Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
Case Conference
The Banner Alzheimer’s Institute Case Conference is a weekly event in which physicians and staff discuss challenging and/or teaching cases of patients seen at the Institute’s Memory Disorders Clinic. These conferences are attended by a multidisciplinary group that includes Banner Alzheimer’s Institute dementia specialists, community physicians (internal medicine, family medicine, and radiology), physician assistants, social workers, nurses, medical students, residents, and fellows.
About the Banner Alzheimer’s Institute
The Banner Alzheimer’s Institute located in Phoenix, Arizona, has an unusually ambitious mission: to end Alzheimer’s disease without losing a generation, set a new standard of care for patients and families, and forge a model of collaboration in biomedical research. The Institute provides high-level care and treatment for patients affected by Alzheimer’s disease, dementia, and related disorders. In addition, the Institute offers extensive support services for families and many unique and rewarding research opportunities.
Received: April 22, 2011; accepted April 22, 2011.
Published online: June 30, 2011 (doi:10.4088/PCC.11alz01204).
Prim Care Companion CNS Disord 2011;13(3):e1-e10
© Copyright 2011 Physicians Postgraduate Press, Inc.
AUTHORS
Roy Yaari, MD, MAS, a neurologist, is director of the Memory Disorders Clinic of Banner Alzheimer’s Institute and a clinical professor of neurology at the College of Medicine, University of Arizona, Tucson.
Adam S. Fleisher, MD, MAS, is associate director of Brain Imaging at the Banner Alzheimer’s Institute, a neurologist at the Institute’s Memory Disorders Clinic, and an associate professor in the Department of Neurosciences at the University of California, San Diego.
Anna Burke, MD, is a geriatric psychiatrist and dementia specialist at the Memory Disorders Clinic of Banner Alzheimer’s Institute.
Helle Brand, PA, is a physician assistant at the Memory Disorders Clinic of Banner Alzheimer’s Institute.
Jan Dougherty, RN, MS, is director of Family and Community Services at Banner Alzheimer’s Institute.
James Seward, PhD, ABPP, is a clinical neuropsychologist at Banner Alzheimer’s Institute.
Pierre N. Tariot, MD, a geriatric psychiatrist, is director of Banner Alzheimer’s Institute and a research professor of psychiatry at the College of Medicine, University of Arizona, Tucson.
Corresponding author: Roy Yaari, MD, MAS, 901 E Willetta St, Phoenix, AZ 85006 ([email protected]).
Participate in the following activity, based on a case conference, and answer questions about diagnosis and treatment.
CME Objective
After studying this case, you should be able to:
- Evaluate an older patient with REM sleep behavior disorder for mild cognitive impairment or early-stage dementia and initiate evidence-based treatment
Accreditation Statement
The CME Institute of Physicians Postgraduate Press, Inc., is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Credit Designation
The CME Institute of Physicians Postgraduate Press, Inc., designates this journal-based CME activity for a maximum of 1.0 AMA PRA Category 1 Creditâ„¢. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Date of Original Release/Review
This educational activity is eligible for AMA PRA Category 1 Credit through June 30, 2014. The latest review of this material was June 2011.
Financial Disclosure
All individuals in a position to influence the content of this activity were asked to complete a statement regarding all relevant personal financial relationships between themselves or their spouse/partner and any commercial interest. The CME Institute has resolved any conflicts of interest that were identified. In the past year, Larry Culpepper, MD, MPH, Editor in Chief, has been a consultant for AstraZeneca, Labopharm, Pfizer, and Trovis; has been a member of the speakers/advisory board for Merck; and has held stock in Labopharm. No member of the CME Institute staff reported any relevant personal financial relationships. Faculty financial disclosure appears at the end of the activity.
HISTORY OF PRESENTING ILLNESS
A 66-year-old man presented for a new patient evaluation at the Banner Alzheimer’s Institute with his wife, who supplemented the clinical history. The chief complaint was “memory problems.” He had been laid off from work 6 years before, as his company was downsizing. At that time, there were no cognitive changes noted, although there were mild depressive symptoms for which the patient was seeing a therapist. Approximately 2 years ago, he began having mild word-finding and name-finding difficulties. He would misplace items including keys. Previously an avid traveler, he became disinterested in traveling and even seemed somewhat fearful of doing so.
