Patient Outcomes Within Schizophrenia Treatment: A Look at the Role of Long-Acting Injectable Antipsychotics
Compliance is a critical issue across all chronic conditions, including schizophrenia. Compliance is not an all-or-nothing phenomenon, with a continuum from taking all medications as prescribed to partial compliance to complete noncompliance. Partial compliance is a serious problem that may result in abrupt dose changes leading to unanticipated adverse effects and can demoralize the patient. Further, there is a nearly 5-fold increase in the risk of relapse in first-episode patients when antipsychotic drug treatment is discontinued. Taken together, these data indicate that it is critical to ensure continuous delivery of antipsychotic treatment. Atypical antipsychotic medications were expected to result in better adherence, primarily because of the anticipated improved efficacy and safety profile. However, atypical agents have poor adherence, irrespective of the type of atypical medication, making it difficult to predict which patients are taking their oral medications. Long-acting injectable (LAI) agents may minimize the fluctuations in peak and overall plasma levels compared with oral agents, indicating they may allow more consistent and predictable administration. Based on clinical experience in my practice, several important observations regarding LAI use in patients with schizophrenia have been identified. First, there are potential advantages to using LAIs, including assistance in understanding reasons for poor response, the possibility of eliminating daily pill ingestion, and the elimination of the abrupt loss of medication coverage. There are also several potential obstacles to the use of LAIs, including a lack of infrastructure for the delivery and disposal of syringes and the ease of use with the oral agents. Several strategies can be used to increase patient willingness to initiate and continue LAI therapy. Strategies to improve acceptance involve presenting the option with enthusiasm, ensuring proper goal setting, educating the patient that this treatment is not equivalent to emergency injections, and repeatedly recommending LAI therapy. Adherence can be improved by ensuring samples are available in the clinical setting at all times.
(J Clin Psychiatry 2014;75[suppl 2]:30-33)
Corresponding author: Rimal B. Bera, MD, Department of Psychiatry and Human Behavior, University of California, Irvine, 101 City Dr, Orange, CA 92868 ([email protected]).
doi:10.4088/JCP.13065su1c.07
© Copyright 2014 Physicians Postgraduate Press, Inc.
Although there are numerous treatment options for schizophrenia patients, outcomes are not always optimal and could be improved. The goal of optimizing treatments in schizophrenia has been beset with many challenges. However, recent new delivery methods may be able to help improve these outcomes.
A major advance in the treatment of schizophrenia and other psychoses was the introduction of long-acting depot formulations of antipsychotics.1 Depot agents have a number of advantages over oral medications, including avoidance of first-pass metabolism and assured medication delivery.2,3 An additional benefit is that the guesswork is eliminated for the treatment team regarding the date of onset of nonadherence. The steady rate of drug delivery of depot agents may also reduce the variation between peak and trough plasma drug levels.2 The consistency that is characteristic of the depot formulation may help improve response to treatment, and the lower peak plasma drug levels with depot compared with oral antipsychotics may potentially reduce the incidence of adverse events.2-6
The present article examines some of the current data on improving outcomes, specifically looking at the role of long-acting injectable (LAI) treatment for schizophrenia patients. Additionally, the important issue of compliance is discussed in the context of treatment outcomes. Further, the potential advantages and disadvantages of LAI antipsychotics are examined. Finally, research involving LAIs is reviewed.
What Is Compliance?
The word compliance is derived from the Latin word complire, meaning to fill up and hence to complete an action, transaction, or process. In modern English, it has come to mean acting in accordance with advice, which in this context refers to when a patient follows the treatment recommendation of his doctor. Conversely, noncompliance refers to the difference between what is recommended and what is actually taken. Further, when noncompliance occurs, it may impact patient outcomes. It is critical to remember that compliance is not an all-or-nothing phenomenon. Patients with illnesses that require regular dosing of medications may vary in compliance in a spectrum from taking all medications as prescribed to partial compliance to complete noncompliance.7
Partial compliance is common not only among patients with schizophrenia,8 but also in patients with other diseases requiring long-term treatment.9 Partial compliance is a serious problem that is often underappreciated by physicians. It is virtually impossible to determine the threshold at which erratic consumption of medication will trigger an exacerbation. Unprescribed and abrupt changes in dose can lead to unanticipated and unexplained adverse effects. For the patient with schizophrenia, the potential consequences of poor compliance can include relapse and hospitalization, demoralization, loss of confidence, loss of job, family discord, and finally danger to self or others.10 The risk of relapse in first-episode patients has been shown to increase almost 5-fold (risk ratio = 4.89) when antipsychotic drug treatment was discontinued.11 Thus, there is a critical need to ensure continuous delivery of antipsychotic treatment for schizophrenia patients with a minimum of treatment interruptions.
