ABSTRACT
The global prevalence of type 2 diabetes mellitus (T2DM) is increasing. T2DM is more common in patients with psychiatric disorders and those who take certain psychotropic drugs. T2DM occurs in 2%–7% of women of reproductive age. The prevalence of pregestational diabetes is 0.5%–2.4%, and that of gestational diabetes is 7%–28%, depending on geographical region. About 20%–50% of pregnancies, across the world, are unplanned; this figure is higher, at about 65%, in women with psychiatric disorders. As a result, many women of reproductive age who have diabetes, including women who do not know that they have diabetes, may unintentionally become pregnant, thus unknowingly exposing their pregnancy to diabetes and its treatment. Exposure of pregnancy to pregestational and gestational diabetes is associated with risks to the mother as well as risks to the child. Risks to the mother include obesity, hypertension, and preeclampsia. Risks to the child include spontaneous abortion, fetal death, macrosomia, major congenital malformations (MCMs), preterm delivery, neonatal hypoglycemia, neonatal hyperbilirubinemia, and neonatal respiratory distress syndrome. A recent large retrospective cohort study with data from 6 countries in Europe, Asia, and North America found that, in about 51,000 women with pregestational T2DM, neither MCMs nor cardiac malformations were more prevalent in offspring of children periconceptionally exposed to second-line antidiabetic treatments relative to exposure to insulin. These findings are reassuring but have limitations that are discussed. A reasonable conclusion from a reading of the reviewed literature is that pregestational and gestational diabetes are best treated during pregnancy, that insulin is a first-line treatment, that metformin is an increasingly accepted alternative, and that safety data on second-line antidiabetic treatments are, so far, reassuring.
J Clin Psychiatry 2024;85(1):24f15318
Author affiliations appear at the end of this article
The prevalence of type 2 diabetes mellitus (T2DM) is increasing across the world. It was estimated that the age-standardized global prevalence of total diabetes grew from 3.2% in 1990 to 6.1% in 2021. In 2021, estimates of the prevalence of diabetes were 6.5% for males and 5.8% for females, with T2DM comprising 96% of cases. The prevalence was lowest, at < 1%, at age < 20 years and peaked at nearly 25% at age 75–79 years. The highest prevalences, in middle age and later, were observed in North Africa and in the Middle East, followed by South Asia.1
Diabetes and Psychiatric Disorders
T2DM is common in patients with psychiatric disorders. This is partly because some disorders, such as mood disorders and schizophrenia, are associated with eating disturbances and sedentariness, and partly because some psychotropic drugs, such as some antipsychotics, antidepressants, anxiolytics, and mood stabilizers, are associated with increased appetite, weight gain, and even the metabolic syndrome. An umbrella review and meta-analysis found pooled T2DM prevalences of 40% in sleep disorder, 21% in binge eating disorder, 16% in substance use disorder, 14% in anxiety disorder, 11% in bipolar disorder, 10% in schizophrenia, 9% in depression, and 8% in intellectual disability.2
Among psychotropic drugs, as examples, antidepressants (pooled hazard ratio [HR], 1.24; 95% confidence interval [CI], 1.18–1.31), and specifically tricyclic antidepressants (pooled relative risk [RR], 1.39; 95% CI, 1.17–1.65), have been associated with new-onset T2DM.3 Antipsychotic drugs have more particularly been associated with an increased risk of T2DM (pooled odds ratio [OR], 2.58; 95% CI, 1.56–4.24)4 and gestational diabetes (pooled RR, 1.24; 95% CI, 1.09–1.42).5 Some antipsychotics, such as olanzapine (OR, 5.8; 95% CI, 2.0–16.7),6 have specifically been identified to carry greater risk.
