Ketamine and esketamine produce rapid antidepressant effects in treatment-resistant depression (TRD), offering a promising alternative to conventional therapies.1,2 Few studies have examined whether the choice of oral antidepressant concurrent to ketamine/esketamine differentially affects clinical outcomes.3 To investigate the relationship between concurrent antidepressant class and clinical outcomes in ketamine/ esketamine treatment, we analyzed data from all patients with TRD treated with intravenous (IV) ketamine or intranasal (IN) esketamine at the Yale Interventional Psychiatry Service between March 2015 and August 2023.
Methods
The treatment setting, clinical approach, and data collection of this report have been described elsewhere.4,5 The Yale Institutional Review Board waived informed consent per the Common Rule. The primary exposure variable was antidepressant class (selective serotonin reuptake inhibitor [SSRI], serotonin–norepinephrine reuptake inhibitor [SNRI], or another oral antidepressant [AD]), relative to no antidepressant. Outcomes included mean changes in the Montgomery-Asberg Depression Rating Scale (MADRS) and Quick Inventory of Depressive Symptomatology (QIDS) scales, response (≥50% improvement on MADRS) and remission (≤10 on MADRS). Outcomes were assessed at first treatment, treatment 4, and final treatment (treatment 8 or last treatment carried forward). Linear mixed models were used to assess change in MADRS and QIDS over time; logistic regressions were used for remission and response and were adjusted for age, sex, race/ethnicity, concomitant benzodiazepine or