A Case of Acute Renal Failure in a Patient Recently Treated With Clozapine and a Review of Previously Reported Cases
Clozapine has been reported to cause acute renal failure due to acute interstitial nephritis. We discuss a case of clozapine-induced acute renal failure and compare it to 7 other cases reported in the literature. We review the signs and symptoms of the hypersensitivity response, such as fever and eosinophilia, caused by clozapine and make recommendations for early detection. Early detection and prompt discontinuation of clozapine can prevent renal damage, as can the avoidance of other nephrotoxic drugs like antibiotics.
Prim Care Companion CNS Disord 2011;13(3):e1-e5
© Copyright 2011 Physicians Postgraduate Press, Inc.
Submitted: October 1, 2010; accepted December 21, 2010.
Published online: May 12, 2011 (doi:10.4088/PCC.10br01091).
Corresponding author: Mary E. Woesner, MD, Department of Psychiatry, 1500 Waters Pl, Bronx, NY 10461 ([email protected]).
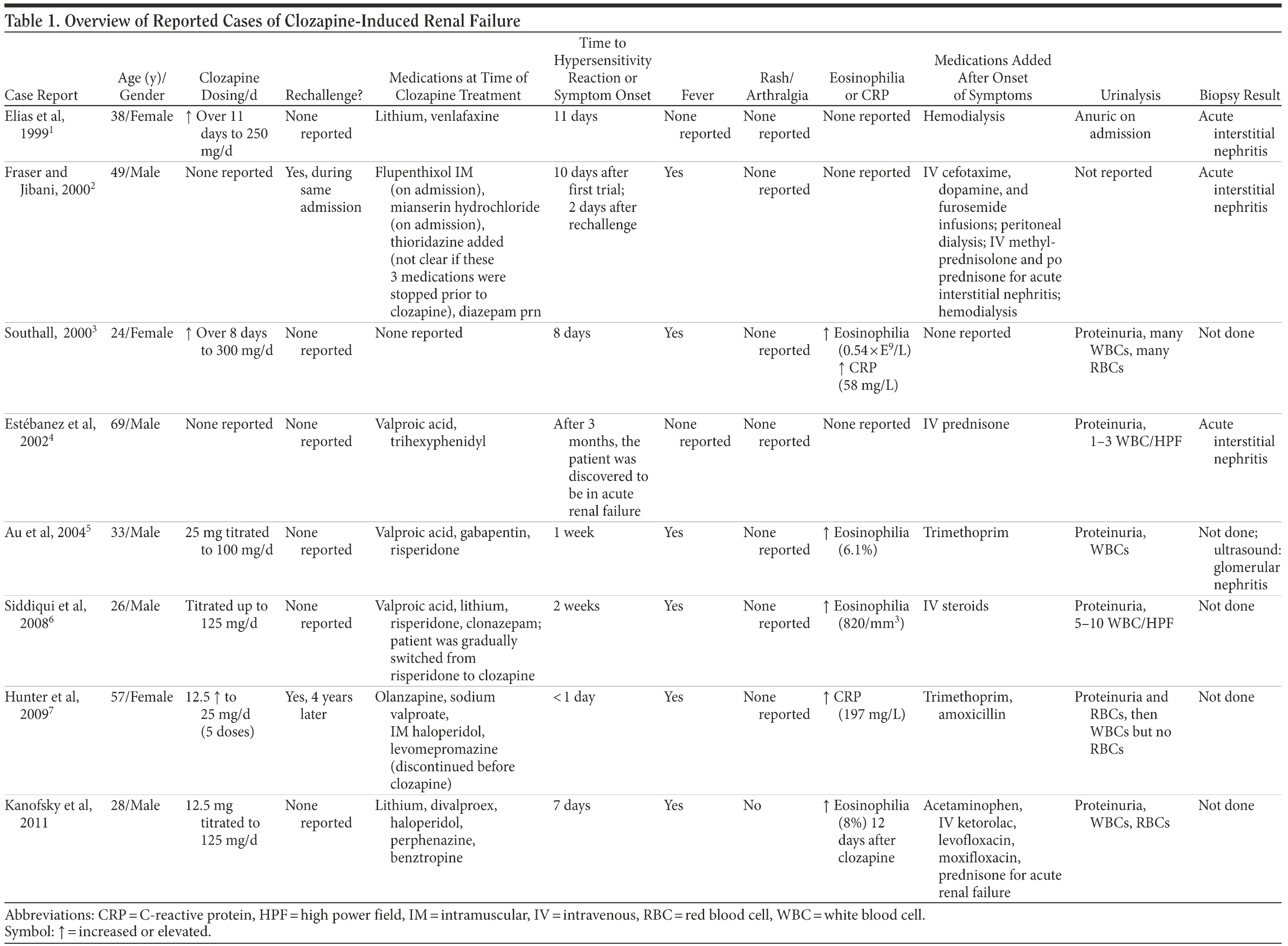
Clozapine has been shown, on rare occasions, to cause acute renal failure due to acute interstitial nephritis.1-7 Despite the growing number of case reports that implicate clozapine as a cause of acute interstitial nephritis, many clinicians remain unaware of this fact. We report on a case of acute renal failure associated with the onset of clozapine therapy. We also speculate that clozapine may have interacted with other medications to create the conditions for acute renal failure. Table 1 gives an overview of previously reported cases.

CASE REPORT
Mr A, a 28-year-old white man with schizoaffective disorder, is hospitalized in 2008 at an acute psychiatric facility for suicidal ideation and a worsening thought disorder. Three weeks prior to admission, he is reported to have a normal eosinophil count at 400/mm3 (4.1%). On admission, his