Diabetes in Women of Reproductive Age
A population estimate of the prevalence of diabetes in US women aged 24–32 years was surprisingly high at 6.5% as far back as in 2007–2008; the data were drawn from the nationally representative National Longitudinal Study of Adolescent to Adult Health, Wave IV.7 In a systematic review and meta-analysis of 39 studies from 27 African countries, the pooled prevalence of T2DM was even higher, at 7.2%, in women of childbearing age8; this figure may have been incorrectly high because the upper age limit for inclusion of women was 54 years. In an Indian study using data from the fourth round of the National Family Health Survey (2015–2016), the prevalence of diabetes in women aged 15–49 years was more modest and ranged from 1.8% to 4.4% in 8 states in northeast India.9
These data are important because a large proportion of pregnancies are unplanned, and because undetected and untreated diabetes, and perhaps even detected and treated diabetes, may adversely affect pregnancy and offspring outcomes, both before and after birth. These issues are discussed in later sections.
Prevalence of Pregestational and Gestational Diabetes
A small but clinically significant number of already diabetic women become pregnant. In a systematic review and meta-analysis of 42 data sources (pooled N = 78,943,376) from 17 countries in North America, Europe, Africa, the Middle East, Asia, and Australasia, the pooled prevalence of pregestational diabetes was found to range from a low of 0.5% in Europe to a high of 2.4% in the Middle East and North Africa. The overall prevalence doubled from 0.5% to 1.0% during 1990–2020.10
A larger proportion of women develop gestational diabetes. In a systematic review and meta-analysis of 57 studies, the pooled prevalence of gestational diabetes ranged from a low of 7.1% in North America and the Caribbean to a high of 27.6% in the Middle East and North Africa. Pooled standardized prevalences were 12.7% in low income countries, 9.2% in middle income countries, and 14.2% in high income countries. The global pooled standardized prevalence was 14.0%.11
Unplanned (Unintended) Pregnancy
In different systematic reviews and meta-analyses, the pooled prevalence of unintended pregnancy was 20% in Iran,12 28% in Ethiopia,13 and 26.5% in 61 low and middle income countries14; these “low” numbers may be driven by sociocultural influences on reproductive behavior. In developed countries, the numbers are higher; for example, the figure was 45%–51% in the United States during 2008–2011.15
In a systematic review and meta-analysis of 11 studies (pooled N = 18,681), the weighted prevalence of unintended pregnancy was 65% among women with anxiety disorders, mood disorders, psychosis, substance use disorders, conduct disorder, and eating disorders. These women were at higher risk of unintended pregnancy relative to women without psychiatric conditions (OR, 1.34; 95% CI, 1.08–1.67).16
The data in this and in previous sections prompt the realization that many women of reproductive age who have diabetes, including women who do not know that they have diabetes, may unintentionally become pregnant, thus unknowingly exposing their pregnancy to diabetes and its treatment.
Adverse Outcomes Associated With Exposure of Pregnancy to Diabetes
Having diabetes during pregnancy is associated with risks to the mother as well as risks to the child. As an example, in a systematic review and meta-analysis of 59 population-based studies (pooled N = 80,437,056), 3.0% of women had pregestational diabetes. Their pregnancies were at increased risk of overall congenital anomalies (RR, 1.99; 95% CI, 1.82–2.17) as well as congenital heart defects (RR, 3.46; 95% CI, 2.77–4.32). In both regards, risks were higher in pregnancies exposed to pregestational diabetes relative to pregnancies exposed to gestational diabetes.17
There are many other risks, too. Risks to the mother include obesity, hypertension, and preeclampsia. Risks to the child before birth include spontaneous abortion, fetal death, macrosomia, and preterm delivery. Risks to the child after birth include neonatal hypoglycemia, neonatal hyperbilirubinemia, and neonatal respiratory distress syndrome.18,19
There are several mechanisms suggested through which uncontrolled hyperglycemia during pregnancy may harm the fetus and the developing brain. These include fetal hyperinsulinism, neuroinflammation, iron deficiency, dysregulation of lipid metabolism, epigenetic changes, and others. In consequence, as reported in observational studies, children exposed to diabetes during pregnancy are at increased risk of delays or impairments in motor, language, cognitive, and general intelligence domains, as well as increased risk of diagnosis of autism spectrum disorder (but not attention-deficit/hyperactivity disorder) and behavioral and psychiatric disorders.20,21
Adverse Outcomes Associated With Gestational Diabetes
In a systematic review and meta-analysis of 59 population-based studies (pooled N = 80,437,056), 2.9% of women had gestational diabetes. Their offspring were at increased risk of overall congenital anomalies (RR, 1.18; 95% CI, 1.13–1.23) as well as congenital heart defects (RR, 1.50; 95% CI, 1.38–1.64). In both regards, risks were lower than in pregnancies exposed to pregestational diabetes.17
Ye et al22 described a systematic review and meta-analysis of 156 studies (pooled N = 7,506,061 pregnancies) of adverse outcomes associated with exposure to gestational diabetes. Important findings from the meta-analysis are presented in Table 1. Readers may note that the presence of adverse outcomes despite insulin treatment could be because the treatment was not adequate in all patients and/or that adjustment for confounding by severity of indication (and for other sources of confounding) was inadequate. Similar findings were described by Greco et al23 in a systematic review and meta-analysis of 85 singleton and 27 twin pregnancy studies exposed to gestational diabetes.