Another key aspect to discuss is the subtle connotation of the term compliance. The definition of compliance is more closely aligned to obedience than it is to efficacy. Further, some believe that this word suggests a paternalistic attitude toward the patient on the prescriber’s part.12 This situation creates a potential power imbalance in which the clinician has power over the patient, and the patient may feel powerless or overwhelmed. In this context, noncompliance demonstrates a form of disobedience, is socially undesirable, and also does not always relate to efficacy.
Moving From Compliance to Adherence to Alliance: Did the Advent of the Atypical Antipsychotics Introduce Change?
Given the potential connotation of the word compliance, the term adherence has been substituted. The word adherence comes from the Latin word adhaerere, meaning "to cling to" or "to remain constant." The term currently refers to being persistent in a practice or tenet, with steady observance or maintenance. From a medical standpoint, adherence means that patients must constantly maintain their therapeutic regimen.
Regardless of the word used to describe adhering to a treatment regimen, it is clear that maintaining the prescribed medication regimen is critical for positive treatment outcome. One current notion about how to improve outcomes through increasing adherence involves changing the concept of compliance to adherence and thus shifting the relationship from one that is paternalistic to one that is more of an alliance. This notion involves developing a therapeutic alliance with both the patient and his or her family. Within the context of improving the relationship, it is also important to destigmatize missing or stopping medications. Virtually all patients have at some point stopped taking medications or demonstrated partial adherence. All of these factors combine to suggest that clinicians should move away from the obedience model and move toward a collaborative model with the patient.
The arrival of the atypical antipsychotic medications renewed hope that there would be more acceptance for injectable medications and thus better adherence. The hope when these drugs were launched was that the reduced neurologic side effect burden would boost adherence rates. The introduction of the atypical antipsychotics did result in a substantial change in the paradigm for treatment of schizophrenia patients. The difference in side effect burden of the atypical agents over conventional medications has been well documented.13
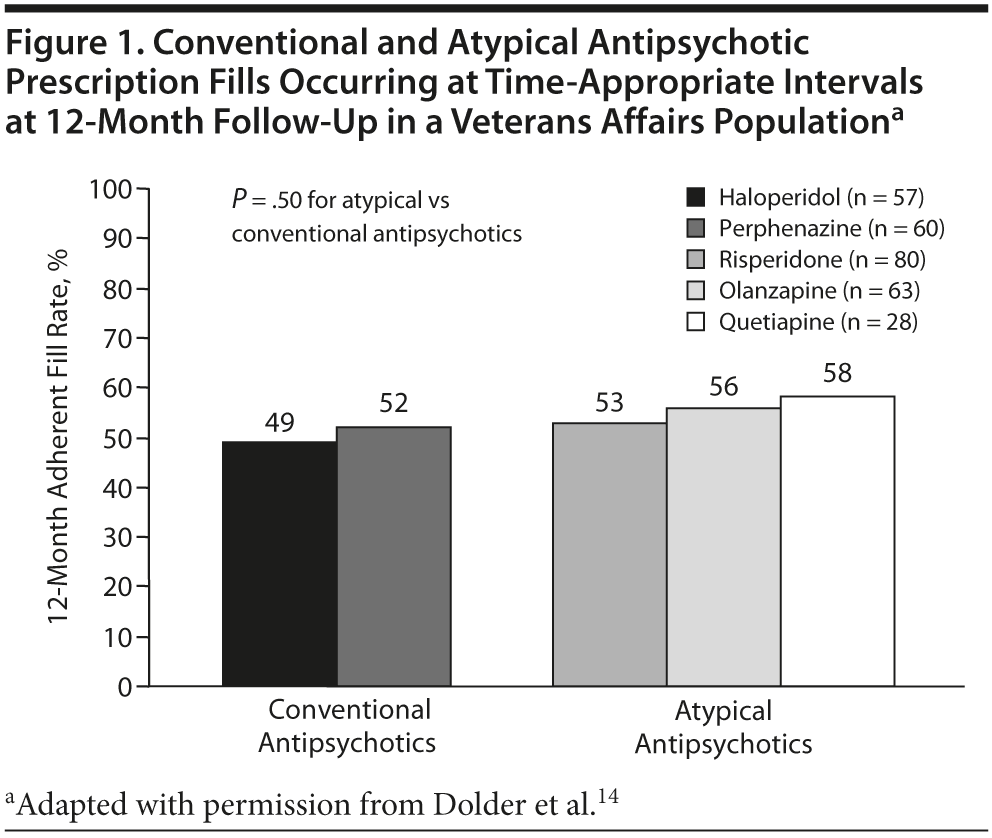
However, despite these improvements in other areas of treatment, poor compliance is common even with the atypical agents. One study14 demonstrated that medication adherence was slightly better with atypical antipsychotics in schizophrenia patients, but poor adherence was considerable even among those receiving these newer agents. According to 12-month pharmacy refill records, 117 outpatients receiving the older agents were without medication for approximately 7 days per month, while 171 patients receiving the atypical agents were without medication for approximately 4 days per month. The difference in mean percentage compliance rates between atypical and typical antipsychotics was significant at 6 months (57% vs 50%, respectively; P = .05) but not at 12 months (55% vs 50%; P = .11; Figure 1). No significant differences were found in adherence between individual medications (P = .50). Regardless of the type of medication, it was clear that it was difficult to predict which patients were taking their oral medications.
