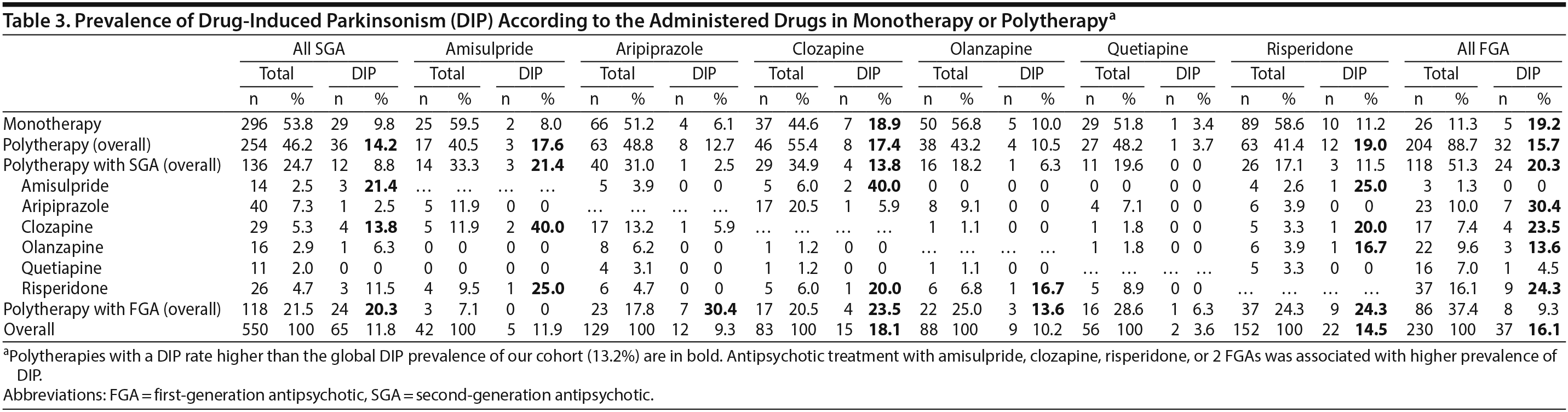
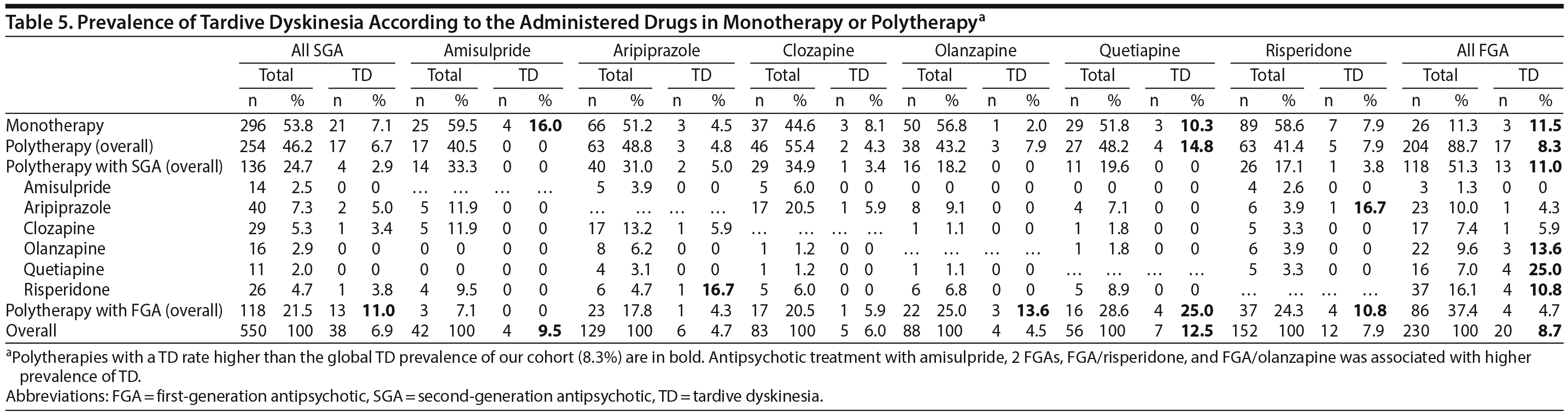
The details of the administered antipsychotics and the related proportions of patients with DIP are presented in Table 3. In the analysis of all drugs, patients with DIP had lower prescription rates of quetiapine than those without DIP (2.2% vs 8.6%, P = .047).
Tardive Dyskinesia
The overall prevalence of tardive dyskinesia was 8.3% (56/674). Thirteen patients (1.9%) had both of these 2 extrapyramidal side effects (DIP and TD).