Interpretation of Findings
It is not clear to what extent the adverse outcomes associated with exposure of pregnancy to pregestational or gestational diabetes are mediated by diabetes vs the treatment thereof. However, given that the first-line treatment of diabetes during pregnancy is the naturally occurring hormone insulin,18 and given that currently available insulin preparations do not cross the placental barrier,18 it is reasonable to assume that diabetes, the genetic predispositions thereto, and the metabolic and other disturbances associated therewith, may drive the risk. It is therefore reasonable to emphasize adequate treatment of diabetes during pregnancy.
Treatment of Diabetes During Pregnancy
In addition to lifestyle modification, insulin is traditionally recommended for the treatment of diabetes during pregnancy.18 This is partly because insulin is a naturally occurring hormone that regulates blood glucose levels in healthy persons, including healthy pregnant women, and partly because, as already stated, insulin does not cross the placental barrier. Metformin, however, is increasingly being prescribed for diabetes during pregnancy, and for gestational diabetes, as well.24,25 Recent data from the US, Europe, and Asia suggest that other, including newer, antidiabetic medications are being prescribed in about 15% of pregnancies and that the prescription rates for these drugs have increased in recent years.26 These drugs include sulfonylureas (eg, glyburide, glimepride), dipeptidyl peptidase 4 (DPP-4) inhibitors (eg, linagliptin, sitagliptin), glucagon-like peptide 1 receptor agonists (GLP-1 RAs; eg, liraglutide, semaglutide), and sodium-glucose cotransporter 2 (SGLT2) inhibitors (eg, canagliflozin, dapagliflozin).
Given that diabetes is common in women of childbearing age, that many pregnancies are unplanned, that diabetes is common during pregnancy, that diabetes in pregnancy needs to be treated, and that non-insulin treatments are now increasingly being used to manage diabetes during pregnancy, it is likely that many pregnancies are unknowingly or knowingly exposed to non-insulin antidiabetic treatments during the period of organogenesis in the first trimester. The safety of such treatments during the first trimester therefore needs to be examined. In this context, Cesta et al26 described a population-based cohort study of major congenital malformations (MCMs) following first-trimester gestational exposure to pregestational T2DM and antidiabetic treatments.