antipsychotic use, history of electroconvulsive therapy or psychiatric hospitalization, comorbid psychiatric disorders, insurance type (public vs private), and treatment modality (IV ketamine vs IN esketamine). Analyses were conducted using Stata version 19/MP (StataCorp).
Results
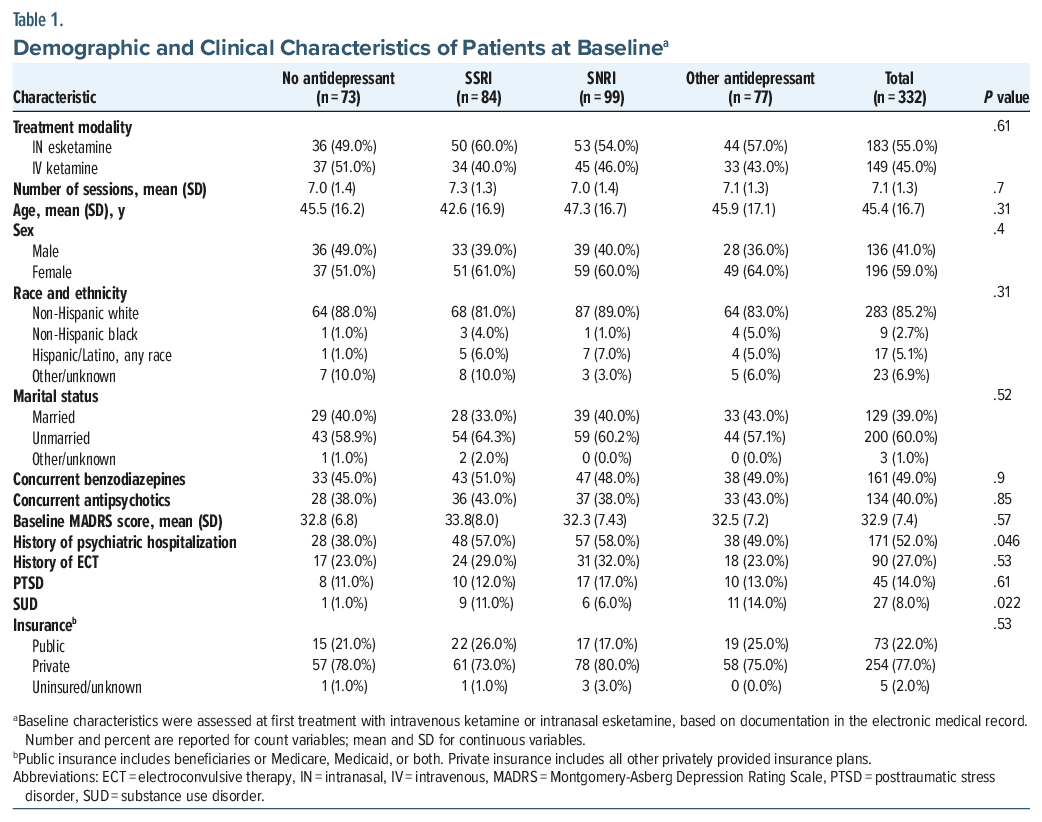
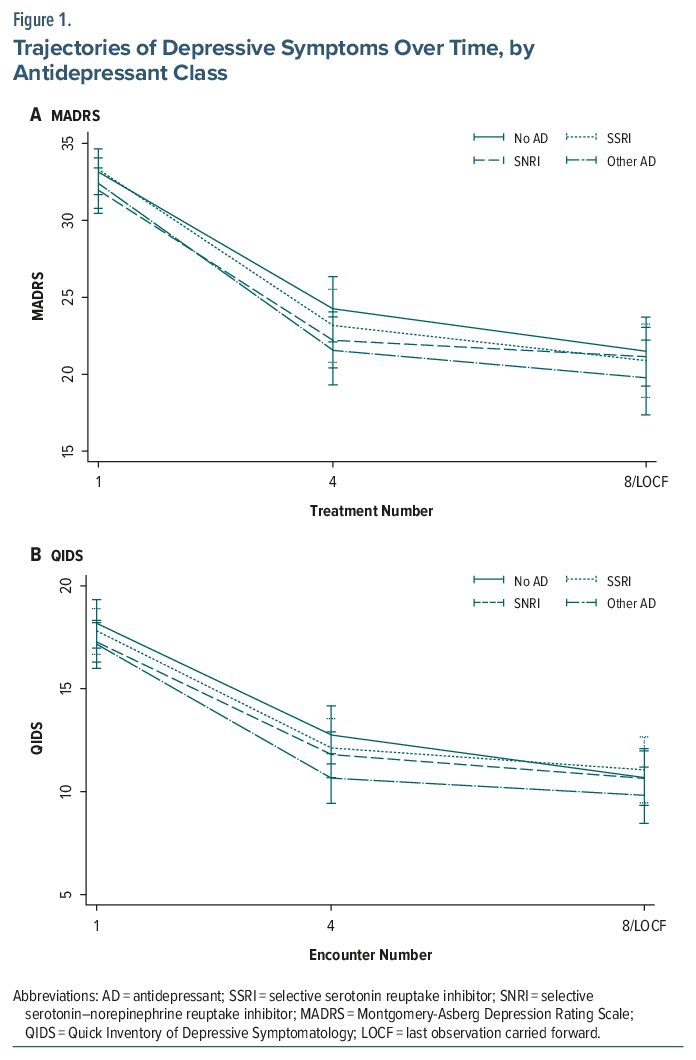
Overall, 332 patients were included in the analysis, of whom 149 received ketamine and 183 received esketamine. At the time of treatment, 84 were prescribed an SSRI, 98 an SNRI, 77 another AD, and 73 no AD. The groups were similar at baseline (Table 1). Using linear mixed-effects models, the mean adjusted MADRS score significantly decreased across visits but did not differ by AD class (SSRI: unstandardized coefficient (b), 0.19 [95% CI, −2.10 to 2.49]; P = .87; SNRI: b, −1.21 [−3.32 to 0.91]; P = .26; other AD: b, −0.74 [−2.97 to 1.49]; P = .52). Results for QIDS were similar (SSRI: b, −0.37 [−1.95 to 1.22]; P= .65; SNRI: b, −0.90 [−2.42 to 0.63]; P = .25; other AD: b, −1.00 [−2.67 to 0.67]; P = .24) (Figure 1).
The adjusted odds of response and remission also did not differ significantly by AD class. For response, odds ratios were 1.29 (95% CI, 0.64–2.58; P = .31) for SSRI, 0.79 (0.40–1.57; P = .52) for SNRI, and 1.74 (0.64–2.65; P = .15) for other AD. For remission, odds ratios were 2.00 (0.83–4.83; P = .12), 1.06 (0.45–2.51; P = .89), and 1.43 (0.56–3.61; P = .45), respectively. A subgroup analysis was conducted in which regressions were repeated within esketamine or ketamine subgroups; results were not meaningfully different.