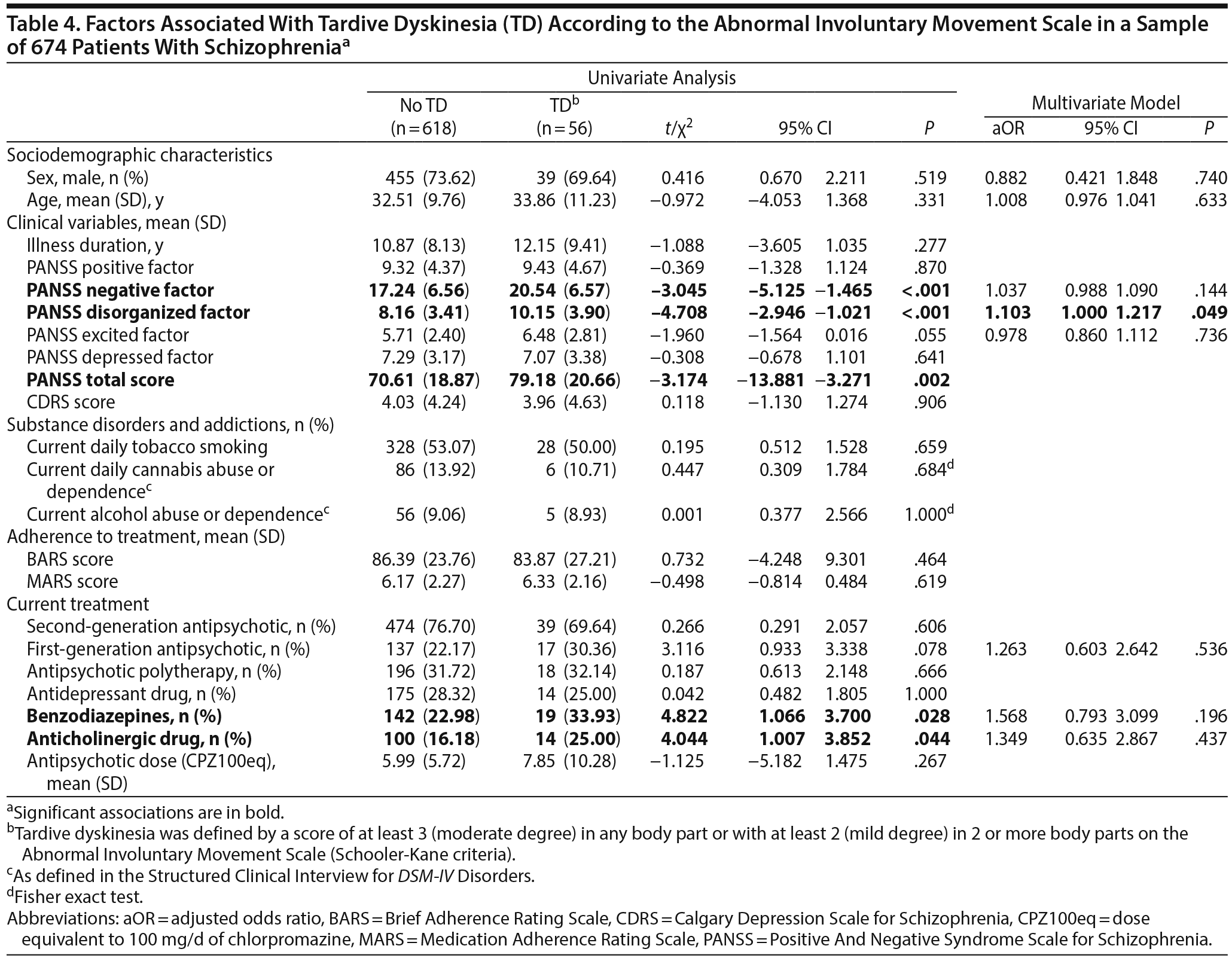
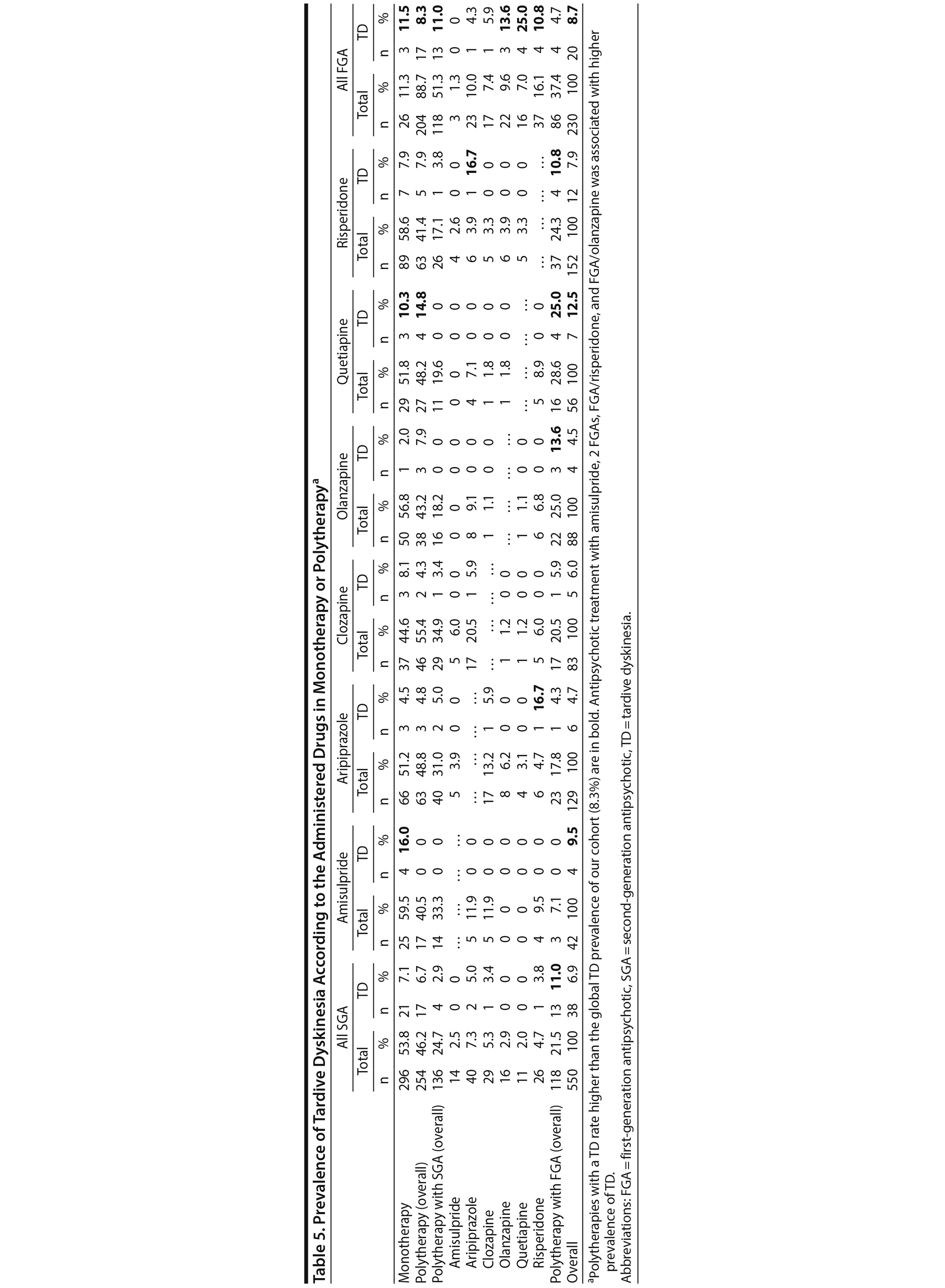
In the multivariate analysis, after adjustment for confounding factors (sex, age, negative symptoms, excitation, FGA prescription, and benzodiazepine and anticholinergic drug administration), TD remained significantly associated with higher disorganization level (PANSS subscore, aOR = 1.103, [1.000-1.217], P = .049) (Table 4). The PANSS disorganized factor is composed of P2 (Conceptual disorganization), N5 (Difficulty in abstract thinking), and G11 (Poor attention) items, which were significantly higher in patients with TD.
The TD frequency according to antipsychotic classes is presented in Table 5. In the analysis of all drugs, patients with TD had higher prescription rates of chlorpromazine (3.6% vs 0.3%, P = .032) and haloperidol (16.1% vs 7%, P = .013) than those without TD.
DISCUSSION
Our major findings may be summarized as follows: the overall prevalence of DIP and TD was, respectively, 13.2% and 8.3% in a national stabilized community-dwelling sample of 674 outpatients with schizophrenia. After adjustment for confounding factors, DIP remained significantly associated with higher negative symptoms level, FGA treatment, and anticholinergic administration, while TD remained significantly associated only with higher disorganization symptoms level.
The DIP rate of 13.2% found in this study is the lowest rate of all published studies to date (with DIP ranging from 14% to 40%6,31-34). The different scales and thresholds, different treatments, and different population characteristics may explain this discrepancy. In the current study, the threshold of 0.65 was used to increase the scale’s specificity (62%).29 This choice may contribute to an underestimation of DIP prevalence in this sample of community-dwelling stabilized outpatients. DIP has been previously associated with