psychiatric medications are lithium, divalproex, haloperidol, perphenazine, and benztropine. He is taking amoxicillin clavulanate for a foot infection. His white blood cell count is elevated at 13,400/mm3, eosinophil count is mildly elevated at 670/mm3 (5%), and neutrophil count is elevated at 10,320/mm3 (75%). His lithium level is 0.49 mEq/L, serum urea nitrogen is 11 mg/dL, serum creatinine is 0.7 mg/dL, total serum protein is 7.4 g/dL, serum albumin is 4.7 mg/dL, and serum calcium is 10.1 mg/dL, all within normal range. Urinalysis on admission is within normal limits.
Clinical Points
- If fever or eosinophilia occurs after initiating clozapine treatment, monitor the patient’s renal function.
- Proteinuria may be the earliest indicator of renal damage.
- Treatment with an antibiotic could exacerbate or precipitate renal failure.
Day 6 of hospitalization: Clozapine 12.5 mg/d is added to Mr A’s medication regimen and is eventually titrated to 125 mg/d. His white blood cell count is normal at 9,100/mm3 and eosinophil count is normal at 400/mm3 (4.4%).
Day 9: Amoxicillin clavulanate is discontinued.
Days 10-12: Mr A complains of headache and is medicated 4 times with acetaminophen.
Day 12: Mr A becomes febrile, with a temperature spike to 103°F. At this time, Mr A is transferred to a medical emergency room (ER) with a chief complaint of fever. Upon admission to the ER, complete blood cell count, serum urea nitrogen (8 mg/dL), and serum creatinine (0.7 mg/dL) are normal and serum calcium is low (7.6 mg/dL). The white blood cell count is 6,800/mm3, and the eosinophil count is not elevated at 140/mm3 (2%). A viral infection is diagnosed and treated with a 30-mg intravenous dose of the nonsteroidal anti-inflammatory drug ketorolac. Urinalysis and serum albumin and total protein levels are not obtained. Mr A is discharged back to the psychiatric facility.