The Study by Cesta et al26
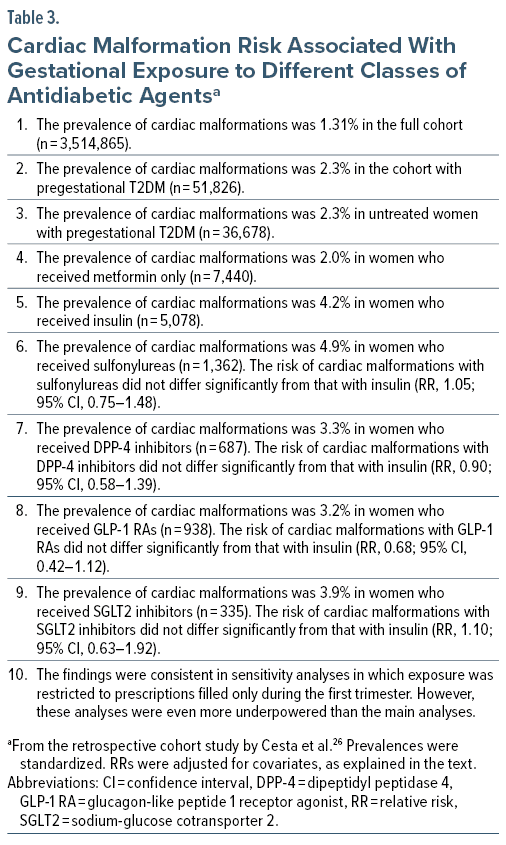
These authors obtained data from each of 4 Nordic countries (Finland, Iceland, Norway, Sweden; 2009–2020), the US MarketScan Database (2012–2021), and the Israeli Maccabi Health Services database (2009–2020). After excluding children with genetic or chromosomal abnormalities and pregnancies exposed to known teratogens, there were 3,514,865 pregnancies with live births among which 1.5% (n = 51,826) were in women with pregestational T2DM. Infants from these pregnancies were followed for up to 1 year for detection of MCMs.
Among the women with pregestational T2DM, 70.8% (n = 36,678) received no antidiabetic treatment during the periconceptional period; the rest (n = 15,148; 29.2%) received metformin monotherapy (14.4%), insulin (9.8%), sulfonylureas (2.6%), DPP-4 inhibitors (1.3%), GLP-1 RAs (1.8%), or SGLT2 inhibitors (0.6%); use of these medications was highest in the US. In this context, periconceptional exposure was defined as 1 or more prescriptions filled between 90 days before pregnancy and the end of the first trimester. For each geographical area, the MCM risk was calculated for each drug group, relative to insulin, after adjusting for covariates such as maternal age, comorbid conditions, and use of other medications. HbA1c levels were available for only some pregnancies and so could not be adjusted for. Risks for the 3 geographical areas were subsequently pooled in meta-analysis.
Important findings from the study are presented in Table 2 and Table 3. In summary, although prevalences of MCMs and cardiac malformations were numerically high in treated pregnancies, relative to treatment with insulin, no second-line antidiabetic agent was associated with a significantly increased risk of either total MCMs or cardiac malformations.
Observations on the Study by Cesta et al26
The findings of the study are reassuring. However, the findings are not definitive. One limitation of the study is that, as stated below, it is unlikely that adjustment for confounding by indication, confounding by severity of indication, and confounding by other unbalanced covariates was adequate. As an example, adjustment for HbA1c levels was not performed. Reassuringly, though, better adjustment could move the findings closer to the null. A more important limitation is that many of the analyses were conducted on very small numbers of malformations in small numbers of exposed women; these analyses were therefore underpowered to identify true differences. On an optimistic note, the upper limits of the 95% CIs (Tables 2 and 3) suggested that even if the study estimates were underestimates, the population value would not be much higher.
The noticeably different MCM rates for different classes of antidiabetic agents could have arisen from confounding by indication. As examples, GLP-1 RAs were more likely to have been prescribed to women with obesity and polycystic ovaries, SGLT2 inhibitors to women with cardiovascular conditions, and SGLT2 inhibitors and insulin to women with complications of diabetes. Furthermore, many women who received these second-line treatments also received metformin or insulin, again indicating more severe diabetes. So, it is unclear to what extent the magnitude of adverse outcomes recorded were a consequence of more severe illness and complications thereof vs medications and combinations thereof.
The study could not provide safety information for certain classes of antidiabetic medications, such as the glitazones, because these had been very infrequently used.
General Observations
Overall safety observed for a class of medications does not mean that all drugs within that class are safe. This is because, for example, drugs within a class may have additional mechanisms beyond their shared mechanism. Drugs would therefore need to be studied individually in adequately powered analyses for more definitive guidance.