antipsychotic daily dose.34 However, antipsychotic daily dose was not associated with higher DIP rates in our results, which suggests that other factors may be involved, including other psychotropic drugs, addictive behavior (such as tobacco smoking), lifestyle habits, and diet. The young mean age and the high proportion of men in our cohort may explain our low prevalence, which could be a limitation. No association of tobacco smoking with DIP was found in the present study, which is not consistent with previous studies suggesting that an inducer effect of daily tobacco smoking may lower the blood antipsychotic level.35,36
The present results confirm that the risk for DIP was associated with current FGA administration in real-world schizophrenia. This finding is consistent with the results of a large meta-analysis including only randomized controlled trials with highly selected patients (ie, with good compliance and without comorbidities and suicide risk).16 Our descriptive analyses show that patients with DIP are most often (56.4%) treated with antipsychotic combination therapy (18.2% with an FGA/FGA combination, 27.3% with an SGA/SGA combination, and 54.5% with an FGA/SGA combination), and 43.6% are treated with a single antipsychotic (14.7% with an FGA monotherapy and 85.3% with an SGA monotherapy). While these results are only cross-sectional, it may be reasonably suggested that antipsychotic combination should be avoided as soon as possible in patients with DIP, especially when associated with negative symptoms. Polytherapy has never, to date, been associated with higher effectiveness,37 which suggests that the benefit/risk ratio is in favor of monotherapy, especially in cases of DIP and negative symptoms. Future interventional studies should determine if switching from polytherapy to antipsychotic monotherapy might alleviate both DIP and negative symptoms.