CLINICAL INSIGHTS: MY PERSONAL EXPERIENCES USING LAI ANTIPSYCHOTICS IN PATIENTS WITH SCHIZOPHRENIA
Clinical Observations Regarding the Potential Advantages of LAI Antipsychotics
Given the adherence issues seen with both oral atypical and conventional antipsychotics, there exists room for improvement in treatment options for schizophrenia patients. LAI agents bring the advantage of long-acting formulations to the treatment armamentarium. Pharmacokinetic studies have shown that fluctuations in plasma drug levels and peak plasma drug levels are lower with LAI compared with oral agents,15 indicating that these new formulations may provide more consistent and predictable plasma drug levels.
Based on clinical observation, there are several potential advantages in using LAIs: (1) dosage deviations are reduced; (2) guessing about adherence status is eliminated; (3) a start date of adherence/nonadherence can be documented; (4) they aid in disentangling reasons for poor response to medication; (5) the possible need for the patient to remember to take a pill daily is eliminated; (6) prescribers can avoid first-pass metabolism; (7) plasma levels are predictable and stable; (8) abrupt loss of efficacy from a missed dose is eliminated; and (9) possible patient preference.
Clinical Observations Regarding the Potential Obstacles to LAI Antipsychotics
There are, however, several potential obstacles to the use of LAIs that have been observed in our clinical experience. The first concerns the infrastructure in which patients receive treatment. A large number of schizophrenia patients are still treated in private practice offices. These clinicians may not prefer giving injections. In addition to this personal preference, many clinicians lack the logistics necessary to deliver these medications, such as a refrigerator or a place to store and discard needles. This just highlights that many schizophrenia patients are receiving treatment in settings that may be problematic for LAI use.
Another potential obstacle involves the possibility of overburdened public agencies.16 Given the financial and time constraints of these facilities, it is usually much easier to simply prescribe an oral agent for most patients. Further, the fact that these patients and their families will have to come to the clinic every 2 to 4 weeks may be inconvenient to both the staff and the families. Finally, in the United States, some antishot sentiments still exist, particularly in comparison to European counterparts.17
Clinical Observations of Strategies for Switching Patients From an Oral Antipsychotic Medication to a LAI
There are several strategies we have used in our clinical practice to assist in switching patients from oral antipsychotic medications to LAIs. However, almost all of these strategies are derived from studies that were part of the clinical trials leading to approval of LAIs, which do not always translate to front-line clinical settings.
One particular issue related to switching patients involves the potential problems with rapidly switching from oral medications to LAIs. Based on clinical experience, the worst result is that the patient will initiate treatment and report that symptoms are worsening following the initial administration of LAIs. This worsening will most likely be associated with the new medication in the minds of both the patient and caretaker. This scenario suggests that a more gradual titration may be necessary for some patients, potentially across weeks or months, to decrease the likelihood of symptom worsening.