Discussion
This analysis did not detect group differences between concurrent oral antidepressant class and clinical outcomes among patients treated with IV ketamine or IN esketamine. This finding was consistent across different outcome measures assessed and models employed. The result suggests that concurrent antidepressant use does not meaningfully alter initial treatment outcomes from ketamine/ esketamine. This may be due to ketamine/esketamine’s unique mechanism of action that acts independently from oral antidepressants. Alternatively, the substantial nonspecific effects inherent in ketamine/esketamine treatment may mask subtle differential effects from oral antidepressants.6
Limitations of this study are the observational study design, the short follow-up period, and the lack of formal assessment for treatment resistance. This last weakness is mitigated in part by the fact that third-party payers generally require 2 failed antidepressant trials prior to authorizing treatment; the vast majority of patients at our clinic receive insurance coverage for both ketamine and esketamine. Longer-term, randomized trials are needed to confirm whether oral antidepressant class differentially affects efficacy and durability of response to ketamine/esketamine treatment. If confirmed, such findings have relevant clinical implications: concurrent prescribing during ketamine/esketamine treatment may be guided more by tolerability than clinical expectations of differential efficacy.
Article Information
Published Online: April 27, 2026. https://doi.org/10.4088/JCP.25br16294
© 2026 Physicians Postgraduate Press, Inc.
J Clin Psychiatry 2026;87(2):25br16294
Submitted: December 22, 2025; accepted March 6, 2026.
To Cite: Curran E, Hardy M, Katz R, et al. Concurrent SSRI, SNRI, or other antidepressant use not associated with differential outcomes in ketamine or esketamine treatment. J Clin Psychiatry 2026;87(2):25br16294.
Author Affiliations: Department of Psychiatry, Yale School of Medicine, New Haven, Connecticut.
Corresponding Author: Samuel Wilkinson, MD, Yale University, 184 Liberty St, New Haven, CT 06519 ([email protected]).
Ms Curran and Dr Hardy contributed equally to this work and share first authorship.
Relevant Financial Relationships: Yale University has a financial relationship with Janssen Pharmaceuticals and may receive financial benefits from this relationship. Dr Wilkinson has received contract research funding (administered through Yale University) from Oui Therapeutics and has served as a consultant or provided data safety and monitoring board services for Joyous, LivaNova, Mind Medicine, Neumora, Sooma Medical, and Sage Therapeutics. The other authors report no financial relationships with commercial interests.
Funding/Support: None.
References (6)

- Popova V, Daly EJ, Trivedi M, et al. Efficacy and safety of flexibly dosed esketamine nasal spray combined with a newly initiated oral antidepressant in treatment-resistant depression: a randomized double-blind active-controlled study. Am J Psychiatry. 2019;176(6):428–438. PubMed CrossRef
- Zarate CA Jr, Singh JB, Carlson PJ, et al. A randomized trial of an N-methyl-D-aspartate antagonist in treatment-resistant major depression. Arch Gen Psychiatry. 2006;63(8):856–864. PubMed CrossRef
- Del Casale A, Spirito S, Arena JF, et al. Esketamine combined with SSRI or SNRI for treatment-resistant depression. JAMA Psychiatry. 2025;82(8):810–817. PubMed CrossRef
- Nikayin S, Rhee TG, Cunningham ME, et al. Evaluation of the trajectory of depression severity with ketamine and esketamine treatment in a clinical setting. JAMA Psychiatry. 2022;79(7):736–738. PubMed CrossRef
- Janik A, Qiu X, Lane R, et al. Esketamine monotherapy in adults with treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry. 2025; 82(9):877–887. PubMed CrossRef
- Matsingos A, Wilhelm M, Noor L, et al. Hype or hope? High placebo response in major depression treatment with ketamine and esketamine: a systematic review and meta-analysis. Front Psychiatry. 2024;15:1346697. PubMed CrossRef
This PDF is free for all visitors!