Patients with anticholinergic treatment had significantly more DIP. This result is plausibly due to the prescription bias: patients with higher DIP are more prone to being prescribed anticholinergic medication by their psychiatrists. However, these results suggest that anticholinergics are not sufficient to correct DIP in real-world patients with schizophrenia, due to potential underdosage or insufficient effectiveness. This result is consistent with the results from a recent meta-analysis,16 which found that clozapine and olanzapine induced significantly fewer EPS than the FGA-anticholinergic association. Moreover, anticholinergic drugs have been associated with cognitive impairment in patients with schizophrenia.38 Altogether, these results suggest that if persistent DIP occurs during FGA and anticholinergic drug prescription, the treatment should be switched to SGA if possible.
DIP has been associated with higher negative symptoms in the present sample, which is consistent with the results of previous studies. This co-occurrence has been suggested to be due to a common neurobiological basis.39,40 The strong associations between negative and motor features in antipsychotic-treated subjects may be explained by drug-induced negative symptoms and motor signs as a consequence of drug-related dopamine blockade41 with a possible direct neurotoxic effect of antipsychotics on dopamine neurons.42 Motor symptoms may be related to the dysfunction of the nigrostriatal dopaminergic system, while negative symptoms may be the consequence of blocking receptors of the meso-cortico-limbic dopaminergic system. Beyond motor symptoms, patients with Parkinson’s disease also show bradyphrenia (G7), as well as many of the negative symptoms (N1 [Blunted affect], N2 [Emotional withdrawal], N4 [Passive/apathetic social withdrawal], N5 [Difficulty in abstract thinking], and N6 [Lack of spontaneity and flow of conversation]), at least in part because of impaired function of meso-cortico-limbic neurons. Similarly to Parkinson’s disease, degeneration of dopaminergic neurons has been identified in 45% of patients with schizophrenia with a 2-year single-photon emission computerized tomography follow-up, which suggested the potential benefit of levodopa therapy in this subgroup.9
The TD rate was 8.3% in the present study, and 27.2% of this sample was currently prescribed at least 1 FGA. The prevalence of TD was lower than expected, which may be due to sociodemographic characteristics, especially the relatively young age of the sample (mean age of 32.6 years). Younger age may also explain the absence of significant association between TD and age in the present sample, which is inconsistent with previous results.15,20,43 In a recent meta-analysis including 41 studies (11,493 patients, mean age = 42.8 years) using the same scale and cutoff (AIMS with Schooler-Kane criteria), the global mean TD prevalence was 25.3% (95% CI = 22.7%-28.1%) vs 7.2% in the 4 studies including only FGA-naive patients.20 The present study did not report the lifetime prescription of antipsychotic, which is a limitation. Future studies should include the lifetime number of antipsychotic treatments and the mean dose/mean duration of exposure for each antipsychotic treatment. Altogether, these studies combined with the present study underlie the difficulty of defining and assessing TD.44 These inconsistent results are due to differences in factors including population age (higher age), sex, region (high geographical variation), medication type and dosing (higher rates during FGA than SGA treatment), and illness and treatment duration.15,20,43 The prevalence of EPS should be interpreted in relation to the relatively low illness duration in the present sample (11 years). TD has been shown to increase with age and lifetime duration of antipsychotic treatment.15,20 No gender effect has been found in the present study, which is consistent with previous findings.20,45
TD was associated with higher disorganized/cognitive score in the current study.23 This factor was highly correlated with cognitive functions evaluated with the Wechsler Adult Intelligence Scale IQ and general cognitive ability.23 This is consistent with previous studies suggesting that TD was associated with cognitive impairment such as orientation, memory, silence, attention, and muteness in schizophrenia.44,46-48 It has been suggested that this association was mediated by decreased brain-derived neurotrophic factor, which may play a critical role in cognitive function and may reflect a compensatory response to severe cerebral damages.49,50 Future studies should explore the specific cognitive functions associated with TD in real-world schizophrenia and determine which intervention may be effective in improving both TD and cognitive functioning in patients suffering from schizophrenia with TD. It has been hypothesized that TD may result primarily from antipsychotics’ induced dopamine supersensitivity in the nigrostriatal pathway (D2 dopamine receptor).51 This would be consistent with observations of Parkinson’s disease in which dyskinesia is induced by an “overdose” of dopaminergic treatment, which may also result in positive psychotic symptoms.52 Cognitive evaluations should be carefully evaluated when evaluating the benefit/risk ratio of switching or adjusting antipsychotic treatment in cases of iatrogenic TD.
Our study has several limitations. The cross-sectional design does not allow us to infer the causal nature of the observed associations. Secondary negative symptoms were not differentiated from primary negative symptoms in the present study. Differentiating primary from secondary negative symptoms remains difficult in populations with long duration of illness due to the memory bias and is adjusted for in prospective studies. No significant associations were found between EPS and depression (CDRS). Previous studies suggested that antipsychotics could increase depressive symptoms due to their D2 high affinity,18 and this point should be explored in future studies. As mentioned above, lifetime exposure to antipsychotics was not reported due to memory bias, as some patients had more than 10 years of illness duration.45 Finally, parkinsonism was defined according to the SAS score based on trained clinician assessment. A DaTscan examination should be considered in future studies for patients with DIP or TD to confirm alterations in dopamine circuits and discriminate DIP from other causes of parkinsonism including Parkinson’s disease.
