Objective: Valproic acid (VPA) use during pregnancy increases fetal risk of major congenital malformations and cognitive impairment. Given these risks, several medical societies have put forth guidelines suggesting to either limit the use of VPA or take certain precautions, such as making sure effective contraception practices and/or appropriate folic acid supplementation are in place, when treating reproductive-aged women. Our study aimed to review and assess adherence to these guidelines.
Methods: Using electronic medical record (EMR) and administrative claims data over a 19-month period (January 1, 2013-July 31, 2014), a retrospective chart review was conducted of all reproductive-aged female patients at a major medical center in the Midwest who were prescribed VPA as treatment for their psychiatric illness (n = 190; aged from 15 to 49 years). Psychiatric diagnoses were determined via ICD-9 billing codes. We assessed 3 variables of interest as an index of adherence to guidelines: chart documentation of provider-patient discussion regarding potential teratogenicity associated with VPA use, prescription of contraceptives, and co-prescription of folic acid.
Results: EMR documentation of provider-patient discussions regarding possible teratogenicity of VPA was rare (13.2%), as was documentation of contraception use (30%) and co-prescription of folate (7.9%). Neither patient demographic characteristics nor diagnoses were associated with outcomes. Among those not receiving treatment in the inpatient setting, patients who were seen by outpatient psychiatry or neurology clinics (rather than other outpatient settings) were more likely to have documented discussions about teratogenicity (23% and 30%, respectively; P = .003), and patients receiving neurologic care were more likely to be prescribed folate than those seen by other providers (26%, P = .004). Women who had contact with inpatient psychiatric services were less likely to be taking contraception (n = 12 [20%], P = .041). Only 22% of women under 34 years of age were documented as using contraception (P = .03).
Conclusions: Adherence to standard guidelines is low even at an academic tertiary care center. To the extent that there is any documentation or co-prescription of folate, it varies by provider specialty.
See related commentary by Balon and Riba
This work may not be copied, distributed, displayed, published, reproduced, transmitted, modified, posted, sold, licensed, or used for commercial purposes. By downloading this file, you are agreeing to the publisher’s Terms & Conditions.

Guideline Adherence for Mentally Ill Reproductive-Aged Women on Treatment With Valproic Acid:
A Retrospective Chart Review

ABSTRACT
Objective: Valproic acid (VPA) use during pregnancy increases fetal risk of major congenital malformations and cognitive impairment. Given these risks, several medical societies have put forth guidelines suggesting to either limit the use of VPA or take certain precautions, such as making sure effective contraception practices and/or appropriate folic acid supplementation are in place, when treating reproductive-aged women. Our study aimed to review and assess adherence to these guidelines.
Methods: Using electronic medical record (EMR) and administrative claims data over a 19-month period (January 1, 2013-July 31, 2014), a retrospective chart review was conducted of all reproductive-aged female patients at a major medical center in the Midwest who were prescribed VPA as treatment for their psychiatric illness (n = 190; aged from 15 to 49 years). Psychiatric diagnoses were determined via ICD-9 billing codes. We assessed 3 variables of interest as an index of adherence to guidelines: chart documentation of provider-patient discussion regarding potential teratogenicity associated with VPA use, prescription of contraceptives, and co-prescription of folic acid.
Results: EMR documentation of provider-patient discussions regarding possible teratogenicity of VPA was rare (13.2%), as was documentation of contraception use (30%) and co-prescription of folate (7.9%). Neither patient demographic characteristics nor diagnoses were associated with outcomes. Among those not receiving treatment in the inpatient setting, patients who were seen by outpatient psychiatry or neurology clinics (rather than other outpatient settings) were more likely to have documented discussions about teratogenicity (23% and 30%, respectively; P = .003), and patients receiving neurologic care were more likely to be prescribed folate than those seen by other providers (26%, P = .004). Women who had contact with inpatient psychiatric services were less likely to be taking contraception (n = 12 [20%], P = .041). Only 22% of women under 34 years of age were documented as using contraception (P = .03).
Conclusions: Adherence to standard guidelines is low even at an academic tertiary care center. To the extent that there is any documentation or co-prescription of folate, it varies by provider specialty.
J Clin Psychiatry 2016;77(4):527-534
dx.doi.org/10.4088/JCP.15m10046
© Copyright 2016 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry, University of Michigan, Ann Arbor
bUniversity of Michigan Medical School, Ann Arbor
cDepartment of Internal Medicine, Division of Gastroenterology, University of Michigan, Ann Arbor
*Corresponding author: Maria Muzik, MD, MS, University of Michigan, Department of Psychiatry, Rachel Upjohn Bldg, 4250 Plymouth Rd, Ann Arbor, MI 48109-5765 ([email protected]).