MCMs are not the only adverse outcomes associated with diabetes and its treatment during pregnancy; other adverse outcomes, such as those listed in Table 1, were referred to in earlier sections of this article. Studies of these outcomes for individual antidiabetic treatments or treatment classes have been published; metformin is perhaps the best researched.24 A review of studies of these treatments is out of the scope of the present article.
Parting Note
A detailed discussion on the management of diabetes during pregnancy, including gestational diabetes, is out of the scope of this article. Interested readers may wish to consult other resources, such as the article by ElSayed et al,18 or region-specific guidelines.
Article Information
Published Online: March 11, 2024. https://doi.org/10.4088/JCP.24f15318
© 2024 Physicians Postgraduate Press, Inc.
To Cite: Andrade C. Exposure of pregnancy to pregestational diabetes, gestational diabetes, and antidiabetic medications with especial focus on major congenital and cardiac malformations in offspring. J Clin Psychiatry. 2024;85(1):24f15318.
Author Affiliations: Department of Clinical Psychopharmacology and Neurotoxicology, National Institute of Mental Health and Neurosciences, Bangalore, India ([email protected]).
 Each month in his online column, Dr Andrade considers theoretical and practical ideas in clinical psychopharmacology with a view to update the knowledge and skills of medical practitioners who treat patients with psychiatric conditions.
Each month in his online column, Dr Andrade considers theoretical and practical ideas in clinical psychopharmacology with a view to update the knowledge and skills of medical practitioners who treat patients with psychiatric conditions.Department of Clinical Psychopharmacology and Neurotoxicology, National Institute of Mental Health and Neurosciences, Bangalore, India ([email protected]).
Financial disclosure and more about Dr Andrade.
References (26)

- GBD 2021 Diabetes Collaborators. Global, regional, and national burden of diabetes from 1990 to 2021, with projections of prevalence to 2050: a systematic analysis for the Global Burden of Disease Study 2021. Lancet. 2023;402(10397):203–234. PubMed CrossRef
- Lindekilde N, Scheuer SH, Rutters F, et al. Prevalence of type 2 diabetes in psychiatric disorders: an umbrella review with meta-analysis of 245 observational studies from 32 systematic reviews. Diabetologia. 2022;65(3):440–456. PubMed CrossRef
- Wang Y, Liu D, Li X, et al. Antidepressants use and the risk of type 2 diabetes mellitus: A systematic review and meta-analysis. J Affect Disord. 2021;287:41–53. PubMed CrossRef
- Galling B, Roldán A, Nielsen RE, et al. Type 2 diabetes mellitus in youth exposed to antipsychotics: a systematic review and meta-analysis. JAMA Psychiatry. 2016;73(3):247–259. PubMed CrossRef
- Wang Z, Wong ICK, Man KKC, et al. The use of antipsychotic agents during pregnancy and the risk of gestational diabetes mellitus: a systematic review and meta-analysis. Psychol Med. 2021;51(6):1028–1037. PubMed CrossRef
- Koro CE, Fedder DO, L’Italien GJ, et al. Assessment of independent effect of olanzapine and risperidone on risk of diabetes among patients with schizophrenia: population based nested case-control study. BMJ. 2002;325(7358):243. PubMed CrossRef
- Britton LE, Berry DC, Hussey JM. Comorbid hypertension and diabetes among US women of reproductive age: prevalence and disparities. J Diabetes Complications. 2018;32(12):1148–1152. PubMed CrossRef
- Chivese T, Werfalli MM, Magodoro I, et al. Prevalence of type 2 diabetes mellitus in women of childbearing age in Africa during 2000-2016: a systematic review and meta-analysis. BMJ Open. 2019;9(5):e024345. PubMed CrossRef
- Meitei WB, Singh SR. Prevalence and determinants of diabetes among women of reproductive age in northeast India. Demogr India. 2021;50(2):55–64.
