REM Sleep Behavior Disorder in Psychiatric Populations
To the Editor: Rapid eye movement (REM) sleep behavior disorder (RBD) is a parasomnia characterized by a loss of REM-related muscle atonia and abnormal motor activities during REM sleep with consequent sleep-related injuries.1,2 It has been increasingly reported among psychiatric populations and has a potential association with use of psychotropics, particularly selective serotonin reuptake inhibitor (SSRI), tricyclic, and serotonin and noradrenergic reuptake inhibitor (SNRI) antidepressants.3-8
A previous case report5 found that cessation of SSRI treatment did not result in complete resolution of clinical RBD symptoms and polysomnographic (PSG) abnormalities. Our recent clinical epidemiologic study8 found that 3.8% of psychiatric outpatients might have RBD features over the past year. The prevalence is 10 times more common than that of the typical RBD in the elderly general population.9 In addition, the RBD features could result in sleep-related injuries and violence, with potential medicolegal repercussions.8,10
In the current study, we conducted a comprehensive evaluation of clinical and PSG features as well as follow-up response to an open intervention of antidepressant regimen modification.
Method. Fifteen depressed subjects with concurrent antidepressant treatment and features suggestive of RBD were included in this case series, which spans from December 2006 to March 2009. They were interviewed by experienced psychiatrists and neurologists for a thorough clinical evaluation, which included a semistructured psychiatric interview and physical examination with Hoehn and Yahr staging for parkinsonism.11 They also underwent video-polysomnographic assessments at baseline and follow-up.
Modification of their current antidepressant regimen was suggested to all subjects. Bupropion was offered as the treatment of choice in this open intervention, as it has rarely been associated with RBD in previous reports. The subjects’ prescription history of benzodiazepines was also recorded, as benzodiazepines, especially clonazepam, are a common treatment option for RBD.