Valproic acid (VPA) use during pregnancy increases fetal risk of neural tube defects and cognitive impairment.1 Other fetal malformations attributed to VPA exposure in utero include urogenital, craniofacial, cardiac, and digit abnormalities.2 Studies examining VPA exposure during the second and third trimester of pregnancy demonstrate an increased risk for autism, lower intelligence, and behavioral problems in school-aged children.3,4 There is a dose-dependent risk of cognitive deficits associated with in utero exposure (8-9 IQ points lower).5 Multiple studies, including a recent meta-analysis of women with epilepsy taking antiepileptic drugs, show the risk of major congenital malformations for VPA monotherapy to be 10.7%, which is above the baseline rate of 3% in healthy women.5-8 According to a recent publication of data from an international registry documenting outcomes of almost 4,000 antiepileptic-exposed pregnancies, odds ratios (ORs) for major congenital malformations are greater than 16 for VPA daily doses exceeding 1,500 mg (24%), and, for daily doses between 700 and 1,500 mg, there is a greater than 10% risk of malformation (OR = 5.8).2 For doses less than 700 mg/d, almost 6% of births showed major congenital malformations (OR = 2.8). This risk is greater than that of all other alternative medication options, including polytherapy with mood stabilizers or antiepileptics other than VPA and antipsychotic medications.2 A recent estimate suggests that, each year in the United States, 40 cases of spina bifida and 35 cases of cleft palate could be avoided if VPA would never be given during pregnancy.9 VPA also induces features of polycystic ovarian syndrome, a hormonal imbalance that can cause pelvic pain, abnormal periods, body hair growth, and acne in the short term and can lead to heart disease and diabetes in the long term. Features of polycystic ovarian syndrome affect approximately 7% of reproductive-aged women taking VPA.10 However, despite these risks, and its being a known teratogen with greater risk than its alternatives, VPA continues to be prescribed for women of reproductive age with psychiatric illnesses and without adequate levels of counseling about teratogenic risk and contraception.11-14
Prior to its serendipitous discovery as an antiepileptic in rats, VPA was used for decades in laboratories as an “inert” solvent for organic compounds. It was later found to have antimigraine and mood-stabilizing properties and was approved for use in pharmaceuticals in 1967,15 and, today, it has US Food and Drug Administration indications for use in broad-spectrum seizure disorders, as a mood stabilizer in the treatment of bipolar affective disorder, and in prophylaxis for migraine headaches. VPA has utility in bipolar depression and is commonly initiated in conjunction with an antipsychotic medication in the context of acute mania despite studies suggesting that antipsychotic monotherapy is equally as effective and poses significantly less risk in pregnancy (including less risk of major congenital malformations and fewer cognitive deficits)16-18 Despite these possible risks, a 2013 study19 looking at close to 9 million VPA prescriptions over 12 years found that 74% of VPA prescriptions were for psychiatric disorders and that use of this medication for reproductive-aged women tripled between the late 1990s and 2007.
In efforts to protect patients and help physicians, many relevant organizations, including the American Psychiatric Association (APA), the American Congress of Obstetrics and Gynecologists, the National Institute of Health and Care Excellence (NICE), and the American Academy of Neurology and American Epilepsy Society, have formulated prescribing guidelines for VPA in this population (Table 1).6,20-23 These guidelines are informed by expert opinions, and, while there are differences in the specific recommendations and some inconsistencies remain, they all share concerns regarding VPA’s teratogenicity and its use in reproductive-aged and pregnant women. The wording in these guidelines varies in the strength with which the prescription of VPA is discouraged, ranging from voicing serious concern about use of VPA, calling for a chart-documented provider-patient discussion regarding VPA’s potential teratogenicity, and urging attention to contraceptive use and co-prescription of folic acid13,15,16 to a very recent explicit statement in guidelines from the United Kingdom of not prescribing or discontinuing VPA when pregnancy is affirmed or planned.14 This explicit NICE guideline is very recent and basically prohibits VPA use in this population (in the United Kingdom); however, the use of VPA in pregnancy or in reproductive years, per US-based guidelines,13,15,16 is not prohibited.
Therefore, the aim of our study was to examine whether those guidelines recommended in the United States and based on VPA’s known teratogenic risk, would be closely followed at a US-based tertiary care medical center for women in childbearing age who are psychiatric patients seen in psychiatric care and whose providers have chosen to start or continue this medicine. We were especially interested in those female patients with care at a higher intensity level and greater provider contact (eg, treated in inpatient psychiatry units), hypothesizing that in those patients, treatment decision and follow-through with guidelines would be more deliberate. To investigate whether guidelines were followed and to determine if there was any relationship between guideline adherence and patient or prescriber characteristics, we explored the prescription patterns of VPA for psychiatric illness in women of childbearing age (aged 15-49 years) at a large academic tertiary care center.

- Given known risks, valproic acid is given to reproductive-aged women with mental illness at alarmingly high rates.
- Prescribers considering starting or continuing VPA in a reproductive-aged woman should first reevaluate alternative medications or therapies; if the benefits of valproic acid are deemed to outweigh the risks for the particular patient, the risk discourse should be documented in the chart and additional protections such as contraception and folate use initiated.