Day 13: Mr A is transported back to the medical ER 17 hours later when the fever returns. Urinalysis reveals 3+ proteinuria and 10-20 white blood cells/high power field. A urine eosinophil count is not performed. Mr A’s total serum protein is low at 5.6 g/dL and serum calcium is low at 8.1 mg/dL. Serum albumin is 3.5 mg/dL and has fallen from the day 1 level (4.7 mg/dL) consistent with the protein loss found in the urinalysis. Liver enzymes and bilirubin levels are normal. The serum urea nitrogen level is 14 mg/dL, serum creatinine level is 1.1 mg/dL (increased from the baseline of 0.7 mg/dL), and serum lithium level is within therapeutic range at 0.97 mmol/L. The white blood cell count is 9,400/mm3 with no eosinophilia at 290/mm3 (3%). A urine culture is taken; Mr A is presumed to have a urinary tract infection and is treated with 1 to 2 doses of the antibiotic levofloxacin and an additional IV dose of ketorolac. The clozapine dose at this time is 125 mg/d and is discontinued. Mr. A is returned to the acute care psychiatric hospital.
Days 13-15: In the psychiatric facility, the results of the urine culture are reported as negative. The patient continues to receive prn acetaminophen.
Day 15: Mr A is afebrile, but the antibiotic moxifloxacin is added to treat a suspected sinus or pulmonary infection. No respiratory symptoms are recorded.
Day 16: The white blood cell count is elevated at 13,200/mm3 and eosinophil count is minimally elevated at 530/mm3 (4%); however, 5 hours later, it rises to 910/mm3 (8%). The neutrophil count is 8,550/mm3 (77%). The lithium level is 1.9 mEq/L and serum creatinine and serum urea nitrogen levels eventually reach 7.1 mg/dL and 92 mg/dL, respectively. Urinalysis reveals 2+ proteinuria and 10-20 white blood cells/high power field. Blood and urine cultures are negative. A urine protein-creatinine ratio is not obtained. Acute renal failure is diagnosed, and Mr A is admitted to the medical facility. At the medical facility, lithium and moxifloxacin are discontinued, and the patient is hydrated and given prednisone 60 mg with slow taper. No renal biopsy is performed.
Mr A recovers from his acute renal failure and is discharged on divalproex and perphenazine. His schizoaffective disorder remains in remission.
DISCUSSION
This patient had longstanding schizoaffective disorder and had been treated with lithium and divalproex. He was placed on clozapine and had a sensitivity reaction within 7 days, with a fever of 103°F. There was no skin rash or arthralgia. He was diagnosed with acute renal failure within 11 days of starting clozapine. Clozapine and lithium were discontinued. No renal biopsy was obtained, and the clinical picture of the acute renal failure was suggestive of drug-induced acute interstitial nephritis.
Acute interstitial nephritis is a disease with heterogeneous clinical manifestations: macular or maculopapular skin rash, fever, proteinuria, sterile pyuria, arthralgias, and eosinophilia. Acute interstitial nephritis is induced by different medications, often with an incomplete clinical picture. Each of the typical symptoms is usually present in fewer than 50% of patients, and all of them together are present in fewer than 5% of cases. Acute interstitial nephritis can be infection related, drug induced, or idiopathic.8
No acute infection was discovered in Mr A. His foot infection resolved, as evidenced by a lack of clinical symptoms and the resumption of normal white blood cell counts on days 6, 12, and 13 of hospitalization. He was not known to have any systemic diseases, such as lupus, that can cause acute interstitial nephritis. However, he was taking nephrotoxic medications, several of which are known to cause acute interstitial nephritis.
