Neurotransmitter Targeting in the Treatment of Depression
Residual symptoms are a common hindrance to daily life for patients with major depressive disorder. Even after antidepressant treatment has led patients to meet remission criteria, almost all patients have at least 1 symptom that remains unresolved. These symptoms can increase the risk for relapse, a chronic course, and suicide attempts. Residual symptoms are lingering symptoms that do not resolve with treatment of the depressive episode, and they should be distinguished from symptoms of comorbid psychiatric or medical conditions and medication side effects. By understanding how various antidepressants affect the 3 monoamine systems of serotonin, norepinephrine, and dopamine, clinicians can select treatments based on the most effective mechanism of action. Dual-action agents show promise for alleviating depressive symptoms that do not resolve with single-action agents. Medications that increase norepinephrine or dopamine neurotransmission may improve several common residual symptoms left after treatment with serotonin-specific agents. Treatment strategies like adjunctive therapies and dosing options are given for common residual symptoms, including sleep difficulties, sexual dysfunction, and pain. For patients to truly regain their quality of life, clinicians must target residual symptoms.
(J Clin Psychiatry 2013;74[suppl 2]:19-24)
From the Department of Psychiatry and Cellular/Molecular Medicine, University of Ottawa, and Mood Disorders Research, University of Ottawa Institute of Mental Health Research, Ottawa, Ontario, Canada.
This article is derived from the planning teleconference series “Depression: Managing the Full Range of Symptoms to Achieve Lasting Remission,” which was held in May and June 2013 and supported by an educational grant from Takeda Pharmaceuticals International, Inc., US Region and Lundbeck.
Dr Blier has received grant funding from Labopharm, Janssen, Lundbeck, AstraZeneca, Pfizer, Bristol-Myers Squibb, Servier, and Merck; has received speaker honoraria from Eli Lilly, Janssen, Lundbeck, AstraZeneca, Pfizer, Bristol-Myers Squibb, and Merck; and has served as a consultant for Eli Lilly, Janssen, Lundbeck, AstraZeneca, Pfizer, Takeda, Bristol-Myers Squibb, Servier, and Merck.
Corresponding author: Pierre Blier, MD, PhD, Institute of Mental Health Research, 1145 Carling Ave, Ottawa, ON K1Z 7K4, Canada ([email protected]).
doi:10.4088/JCP.12084su1c.04
© Copyright 2013 Physicians Postgraduate Press, Inc.
After responding to antidepressant treatment, many patients with major depressive disorder (MDD) often have lingering symptoms but may no longer meet the criteria for a major depressive episode. According to the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5), a major depressive episode requires the presence of 5 or more symptoms during a consecutive 2-week timeframe.1 Therefore, patients can be in remission and not meet MDD criteria while still experiencing residual, subthreshold symptoms.

- Residual symptoms occur in most patients who have been treated for a major depressive episode and achieved remission.
- Many antidepressants increase 5-HT and trigger a decrease in NE and DA.
- Symptoms that did not respond to the initial antidepressant may respond to a medication with a different mechanism of action.
- Effective strategies for treating common residual symptoms include switching to a different antidepressant and adding adjunctive medications to target the symptom(s).
Patients with unresolved symptoms have more functional and social impairments, more time with subthreshold depressive symptoms, and an increased risk of relapse, chronic course of illness, and suicide attempts than those with depression without residual symptoms.2-5
Therefore, the treatment goal for patients with MDD must be complete remission of symptoms rather than response. Response to antidepressant treatment is typically considered a 50% or more reduction in symptoms, often measured by a standardized rating scale, such as the Hamilton Depression Rating Scale (HDRS) or the Montgomery-Asberg Depression Rating Scale (MADRS).2 Although that reduction may be a statistically significant improvement, many patients with MDD are left with clinically significant symptoms, showing that even the goal of remission does not guarantee that all depressive symptoms are resolved. In the Sequenced Treatment Alternatives to Relieve Depression (STAR*D) trial,6 more than 90% of patients who met remission criteria had at least 1 residual symptom (median of 3 symptoms). Patients who remitted before 6 weeks of treatment had fewer residual symptoms than later remitters at 1 year, which lessened their risk of relapse, emphasizing why complete remission should be the treatment goal.
To help patients achieve asymptomatic remission, clinicians must recognize residual symptoms, understand the connection between monoamine systems and antidepressant treatments, and target common residual symptoms during treatment.