CONCLUSION
The present findings suggest a high prevalence of DIP and TD in a relatively young sample of patients with schizophrenia with a mean illness duration of approximately 11 years and a complex association of negative (for DIP) and disorganized/cognitive (for TD) symptomatology. More than one-fourth of the patients were administered at least 1 FGA, and these patients were found to have higher DIP levels despite the prescription of anticholinergic medications. The present findings indicate that the choice of monotherapy with SGAs should be recommended as soon as possible to prevent EPS onset in patients with schizophrenia.
Submitted: March 15, 2018; accepted July 9, 2018.
Published online: January 8, 2019.
Disclosure of off-label usage: The authors have determined that, to the best of their knowledge, no investigational information about pharmaceutical agents that is outside US Food and Drug Administration-approved labeling has been presented in this article.
Author contributions: Mr Tessier and Dr Daubigney performed the statistical analysis. Mr Tessier and Drs Daubigney and Misdrahi wrote the first complete manuscript. Drs Llorca, Fond, and Meissner edited earlier versions of the manuscript for important intellectual content. All authors were involved in the patients’ recruitment, the clinical evaluation, and acquisition of the clinical data and approved the final manuscript.
FACE-SZ Group: M. Andrianarisoa, MDa,e; B. Aouizerate, MD, PhDa,b; N. Bazin, MDa,l; F. Berna, MD, PhDa,c; O. Blanc, MSca,d; L. Brunel, MSca,e; E. Bulzacka, MSca,e; D. Capdevielle, MD, PhDa,f; I. Chereau-Boudet, MDa,d; G. Chesnoy-Servanin, MSca,g; N. Coulon, MD, PhDa,e; J.-M. Danion, MDa,c; T. D’ Amato, MD, PhDa,g; A. Deloge, MDa,h; C. Delorme, MSca,i; H. Denizot, MDa,d; J.-M. Dorey, MDa,g; C. Dubertret, MD, PhDa,j; J. Dubreucq, MDa,i; C. Faget-Agius, MDa,k; C. Fluttaz, MSca,i; G. Fond, MD, PhDa; S. Fonteneau, MSca,l; F. Gabayet, MSca,i; E. Giraud-Baro, MDa,i; M. Jarroir, MSca,l; D. Lacelle, MSca,d; C. Lançon, MD, PhDa,k; H. Laouamri, MSca; M. Leboyer, MD, PhDa,e; T. Le Gloahec, MSca,e; Y. Le Strat, MD, PhDa,j; P.-M. Llorca, MD, PhDa,d; J. Mallet, MDa,j; E. Metairie, MSca,k; D. Misdrahi, MDa,h; I. Offerlin-Meyer, PhDa,c; C. Passerieux, MD, PhDa,l; P. Peri, MSca,k; S. Pires, MSca,d; C. Portalier, MSca,j; L. Ramet, MSca,l; R. Rey, MDa,g; C. Roman, MSca,i; A. Schandrin, MDa,m; F. Schürhoff, MD, PhDa,e; A. Tessier, MSca,h; A.-M. Tronche, MDa,d; M. Urbach, MDa,l; F. Vaillant, MSca,k; A. Vehier, MSca,g; P. Vidailhet, MD, PhDa,c; E. Vil× , MSca,h; H. Yazbek, MSca,f; and A. Zinetti-Bertschy, MSc.a,c
aFondaMental Foundation, Créteil, France
bDepartment of Adult Psychiatry, Charles Perrens Hospital; and Laboratory of Nutrition and Integrated Neurobiology (UMR INRA 1286), University of Bordeaux, Bordeaux, France
cStrasbourg University Hospital, University of Strasbourg, INSERM U1114, Federation of Translational Psychiatry, Strasbourg, France
dCMP B, CHU, EA 7280 Faculty of Medicine, University of Auvergne, Clermont-Ferrand, France
eINSERM U955, Translational Psychiatry Team, Créteil, France, Paris-Est Créteil University, DHU Pe-PSY, Psychiatry and Addictions Department, Henri Mondor Hospital, Créteil, France
fAcademic Department of Adult Psychiatry, La Colombiרre Hospital, CHRU Montpellier, Montpellier University, Inserm 1061, Montpellier, France
gINSERM U1028, CNRS UMR5292, Neurosciences Research Center of Lyon, Claude Bernard University, PSYR2 team, Le Vinatier, Hospital, Bron, France
hDepartment of Adult Psychiatry, Charles Perrens Hospital; University of Bordeaux, CNRS UMR 5287-INCIA, Bordeaux, France
iPsychosocial Rehabilitation Reference Centre, Alpes Isרre Hospital, Grenoble, France
jAP-HP, Department of Psychiatry, Louis Mourier Hospital, Colombes, Inserm U894 Paris Diderot University, Sorbonne Paris Cité, Faculty of Medicine, Paris, France
kAP-HM, Academic Department of Psychiatry, Marseille, France
lDepartment of Adult Psychiatry, Versailles Hospital, Le Chesnay, EA 4047 HANDIReSP, Versailles Saint-Quentin en Yvelines University, Versailles, France
mCarémeau Hospital, N×®mes, France
Financial disclosure: Drs Misdrahi, Daubigney, Meissner, Schurhoff, Boyer, Aouizerate, Andrianarisoa, Berna, Capdevielle, Chereau-Boudet, D’ Amato, Dubertret, Dubreucq, Faget-Agius, Lançon, Mallet, Passerieux, Rey, Schandrin, Urbach, Vidailhet, Llorca, and Fond; Mr Tessier; and Mss Godin and Bulzacka have no personal affiliations or financial relationships with any commercial interest to disclose relative to the article.