METHODS
Study Design
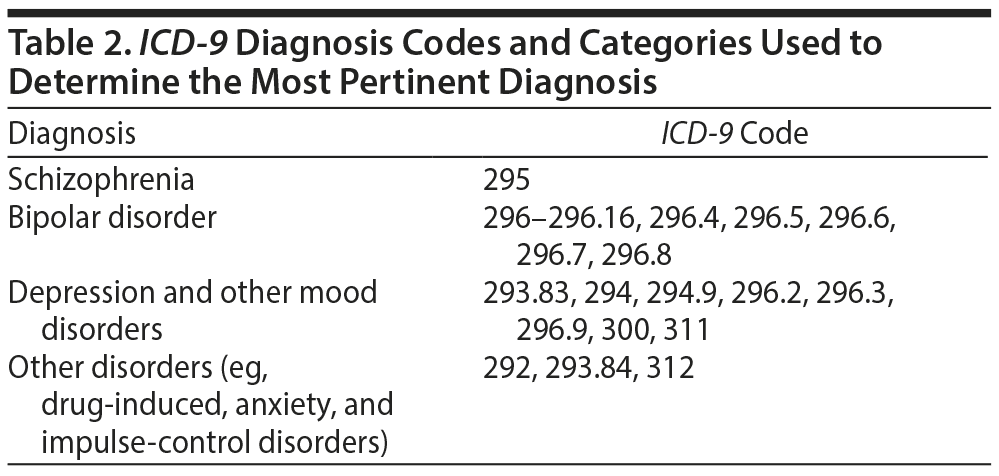
We conducted a retrospective chart review study on all female, reproductive-aged (ie, 15- to 49-year-old) patients who had medical records at a tertiary medical center and who were prescribed VPA (for an associated psychiatric diagnosis, including mood disorders, bipolar disorders, schizophrenia, and impulse-control disorder) according to electronic medical record (EMR) review during the study timeframe of 19 months (January 1, 2013-July 31, 2014). Psychiatric diagnoses were determined via ICD-9 billing codes (see Table 2 for codes). We extracted data (by billing code) for patients with mood, thought, anxiety, and behavioral disorders. Data were extracted from the university EMR system by the institution’s Performance Assessment and Clinical Effectiveness (PACE) office. A total of 190 patients met inclusion criteria. The study was approved by the Institutional Review Board (HUM00090395).
Selected independent variables were extracted from the EMR. These included patient age, insurance type, ethnicity, psychiatric diagnoses, inpatient psychiatric contact (including psychiatric emergency room, inpatient consultation, inpatient psychiatric unit—adult and child/adolescent), and relevant outpatient follow-up at the institution (psychiatry clinic, neurology clinic, primary care, or none of these 3). We also noted if outpatient follow-up was intended within the community mental health system. Outpatient affiliation was determined by text searches and review of progress notes from actual providers, inpatient psychiatry documentation, or emergency department documentation.
We had 3 dependent variables: contraception or infertility (including prescription of oral contraceptive pills, an IUD, prior hysterectomy, or tubal ligation), co-prescription of folic acid, and documentation of provider-patient discussion regarding potential risks associated with VPA use. We performed a text search of all progress notes for key words and information regarding pregnancies, documentation of these discussions, and use of contraception. Terms used in this search included IUD, intrauterine, contracept-, birth control, OCP, salping-, ligation, hysterect-, teratog-, pregnan-, and defect. Chart documentation was assessed as having any or no documentation. Presence of adequate documentation included statements such as, “It was repeatedly emphasized to patient the importance of maintaining a method of birth control given the teratogenicity of valproic acid” or, “We reviewed the side effects of valproic acid in detail.” A generic computer-generated statement (ie, “education was given on treatment plans”) or no documentation at all in the chart was considered insufficient.
If patients had multiple psychiatric diagnoses documented as per billing codes, we assigned one diagnosis based on the following hierarchy: schizophrenia, then bipolar disorder followed by depressive disorders and then other disorders.
Data and Statistical Analysis
Statistical analysis was completed using STATA version 13 (StataCorp). Bivariate analyses using χ2 tests were conducted to assess the relationships between each of our 3 dependent variables and each independent variable. For bivariate analysis, age was dichotomized at the median for statistical analysis. Race was dichotomized as white or nonwhite since the large majority of patients were white. Insurance was dichotomized as private insurance versus public insurance (eg, Medicaid, Medicare) or self-pay. We also calculated the number of documented pregnancies during the study window.