These issues gradually worsened over time. He began to repeat questions and stories. At the time of evaluation, the patient was independent in most activities of daily life. He continued to manage their personal finances. His wife noted that he was “slower with numbers” when preparing taxes. He had recently missed a bill payment, which was unusual for him. The patient admitted to his wife that he became lost while driving at least 1 time, and she noted decreased confidence with geographic orientation. He had some difficulty parking straight in parking spaces and not fully pulling the car in all the way. There were no significant safety concerns while driving. He had some difficulty remembering codes for voice mail and home security. He remained able to do all his household chores, operate his computer and e-mail, and exercise regularly.
Within the last 2 years, he developed episodes during which he would “act out his dreams,” thrashing during sleep and even falling out of bed. His primary care physician initiated mirtazapine for sleep concerns, but this made him feel like a “zombie,” and he “could not function.” The mirtazapine was discontinued with resolution of the undesired effects. These sleep episodes occurred every 2 weeks, and his wife could no longer share the bed with him. He had become increasingly irritable and apathetic. He developed a short temper, which was uncharacteristic. The patient stated that he was aware of cognitive changes, specifically with memory and spelling, which caused anxiety.
He had no visual hallucinations or psychosis. There was no physical aggression or socially inappropriate behavior. He stated that he would often experience light-headedness when standing quickly and had “had that for years.” There was no history of constipation, erectile dysfunction, visual hallucinations, or gait changes; although, he felt that his gait might be a little bit more unsteady than in the past.
PAST MEDICAL HISTORY
The patient had a history of hypercholesterolemia and seasonal allergies. He had what appeared to be an undiagnosed rapid eye movement (REM) sleep behavior disorder. The patient had an appendectomy and tonsillectomy in the past.
ALLERGIES
The patient was allergic to sulfamethoxazole and trimethoprim (caused hives). He had an adverse reaction with mirtazapine, as noted above.
MEDICATIONS
The patient’s current medications were rosuvastatin, cetirizine, and fluticasone nasal spray. He also took fish oil, aspirin 325 mg, a multivitamin, and vitamin E 400 IU.
SOCIAL HISTORY
The patient completed a PhD in a physical science and attended prestigious universities for both undergraduate and graduate school. He worked as a physical scientist and retired in 2004 when his company downsized. He lived with his wife. There was no significant history of alcohol or tobacco use.
FAMILY HISTORY
The patient had a maternal grandmother with “dementia.” The patient’s parents died in their 80s but were noted to be cognitively intact. His father had a similar “sleep thrashing” in his later years, but there was no evidence of a dementia.
Original material is selected for credit designation based on an assessment of the educational needs of CME participants, with the purpose of providing readers with a curriculum of CME activities on a variety of topics from volume to volume. This special series of case reports about dementia was deemed valuable for educational purposes by the Publisher, Editor in Chief, and CME Institute Staff. Activities are planned using a process that links identified needs with desired results.
To obtain credit, read the material and go to PSYCHIATRIST.COM to complete the Posttest and Evaluation online.
The DSM-IV (American Psychiatric Association, 2000) defines dementia as multiple cognitive deficits that include memory impairment and at least 1 of the following cognitive disturbances: aphasia, apraxia, agnosia, or a disturbance in executive functioning. The cognitive deficits must be sufficiently severe to cause impairment in social or occupational functioning and must represent a decline from a previously higher level of functioning. A diagnosis of dementia should not be made if the cognitive deficits occur exclusively during the course of a delirium (American Psychiatric Association, 2000).
Mild cognitive impairment refers to cognitive impairment that does not meet the criteria for normal aging or dementia because the cognitive impairment does not impair activities of daily living. Several criteria for mild cognitive impairment have been proposed (Voisin et al, 2003). Originally, mild cognitive impairment emphasized memory impairment as a precursor state for Alzheimer’s disease (Petersen et al, 1999). It then became apparent that mild cognitive impairment is a heterogeneous entity that affects other cognitive domains and includes the prodromal stages of other dementias. The diagnostic criteria for mild cognitive impairment are not exact and require subjectivity in determining whether a cognitive impairment is present or what constitutes impairment in activities of daily living.
Based on the clinical history alone, do you think:
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. He meets criteria for dementia | 60% |
| B. He is likely to be cognitively normal | 0% |
| C. He possibly has mild cognitive impairment | 40% |
| D. His cognitive issues are likely due to an underlying psychiatric disorder | 0% |
The conference attendees who chose option A stated that there was a history of a decline in social/occupational functioning in that the patient stopped traveling, was having difficulty with codes for voice mail and security, possibly needed assistance with directions when driving, and was showing some subtle changes with finances. Those who chose C felt that, in general, symptoms were quite mild and did not interfere with daily functioning. The treating physician felt that the patient best met criteria for mild cognitive impairment. This split demonstrates the subjectivity and thus interrater variability in diagnosing mild cognitive impairment.