Funding/support: This work was funded by Charles-Perrens Hospital, AP-HP (Assistance Publique des H×´pitaux de Paris), AP-HM (Assistance Publique des H×´pitaux de Marseille), FondaMental Foundation (RTRS Santé Mentale), the Investissements d’ Avenir program managed by the Agence Nationale de la Recherché (ANR) under reference ANR-11-IDEX-0004-02 and ANR-10-COHO-10-01, and INSERM (Institut National de la Santé et de la Recherche Médicale).
Role of the sponsor: The FondaMental Foundation (RTRS Santé Mentale) provided data management support. Charles-Perrens Hospital, AP-HP (Assistance Publique des H×´pitaux de Paris), AP-HM (Assistance Publique des H×´pitaux de Marseille), and Investissements d’ Avenir program had no role in the conduct or publication of the study.
Acknowledgments: The authors express thanks to the nurses and to the patients who were included in the present study. They thank Hakim Laouamri, MSc (FondaMental Foundation) and his team (Stéphane Beaufort, MSc; Seif Ben Salem, MSc; Karmרne Souyris, MSc; Victor Barteau, MSc; and Mohamed Laaidi, MSc) for the development of the FACE-SZ computer interface, data management, quality control, and regulatory aspects. They thank Prof François Tison, MD, PhD, and Prof Dominique Guehl, MD, PhD, of Bordeaux University Hospital, for critical reading and the neurological point of view. The acknowledged individuals report no conflicts of interest with this study.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
REFERENCES
1. Browne S, Roe M, Lane A, et al. Quality of life in schizophrenia: relationship to sociodemographic factors, symptomatology and tardive dyskinesia. Acta Psychiatr Scand. 1996;94(2):118-124. PubMed CrossRef
2. Chang WC, Cheung R, Hui CLM, et al. Rate and risk factors of depressive symptoms in Chinese patients presenting with first-episode non-affective psychosis in Hong Kong. Schizophr Res. 2015;168(1-2):99-105. PubMed CrossRef
3. Munhoz RP, Bertucci Filho D, Teive HAG. Not all drug-induced Parkinsonism are the same: the effect of drug class on motor phenotype. Neurol Sci Off J Ital Neurol Soc Ital Soc Clin Neurophysiol. 2017;38(2):319-324. PubMed CrossRef
4. Hasan A, Falkai P, Wobrock T, et al; WFSBP Task force on Treatment Guidelines for Schizophrenia. World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for biological treatment of schizophrenia, part 2: update 2012 on the long-term treatment of schizophrenia and management of antipsychotic-induced side effects. World J Biol Psychiatry. 2013;14(1):2-44. PubMed CrossRef
5. Kane JM. Pharmacologic treatment of schizophrenia. Biol Psychiatry. 1999;46(10):1396-1408. PubMed CrossRef
6. Novick D, Haro JM, Bertsch J, et al. Incidence of extrapyramidal symptoms and tardive dyskinesia in schizophrenia: thirty-six-month results from the European Schizophrenia Outpatient Health Outcomes study. J Clin Psychopharmacol. 2010;30(5):531-540. PubMed CrossRef
7. Pappa S, Dazzan P. Spontaneous movement disorders in antipsychotic-naive patients with first-episode psychoses: a systematic review. Psychol Med. 2009;39(7):1065-1076. PubMed CrossRef
8. Bower JH, Maraganore DM, McDonnell SK, et al. Incidence and distribution of Parkinsonism in Olmsted County, Minnesota, 1976-1990. Neurology. 1999;52(6):1214-1220. PubMed CrossRef
9. Tinazzi M, Morgante F, Matinella A, et al. Imaging of the dopamine transporter predicts pattern of disease progression and response to levodopa in patients with schizophrenia and parkinsonism: a 2-year follow-up multicenter study. Schizophr Res. 2014;152(2-3):344-349. PubMed CrossRef
10. Tinazzi M, Cipriani A, Matinella A, et al. [123I]FP-CIT single photon emission computed tomography findings in drug-induced parkinsonism. Schizophr Res. 2012;139(1-3):40-45. PubMed CrossRef
11. Rybakowski JK, Vansteelandt K, Remlinger-Molenda A, et al; EUFEST Study Group. Extrapyramidal symptoms during treatment of first schizophrenia episode: results from EUFEST. Eur Neuropsychopharmacol. 2014;24(9):1500-1505. PubMed CrossRef
12. Othman Z, Ghazali M, Razak A, et al. Severity of tardive dyskinesia and negative symptoms are associated with poor quality of life in schizophrenia patients. Int Med J. 2013;20:677-680.