RESULTS
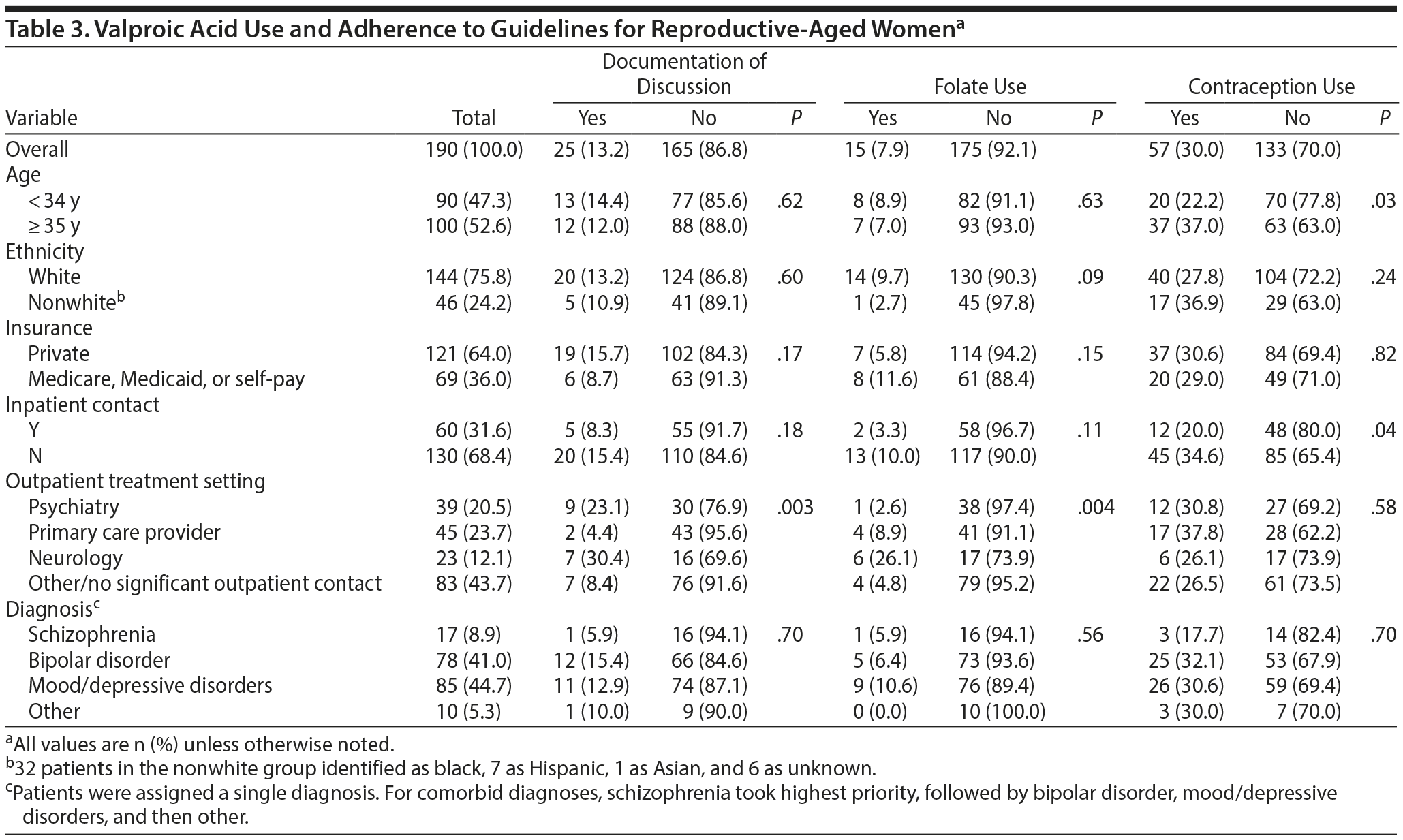
A total of 190 reproductive-aged female patients with at least one psychiatric diagnosis and an active VPA prescription were identified. Table 3 provides bivariate assessments between dependent and independent variables. Patients had a median age of 34 years (SD = 9.5 years) and were predominantly white (76%), and 64% were privately insured. Psychiatric diagnoses included mood/depressive disorders (45%), bipolar disorder (41%), schizophrenia (9%), and other psychiatric illness (5%). Sixty patients (32%) had contact with inpatient psychiatric service. The most common follow-up was outside of our system (n = 83, 44%), and a quarter of patients (n = 45, 24%) received primary care at the medical institution. Only 39 (21%) received follow-up outpatient psychiatric care and 23 (12%) received follow-up outpatient neurologic care at the institution. Thirty-four patients (41%) with follow-up care in the community were community mental health clients.
We found that only 25 women (13.2%) on VPA treatment had adequate documentation regarding teratogenic risk, only 15 (7.9%) were prescribed folate, and only 57 (30%) had documented contraception use.
Based on findings from χ2 analyses, documentation of adequate provider-patient discussions about VPA teratogenicity risk was not associated with patient characteristics with one exception; receiving outpatient services through psychiatry or neurology clinics at the institution (in contrast to receiving psychiatric care primarily through community psychiatry or primary care) significantly increased the likelihood of appropriate documentation of VPA risk (P = .003). Specifically, 16 documented discussions (64%) occurred in institution-based psychiatry or neurology clinics, despite the fact that this group represented only 33% of total study patients. Similarly, folate co-prescription was significantly associated with prescriber specialty. Patients treated at outpatient neurology clinics had the highest rates (n = 6, 26%), and, in contrast, those treated at outpatient psychiatry clinics had the lowest (n = 1, 2.6%, P = .004). Women who had contact with the inpatient psychiatry service were less likely to be using contraception (n = 12, 20%, P = .041). However, women with inpatient psychiatry contact were not more likely to have chart documentation of provider-patient discussions of VPA risk (n = 5, 8%, P = .181), nor did they have greater co-prescription of folate (n = 2, 3%, P = .113). Of patients who were pregnant and on VPA treatment (n = 3), none had documented contraception use, 2 were receiving folate, and all 3 had documentation of provider-patient discussion about the associated teratogenic risks in their chart. After receiving news of pregnancy status in the emergency department, 1 woman reported that she planned to terminate the pregnancy.