Recognize and Differentiate Residual Symptoms
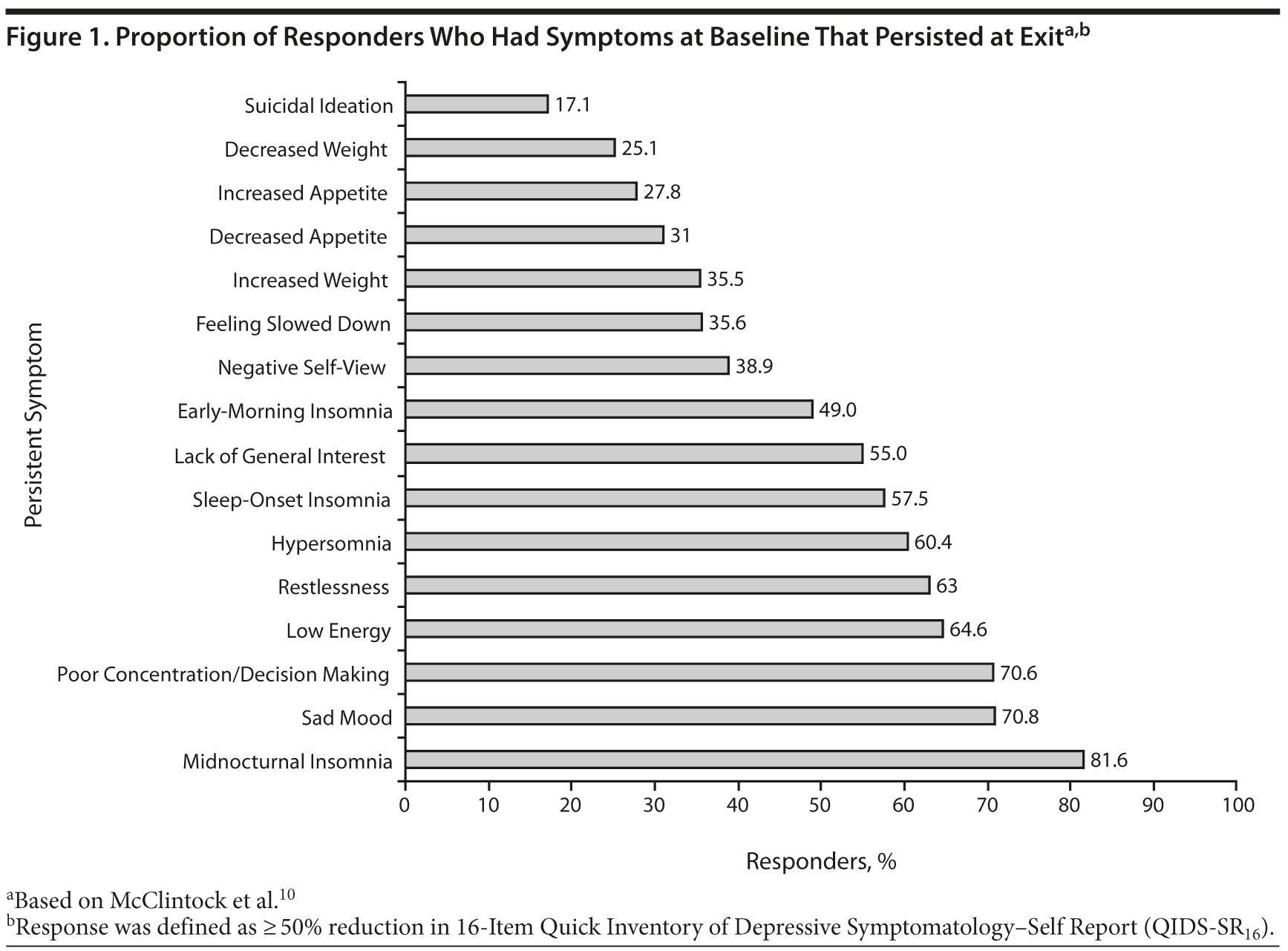
Residual symptoms are often milder versions of typical depressive symptoms7 and fall into the same domains: emotional, physical, and cognitive.1 Of these 3 symptom domains, the cognitive aspects of MDD are probably the most neglected in treatment. Cognitive impairments can include problems in attention, working memory, and executive function (eg, problem solving and task planning).8 For example, difficulty concentrating or thinking has been cited as the third most common symptom for patients with MDD, behind only sleep problems and depressed mood (Figure 1).9,10
Residual symptoms in patients with MDD may be unresolved depressive symptoms or caused by a co-occurring psychiatric or medical condition or adverse events from medication. For example, patients with MDD often have comorbid substance dependence, anxiety disorders, personality disorders, cardiometabolic conditions, and diabetes.11,12 Secondary psychiatric or medical conditions, as well as their treatments, may produce symptoms such as fatigue, cognitive impairment, or sexual dysfunction, which may also be residual symptoms of MDD.7 Additionally, antidepressant-induced side effects may mimic residual symptoms and contribute to treatment discontinuation, causing previously controlled symptoms to emerge.13 Common adverse effects that lead to discontinuation include drowsiness/fatigue, insomnia, sexual dysfunction, gastrointestinal symptoms, anxiety, headache, weight changes, nausea, and rash.14,15
Making a complete baseline assessment of depressive symptoms before initiating treatment can help clinicians differentiate between unresolved residual symptoms, comorbid conditions, and treatment-emergent side effects. If new symptoms arise after treatment is initiated, they are likely to be medication side effects or a result of a comorbidity such as substance use.7 Whatever the cause of persistent or new-onset symptoms in patients being treated for MDD, clinicians must recognize and treat them to improve adherence and both short- and long-term outcomes.