13. Lerner PP, Miodownik C, Lerner V. Tardive dyskinesia (syndrome): current concept and modern approaches to its management. Psychiatry Clin Neurosci. 2015;69(6):321-334. PubMed CrossRef
14. Owens DG, Johnstone EC, Frith CD. Spontaneous involuntary disorders of movement: their prevalence, severity, and distribution in chronic schizophrenics with and without treatment with neuroleptics. Arch Gen Psychiatry. 1982;39(4):452-461. PubMed CrossRef
15. Miller DD, McEvoy JP, Davis SM, et al. Clinical correlates of tardive dyskinesia in schizophrenia: baseline data from the CATIE schizophrenia trial. Schizophr Res. 2005;80(1):33-43. PubMed CrossRef
16. Leucht S, Corves C, Arbter D, et al. Second-generation versus first-generation antipsychotic drugs for schizophrenia: a meta-analysis. Lancet. 2009;373(9657):31-41. PubMed CrossRef
17. Rummel-Kluge C, Komossa K, Schwarz S, et al. Second-generation antipsychotic drugs and extrapyramidal side effects: a systematic review and meta-analysis of head-to-head comparisons. Schizophr Bull. 2012;38(1):167-177. PubMed CrossRef
18. Yoshida K, Bies RR, Suzuki T, et al. Tardive dyskinesia in relation to estimated dopamine D2 receptor occupancy in patients with schizophrenia: analysis of the CATIE data. Schizophr Res. 2014;153(1-3):184-188. PubMed CrossRef
19. Berna F, Misdrahi D, Boyer L, et al; FACE-SZ (FondaMental Academic Centers of Expertise for Schizophrenia) group. Akathisia: prevalence and risk factors in a community-dwelling sample of patients with schizophrenia: results from the FACE-SZ dataset. Schizophr Res. 2015;169(1-3):255-261. PubMed CrossRef
20. Carbon M, Hsieh C-H, Kane JM, et al. Tardive dyskinesia prevalence in the period of second-generation antipsychotic use: a meta-analysis. J Clin Psychiatry. 2017;78(3):e264-e278. PubMed CrossRef
21. Schürhoff F, Fond G, Berna F, et al; FondaMental Academic Centers of Expertise for Schizophrenia (FACE-SZ) collaborators. A national network of schizophrenia expert centres: an innovative tool to bridge the research-practice gap. Eur Psychiatry. 2015;30(6):728-735. PubMed CrossRef
22. Kay SR, Fiszbein A, Opler LA. The Positive and Negative Syndrome Scale (PANSS) for schizophrenia. Schizophr Bull. 1987;13(2):261-276. PubMed CrossRef
23. Wallwork RS, Fortgang R, Hashimoto R, et al. Searching for a consensus five-factor model of the Positive and Negative Syndrome Scale for schizophrenia. Schizophr Res. 2012;137(1-3):246-250. PubMed CrossRef
24. Lançon C, Auquier P, Reine G, et al. Study of the concurrent validity of the Calgary Depression Scale for Schizophrenics (CDSS). J Affect Disord. 2000;58(2):107-115. PubMed CrossRef
25. Byerly MJ, Nakonezny PA, Rush AJ. The Brief Adherence Rating Scale (BARS) validated against electronic monitoring in assessing the antipsychotic medication adherence of outpatients with schizophrenia and schizoaffective disorder. Schizophr Res. 2008;100(1-3):60-69. PubMed CrossRef
26. Misdrahi D, Tessier A, Swendsen J, et al; FACE-SZ (FondaMental Academic Centers of Expertise for Schizophrenia) Group. Determination of adherence profiles in schizophrenia using self-reported adherence: results from the FACE-SZ dataset. J Clin Psychiatry. 2016;77(9):e1130-e1136. PubMed CrossRef
27. Leucht S, Samara M, Heres S, et al. Dose equivalents for second-generation antipsychotics: the minimum effective dose method. Schizophr Bull. 2014;40(2):314-326. PubMed CrossRef
28. Knol W, Keijsers CJPW, Jansen PAF, et al. Systematic evaluation of rating scales for drug-induced parkinsonism and recommendations for future research. J Clin Psychopharmacol. 2010;30(1):57-63. PubMed CrossRef
29. Janno S, Holi MM, Tuisku K, et al. Validity of Simpson-Angus Scale (SAS) in a naturalistic schizophrenia population. BMC Neurol. 2005;5(1):5. PubMed CrossRef
30. Bark N, Florida D, Gera N, et al. Evaluation of the routine clinical use of the Brief Psychiatric Rating Scale (BPRS) and the Abnormal Involuntary Movement Scale (AIMS). J Psychiatr Pract. 2011;17(4):300-303. PubMed CrossRef
31. Czobor P, Van Dorn RA, Citrome L, et al. Treatment adherence in schizophrenia: a patient-level meta-analysis of combined CATIE and EUFEST studies. Eur Neuropsychopharmacol. 2015;25(8):1158-1166. PubMed CrossRef