DISCUSSION
This exploratory study sought to describe VPA prescribing practices for reproductive-aged women with psychiatric diagnoses who were receiving treatment in a large tertiary care health system. Overall documentation of provider-patient discussion and contraception and folate use was low, and the study results were not associated with patients’ demographics. Where the patients received their care did have a significant impact insofar as patients who received outpatient care in neurology and psychiatry clinics were twice as likely to have a documentation of discussion regarding VPA teratogenicity as women cared for by other providers in the health system (ie, primary care) or outside the system (ie, community). We also found that folate co-prescription with VPA was low and was most commonly present when neurologic care was the outpatient provider type and least likely when psychiatric was the provider type. We found that women who became/were pregnant (although the number was very small) were more likely to be prescribed folate and always have a documentation of discussion regarding VPA teratogenicity. In contrast to expectation, patients who had inpatient psychiatric treatment were less likely to be on contraception and, though not statistically significant, less likely to have chart documentation regarding VPA risk or be prescribed folate. We suspect our findings could generalize to other tertiary care referral centers.
There are several possible explanations for low levels of documentation of discussion and contraception and folate use. First, while there are guidelines in place for VPA prescriptions to childbearing-aged or pregnant women, the guidelines seem inconsistent and vague for helping physicians take care of patients for whom this issue is relevant (Table 1). APA guidelines recommend that women with bipolar disorder who choose to remain on regimens including VPA during pregnancy should receive additional screening for neural tube defects. APA guidelines also recommend that all women with bipolar disorder of reproductive potential who are taking medications be encouraged to practice “effective contraception.”20 The American Congress of Obstetrics and Gynecologists notes the teratogenicity of VPA in their guidelines and recommends that VPA be avoided in pregnancy. NICE is the most definitive, stating in the most recent guidelines for antenatal care from December 2014, “Do not offer valproate for acute or longterm treatment of a mental health problem in women who are planning a pregnancy or are pregnant” (www.nice.org.uk/guidance/cg192/chapter/1-recommendations) and, in their September 2014 guidelines, “Do not offer VPA to young girls or women of childbearing potential.”21 Finally, the American Academy of Neurology and American Epilepsy Society recommend avoidance of VPA, if possible, during pregnancy.6 The Subcommittee of the American Academy of Neurology and the American Epilepsy Society also recommends that, despite unclear benefit24 and given low risk, women taking VPA should be given folic acid,25 and, for the same reasons, NICE guidelines suggest all women receiving antiepileptic drugs take 5 mg of folic acid each day.23
Expert opinions on this matter echo elements of these guidelines and very likely informed them. Expert opinions suggest that valproic acid should be avoided throughout pregnancy and by women of childbearing age who are not using effective contraception,26 and Yonkers et al state that “clinicians should discuss the issue of pregnancy and its management with every patient with bipolar disorder who has childbearing potential, regardless of future reproductive plans.”27(p617) A recent report on global opportunities to prevent birth defects claims an “urgent necessity” to educate neurologists and psychiatrists to eliminate VPA prescriptions among women of childbearing potential.28 Despite limited evidence to suggest that folic acid ameliorates the risk of neural tube defects with VPA, other experts continue to make recommendations reinforcing the importance of folic acid in reproductive-aged women taking antiepileptic agents.29,30 Some experts recommend alternative agents and planned pregnancies when possible to minimize risk—especially given that many of the malformations associated with VPA occur in weeks 4 to 5 of pregnancy.12 There are many important recommendations, but a clear and unequivocal set of required and binding rules of “what to do” and “when to do” is missing, at least in US-based guidelines.
Our findings are in line with other studies showing that guidelines are not routinely followed. A similar retrospective chart review from Scotland identified 67 reproductive-aged female psychiatric patients on VPA treatment and also found low levels of discussions about teratogenic risk on initiation of medication (22%) and low levels of discussions about contraception use (13%).14 Our study thus replicates prior work showing inadequate documentation of patient discussions regarding teratogenicity and contraception use in a larger sample and in a different country and also extends the scope by including additional focus on folate co-prescriptions. Another study found that many reproductive-aged women are prescribed VPA without adequate counseling, and yet another study at Veterans’ hospitals suggested that female veterans overestimated their physicians’ attentiveness to notifying them about teratogenicity of any medication.31-34 After the NICE guidelines were generated, a study in the United Kingdom reviewed charts of all childbearing-aged women on treatment with antiepileptic drugs at 3 teaching hospital psychiatry departments and noted poor adherence to their guidelines.13 A national study reviewing close to 9 million prescriptions over 12 years looked at valproic acid prescribing practices in non-epileptic women and, after noting that 74% of prescriptions written for VPA were for women with psychiatric diagnoses, recommended decreasing valproic acid prescriptions in reproductive-aged women to decrease birth defects and cognitive defects.19 A study of New York State Medicaid claims reviewed 40,000 patients taking antiepileptic drugs and found VPA to be the number one antiepileptic drug prescribed for young women—23.4% of prescriptions.33 Another study looking at psychotropic prescribing practices for the New York State Medicaid population (217,216 charts reviewed) noted that VPA accounted for more than 30% of antiepileptic drugs prescribed for reproductive-aged women and cited this as a “clinically questionable prescribing practice.”34 This finding is particularly concerning when one takes into consideration that the rate of unintended pregnancy in the general population is estimated to be greater than 50%35 and women with bipolar illness are not prescribed contraception at the same rates as their age-matched counterparts.36
Considering known risks and available (yet inconsistent or nonbinding) guidelines in conjunction with apparently less risky alternatives to managing symptoms of serious mental illness in reproductive-aged women, it remains an open question why VPA prescriptions for this population continue—and without adequate provider-patient documentation of informed discussions about teratogenicity and contraception use. Folic acid, given its low risk profile and potential utility, is also not used widely enough. Inherent difficulties in tracking all of our study variables for patients with complex psychiatric care limited this study and may provide some clues as to why our rates of guideline adherence were so low. Many of the patients in the study have Medicare or Medicaid, suggesting, given their age, that they are either impoverished or permanently disabled. A significant number of patients had only brief contact with inpatient psychiatric services, such as exclusively with psychiatric emergency services, and follow-up with community mental health care providers, suggesting severe psychopathology and limited resources. Thus, these patients had no significant follow-up within the medical tertiary care institution, limiting the ability to determine to what extent guidelines may be followed by their community providers; yet, even if such discussions are happening, they are not inquired about during such inpatient encounters and thus not documented. Thus, an important opportunity for securing a follow-through with guidelines seems missed. It also is very plausible that acute psychiatric care with limited institutional follow-up is a trend at tertiary care centers and diminishes follow-through with such guidelines.