Electromyogram (EMG) activities during REM sleep were determined by a quantitative criterion of REM-related EMG activities (REMREEA) on chin muscles. Details of this quantitative scoring method have been described elsewhere.2 In brief, REMREEA is the summation of the percentage of both tonic and phasic chin EMG activities during REM sleep. EMG activities associated with respiratory events, periodic leg movements, spontaneous arousal, and signal artifacts were excluded from the analysis. The scorer of the REMREEA muscle activities was blind to the sleep diagnoses. The study was approved by the institutional ethics committee, and informed consent was obtained from all study subjects.
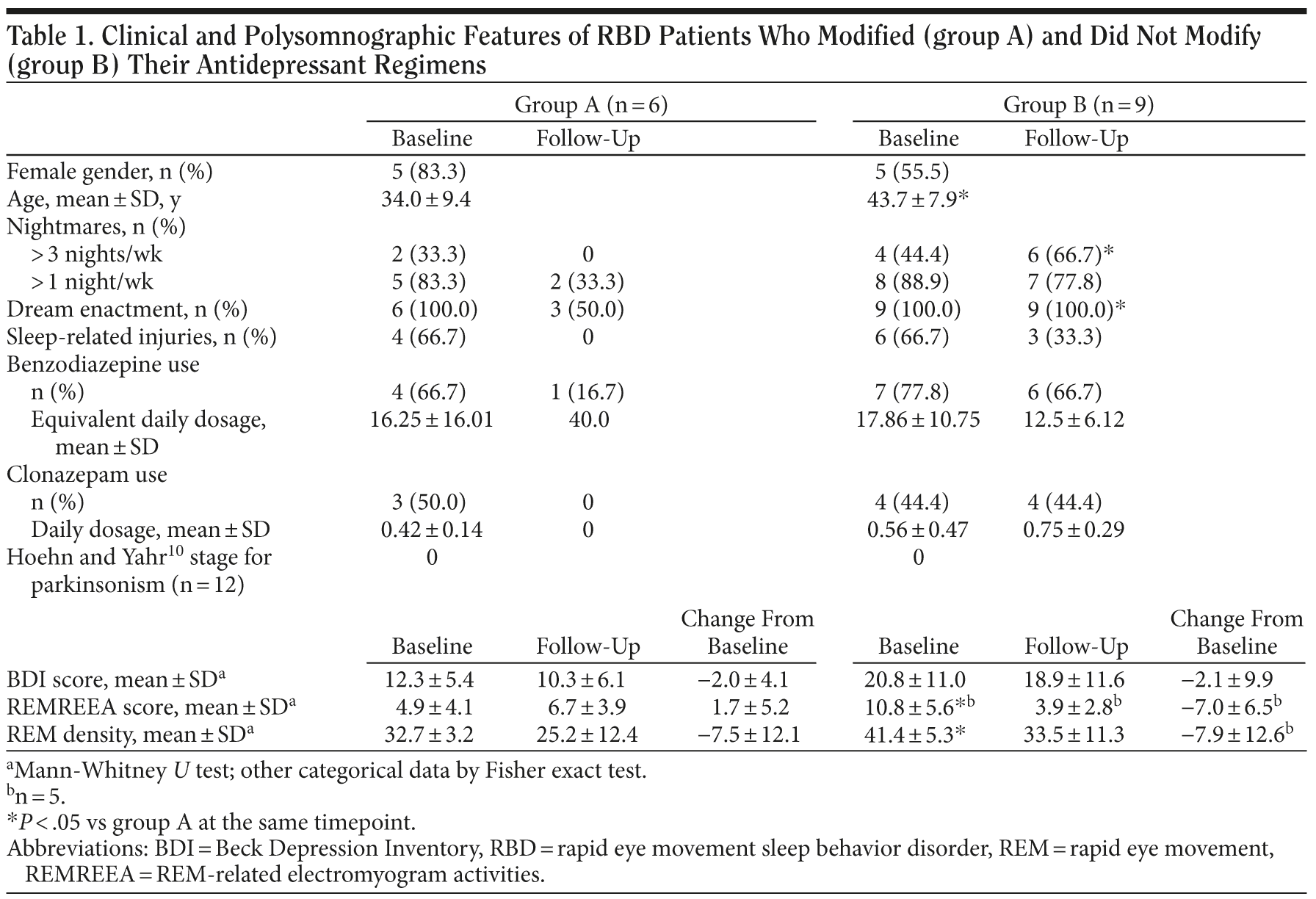
Results. At baseline, the majority of subjects (n = 12) were taking an SSRI, and 3 were taking an SNRI. The dosages of antidepressants were all within therapeutic range. None of the subjects had any clinical neurologic disorder or sign of parkinsonism. Six of the subjects (group A) agreed to alter their treatment, including switching to bupropion (n = 3) and discontinuing antidepressant use (n = 3) (in view of clinical remission). The remaining 9 subjects (group B) opted to keep their usual treatments, and only 5 completed the follow-up PSG. The mean duration of the follow-up period was 13 months (range, 9-19 months), with similar duration between the 2 groups.
For patients who continued their usual antidepressant treatments, there was no significant difference in the mean equivalent daily dosage of antidepressants between baseline and follow-up. Eleven patients were receiving benzodiazepine treatment at baseline (group A, n = 4, mean equivalent dosage of diazepam = 16.25 ± 16.01; group B, n = 7, mean equivalent dosage of diazepam = 17.86 ± 10.75). Among them, 7 subjects were prescribed clonazepam at a mean dosage of 0.5 mg/d (group A, n = 3; group B, n = 4). At follow-up, only 1 subject in group A continued to receive benzodiazepine treatment (same dosage as at baseline), while 6 subjects in group B continued to receive benzodiazepine treatment (including 4 subjects taking clonazepam). Their mean equivalent dosage of benzodiazepine did not differ from baseline.
At baseline, both groups had comparable clinical features of RBD with respect to frequency of nightmares, dream enactments, and sleep-related injuries (Table 1). However, group B subjects were older and had slightly more REMREEA. At follow-up, group A showed improvement in clinical symptoms of RBD, especially in frequency of nightmares and dream enactments (Table 1). They also had a nearly significant reduction in sleep-related injuries. However, their PSG still showed residual REMREEA.