32. Hansen TE, Casey DE, Hoffman WF. Neuroleptic intolerance. Schizophr Bull. 1997;23(4):567-582. PubMed CrossRef
33. Janno S, Holi M, Tuisku K, et al. Prevalence of neuroleptic-induced movement disorders in chronic schizophrenia inpatients. Am J Psychiatry. 2004;161(1):160-163. PubMed CrossRef
34. Parksepp M, Ljubajev ל, Tפht K, et al. Prevalence of neuroleptic-induced movement disorders: an 8-year follow-up study in chronic schizophrenia inpatients. Nord J Psychiatry. 2016;70(7):498-502. PubMed CrossRef
35. Kroon LA. Drug interactions with smoking. Am J Health Syst Pharm. 2007;64(18):1917-1921. PubMed CrossRef
36. Wu T-H, Chiu C-C, Shen WW, et al. Pharmacokinetics of olanzapine in Chinese male schizophrenic patients with various smoking behaviors. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32(8):1889-1893. PubMed CrossRef
37. Correll CU, Rummel-Kluge C, Corves C, et al. Antipsychotic combinations vs monotherapy in schizophrenia: a meta-analysis of randomized controlled trials. Schizophr Bull. 2009;35(2):443-457. PubMed CrossRef
38. Minzenberg MJ, Poole JH, Benton C, et al. Association of anticholinergic load with impairment of complex attention and memory in schizophrenia. Am J Psychiatry. 2004;161(1):116-124. PubMed CrossRef
39. Peralta V, Cuesta MJ. Negative parkinsonian, depressive and catatonic symptoms in schizophrenia: a conflict of paradigms revisited. Schizophr Res. 1999;40(3):245-253. PubMed CrossRef
40. Docx L, Morrens M, Bervoets C, et al. Parsing the components of the psychomotor syndrome in schizophrenia. Acta Psychiatr Scand. 2012;126(4):256-265. PubMed CrossRef
41. Heinz A, Knable MB, Coppola R, et al. Psychomotor slowing, negative symptoms and dopamine receptor availability—an IBZM SPECT study in neuroleptic-treated and drug-free schizophrenic patients. Schizophr Res. 1998;31(1):19-26. PubMed CrossRef
42. Rollema H, Skolnik M, D’ Engelbronner J, et al. MPP(+)-like neurotoxicity of a pyridinium metabolite derived from haloperidol: in vivo microdialysis and in vitro mitochondrial studies. J Pharmacol Exp Ther. 1994;268(1):380-387. PubMed
43. Jeste DV, Lacro JP, Palmer B, et al. Incidence of tardive dyskinesia in early stages of low-dose treatment with typical neuroleptics in older patients. Am J Psychiatry. 1999;156(2):309-311. PubMed CrossRef
44. Wegner JT, Catalano F, Gibralter J, et al. Schizophrenics with tardive dyskinesia: neuropsychological deficit and family psychopathology. Arch Gen Psychiatry. 1985;42(9):860-865. PubMed CrossRef
45. Yassa R, Jeste DV. Gender differences in tardive dyskinesia: a critical review of the literature. Schizophr Bull. 1992;18(4):701-715. PubMed CrossRef
46. Waddington JL, Youssef HA. An unusual cluster of tardive dyskinesia in schizophrenia: association with cognitive dysfunction and negative symptoms. Am J Psychiatry. 1986;143(9):1162-1165. PubMed CrossRef
47. Waddington JL, Youssef HA, Dolphin C, et al. Cognitive dysfunction, negative symptoms, and tardive dyskinesia in schizophrenia: their association in relation to topography of involuntary movements and criterion of their abnormality. Arch Gen Psychiatry. 1987;44(10):907-912. PubMed CrossRef
48. Byne W, White L, Parella M, et al. Tardive dyskinesia in a chronically institutionalized population of elderly schizophrenic patients: prevalence and association with cognitive impairment. Int J Geriatr Psychiatry. 1998;13(7):473-479. PubMed CrossRef
49. Wu JQ, Chen DC, Tan YL, et al. Cognition impairment in schizophrenia patients with tardive dyskinesia: association with plasma superoxide dismutase activity. Schizophr Res. 2014;152(1):210-216. PubMed CrossRef
50. Wu JQ, Chen DC, Tan YL, et al. Altered BDNF is correlated to cognition impairment in schizophrenia patients with tardive dyskinesia. Psychopharmacology (Berl). 2015;232(1):223-232. PubMed CrossRef
51. Seeman P. Schizophrenia and dopamine receptors. Eur Neuropsychopharmacol. 2013;23(9):999-1009. PubMed CrossRef
52. Hinkle JT, Perepezko K, Rosenthal LS, et al. Markers of impaired motor and cognitive volition in Parkinson’s disease: correlates of dopamine dysregulation syndrome, impulse control disorder, and dyskinesias. Parkinsonism Relat Disord. 2018;47:50-56. PubMed. CrossRef
This PDF is free for all visitors!