Theta-Burst Repetitive Transcranial Magnetic Stimulation for Treatment-Resistant Obsessive-Compulsive Disorder With Concomitant Depression
To the Editor: Repetitive transcranial magnetic stimulation (rTMS) is a potential therapy for major depressive disorder and obsessive-compulsive disorder that acts by modulating cortical activities.1,2 Recently, Huang et al3 have developed a novel paradigm of theta-burst stimulation (TBS)—low-intensity bursts of rTMS at 50 Hz—as a safer, more controllable, more consistent, and longer-lasting rTMS. The continuous TBS (cTBS) inhibits, while the intermittent TBS (iTBS) enhances, excitability in the motor cortex.3 To the best of our knowledge, TBS has yet to be applied to the treatment of psychiatric disorders.
Here, we report a patient with treatment-resistant obsessive-compulsive disorder and concomitant major depressive disorder who responded dramatically to augmentation treatment with TBS. The improvement in obsessive-compulsive disorder symptoms was associated with a marked reduction in regional blood oxygen level-dependent (BOLD) activity in right dorsolateral prefrontal cortex in functional magnetic resonance imaging (fMRI) acquired during symptom provocation. The results suggest that TBS could be a potential treatment for treatment-resistant obsessive-compulsive disorder and major depressive disorder.
Case report. Mr A, a 33-year-old man, presented with obsessive symptoms of pathological doubt and compulsive behaviors that he had not experienced since the age of 17.
At the time of the previous symptoms, he experienced a full remission of the obsessive symptoms with clomipramine 75 mg/d. Five years ago, having been in stable condition with good psychosocial and occupational function for more than 10 years, he discontinued his medication. Consequently, in October 2005, he experienced a recurrence of severe obsessive symptoms, including intrusive thoughts about the color of everything he saw. Moreover, he experienced a major depressive episode with suicidal attempts via drug overdose and wrist-slitting.
Immediately, he was prescribed clomipramine 75 mg/d for 8 weeks; however, it did not appear to be as effective as before. Furthermore, the subsequent treatments with amitriptyline (125 mg/d for 16 weeks) and fluoxetine (60 mg/d for 8 weeks) were also ineffective. With a DSM-IV diagnosis of treatment-resistant obsessive-compulsive disorder and concomitant major depressive disorder, he was admitted to our acute psychiatric ward for further treatment.
On admission, physical and neurologic examinations revealed no abnormal findings. He had no abnormal hematologic, liver, or renal function except hyperuricemia (uric acid: 11 mg/dL) and dyslipidemia (cholesterol/triglycerides: 241/438 mg/dL). Results of a routine electroencephalography examination were also normal. His personal and family histories were negative for epilepsy. During hospitalization, he responded poorly to combination therapy with 10 mg/d olanzapine and 60 mg/d fluoxetine for more than 8 weeks. Alternative treatment with TBS therapy was suggested to the patient and his family by the consultant psychiatrists and neurologists. He signed an informed consent statement after gaining a full understanding of the experimental procedures and their risk.
In the first stage, Mr A underwent 10 sessions of cTBS over his right dorsolateral prefrontal cortex for his obsessive-compulsive disorder symptoms. The first 6 sessions was performed twice per week, and the last 4 sessions was performed once per day. Stimuli were delivered by means of a figure 8-shaped coil placed over the frontal scalp area overlying the dorsolateral prefrontal cortex. The basic pattern of TBS consists of 3 pulses at 50 Hz with a 200-ms interval. For the cTBS paradigm, 4 continuous sessions of 20-s TBS trains were given (1,200 pulses). A total of 10 sessions (12,000 pulses at an intensity of 80% active motor threshold) of cTBS were given within a 4-week period.
One week after finishing the first stage, Mr A received the second stage of experimental treatment, 10 sessions of iTBS over his left dorsolateral prefrontal cortex for his depressive symptoms. In this iTBS course, a 2-second train of TBS was used and repeated every 10 seconds for a total of 400 seconds (1,200 pulses). Every session was given at a 2-day interval. A total of 10 sessions (12,000 pulses at an intensity of 80% active motor threshold) of iTBS were given within a 2-week period.