Understand the Connection Between Monoamine Systems and Antidepressant Treatments
The 3 main monoamine systems associated with the pathophysiology of depression and the therapeutic effects of antidepressants are serotonin (5-HT), norepinephrine (NE), and dopamine (DA). These systems control specific aspects related to impulsivity, arousal, and reward effects in the brain, respectively. For example, people with 5-HT hypofunction can be at greater risk for committing suicide because of the 5-HT system’s association with regulating impulsive aggression.16,17
Although each of these 3 neurotransmitter systems has specific associations, significant overlap exists in the signs and symptoms of depression. While an antidepressant may control some symptoms, such as depressed mood, new symptoms like anxiety or fatigue may be triggered by the medication’s mechanism of action on 1 or more systems.
In the human brain, the cell bodies of these monoaminergic neurons are centered in the brainstem or in the mesencephalon, or midbrain. Specifically, 5-HT cell bodies are contained in the dorsal and medial raphe nuclei, NE neurons are located in the locus ceruleus (LC), and DA neurons are in the ventral tegmental area (VTA).
The 5-HT, NE, and DA neurons are interconnected both anatomically and functionally. These cells send reciprocal interactions and projections to common targets. Both the limbic system and the cortex are innervated by these 3 systems. The mechanism of action at various 5-HT, NE, and DA receptors affects the efficacy and tolerability of many depression treatments, including antidepressants such as selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs).
Selective Serotonin Reuptake Inhibitors
An SSRI increases 5-HT transmission, which is sufficient to control depressive symptoms for some patients, but it can cause incomplete response or may not completely resolve all depressive symptoms. This increase in 5-HT inhibits NE neurons and decreases their firing rate, resulting in decreases in DA neuronal activity as well. Rat studies18 have confirmed these effects with sustained use of SSRIs. When rats were administered citalopram or paroxetine for 2, 14, or 21 days, a gradual decrease in spontaneous firing activity of NE neurons in the LC resulted. Rats treated daily for 14 days with citalopram also showed reduction in NE levels in the basolateral nucleus of the amygdala and the LC.19
Another rat study20 demonstrated the effects of SSRIs on DA neuronal firing in the VTA. The SSRI escitalopram, which has greater 5-HT reuptake inhibition potency than citalopram, decreased the firing rate and burst activity of DA neurons with sustained administration in anesthetized rats, but citalopram attenuated only burst activity, not the firing rate. Antagonists at 5-HT2C receptors and agonists at DA receptors may prove to be effective adjunctive therapy because of their complementary mechanism of action with SSRIs.20
The effects of antidepressants on NE and DA receptors relate directly to depressive symptoms like cognitive impairment, anxiety, and fatigue.21 NE and DA in the prefrontal cortex (PFC) regulate working memory and task performance, and too much or too little can lead to cognitive impairment. For example, excessive NE or DA release impairs PFC regulation and may cause anxiety or psychosis, whereas inadequate excitation of these receptors can lead to inattention and drowsiness.21
As more research is done on specific receptor subtypes within the 5-HT, NE, and DA systems, new pharmacologic options should emerge for treating partial responders and nonresponders to antidepressant treatment. For example, administering atypical antipsychotics at subtherapeutic doses can provide helpful supplemental pharmacologic properties without decreasing the D2 transmission that is the basis for antipsychotic response.22 Atypical antipsychotics may be used to block 5-HT2A receptors, which are the pathways used by SSRIs to decrease NE transmission.23 Therefore, using an adjunctive atypical antipsychotic to block the 5-HT2A specific receptor subtype could prevent the potentially negative impact of SSRIs on NE transmission.
Dual-Acting Agents
Agents that target more than 1 monoamine system have the potential to more effectively treat depressive symptoms than single-action agents. For example, SNRIs increase the function of the 5-HT system, even at minimal doses, but higher doses also block NE transporters. Even though the NE firing will be decreased, the transporters at the other end of the neuron will be blocked.24 This action is very different from an SSRI. Whereas an SSRI can increase only 5-HT, an SNRI can increase both 5-HT and NE transmission as long as it is administered at an adequate dose. The ability of SNRIs and norepinephrine reuptake inhibitors (NRIs) to enhance NE neurotransmission may improve residual symptoms of anhedonia and psychomotor retardation.
Norepinephrine reuptake inhibitors also affect DA. Norepinephrine is synthesized from DA, and the only chemical difference between them is that NE has an extra hydroxyl group on the side chain. The PFC has few DA transporters, and NE transporters are probably the primary mechanism for DA reuptake in the PFC.25 Therefore, blocking NE reuptake transporters with an NRI (eg, desipramine, reboxetine, atomoxetine) can elevate levels of DA in the PFC or hippocampus. Reboxetine enhances DA output in the PFC but not in the nucleus accumbens.26 A study27 comparing agents (eg, fluoxetine, clomipramine, imipramine, and desipramine) that increase extracellular concentrations of DA in the PFC but not in the medial nucleus accumbens revealed that stimulating DA transmission in the PFC may have a role in the effectiveness of these drugs independent of an action in the nucleus accumbens.