Currently Ongoing Studies With Unpublished Data Examining LAIs as a Treatment Option in Our Laboratory
In our research laboratory, there are several ongoing studies. In one of these studies, polypharmacy was reduced after 1 year in those receiving LAI treatment compared with those taking oral medication (data not shown). This reduction in concurrent medication use was not limited to antipsychotics but also resulted in reduced overall medication use, including other psychotropic medications. In addition to the reduction in medication usage, this finding suggests that there is potential for lower overall drug costs. The possibility exists that those taking LAIs for an extended time period may maintain positive outcomes while also reducing drug use, resulting in a cost savings down the road.
Another area of study in our laboratory concerns identifying where the inflection point in adherence is with LAIs. The tipping point for achieving long-term adherence does not appear to be after the first or even second injection. Based on our research, the third injection appears to be the point at which adherence becomes more of a habit or routine instead of requiring effort. Our clinical experience suggests that the potential mechanism for this inflection point likely lies in the gradual reduction in the patient’s concerns over pain and/or side effects. These unpublished data from our laboratory indicate that extra attention should be paid to the patient by the clinician and staff during this period prior to the third injection.
Clinical Observations of Strategies for Presenting LAIs as a Treatment Option
On the basis of clinical experience, several strategies have been identified that increase a patient’s willingness to initiate therapy and adherence when recommending LAIs. The first strategy designed to improve patient acceptance originates with the presenter of the treatment. The degree of enthusiasm exhibited by the presenter appears to enhance the patient’s desire to agree to treatment. The next useful acceptance strategy is to ensure proper documentation of the attainable goals for the patient. These include such things as helping the patient to live independently and be capable of working, taking less medication, and developing relationships. Another strategy employed is encouraging the patient to try the injection for 1 to 2 times, particularly at the beginning of treatment. Another strategy focuses on reassuring the patient that LAI treatment is not the same as a short-acting emergency intramuscular dose. Many patients have undergone this often traumatic experience and will not differentiate these 2 treatments on their own. This suggests that education is vital to assure them that LAI treatment is not an emergency situation, but a potential treatment to improve outcome. The last strategy suggested involves repeatedly presenting LAI as an option at future appointments even after the patient declines the initial recommendation.
Strategies have also been developed to improve adherence in our clinical practice. One important strategy involves ensuring that samples are available for the patients, especially at the beginning of treatment. It is important to have them available so that LAI can be administered immediately upon consent as opposed to having to wait for delivery from the pharmacy. This ensures that there is a minimal amount of time for patients to reconsider their decision to consent to LAI administration.
CONCLUSIONS
Compliance is an important consideration for all chronic conditions, including schizophrenia. Partial compliance represents a serious problem that may result in abrupt dose changes leading to adverse effects and diminished efficacy, suggesting that it is important to ensure continuous delivery of schizophrenia medication. LAI agents have particular promise in minimizing the fluctuations in plasma drug levels associated with oral agents. Although these agents have a variety of advantages such as reducing dosage deviations, there may be obstacles to the use of LAIs, including a lack of infrastructure in clinical settings. Based on my clinical experience, several strategies have been developed that may increase the patient’s willingness to both initiate and continue LAI therapy. These include ensuring clinician enthusiasm about the treatment, minimizing anxiety surrounding injections, and informing patients that LAI therapy is not equivalent to emergency injections.
Author affiliation: Department of Psychiatry and Human Behavior, University of California, Irvine.
Potential conflicts of interest: Dr Bera received a fee for service from Otsuka America Pharmaceutical, Inc., and Lundbeck for participation in the meeting and preparation of this manuscript; has received grant/research support from Novartis and Otsuka America Pharmaceutical, Inc.; and has been on the speakers/advisory boards of Novartis, Otsuka America Pharmaceutical, Inc., AstraZeneca, Forest, Sunovion, and Teva.
Acknowledgments: This article is derived from a roundtable meeting titled "Understanding the lifetime course of schizophrenia: a longitudinal perspective on neurobiology to promote better outcomes and recovery," which was held October 15, 2013. Editorial assistance in developing the manuscript was provided by Healthcare Global Village.