It is of great interest that some signs of acute interstitial nephritis/acute renal failure (proteinuria, hypoproteinemia, white cells in the urine, and hypocalcemia) developed coincident with the clozapine-induced fever. This preceded the presence of frank acute renal failure (serum urea nitrogen and serum creatinine levels were not elevated). It is conceivable that the 1 dose of ketorolac given during the first ER visit could be responsible for the urine and serum protein abnormalities noted during the second visit. Ketorolac can be nephrotoxic. No urinalysis and no serum protein levels were taken during the first ER visit; however, a low serum calcium level of 7.6 mg/dL was noted. This points to clozapine, rather than ketorolac, as the toxic agent, because the fever and the low serum calcium level preceded the dose of ketorolac given during the first ER visit. Hypocalcemia is a frequent consequence of a drop in serum proteins, which includes a drop in calcium-binding protein moieties. At the second ER visit, there was evidence of heavy hyperproteinuria and sterile pyuria. Both are common in allergic interstitial nephritis and are compatible with our clozapine hypothesis and the facts of this case. We believe this case is unique in documenting the evolution of a clozapine-induced episode of acute interstitial nephritis.
The close temporal relationship of the initiation of clozapine treatment to the onset of renal failure and development of fever prior to the initiation of other medications point to clozapine being the possible culprit in the case of Mr A. In published cases, the onset of hypersensitivity reactions (eosinophilia and/or fever) occurred from 1 to 2 days with clozapine rechallenge,2,7 and up to 14 days after initial exposure to clozapine.1-3,5,6 This is consistent with our finding of 7 days.
We do not believe that the lithium precipitated the acute renal failure. The patient took lithium for years without renal complications. Classically, lithium causes a chronic insidious nephritis, not an acute and precipitous change in renal function. However, we cannot rule out the possibility of an interaction between clozapine and lithium. In 2 published cases,1,6 the patient was taking lithium and clozapine at the time of onset of acute interstitial nephritis/acute renal failure. Sodium valproate has also been reported as an agent responsible for acute interstitial nephritis.9-11 In our case, as was true with lithium, sodium valproate was given for an extended period prior to the onset of renal failure, and the patient continued sodium valproate after discharge without any adverse reactions. In 3 published cases,4-6 patients were taking sodium valproate at the time of onset of acute interstitial nephritis/acute renal failure. As in our case, there is 1 other published case report6 of a patient who developed acute renal failure while taking a combination of lithium, sodium valproate, and clozapine.
Amoxicillin/clavulanic acid can also cause acute interstitial nephritis, but the patient’s 10-day course of therapy was discontinued 3 days prior to the onset of fever. As noted, ketorolac can be nephrotoxic.12 However, fever and hypocalcemia preceded the dose of ketorolac given during the first ER visit.
We used the Naranjo scores13 to estimate the probability of an acute interstitial nephritis drug reaction for each of the suspected medications, and the scores are as follows:
1. Clozapine: score 4
2. Ketorolac: score 2
3. Moxifloxacin: score 0
4. Levofloxacin: score -1
5. Sodium valproate: score -3
The Naranjo score is interpreted as follows: 1-4 = possible causative drug, ≤ 0 = doubtful. The highest score points to the most likely offending agent. This suggests that clozapine is the drug most likely to have caused acute interstitial nephritis in Mr A.
There is the possibility that clozapine sensitizes the kidneys to the effects of other drugs. For instance, Fraser and Jibani2 report on the case of a patient who was treated with clozapine and developed renal failure. Clozapine was discontinued, and he was placed on the antibiotic cefotaxime in response to an elevated white blood cell count. Many antibiotics have a relatively high risk of causing acute interstitial nephritis.9 In this case,2 the patient’s renal function deteriorated after the initiation of cefotaxime, and he was placed on peritoneal dialysis. Did the cefotaxime cause a greater loss of renal function? In at least 2 other case reports,5,7 antibiotics were prescribed around the time acute renal failure was diagnosed. Was the antibiotic causative? Was it a contributing factor?