In addition to SNRI and NRI agents, the antidepressant mirtazapine is a presynaptic α2 antagonist with dual action on NE and 5-HT neurotransmission.28 Mirtazapine has postsynaptic antagonism at 5-HT2 and 5-HT3 receptors, which contributes to its antidepressant effects without typical SSRI side effects.28
Target Common Residual Symptoms
Applying what is known about the effects of drugs on the 5-HT, NE, and DA systems, clinicians can target specific residual symptoms that are the most common or troublesome for patients with MDD.
Fatigue/Sleepiness
Agents that increase NE and DA neurotransmission in relevant pathways may improve symptoms of fatigue, sleepiness, and cognitive impairment. Fatigue may be a symptom of the depression or a side effect of antidepressant treatment.29,30 Although tricyclic antidepressants (TCAs) are more commonly associated with sedating effects, some SSRIs and SNRIs can also cause treatment-emergent fatigue.31 For patients who present with fatigue at baseline, antidepressant medications that are more likely than others to resolve fatigue symptoms can be used as monotherapy. These include bupropion, venlafaxine, sertraline, and fluoxetine.32 As a norepinephrine-dopamine releaser, bupropion has been shown to significantly improve fatigue scores compared with SSRIs and placebo in patients with MDD.33 Patients who experienced remission with bupropion also had fewer residual hypersomnia or fatigue symptoms compared with SSRI remitters.33 SSRI treatment for fatigue comes mainly from studies in special populations.34
Another treatment strategy is to use adjunctive therapy with agents that increase NE and/or DA neurotransmission to target fatigue and sleepiness.31 Adjunctive treatments include psychostimulants (eg, methylphenidate), atomoxetine, or atypical antipsychotics (at lower doses).7,35 Some atypical antipsychotics (eg, olanzapine, risperidone, quetiapine) can alleviate daytime fatigue by improving sleep quality at night.36 Similarly, pramipexole, a presynaptic DA agonist, has been shown to safely improve poor sleep quality in patients with restless leg syndrome and in patients with MDD.37,38
In contrast to the sedative effects of atypical antipsychotics and pramipexole, atomoxetine is an NRI used to treat attention deficit/hyperactivity disorder (ADHD). A study39 of adjunctive atomoxetine in 12 patients with MDD suggests a promising role for treating residual fatigue. Patients taking atomoxetine had significant reductions on the fatigue measure, and 5 of the 12 patients had a 50% or more decrease in fatigue measurement scores. All 12 patients remitted by follow-up.
An adjunctive treatment that increases histamine (H) and DA is the wake-promoting agent modafinil, which can improve fatigue and sleepiness in patients with MDD.32,35 In partial responders to antidepressant therapy, adjunctive modafinil significantly improved sleepiness and residual fatigue.40 Because modafinil (200-400 mg/d) interferes with DA reuptake inhibition and may be associated with dependence and abuse, it should be used with caution in certain populations.41
Insomnia
Another common sleep disturbance that often persists in patients with MDD even after remission is insomnia.42 In an 8-week study43 of residual MDD symptoms in responders to a fixed dose of fluoxetine, 92% of patients with posttreatment insomnia had pretreatment insomnia. The persistence of insomnia throughout treatment indicates a significant problem.
Clinicians may select sedative or sleep-promoting antidepressants or atypical antipsychotics to help patients with MDD and insomnia. Trazodone at low doses (25-150 mg) at bedtime could be used as an adjunctive treatment because it blocks 5-HT2A/C receptors as well as α1 receptors. Blocking H1 receptors may also cause sedating effects, therefore doxepin, a probable H1 and 5-HT2 antagonist, in small doses (eg, 10-20 mg/d) and quetiapine, a probable H1, 5-HT2, and α1 antagonist, (25-50 mg/d) could be used in conjunction with depression treatment to alleviate insomnia. Benzodiazepine hypnotics can improve sleep disturbances, although some are more selective than others. For example, zolpidem augmentation (10 mg/qhs) of an SSRI (ie, fluoxetine, sertraline, or paroxetine) improved sleep quality and duration in patients with persistent insomnia.44
Cognitive Impairment
Although cognitive symptoms usually improve with antidepressant treatment, they may remain unresponsive or emerge as a treatment side effect.29,31 Attention and memory problems are often present in patients with MDD,45 although prevalence rates for residual cognitive impairment have not been established for patients with remitted MDD. Anecdotal augmentation strategies to alleviate cognitive symptoms include cholinesterase inhibitors, bupropion, DA receptor agonists, psychostimulants, and modafinil.29 Like the treatment of fatigue and sleepiness, cognitive impairment should improve with increased NE and DA neurotransmission. For example, augmenting SSRI treatment with the NRI atomoxetine, although not FDA-approved for the treatment of depression, has alleviated concentration and sleep problems in a case study including 3 patients who could not tolerate dual-acting medications.46 By augmenting an SSRI with an NRI, clinicians can also create a dual-acting regimen.
