Brief Report
Propranolol, Traumatic Memories, and Amnesia: A Study of 36 Cases
To the Editor: Studies1-3 have shown that treatment with propranolol hydrochloride may be helpful in decreasing physiologic responses and preventing the development of posttraumatic stress disorder (PTSD) in those who have experienced a traumatic event. This clinical study was done to determine whether treatment of traumatic memories with 1 or 2 doses of propranolol improves symptoms of PTSD in psychiatric patients.
Method. Between September 2008 and October 2010, 36 psychiatric outpatients with DSM-IV-defined PTSD (75% [n = 27]) or subsyndromal PTSD (25% [n = 9]) gave informed consent to undergo brief treatment with propranolol. These patients were still haunted and distressed by traumatic experiences, which had occurred between 3 months and 50 years earlier. They were all diagnosed with at least 1 other mental disorder. They received either 1 dose of propranolol (regular formulation) 40 mg (n = 11) or 1 dose of propranolol (regular formulation) 40 mg followed 2 hours later by 1 dose of propranolol (long-acting) 60 mg (n = 25). Patients received 1 dose if they had a history of possible, yet unconfirmed, asthma (n = 10) or a history of hypersensitivity to medicines in general (n = 1). They took propranolol after reactivation of their traumatic memory, which was achieved by asking patients to write and talk about their traumatic experience. Patients were assessed approximately 3 weeks later.
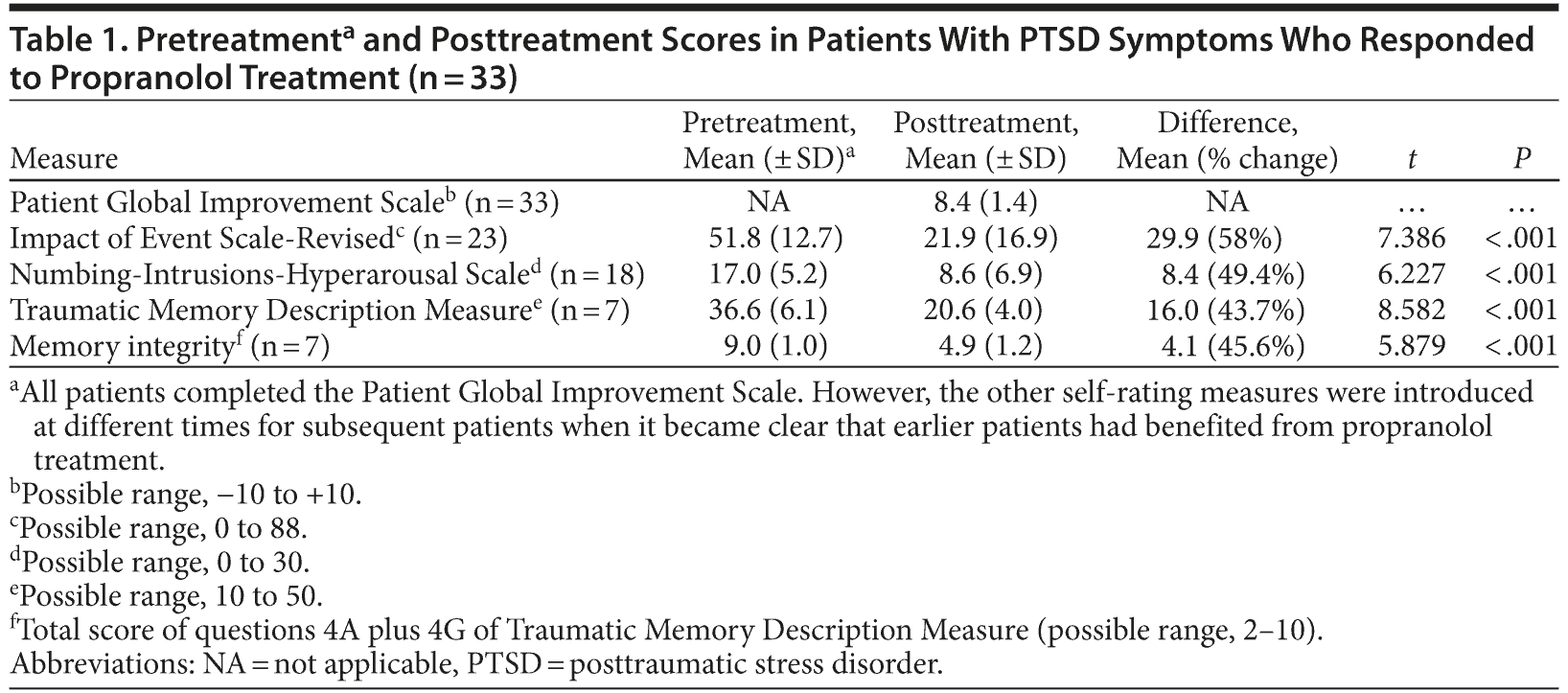
A specific instrument was developed, the Traumatic Memory Description Measure (available on request from the author), to assist the patient in providing as much detail about the event as possible (part A) and record the patients’ perception of the quantity and quality of the memory, including the vividness of colors, smells, sounds, and tastes (part B). Two questions relate specifically to the overall integrity of the memory on a 5-point scale: question 4A, "How well do you remember the traumatic event?" (poorly = 1, very well = 5) and question 4G, "My overall memory for the traumatic event is’ ¦" (absent = 1, extremely detailed = 5). In addition, the author recorded the patients’ self-reports of any changes they experienced following treatment. Other measures consisted of the Impact of Event Scale-Revised4; Numbing-Intrusions-Hyperarousal Scale (unpublished scale, D. V. Sheehan, MD, MBA; J. R. T. Davidson, MD; 2000); and Patient Global Improvement Scale (PGIS; unpublished scale, D. V. Sheehan, MD, MBA; 1985). All patients completed the PGIS, which was the primary outcome measure. However, the other self-rating measures were introduced at different times when it became clear that propranolol was being effective. This was done to evaluate the extent of the treatment responses and accounts for the different numbers of patients in the 4 measures used.