Providers may lack the information needed to make informed decisions about how to take care of these patients. Atypical antipsychotics are known to be more effective than antiepileptics in the management of acute mania, but perhaps there are still compelling reasons providers decide to start treatment with mood stabilizers in acute settings.16 Additionally, although there are studies showing that, unlike VPA, antipsychotics are not thought to have major teratogenic potential, most of these studies are small and there are limited data about their use during pregnancy.17 In addition, although all antiepileptic drugs have a dose-dependent increased risk of teratogenicity, it may not be widely known that, for VPA, this teratogenicity is most robust.2
We encountered other significant limitations. We did not include patients who were taking VPA exclusively for neurologic indications. While this reduced the number of patients that we included in this study, it allowed for a more homogeneous sample of psychiatric patients. Given that this was an observational study at a single site with a small sample size, it is also possible that the available data underestimate documented discussions and folate and contraception use. Since many patients received treatment outside of our institution, some may have been prescribed contraception or received counseling about teratogenicity elsewhere, yet this still does not explain the lack of documentation of such. Additionally, though potentially useful in this scenario, we note that folic acid by itself is unlikely to offer the protective benefit needed to ameliorate teratogenic risk. At the same time, we recognize that some patients may take folate that is not recorded or alternatively were prescribed folic acid but did not take it as directed. We also did not collect information about the dosing of VPA or concomitantly prescribed antiepileptic drugs—both of which are linked to overall teratogenic risk. Without pharmacy data confirming prescription refills or laboratory data showing blood levels of medication, it is difficult to determine that the patients who are prescribed VPA are actually taking it. Lastly, our data precluded obtaining demographic information about the prescribers, and thus we are unable to investigate whether male and female prescribers differed in their approach. Future studies would benefit from information on gendered prescription and documentation patterns on VPA for childbearing women.
There is not a 100% chance of exposure to VPA leading to teratogenicity and, despite equally effective alternative agents, there may still be cases in which the benefits of VPA in a reproductive-aged woman outweigh the risks. However, in examining the risks, guidelines, and current clinical practices at a tertiary care center, it seems that, to improve patient safety in this inherently vulnerable population, a new approach is needed. In psychiatry—and particularly for diagnostic entities such as schizophrenia and bipolar disorder—patient behavior, judgment, and insight are inherently linked to disease processes. A patient could easily become pregnant and, depending on her state of mind, unknowingly or carelessly expose her fetus to a teratogenic medication and bring about subsequent serious lifelong medical consequences for her child. This possibility highlights the unique issues of impaired judgment and insight that are central to so many psychopathological processes. These nuanced differences in disease processes are not readily woven into the decision-making process and are hard to standardize. Given the obvious risks and clear alternate options, we recommend alternative measures be considered to ensure reproductive-aged women either are not prescribed VPA or are adequately educated about teratogenicity and contraception when VPA is being recommended. The patient’s spouse/partner or other significantly involved person in the patient’s life should also be included in such discussions. If a reproductive-aged patient must take this medication for some extenuating circumstance, she ought to be taking folic acid with it, and a discussion about contraception needs to be standard practice as well. We wonder whether current standards of trainee and continuing medical education adequately address the complex issue of VPA prescription. Research surveys could investigate the knowledge base and perceived preparedness at various levels of training and lead to establishment of practice-relevant teaching curricula. Another possibility is to consider safeguards such as those used for isotretinoin and clozapine in order to protect fertile women with schizophrenic, bipolar, or depressive disorders when they are using this medically potent yet potentially teratogenic medication. Lastly, electronic ordering systems may be used to establish appropriate warnings for providers who are prescribing VPA to these patients.