This study provides further insights about RBD in psychiatric populations. First, the hypothesis of antidepressant treatment as the sole cause of RBD among psychiatric patients was not fully supported. Upon withdrawal or switch of antidepressants, the clinical symptoms improved, but the PSG features persisted. Although the study was limited by a small sample size and open intervention, the preliminary findings implied that, among vulnerable subjects, antidepressants might serve as precipitating agents for RBD symptoms instead of being the sole causative agent. A recent study of a series of patients with early-onset RBD reported a rather similar demographic background: female subjects with psychiatric morbidities and psychotropic usage.12 In particular, the longitudinal course of early-onset RBD and its relationship with typical RBD, especially with respect to a neurodegenerative etiologic basis, are unclear.8 In light of these distinct demographics of early onset and female predominance, and the potentially complex etiologic basis of the symptoms, we suggested that RBD in psychiatric populations would be better labeled as "atypical RBD" instead of "drug-induced RBD."
Second, our study suggested a possible synergistic mechanism of serotonergic antidepressants in precipitating RBD symptoms by modulation of the dream mechanism and REM sleep-related muscle atonia. Previous postulation of the mechanism focused on the effects of antidepressants on REM-related muscle atonia, including enhancement of EMG activities by SSRIs during REM sleep.13 However, both our study and a previous study5 found that REM-related muscle atonia was not completely restored after withdrawal of the antidepressants. The authors of the other study proposed an inhibitory role of antidepressants on extrapyramidal dopaminergic neurons,5 but our neurologic examination detected no clinical parkinsonism or other extrapyramidal side effects. Nonetheless, subtle dopaminergic transmission disturbance could not be excluded, and further neuroimaging study will be needed.14
As seen in the typical RBD cases, nightmares are a consistent and dominating feature that may act as an emotional drive for dream enactment.1,15 Changes in dream content with more vivid and intense dreams and nightmares have been reported among subjects taking serotonergic antidepressants.16-18 In our study, there was a parallel diminution of clinical RBD symptoms and severity of nightmares and dream enactments upon amendment of the antidepressant regimen. Thus, a distinct possibility remained that a serotonergic antidepressant could partially exert its effect by the modulation of dream mechanism. In addition, nightmares have been closely associated with mental states and psychiatric disorders (especially depression and posttraumatic stress disorder).19,20 In this regard, further study will be needed to examine the etiologic role of nightmares in both typical and atypical RBD as well as their associations with mental states and antidepressants.
References
1. Wing YK, Lam SP, Li SX, et al. REM sleep behaviour disorder in Hong Kong Chinese: clinical outcome and gender comparison. J Neurol Neurosurg Psychiatry. 2008;79(12):1415-1416. PubMed doi:10.1136/jnnp.2008.155374
2. Zhang J, Lam SP, Ho CKW, et al. Diagnosis of REM sleep behavior disorder by video-polysomnographic study: is one night enough? Sleep. 2008;31(8):1179-1185. PubMed