Sexual Dysfunction
Sexual dysfunction is a common side effect of agents that block the 5-HT reuptake transporter.47 This means that many antidepressants are associated with sexual problems related to desire, arousal, and orgasm/ejaculation.48 Many patients are unlikely to spontaneously report sexual dysfunction to their physician, and up to 42% of patients waited for the symptoms to resolve themselves.47 As a result, sexual dysfunction symptoms contribute to patient dissatisfaction and treatment noncompliance.49
Several strategies are available to treat sexual dysfunction, although many lack substantial evidence for efficacy. For example, adding a medication to an antidepressant has yielded mostly negative results with the exceptions of bupropion and sildenafil.50,51
Dosing reductions may be used to eliminate sexual dysfunction, as long as antidepressant efficacy is not sacrificed.48 When titrating down the antidepressant dose, clinicians should always monitor depressive symptoms to ensure that efficacy is maintained and relapse is prevented.
Another strategy is to switch to another antidepressant that does not have a propensity to cause sexual dysfunction. For example, nefazodone, bupropion, and mirtazapine are associated with less sexual dysfunction than SSRIs and venlafaxine.52,53 Of course, any switching strategy requires careful monitoring for discontinuation symptoms or loss of efficacy.49 An optimal strategy is a plateau switch in which the drug inhibiting 5-HT reuptake is continued while the new antidepressant is introduced and titrated up to a therapeutic dose. This approach must not be used with a monoamine oxidase inhibitor (MAOI) because monoamine oxidase should not be inhibited at the same time as blocking the reuptake of 5-HT. The initial antidepressant would have to be completely discontinued, typically with a washout period, before starting an MAOI.
Pain
Patients with MDD often have comorbid pain symptoms. A review54 found that an average of 65% of patients with depression experienced 1 or more pain complaints (eg, headache, back ache, joint pain, or general pain). Among patients whose MDD had remitted after 3 months of treatment, about 10% reported residual pain symptoms (Figure 2).42
When antidepressants increase levels of NE and 5-HT in certain brain areas, they help modulate pain signals.54 This explains the efficacy of certain antidepressants in alleviating pain symptoms. But, if an antidepressant has not reduced pain symptoms, clinicians must consider other options. If the patient was taking an SSRI, then a crossover to a dual reuptake inhibitor (eg, duloxetine, milnacipran, venlafaxine) can be used. The SNRI dose should be increased to the upper range to ensure significant NE reuptake inhibition along with 5-HT reuptake inhibition.55 In a study56 of patients with MDD and painful physical symptoms (N = 86), venlafaxine extended-release (75-225 mg/d) significantly decreased total HDRS17 scores (P < .0001) and overall pain (P < .0001) at every visit. Pain responders demonstrated a better depression remission rate than pain nonresponders (56% vs 20%; P = .0001).
Of a similar structure to venlafaxine, tramadol is a 5-HT and NE reuptake inhibitor and a μ-opoid agonist available in slow-release formulations. Widely prescribed as an analgesic comparable to codeine, tramadol acts only on the μ-opioid receptors, unlike other opioids that act on multiple opioid receptors. Due to its unique mechanism of action, tramadol shows promise for patients with depression and chronic pain.57 Although the abuse incidence for tramadol is low, agonists of μ-receptors still carry a risk for addiction.57,58 Serotonin syndrome is another risk if tramadol is combined with other agents that increase 5-HT activity,57 but it is not necessarily contraindicated with SSRIs if it is uptitrated slowly.
The TCAs amitriptyline or nortriptyline could be used adjunctively at low doses (ie, 25-50 mg/d) for pain management,59 and gabapentin and pregabalin, anticonvulsants that target the α2δ subunit of voltage-gated calcium channels, may be beneficial for pain as well as for comorbid anxiety.60 Gabapentin and pregabalin lower neurotransmission in pain pathways and are FDA-approved to treat pain in a variety of conditions.60
Conclusion
Residual symptoms are a common and problematic issue in patients with MDD who remit with antidepressant treatment. Residual symptoms increase the risk of relapse, suicide, and continued functional impairments in relationships and work. Although the severity of the symptoms may be decreased through treatment, many physical, emotional, and cognitive symptoms can remain. These unresolved symptoms from the major depressive episode should be distinguished from symptoms of comorbid psychiatric or medical conditions and from side effects of treatment.
Most antidepressant medications alleviate depressive symptoms by acting at 5-HT receptors in the brain. Because the 5-HT system is interconnected with the NE and DA systems, medications that increase 5-HT often decrease NE and DA, resulting in certain persistent symptoms. Dual-action medications, like SNRIs, are more effective than SSRIs at controlling some residual symptoms. For patients who cannot tolerate dual-action agents, clinicians can target specific residual symptoms using a variety of adjunctive medications or dosing strategies. Adding low doses of medications with a different mechanism of action can supplement the antidepressant effects by targeting unresolved symptoms such as fatigue, insomnia, sexual dysfunction, or pain.