In summary, the practitioner should try other mood-stabilizing agents before prescribing VPA to a reproductive-aged woman or girl. If there is a compelling reason or are extenuating circumstances that require use of VPA, we recommend extensive counseling about teratogenic risk, encouraged use of contraception, and concomitant use of folate. Providers should also make a habit of documenting a discussion of teratogenic risk.
Submitted: April 11, 2015; accepted June 30, 2015.
Drug names: clozapine (Clozaril, FazaClo, and others), isotretinoin (Claravis, Amnesteem, and others), valproic acid (Depakene and others).
Disclosure of off-label usage: The authors have determined that, to the best of their knowledge, no investigational information about pharmaceutical agents that is outside US Food and Drug Administration-approved labeling has been presented in this article.
Financial disclosure: Drs Gotlib, Kurlander, Zivin, Riba, and Muzik and Ms Perelstein have no personal affiliations or financial relationships with any commercial interest to disclose relative to the article.
Funding/support: There was no external funding for this study.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
REFERENCES
1. Adab N, Kini U, Vinten J, et al. The longer term outcome of children born to mothers with epilepsy. J Neurol Neurosurg Psychiatry. 2004;75(11):1575-1583. PubMed doi:10.1136/jnnp.2003.029132
2. Tomson T, Battino D, Bonizzoni E, et al; EURAP study group. Dose-dependent risk of malformations with antiepileptic drugs: an analysis of data from the EURAP epilepsy and pregnancy registry. Lancet Neurol. 2011;10(7):609-617. PubMed doi:10.1016/S1474-4422(11)70107-7
3. Gentile S. Risks of neurobehavioral teratogenicity associated with prenatal exposure to valproate monotherapy: a systematic review with regulatory repercussions. CNS Spectr. 2014;19(4):305-315. PubMed doi:10.1017/S1092852913000990
4. Christensen J, Gr׸nborg TK, S׸rensen MJ, et al. Prenatal valproate exposure and risk of autism spectrum disorders and childhood autism. JAMA. 2013;309(16):1696-1703. PubMed doi:10.1001/jama.2013.2270
5. Bromley R, Weston J, Adab N, et al. Treatment for epilepsy in pregnancy: neurodevelopmental outcomes in the child [published online October 30, 2014]. Cochrane Database Syst Rev. doi:10.1002/14651858.cd010236.pub2 PubMed
6. Harden CL, Meador KJ, Pennell PB, et al; American Epilepsy Society. Practice parameter update: management issues for women with epilepsy—focus on pregnancy (an evidence-based review): teratogenesis and perinatal outcomes: report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and American Epilepsy Society. Neurology. 2009;73(2):133-141. PubMed doi:10.1212/WNL.0b013e3181a6b312
7. Wyszynski DF, Nambisan M, Surve T, et al; Antiepileptic Drug Pregnancy Registry. Increased rate of major malformations in offspring exposed to valproate during pregnancy. Neurology. 2005;64(6):961-965. PubMed doi:10.1212/01.WNL.0000154516.43630.C5
8. Meador K, Reynolds MW, Crean S, et al. Pregnancy outcomes in women with epilepsy: a systematic review and meta-analysis of published pregnancy registries and cohorts. Epilepsy Res. 2008;81(1):1-13. PubMed doi:10.1016/j.eplepsyres.2008.04.022
9. Gilboa SM, Broussard CS, Devine OJ, et al. Influencing clinical practice regarding the use of antiepileptic medications during pregnancy: modeling the potential impact on the prevalences of spina bifida and cleft palate in the United States. Am J Med Genet C Semin Med Genet. 2011;157C(3):234-246. PubMed doi:10.1002/ajmg.c.30306
10. Joffe H, Hayes FJ. Menstrual cycle dysfunction associated with neurologic and psychiatric disorders: their treatment in adolescents. Ann N Y Acad Sci. 2008;1135(1):219-229. PubMed doi:10.1196/annals.1429.030
11. Curtis V. Women are not the same as men: specific clinical issues for female patients with bipolar disorder. Bipolar Disord. 2005;7(suppl 1):16-24. PubMed doi:10.1111/j.1399-5618.2005.00190.x
12. Cohen LS. Treatment of bipolar disorder during pregnancy. J Clin Psychiatry. 2007;68(suppl 9):4-9. PubMed
13. Wieck A, Rao S, Sein K, et al. A survey of antiepileptic prescribing to women of childbearing potential in psychiatry. Arch Women Ment Health. 2007;10(2):83-85. PubMed doi:10.1007/s00737-007-0175-y
14. Langan J, Perry A, Oto M. Teratogenic risk and contraceptive counselling in psychiatric practice: analysis of anticonvulsant therapy. BMC Psychiatry. 2013;13(1):234. PubMed doi:10.1186/1471-244X-13-234
15. Lloyd KA. A scientific review: mechanisms of valproate-mediated teratogenesis. Biosci Horiz. 2013;6:hzt003. doi:10.1093/biohorizons/hzt003
16. Cipriani A, Barbui C, Salanti G, et al. Comparative efficacy and acceptability of antimanic drugs in acute mania: a multiple-treatments meta-analysis. Lancet. 2011;378(9799):1306-1315. PubMed doi:10.1016/S0140-6736(11)60873-8
17. Habermann F, Fritzsche J, Fuhlbrück F, et al. Atypical antipsychotic drugs and pregnancy outcome: a prospective, cohort study. J Clin Psychopharmacol. 2013;33(4):453-462. PubMed doi:10.1097/JCP.0b013e318295fe12
18. Johnson KC, LaPrairie JL, Brennan PA, et al. Prenatal antipsychotic exposure and neuromotor performance during infancy. Arch Gen Psychiatry. 2012;69(8):787-794. PubMed doi:10.1001/archgenpsychiatry.2012.160
19. Adedinsewo DA, Thurman DJ, Luo Y-H, et al. Valproate prescriptions for nonepilepsy disorders in reproductive-age women. Birth Defects Res A Clin Mol Teratol. 2013;97(6):403-408. PubMed doi:10.1002/bdra.23147
20. American Psychiatric Association. APA Practice Guidelines for the Treatment of Psychiatric Disorders: Comprehensive Guidelines and Guideline Watches. Vol 1. 1st ed. Arlington, VA: American Psychiatric Association; 2006.