Based on the clinical history alone, what would you expect his Mini-Mental State Examination (MMSE) score to be?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. 28-30 | 50% |
| B. 25-27 | 40% |
| C. 22-24 | 10% |
| D. 19-21 | 0% |
| E. 15-18 | 0% |
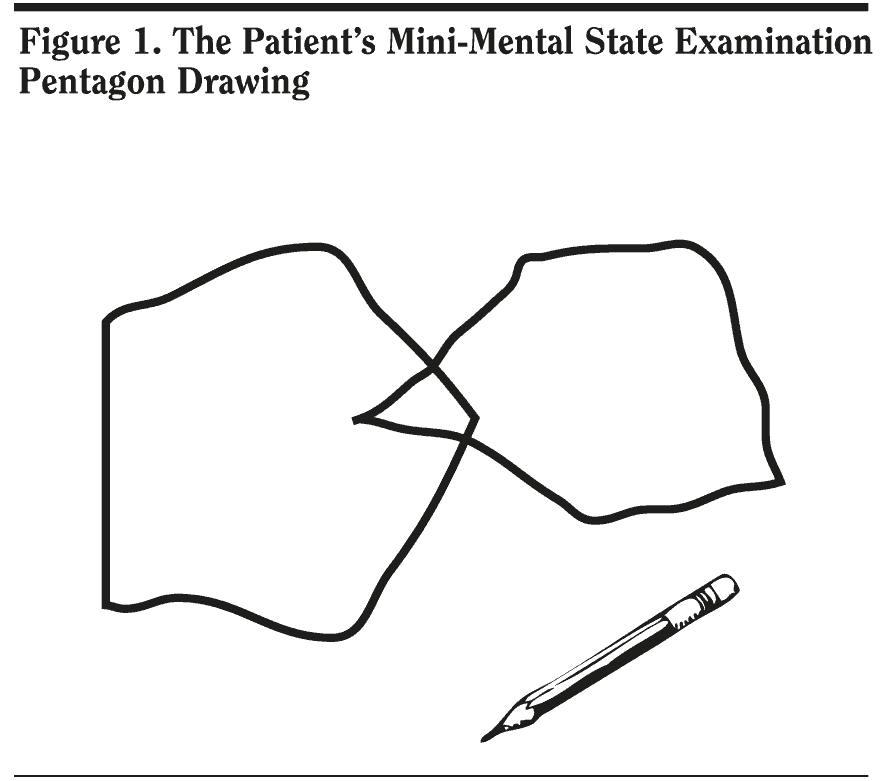
Most responses from case conference attendees were in the upper ranges, indicating that the group felt the patient’s cognitive symptoms were mild on the basis of clinical history. The patient’s MMSE score was 29/30. He missed 1 point on delayed recall. His pentagon copy and written sentence from the MMSE are shown in Figure 1 and 2.