Drug names: atomoxetine (Strattera), bupropion (Wellbutrin, Aplenzin, and others), citalopram (Celexa and others), clomipramine (Anafranil and others), desipramine (Norpramin and others), doxepin (Zonalon, Silenor, and others), duloxetine (Cymbalta), escitalopram (Lexapro and others), fluoxetine (Prozac and others), gabapentin (Neurontin, Gralise, and others), imipramine (Tofranil and others), methylphenidate (Focalin, Daytrana, and others), milnacipran (Savella), mirtazapine (Remeron and others), modafinil (Provigil and others), nortriptyline (Pamelor, Aventyl, and others), olanzapine (Zyprexa and others), paroxetine (Paxil, Pexeva, and others), pramipexole (Mirapex and others), pregabalin (Lyrica), quetiapine (Seroquel and others), risperidone (Risperdal and others), sertraline (Zoloft and others), sildenafil (Viagra and Revatio), tramadol (Ultram, Ryzolt, and others), trazodone (Oleptro and others), venlafaxine (Effexor and others), zolpidem (Ambien, Zolpimist, and others).
Disclosure of off-label usage: Dr Blier has determined that, to the best of his knowledge, atomoxetine, gabapentin, methylphenidate, milnacipran, modafinil, pramipexole, pregabalin, quetiapine, sildenafil, and trazodone are not approved by the US Food and Drug Administration for the treatment of depression.
References
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed. Arlington, VA: American Psychiatric Association; 2013.
2. Israel JA. The impact of residual symptoms in major depression. Pharmaceuticals. 2010;3(8):2426-2440. doi:10.3390/ph3082426
3. Kennedy N, Paykel ES. Residual symptoms at remission from depression: impact on long-term outcome. J Affect Disord. 2004;80(2-3):135-144. PubMed doi:10.1016/S0165-0327(03)00054-5
4. Kennedy N, Foy K. The impact of residual symptoms on outcome of major depression. Curr Psychiatry Rep. 2005;7(6):441-446. PubMed doi:10.1007/s11920-005-0065-9
5. Paykel ES, Ramana R, Cooper Z, et al. Residual symptoms after partial remission: an important outcome in depression. Psychol Med. 1995;25(6):1171-1180. PubMed doi:10.1017/S0033291700033146
6. Nierenberg AA, Husain MM, Trivedi MH, et al. Residual symptoms after remission of major depressive disorder with citalopram and risk of relapse: a STAR*D report. Psychol Med. 2010;40(1):41-50. PubMed doi:10.1017/S0033291709006011
7. Fava M. Pharmacological approaches to the treatment of residual symptoms. J Psychopharmacol. 2006;20(suppl 3):29-34. PubMed doi:10.1177/1359786806064325
8. Marazziti D, Consoli G, Picchetti M, et al. Cognitive impairment in major depression. Eur J Pharmacol. 2010;626(1):83-86. PubMed doi:10.1016/j.ejphar.2009.08.046
9. Chen LS, Eaton WW, Gallo JJ, et al. Empirical examination of current depression categories in a population-based study: symptoms, course, and risk factors. Am J Psychiatry. 2000;157(4):573-580. PubMed doi:10.1176/appi.ajp.157.4.573
10. McClintock SM, Husain MM, Wisniewski SR, et al. Residual symptoms in depressed outpatients who respond by 50% but do not remit to antidepressant medication. J Clin Psychopharmacol. 2011;31(2):180-186. PubMed doi:10.1097/JCP.0b013e31820ebd2c
11. Hasin DS, Goodwin RD, Stinson FS, et al. Epidemiology of major depressive disorder: results from the National Epidemiologic Survey on Alcoholism and Related Conditions. Arch Gen Psychiatry. 2005;62(10):1097-1106. PubMed doi:10.1001/archpsyc.62.10.1097
12. Egede LE. Major depression in individuals with chronic medical disorders: prevalence, correlates and association with health resource utilization, lost productivity and functional disability. Gen Hosp Psychiatry. 2007;29(5):409-416. PubMed doi:10.1016/j.genhosppsych.2007.06.002
13. Goethe JW, Woolley SB, Cardoni AA, et al. Selective serotonin reuptake inhibitor discontinuation: side effects and other factors that influence medication adherence. J Clin Psychopharmacol. 2007;27(5):451-458. PubMed doi:10.1097/jcp.0b013e31815152a5
14. Prabhakar D, Balon R. How do SSRIs cause sexual dysfunction? Curr Psychiatr. 2010;9(12).
15. Bull SA, Hunkeler EM, Lee JY, et al. Discontinuing or switching selective serotonin-reuptake inhibitors. Ann Pharmacother. 2002;36(4):578-584. PubMed doi:10.1345/aph.1A254
16. Seo D, Patrick CJ, Kennealy PJ. Role of serotonin and dopamine system interactions in the neurobiology of impulsive aggression and its comorbidity with other clinical disorders. Aggress Violent Behav. 2008;13(5):383-395. PubMed doi:10.1016/j.avb.2008.06.003
17. Nordström P, Samuelsson M, Asberg M, et al. CSF 5-HIAA predicts suicide risk after attempted suicide. Suicide Life Threat Behav. 1994;24(1):1-9. PubMed
18. Szabo ST, de Montigny C, Blier P. Progressive attenuation of the firing activity of locus coeruleus noradrenergic neurons by sustained administration of selective serotonin reuptake inhibitors. Int J Neuropsychopharmacol. 2000;3(1):1-11. PubMed doi:10.1017/S1461145700001772
19. Kawahara Y, Kawahara H, Kaneko F, et al. Long-term administration of citalopram reduces basal and stress-induced extracellular noradrenaline levels in rat brain. Psychopharmacology (Berl). 2007;194(1):73-81. PubMed doi:10.1007/s00213-007-0826-8
20. Dremencov E, El Mansari M, Blier P. Effects of sustained serotonin reuptake inhibition on the firing of dopamine neurons in the rat ventral tegmental area. J Psychiatry Neurosci. 2009;34(3):223-229. PubMed
21. Hains AB, Arnsten AFT. Molecular mechanisms of stress-induced prefrontal cortical impairment: implications for mental illness. Learn Mem. 2008;15(8):551-564. PubMed doi:10.1101/lm.921708
22. Blier P. Rational site-directed pharmacotherapy for major depressive disorder [published online ahead of print June 10, 2013]. Int J Neuropsychopharmacol.