Results. Twenty-three of the 36 patients treated were male (63.9%), 31 had concurrent mood disorder (86.1%), and 20 were subjected to actual/threatened assault (55.6%). Patients’ mean age was 40.0 years (SD = 11.2), their traumatic events occurred a mean of 14.4 (SD = 13.6) years earlier, and they were taking a mean of 1.7 (SD = 1.3) psychotropic medicines.
Thirty-three patients (91.7%) responded to treatment; see Table 1 for results. The mean score of 8.4 out of a possible 10 ("major improvement/back to normal self") on the PGIS equates to "markedly improved." These benefits have been sustained for up to 2½ years. No significant side effects were reported, even in patients with a history of possible asthma. The 3 patients who did not report a benefit (score of zero on the PGIS) indicated no deterioration following treatment.
Patients’ self-reports included comments about the quality and quantity of the memory for the event. The posttreatment comments of 17 of 26 patients (excluding the 7 who completed the Traumatic Memory Description Measure and the 3 nonresponders), or 65.4%, included spontaneous references to some aspect of the integrity of their memory.
Understandably, patients found it difficult to write and talk about their traumatic events; however, they were very positive about the benefits of treatment. In addition to fewer reexperiences of the event and diminished emotional distress, patients reported a marked reduction in both the quality and quantity of the integrity of their traumatic memories. Propranolol proved to be an effective, quick, safe, and inexpensive treatment for patients suffering from traumatic memories and associated psychological distress. It also diminished the integrity of these traumatic memories, resulting in a degree of amnesia for the traumatic event.
References
1. Pitman RK, Sanders KM, Zusman RM, et al. Pilot study of secondary prevention of posttraumatic stress disorder with propranolol. Biol Psychiatry. 2002;51(2):189-192.PubMed doi:10.1016/S0006-3223(01)01279-3
2. Vaiva G, Ducrocq F, Jezequel K, et al. Immediate treatment with propranolol decreases posttraumatic stress disorder two months after trauma. Biol Psychiatry. 2003;54(9):947-949.PubMed doi:10.1016/S0006-3223(03)00412-8
3. Brunet A, Orr SP, Tremblay J, et al. Effect of post-retrieval propranolol on psychophysiologic responding during subsequent script-driven traumatic imagery in post-traumatic stress disorder. J Psychiatr Res. 2008;42(6):503-506.PubMed doi:10.1016/j.jpsychires.2007.05.006
4. Weiss DS, Marmar CR. The Impact of Event Scale-Revised. In: Wilson J, Keane TM, eds. Assessing Psychological Trauma and PTSD. New York, NY: Guilford; 1996:399-411.
Editor’s note: Dr Menzies and colleagues are currently conducting a larger clinical trial comparing propranolol and placebo in subjects who have emotional distress associated with reexperiencing of traumatic events (clinicaltrials.gov identifier: NCT01069159).
Author affiliation: Department of Psychiatry, University of Saskatchewan, Saskatoon, Canada. Potential conflicts of interest: None reported. Funding/support: None reported. Previous presentations: Some data were presented at the Canadian Academy of Psychiatry and Law Annual winter conference; March 1, 2010; Banff, Alberta, Canada; and the Canadian Psychiatric Association Annual Meeting; September 23, 2010; Toronto, Ontario, Canada. Acknowledgments: The author thanks Marilyn Baetz, MD, FRCPC, Professor of Psychiatry, University of Saskatchewan, Saskatoon, Canada, who provided statistical assistance, a critical review, and helpful suggestions. Dr Baetz reports no conflict of interest.
doi:10.4088/JCP.11l07121
© Copyright 2012 Physicians Postgraduate Press, Inc.