No adverse event was noted during the whole therapeutic course. The patient was rated at baseline, 24 hours after 10 sessions of cTBS, and 24 hours after 10 sessions of iTBS. There was a marked reduction in obsessive-compulsive disorder symptom severity after 10 sessions of cTBS treatment. The patient’s Yale-Brown Obsessive Compulsive Scale (YBOCS)4 score decreased from 19 to 8. It is interesting to note that a partial remission in depression was also achieved after the first cTBS treatment, with scores on the 24-item Hamilton Depression Rating Scale (HDRS)5 reduced from 49 to 29 after the first stage. After the second stage of 10-session iTBS treatment, he had a further improvement in depressive symptoms (from 29 to 15 in HDRS score) with no worsening in obsessive-compulsive disorder symptoms (from 8 to 9 in YBOCS score). Mr A was followed up for the next 3 months, during which he was in stable psychiatric condition, although HDRS and YBOCS ratings were not measured.
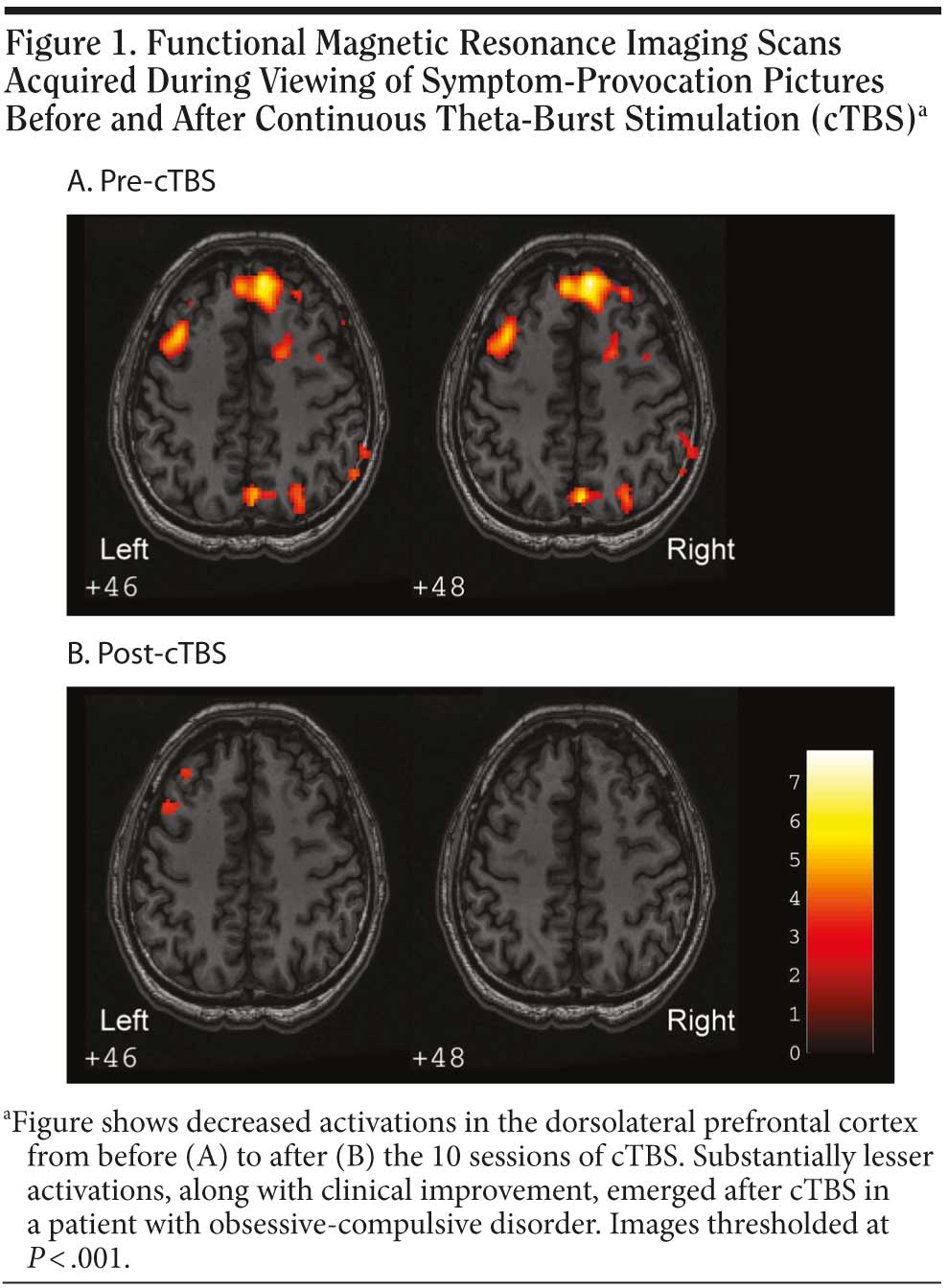
Functional magnetic resonance imaging was performed before and after 10 sessions of cTBS for evaluation of regional BOLD changes. The fMRI data were acquired during viewing of symptom-provocation pictures. A substantial reduction in BOLD activity in the right dorsolateral prefrontal cortex region was observed when stimuli were assessed after the cTBS treatment (Figure 1).