23. Szabo ST, Blier P. Effects of serotonin (5-hydroxytryptamine, 5-HT) reuptake inhibition plus 5-HT2A receptor antagonism on the firing activity of norepinephrine neurons. J Pharmacol Exp Ther. 2002;302(3):983-991. PubMed doi:10.1124/jpet.102.033282
24. Trivedi MH, Hollander E, Nutt D, et al. Clinical evidence and potential neurobiological underpinnings of unresolved symptoms of depression. J Clin Psychiatry. 2008;69(2):246-258. PubMed doi:10.4088/JCP.v69n0211
25. Morón JA, Brockington A, Wise RA, et al. Dopamine uptake through the norepinephrine transporter in brain regions with low levels of the dopamine transporter: evidence from knock-out mouse lines. J Neurosci. 2002;22(2):389-395. PubMed
26. Linnér L, Endersz H, Ohman D, et al. Reboxetine modulates the firing pattern of dopamine cells in the ventral tegmental area and selectively increases dopamine availability in the prefrontal cortex. J Pharmacol Exp Ther. 2001;297(2):540-546. PubMed
27. Tanda G, Carboni E, Frau R, et al. Increase of extracellular dopamine in the prefrontal cortex: a trait of drugs with antidepressant potential? Psychopharmacology (Berl). 1994;115(1-2):285-288. PubMed doi:10.1007/BF02244785
28. Fawcett J, Barkin RL. Review of the results from clinical studies on the efficacy, safety and tolerability of mirtazapine for the treatment of patients with major depression. J Affect Disord. 1998;51(3):267-285. PubMed doi:10.1016/S0165-0327(98)00224-9
29. Fava M. Symptoms of fatigue and cognitive/executive dysfunction in major depressive disorder before and after antidepressant treatment. J Clin Psychiatry. 2003;64(suppl 14):30-34. PubMed
30. Pae CU, Lim HK, Han C, et al. Fatigue as a core symptom in major depressive disorder: overview and the role of bupropion. Expert Rev Neurother. 2007;7(10):1251-1263. PubMed doi:10.1586/14737175.7.10.1251
31. Targum SD, Fava M. Fatigue as a residual symptom of depression. Innov Clin Neurosci. 2011;8(10):40-43. PubMed
32. Demyttenaere K, De Fruyt J, Stahl SM. The many faces of fatigue in major depressive disorder. Int J Neuropsychopharmacol. 2005;8(1):93-105. PubMed doi:10.1017/S1461145704004729
33. Papakostas GI, Nutt DJ, Hallett LA, et al. Resolution of sleepiness and fatigue in major depressive disorder: a comparison of bupropion and the selective serotonin reuptake inhibitors. Biol Psychiatry. 2006;60(12):1350-1355. PubMed doi:10.1016/j.biopsych.2006.06.015
34. Marin H, Menza MA. Specific treatment of residual fatigue in depressed patients. Psychiatry (Edgmont). 2004;1(2):12-18. PubMed
35. Stahl SM, Zhang L, Damatarca C, et al. Brain circuits determine destiny in depression: a novel approach to the psychopharmacology of wakefulness, fatigue, and executive dysfunction in major depressive disorder. J Clin Psychiatry. 2003;64(suppl 14):6-17. PubMed
36. Yamashita H, Mori K, Nagao M, et al. Effects of changing from typical to atypical antipsychotic drugs on subjective sleep quality in patients with schizophrenia in a Japanese population. J Clin Psychiatry. 2004;65(11):1525-1530. PubMed doi:10.4088/JCP.v65n1114
37. Corrigan MH, Denahan AQ, Wright CE, et al. Comparison of pramipexole, fluoxetine, and placebo in patients with major depression. Depress Anxiety. 2000;11(2):58-65. PubMed doi:10.1002/(SICI)1520-6394(2000)11:2<58::AID-DA2>3.0.CO;2-H
38. Aiken CB. Pramipexole in psychiatry: a systematic review of the literature. J Clin Psychiatry. 2007;68(8):1230-1236. PubMed doi:10.4088/JCP.v68n0810
39. Papakostas GI, Petersen TJ, Burns AM, et al. Adjunctive atomoxetine for residual fatigue in major depressive disorder. J Psychiatr Res. 2006;40(4):370-373. PubMed doi:10.1016/j.jpsychires.2005.04.013
40. Fava M, Thase ME, DeBattista C, et al. Modafinil augmentation of selective serotonin reuptake inhibitor therapy in MDD partial responders with persistent fatigue and sleepiness. Ann Clin Psychiatry. 2007;19(3):153-159. PubMed doi:10.1080/10401230701464858