In our case, clozapine was acknowledged as having caused a sensitivity reaction (high fever), but the worsening of the renal failure occurred after the antibiotics levofloxacin and moxifloxacin were added to the medication regimen. There are case reports of nephrotoxicity (acute interstitial nephritis) associated with moxifloxacin use,14,15 and levofloxacin has been reported to cause acute interstitial nephritis as well.8 Did these antibiotics add to the nephrotoxicity of clozapine? Did the kidneys fail only after being exposed to 2 or more medications that could potentially cause acute renal failure? There is speculation that the cause of acute interstitial nephritis could be “multifactorial,” and, in some instances, be the result of drug-drug interactions.16
Drug-drug interactions have been reported in cases of adverse drug reactions as possible contributing factors. For example, patients with drug-induced hypersensitivity syndrome often show unexplained cross-reactivity to multiple drugs with different structures, including those used after the onset of symptoms, frequently leading to the deterioration of the clinical picture.17 Moreover, semisynthetic penicillins, ampicillins, and amoxicillin have only rarely been reported to cause hepatotoxicity when used alone. However, when amoxicillin is used in combination with clavulanic acid, the risk of cholestasis substantially increases.18 It is plausible that clozapine is more likely to cause acute interstitial nephritis in combination with certain drugs, including the antibiotics prescribed and reported in the case reports and in our case. These drugs affect metabolizing potential or modify drug transport by tubuloendothelial cells or could reduce renal clearance. Such changes have been reported to increase the risk for the development of acute interstitial nephritis, for example, in older patients with decreased renal clearance.8
CONCLUSIONS
It is important to try to predict and prevent serious adverse reactions. Mild eosinophilia preceded and later frank eosinophilia coincided with acute renal failure in our patient and was involved in at least 3 other case reports.3,5,6 We recommend that patients taking clozapine have their renal function monitored if there is an elevated or spiking eosinophil count, which will be noted in the weekly complete blood count. Fever is the most commonly mentioned hypersensitivity reaction, occurring in at least 5 of the 7 previous cases2,3,5−7 and in our own case. Urinalysis can provide important diagnostic information. Proteinuria and/or white blood cells in the urine may be the earliest indicators of acute interstitial nephritis/acute renal failure. This was true for our patient, and they were present in at least 5 of the previous cases.3-7
Caution is recommended when adding other medications that cause acute interstitial nephritis/acute renal failure. Also, be cautious when interpreting fever as a sign of infection and adding an antibiotic without clear evidence of infection. As noted, fever can be a sign of a hypersensitivity reaction and not infection.
In summary, clozapine is often the only effective treatment for patients with chronic, nonresponsive psychotic illness. There is mounting evidence that, on rare occasions, acute interstitial nephritis/acute renal failure occurs as a result of a sensitivity reaction to clozapine. More needs to be done to educate physicians about this serious complication and to untangle its etiology.
Drug names: benztropine (Cogentin and others), cefotaxime (Claforan and others), clonazepam (Klonopin and others), clozapine (Clozaril, FazaClo, and others), diazepam (Diastat, Valium, and others), divalproex (Depakote and others), furosemide (Lasix and others), gabapentin (Neurontin and others), haloperidol (Haldol and others), ketorolac (Acular, Acuvail, and others), levofloxacin (Levaquin, Quixin, and others), lithium (Lithobid and others), moxifloxacin (Vigamox, Moxeza, and others), olanzapine (Zyprexa), prednisolone (Prelone and others), risperidone (Risperdal and others), valproic acid (Depakene, Stavzor, and others), venlafaxine (Effexor and others).
Author affiliations: Bronx Psychiatric Center (Drs Kanofsky, Woesner, and Gittens); Department of Psychiatry and Behavioral Sciences, Albert Einstein College of Medicine, Bronx (Drs Kanofsky and Woesner); Department of Psychiatry, Weill Cornell Medical College, New York (Dr Harris); New York Medical College, St Vincent’s Hospital Westchester, Harrison (Dr Kelleher); and Department of Allergy and Immunology, Albert Einstein College of Medicine, Montefiore Medical Center, Bronx (Dr Jerschow), New York.
Potential conflicts of interest: None reported.