- Chivese T, Hoegfeldt CA, Werfalli M, et al. IDF Diabetes Atlas: The prevalence of pre-existing diabetes in pregnancy: a systematic review and meta-analysis of studies published during 2010-2020. Diabetes Res Clin Pract. 2022;183:109049. PubMed CrossRef
- Wang H, Li N, Chivese T, et al. IDF Diabetes Atlas Committee Hyperglycaemia in Pregnancy Special Interest Group. IDF Diabetes Atlas: estimation of global and regional gestational diabetes mellitus prevalence for 2021 by International Association of Diabetes in Pregnancy Study Group’s criteria. Diabetes Res Clin Pract. 2022;183:109050. PubMed CrossRef
- Sayehmiri K, Ebtekar F, Zarei M, et al. Prevalence of unwanted pregnancy among Iranian women: an updated meta-analysis. BMC Pregnancy Childbirth. 2019;19(1):491. PubMed CrossRef
- Alene M, Yismaw L, Berelie Y, et al. Prevalence and determinants of unintended pregnancy in Ethiopia: A systematic review and meta-analysis of observational studies. PLoS One. 2020;15(4):e0231012. PubMed CrossRef
- Aragaw FM, Amare T, Teklu RE, et al. Magnitude of unintended pregnancy and its determinants among childbearing age women in low and middle-income countries: evidence from 61 low and middle income countries. Front Reprod Health. 2023;5:1113926. PubMed CrossRef
- Finer LB, Zolna MR. Declines in unintended pregnancy in the United States, 2008-2011. N Engl J Med. 2016;374(9):843–852. PubMed CrossRef
- Schonewille NN, Rijkers N, Berenschot A, et al. Psychiatric vulnerability and the risk for unintended pregnancies, a systematic review and meta-analysis. BMC Pregnancy Childbirth. 2022;22(1):153. PubMed CrossRef
- Zhang TN, Huang XM, Zhao XY, et al. Risks of specific congenital anomalies in offspring of women with diabetes: A systematic review and meta-analysis of population-based studies including over 80 million births. PLoS Med. 2022;19(2):e1003900. PubMed CrossRef
- ElSayed NA, Aleppo G, Aroda VR, et al, on behalf of the American Diabetes Association. 15. Management of Diabetes in Pregnancy: Standards of Care in Diabetes-2023. Diabetes Care. 2023;46(suppl 1):S254–S266. PubMed CrossRef
- Ghaffari N. Promising safety profile of noninsulin medications for type 2 diabetes in early pregnancy. JAMA Intern Med. 2024;184(2):152–153. PubMed CrossRef
- Rowland J, Wilson CA. The association between gestational diabetes and ASD and ADHD: a systematic review and meta-analysis. Sci Rep. 2021;11(1):5136. PubMed CrossRef
- Rodolaki K, Pergialiotis V, Iakovidou N, et al. The impact of maternal diabetes on the future health and neurodevelopment of the offspring: a review of the evidence. Front Endocrinol (Lausanne). 2023;14:1125628. PubMed CrossRef
- Ye W, Luo C, Huang J, et al. Gestational diabetes mellitus and adverse pregnancy outcomes: systematic review and meta-analysis. BMJ. 2022;377:e067946. PubMed CrossRef
- Greco E, Calanducci M, Nicolaides KH, et al. Gestational diabetes mellitus and adverse maternal and perinatal outcomes in twin and singleton pregnancies: a systematic review and meta-analysis. Am J Obstet Gynecol. 2024;230(2):213–225. PubMed CrossRef
- Paschou SA, Shalit A, Gerontiti E, et al. Efficacy and safety of metformin during pregnancy: an update. Endocrine. 2024;83(2):259–269. PubMed CrossRef
- Zhang M, Zhou Y, Zhong J, et al. Current guidelines on the management of gestational diabetes mellitus: a content analysis and appraisal. BMC Pregnancy Childbirth. 2019;19(1):200. PubMed CrossRef
- Cesta CE, Rotem R, Bateman BT, et al. Safety of GLP-1 receptor agonists and other second-line antidiabetics in early pregnancy. JAMA Intern Med. 2024;184(2):144–152. PubMed CrossRef
This PDF is free for all visitors!