To the best of our knowledge, this is the first report of successful treatment with TBS in a patient with treatment-resistant obsessive-compulsive disorder and major depressive disorder. The mechanisms of effectiveness of TBS are based on the evidence that obsessive-compulsive disorder is often associated with an overactivity of the prefrontal cortex, especially of the right side.6,7 Thus, a suppression of excitatory circuits in the right prefrontal cortex by cTBS might improve obsessive-compulsive disorder symptoms. On the other hand, depression is often associated with hypoactivity over the left dorsolateral prefrontal cortex.1 Hence, a stimulation of the left dorsolateral prefrontal cortex by iTBS may improve depressive symptoms. The findings from fMRI studies support the clinical results. Further double-blind controlled studies on the therapeutic application of TBS in obsessive-compulsive disorder and major depressive disorder are warranted.
References
1. O’ Reardon JP, Solvason HB, Janicak PG, et al. Efficacy and safety of transcranial magnetic stimulation in the acute treatment of major depression: a multisite randomized controlled trial. Biol Psychiatry. 2007;62(11):1208-1216. PubMed doi:10.1016/j.biopsych.2007.01.018
2. Greenberg BD, George MS, Martin JD, et al. Effect of prefrontal repetitive transcranial magnetic stimulation in obsessive-compulsive disorder: a preliminary study. Am J Psychiatry. 1997;154(6):867-869. PubMed
3. Huang YZ, Edwards MJ, Rounis E, et al. Theta burst stimulation of the human motor cortex. Neuron. 2005;45(2):201-206. PubMed doi:10.1016/j.neuron.2004.12.033
4. Goodman WK, Price LH, Rasmussen SA, et al. The Yale-Brown Obsessive Compulsive Scale (Y-BOCS), I: development, use, and reliability. Arch Gen Psychiatry. 1989;46(11):1006-1011. PubMed
5. Hamilton MA. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23(1):56-62. PubMed doi:10.1136/jnnp.23.1.56
6. Rauch SL, Wedig MM, Wright CI, et al. Functional magnetic resonance imaging study of regional brain activation during implicit sequence learning in obsessive-compulsive disorder. Biol Psychiatry. 2007;61(3):330-336. PubMed doi:10.1016/j.biopsych.2005.12.012
7. Saxena S, Rauch SL. Functional neuroimaging and the neuroanatomy of obsessive-compulsive disorder. Psychiatr Clin North Am. 2000;23(3):563-586. PubMed doi:10.1016/S0193-953X(05)70181-7
Author affiliations: Department of Psychiatry, Taichung Hospital, Department of Health, Executive Yuan, Taiwan (Dr Wu); Department of Psychiatry and Mind-Body Interface Laboratory (MBI-Laboratory) (Dr Su), Department of Neurology (Drs Tsai and Lu), Department of Radiology (Mr Chen and Dr Shen), and Graduate Institute of Neural and Cognitive Sciences (Drs Tsai, Shen, and Su), China Medical University & Hospital, Taichung, Taiwan; and Institute of Psychiatry, King’s College London, United Kingdom (Dr Su). Potential conflicts of interest: None reported. Funding/support: The work was supported by the following grants to Dr Su: the NARSAD Young Investigator Award (United States) and the NSC 98-2627-B-039-003 and NSC 98-2628-B-039-020-MY3 from the National Science Council in Taiwan. Acknowledgment: We thank Jenny Peilun Liu for English editing and critical review. Ms Liu reports no financial or other relationship relevant to this letter.
doi:10.4088/JCP.09l05426blu
© Copyright 2010 Physicians Postgraduate Press, Inc.