Funding/support: None reported.
Acknowledgment: The authors wish to thank Deborah R. Liss, MD (Philadelphia, Pennsylvania), for help with editing and translation. Dr Liss reports no conflicts of interest related to the subject of this article.
REFERENCES
1. Elias TJ, Bannister KM, Clarkson AR, et al. Clozapine-induced acute interstitial nephritis. Lancet. 1999;354(9185):1180-1181. PubMed doi:10.1016/S0140-6736(99)01508-1
2. Fraser D, Jibani M. An unexpected and serious complication of treatment with the atypical antipsychotic drug clozapine. Clin Nephrol. 2000;54(1):78-80. PubMed
3. Southall KE. A case of interstitial nephritis on clozapine. Aust N Z J Psychiatry. 2000;34(4):697-698. PubMed
4. Estébanez C, Fernסndez Reyes MJ, Sסnchez Hernסndez R, et al. Acute interstitial nephritis caused by clozapine. Nefrologia. 2002;22(3):277-281. PubMed
5. Au AF, Luthra V, Stern R. Clozapine-induced acute interstitial nephritis. Am J Psychiatry. 2004;161(8):1501. PubMed doi:10.1176/appi.ajp.161.8.1501
6. Siddiqui BK, Asim S, Shamim A, et al. Simultaneous allergic interstitial nephritis and cardiomyopathy in a patient on clozapine. NDT Plus. 2008;1(1):55-62. doi:10.1093/ndtplus/sfm003
7. Hunter R, Gaughan T, Queirazza F, et al. Clozapine-induced interstitial nephritis: a rare but important complication: a case report. J Med Case Reports. 2009;3(1):8574. PubMed doi:10.1186/1752-1947-0003-0000008574
8. Keller M, Spanou Z, Pichler WJ. Drug-induced interstitial nephritis. In: Pichler WJ. Drug Hypersensitivity. Basel, Switzerland: Karger; 2007:295-305.
9. Rossert J. Drug-induced acute interstitial nephritis. Kidney Int. 2001;60(2):804-817. PubMed doi:10.1046/j.1523-1755.2001.060002804.x
10. Lin C-Y, Chiang H. Sodium-valproate-induced interstitial nephritis. Nephron. 1988;48(1):43-46. PubMed doi:10.1159/000184867
11. Yoshikawa H, Watanabe T, Abe T. Tubulo-interstitial nephritis caused by sodium valproate. Brain Dev. 2002;24(2):102-105. PubMed doi:10.1016/S0387-7604(02)00007-4
12. Buck ML, Norwood VF. Ketorolac-induced acute renal failure in a previously healthy adolescent. Pediatrics. 1996;98(2, pt 1):294-296. PubMed
13. Naranjo CA, Busto U, Sellers EM, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30(2):239-245. PubMed doi:10.1038/clpt.1981.154
14. Argirov M, Ricken G, Zecher D, et al. Acute interstitial nephritis associated with moxifloxacin use. Clin Ther. 2005;27(8):1260-1263. PubMed doi:10.1016/j.clinthera.2005.08.008
15. Izzedine H, Brocheriou I, Martinez V, et al. Immune reconstitution inflammatory syndrome and acute granulomatous interstitial nephritis. AIDS. 2007;21(4):534-535. PubMed doi:10.1097/QAD.0b013e32802c7bce
16. Lomaestro BM. Fluoroquinolone-induced renal failure. Drug Saf. 2000;22(6):479-485. PubMed doi:10.2165/00002018-200022060-00006
17. Shiohara T, Takahashi R, Kano Y. Drug-induced hypersensitivity syndrome and viral reactivation. In: Pichler WJ. Drug Hypersensitivity. Basel, Switzerland: Karger; 2007:251-266.
18. Cerny A, Bertoli R. Drug allergic liver injury. In: Pichler WJ. Drug Hypersensitivity. Basel, Switzerland: Karger; 2007:278-294.