41. Volkow ND, Fowler JS, Logan J, et al. Effects of modafinil on dopamine and dopamine transporters in the male human brain: clinical implications. JAMA. 2009;301(11):1148-1154. PubMed doi:10.1001/jama.2009.351
42. Romera I, Pérez V, Ciudad A, et al. Residual symptoms and functioning in depression, does the type of residual symptom matter? a post-hoc analysis. BMC Psychiatry. 2013;13:51. PubMed doi:10.1186/1471-244X-13-51
43. Nierenberg AA, Keefe BR, Leslie VC, et al. Residual symptoms in depressed patients who respond acutely to fluoxetine. J Clin Psychiatry. 1999;60(4):221-225. PubMed doi:10.4088/JCP.v60n0403
44. Asnis GM, Chakraburtty A, DuBoff EA, et al. Zolpidem for persistent insomnia in SSRI-treated depressed patients. J Clin Psychiatry. 1999;60(10):668-676. PubMed doi:10.4088/JCP.v60n1005
45. Goeldner C, Ballard TM, Knoflach F, et al. Cognitive impairment in major depression and the mGlu2 receptor as a therapeutic target. Neuropharmacology. 2013;64:337-346. PubMed doi:10.1016/j.neuropharm.2012.08.001
46. Berigan TR. Atomoxetine used adjunctively with selective serotonin reuptake inhibitors to treat depression. Prim Care Companion J Clin Psychiatry. 2004;6(2):93-94. PubMed doi:10.4088/PCC.v06n0209
47. Clayton AH, Montejo AL. Major depressive disorder, antidepressants, and sexual dysfunction. J Clin Psychiatry. 2006;67(suppl 6):33-37. PubMed
48. Baldwin DS, Palazzo MC, Masdrakis VG. Reduced treatment-emergent sexual dysfunction as a potential target in the development of new antidepressants. Depress Res Treat. 2013;2013:256841.
49. Labbate LA, Croft HA, Oleshansky MA. Antidepressant-related erectile dysfunction: management via avoidance, switching antidepressants, antidotes, and adaptation. J Clin Psychiatry. 2003;64(suppl 10):11-19. PubMed
50. Clayton AH, Warnock JK, Kornstein SG, et al. A placebo-controlled trial of bupropion SR as an antidote for selective serotonin reuptake inhibitor-induced sexual dysfunction. J Clin Psychiatry. 2004;65(1):62-67. PubMed doi:10.4088/JCP.v65n0110
51. Nurnberg HG, Hensley PL. Sildenafil citrate for the management of antidepressant-associated erectile dysfunction. J Clin Psychiatry. 2003;64(suppl 10):20-25. PubMed
52. Montejo AL, Llorca G, Izquierdo JA, et al; Spanish Working Group for the Study of Psychotropic-Related Sexual Dysfunction. Incidence of sexual dysfunction associated with antidepressant agents: a prospective multicenter study of 1022 outpatients. J Clin Psychiatry. 2001;62(suppl 3):10-21. PubMed
53. Clayton AH, Pradko JF, Croft HA, et al. Prevalence of sexual dysfunction among newer antidepressants. J Clin Psychiatry. 2002;63(4):357-366. PubMed doi:10.4088/JCP.v63n0414
54. Bair MJ, Robinson RL, Katon W, et al. Depression and pain comorbidity: a literature review. Arch Intern Med. 2003;163(20):2433-2445. PubMed doi:10.1001/archinte.163.20.2433
55. Trivedi MH. The link between depression and physical symptoms. Prim Care Companion J Clin Psychiatry. 2004;6(suppl 1):12-16. PubMed
56. Huang X, Li C, Luo YL, et al. Efficacy of venlafaxine extended-release monotherapy for first-episode depression with painful physical symptoms. Neuroreport. 2013;24(7):364-369. PubMed doi:10.1097/WNR.0b013e3283601a3e
57. Reeves RR, Burke RS. Tramadol: basic pharmacology and emerging concepts. Drugs Today (Barc). 2008;44(11):827-836. PubMed doi:10.1358/dot.2008.44.11.1308916
58. Barber J. Examining the use of tramadol hydrochloride as an antidepressant. Exp Clin Psychopharmacol. 2011;19(2):123-130. PubMed doi:10.1037/a0022721
59. Maizels M, McCarberg B. Antidepressants and antiepileptic drugs for chronic non-cancer pain. Am Fam Physician. 2005;71(3):483-490. PubMed
60. Stahl SM. Anticonvulsants and the relief of chronic pain: pregabalin and gabapentin as α2δ ligands at voltage-gated calcium channels. J Clin Psychiatry. 2004;65(5):596-597. PubMed doi:10.4088/JCP.v65n0501