Funding/support: The meeting, manuscript preparation, and dissemination of the supplement were sponsored by Otsuka America Pharmaceutical, Inc., and Lundbeck.
REFERENCES
1. Morrissette DA, Stahl SM. Optimizing outcomes in schizophrenia: long-acting depots and long-term treatment. CNS Spectr. 2012;17(suppl 1):10-21. PubMed doi:10.1017/S1092852912000739
2. Barnes TR, Curson DA. Long-term depot antipsychotics: a risk-benefit assessment. Drug Saf. 1994;10(6):464-479. PubMed doi:10.2165/00002018-199410060-00005
3. Nayak RK, Doose DR, Nair NP. The bioavailability and pharmacokinetics of oral and depot intramuscular haloperidol in schizophrenic patients. J Clin Pharmacol. 1987;27(2):144-150. PubMed doi:10.1002/j.1552-4604.1987.tb02175.x
4. Kane JM, Davis JM, Schooler N, et al. A multidose study of haloperidol decanoate in the maintenance treatment of schizophrenia. Am J Psychiatry. 2002;159(4):554-560. PubMed doi:10.1176/appi.ajp.159.4.554
5. Midha KK, Hubbard JW, Marder SR, et al. Impact of clinical pharmacokinetics on neuroleptic therapy in patients with schizophrenia. J Psychiatry Neurosci. 1994;19(4):254-264. PubMed
6. Marder SR, Midha KK, Van Putten T, et al. Plasma levels of fluphenazine in patients receiving fluphenazine decanoate: relationship to clinical response. Br J Psychiatry. 1991;158(5):658-665. PubMed doi:10.1192/bjp.158.5.658
7. Kane JM, Aguglia E, Altamura AC, et al. Guidelines for depot antipsychotic treatment in schizophrenia. European Neuropsychopharmacology Consensus Conference in Siena, Italy. Eur Neuropsychopharmacol. 1998;8(1):55-66. PubMed doi:10.1016/S0924-977X(97)00045-X
8. Cramer JA, Rosenheck R. Compliance with medication regimens for mental and physical disorders. Psychiatr Serv. 1998;49(2):196-201. PubMed
9. Costa FV. Compliance with antihypertensive treatment. Clin Exp Hypertens. 1996;18(3-4):463-472. PubMed doi:10.3109/10641969609088977
10. Keers R, Ullrich S, Destavola BL, et al. Association of violence with emergence of persecutory delusions in untreated schizophrenia. Am J Psychiatry. 2014;171(3):332-339. PubMed doi:10.1176/appi.ajp.2013.13010134
11. Robinson D, Woerner MG, Alvir JM, et al. Predictors of relapse following response from a first episode of schizophrenia or schizoaffective disorder. Arch Gen Psychiatry. 1999;56(3):241-247. PubMed doi:10.1001/archpsyc.56.3.241
12. Jaeger M, Rossler W. Attitudes towards long-acting depot antipsychotics: a survey of patients, relatives and psychiatrists. Psychiatry Res. 2010;175(1-2):58-62. PubMed doi:10.1016/j.psychres.2008.11.003
13. Markowitz JS, Brown CS, Moore TR. Atypical antipsychotics, part I: pharmacology, pharmacokinetics, and efficacy. Ann Pharmacother. 1999;33(1):73-85. PubMed doi:10.1345/aph.17215
14. Dolder CR, Lacro JP, Dunn LB, et al. Antipsychotic medication adherence: is there a difference between typical and atypical agents? Am J Psychiatry. 2002;159(1):103-108. PubMed doi:10.1176/appi.ajp.159.1.103
15. Eerdekens M, Resmussen M, Vermeulen A, et al. Kinetics and safety of a novel risperidone depot formulation. Biol Psychiatry. 2000;47(8):S31. doi:10.1016/S0006-3223(00)00364-4
16. Getzen H, Beasley M, D’ Mello DA. Barriers to utilizing long-acting injectable antipsychotic medications. Ann Clin Psychiatry. 2014;26(1):33-38. PubMed
17. Tiihonen J, Haukka J, Taylor M, et al. A nationwide cohort study of oral and depot antipsychotics after first hospitalization for schizophrenia. Am J Psychiatry. 2011;168(6):603-609. doi:10.1176/appi.ajp.2011.10081224 PubMed