21. Bipolar disorder, the assessment and management of bipolar disorder in adults, children and young people in primary and secondary care: key-priorities-for-implementation. NICE Guidance and Guidelines. NICE Web site. http://www.nice.org.uk/guidance/cg185/chapter/key-priorities-for-implementation. Updated September 2014. Accessed January 5, 2015.
22. ACOG Committee on Practice Bulletins—Obstetrics. ACOG Practice Bulletin: clinical management guidelines for obstetrician-gynecologists number 92, April 2008 (replaces practice bulletin number 87, November 2007): use of psychiatric medications during pregnancy and lactation. Obstet Gynecol. 2008;111(4):1001-1020. 10.1097/AOG.0b013e31816fd910 PubMed
23. The epilepsies: the diagnosis and management of the epilepsies in adults and children in primary and secondary care: 1-recommendations. NICE Guidance and Guidelines. NICE Web site. http://www.nice.org.uk/guidance/cg137/chapter/1-recommendations#women-and-girls-with-epilepsy. Accessed April 20, 2015.
24. Wieck A. The use of anti-epileptic medication in women with affective disorders in early and late pregnancy and during breastfeeding. Curr Womens Health Rep. 2011;7(1):50-57. doi:10.2174/157340411794474739
25. Harden CL, Pennell PB, Koppel BS, et al; American Epilepsy Society. Management issues for women with epilepsy—focus on pregnancy (an evidence-based review), III: vitamin K, folic acid, blood levels, and breast-feeding: Report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsia. 2009;50(5):1247-1255. PubMed doi:10.1111/j.1528-1167.2009.02130.x
26. Valproic acid: long-term effects on children exposed in utero. Prescrire Int. 2009;18(104):253-257. PubMed
27. Yonkers KA, Wisner KL, Stowe Z, et al. Management of bipolar disorder during pregnancy and the postpartum period. Am J Psychiatry. 2004;161(4):608-620. PubMed doi:10.1176/appi.ajp.161.4.608
28. Kancherla V, Oakley GP Jr, Brent RL. Urgent global opportunities to prevent birth defects. Semin Fetal Neonatal Med. 2014;19(3):153-160. PubMed doi:10.1016/j.siny.2013.11.008
29. Begum S, Thomas SV. Women with epilepsy in reproductive age group: special issues and management strategies. J Assoc Physicians India. 2013;61(suppl):48-51. PubMed
30. Weber M, Dib M. Folic acid and prevention of anomalies of foetal neural tube closing in women treated for epilepsy. Rev Neurol (Paris). 2003;159(2):165-170. PubMed
31. Farahi N, Zolotor A. Recommendations for preconception counseling and care. Am Fam Physician. 2013;88(8):499-506. PubMed
32. Schwarz EB, Mattocks K, Brandt C, et al. Counseling of female veterans about risks of medication-induced birth defects. J Gen Intern Med. 2013;28(suppl 2):S598-S603. PubMed doi:10.1007/s11606-012-2240-0
33. Wisner KL, Leckman-Westin E, Finnerty M, et al. Valproate prescription prevalence among women of childbearing age. Psychiatr Serv. 2011;62(2):218-220. PubMed doi:10.1176/ps.62.2.pss6202_0218
34. Essock SM, Covell NH, Leckman-Westin E, et al. Identifying clinically questionable psychotropic prescribing practices for Medicaid recipients in New York State. Psychiatr Serv. 2009;60(12):1595-1602. PubMed doi:10.1176/ps.2009.60.12.1595
35. Finer LB, Zolna MR. Shifts in intended and unintended pregnancies in the United States, 2001-2008. Am J Public Health. 2014;104(suppl 1):S43-S48. PubMed doi:10.2105/AJPH.2013.301416
36. Magalh×£es PV da S, Kapczinski F, Kauer-Sant’ Anna M. Use of contraceptive methods among women treated for bipolar disorder. Arch Women Ment Health. 2009;12(3):183-185. doi:10.1007/s00737-009-0060-y
This PDF is free for all visitors!