3. Schutte S, Doghramji K. REM behaviour disorder seen with venlafaxine. Sleep Res. 1996;25:364.
4. Onofrj M, Luciano AL, Thomas A, et al. Mirtazapine induces REM sleep behavior disorder (RBD) in parkinsonism. Neurology. 2003;60(1):113-115. PubMed
5. Schenck CH, Mahowald MW, Kim SW, et al. Prominent eye movements during NREM sleep and REM sleep behavior disorder associated with fluoxetine treatment of depression and obsessive-compulsive disorder. Sleep. 1992;15(3):226-235. PubMed
6. Nofzinger EA, Reynolds CF 3rd. REM sleep behavior disorder [letter]. JAMA. 1994;271(11):820. PubMed doi:10.1001/jama.271.11.820
7. Parish JM. Violent dreaming and antidepressant drugs: or how paroxetine made me dream that I was fighting Saddam Hussein. J Clin Sleep Med. 2007;3(5):529-531. PubMed
8. Lam SP, Fong SYY, Ho CKW, et al. Parasomnia among psychiatric outpatients: a clinical, epidemiologic, cross-sectional study. J Clin Psychiatry. 2008;69(9):1374-1382. PubMed doi:10.4088/JCP.v69n0904
9. Chiu HF, Wing YK, Lam LCW, et al. Sleep-related injury in the elderly—an epidemiological study in Hong Kong. Sleep. 2000;23(4):513-517. PubMed
10. Schenck CH, Lee SA, Bornemann MA, et al. Potentially lethal behaviors associated with rapid eye movement sleep behavior disorder: review of the literature and forensic implications. J Forensic Sci. 2009;54(6):1475-1484. PubMed doi:10.1111/j.1556-4029.2009.01163.x
11. Hoehn MM, Yahr MD. Parkinsonism: onset, progression and mortality. Neurology. 1967;17(5):427-442. PubMed
12. Teman PT, Tippmann-Peikert M, Silber MH, et al. Idiopathic rapid-eye-movement sleep disorder: associations with antidepressants, psychiatric diagnoses, and other factors, in relation to age of onset. Sleep Med. 2009;10(1):60-65. PubMed doi:10.1016/j.sleep.2007.11.019
13. Winkelman JW, James L. Serotonergic antidepressants are associated with REM sleep without atonia. Sleep. 2004;27(2):317-321. PubMed
14. Albin RL, Koeppe RA, Chervin RD, et al. Decreased striatal dopaminergic innervation in REM sleep behavior disorder. Neurology. 2000;55(9):1410-1412. PubMed
15. Li SX, Wing YK, Lam SP, et al. Validation of a new REM sleep behavior disorder questionnaire (RBDQ-HK). Sleep Med. 2010;11(1):43-48. PubMed doi:10.1016/j.sleep.2009.06.008
16. Pagel JF, Helfter P. Drug induced nightmares—an etiology based review. Hum Psychopharmacol. 2003;18(1):59-67. PubMed doi:10.1002/hup.465
17. Kirschner NT. Medication and dreams: changes in dream content after drug treatment. Dreaming. 1999;9(2/3):195-200. doi:10.1023/A:1021349901234
18. Pace-Schott EF, Gersh T, Silvestri R, et al. SSRI treatment suppresses dream recall frequency but increases subjective dream intensity in normal subjects. J Sleep Res. 2001;10(2):129-142. PubMed doi:10.1046/j.1365-2869.2001.00249.x
19. Plante DT, Winkelman JW. Parasomnias: psychiatric considerations. Sleep Med Clin. 2008;3(2):217-229. doi:10.1016/j.jsmc.2008.01.005
20. Li SX, Lam SP, Yu MWM, et al. Nocturnal sleep disturbances as a predictor of suicide attempts among psychiatric outpatients. J Clin Psychiatry. In press.
Author affiliation: Department of Psychiatry, Shatin Hospital (Drs Lam, Zhang, Tsoh, and Wing; Ms Li; and Mr Ho) and Department of Medicine and Therapeutics, Faculty of Medicine (Drs Mok and Chan), the Chinese University of Hong Kong, Shatin, Hong Kong SAR. Potential conflicts of interest: Mr Ho received honoraria from Covidien (Tyco Healthcare) for a 1-day lecture on clinical polysomnography in December 2008. The other authors report no financial or other relationship relevant to the subject of this letter. Funding/support: This study is supported by the Health and Health Services Research Fund (HHSRF) of Hong Kong SAR (project reference 07080011). Previous presentation: Some of the results in this letter were presented as a poster at the 22nd Annual Meeting of the Professional Sleep Societies (SLEEP 2008); June 7-12, 2008; Baltimore, Maryland.
doi:10.4088/JCP.l05877gry
© Copyright 2010 Physicians Postgraduate Press, Inc.