Based on the clinical history alone, what would you expect his Montreal Cognitive Assessment (MoCA) score to be?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. 28-30 | 50% |
| B. 25-27 | 40% |
| C. 22-24 | 10% |
| D. 19-21 | 0% |
| E. 15-1 | 0% |
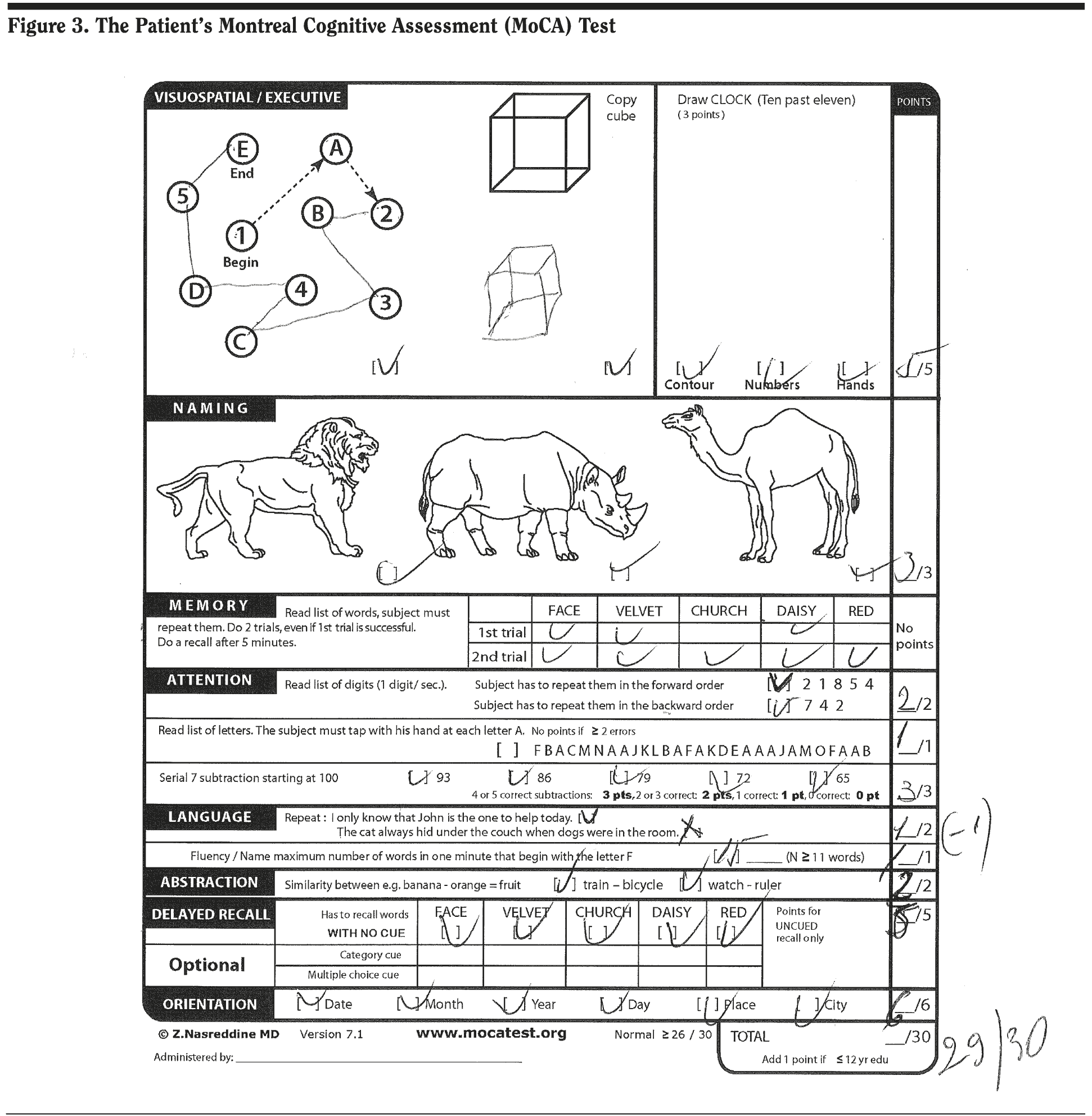
Most responses from those present at the case conference were in the upper ranges, indicating that the group felt the patient’s cognitive symptoms were mild on the basis of the clinical history. His MoCA score was 29/30, missing 1 point on language repetition as shown in Figure 3.
Based on the clinical history alone, what would you expect his Category Retrieval Test score to be?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. 0-5 | 0% |
| B. 6-10 | 0% |
| C. 10-15 | 80% |
| D. 15-20 | 20% |
| E. 21-25 | 0% |
Again, most responses from case conference attendees were in the upper ranges, indicating that the group felt the patient’s cognitive symptoms were mild on the basis of the clinical history. His Category Retrieval Test score was 20, with no repetitions.
PHYSICAL EXAMINATION
The patient’s vital signs were blood pressure: 120/70 mm Hg, pulse: 68 bpm, height: 5 ft 9 in, and weight: 204.3 lb. The patient’s general physical examination was unremarkable.
NEUROLOGIC EXAMINATION
The patient’s neurologic examination was unremarkable except for bilateral hearing aids, occasional bilateral upper extremity arrests with rapid alternating movements, the presence of a mild snout reflex, and mild postural instability with the pull test.
NEUROLOGIC EXAMINATION DISCUSSION
Different dementias may be associated with various physical examination findings. However, most often the physical examination is normal in the early stages. Some subtle general findings can include frontal release signs such as a positive snout, glabellar, or palmomental reflex (Links et al, 2010). Postural instability is tested clinically with the pull test, during which the examiner stands behind the patient and firmly pulls the patient backward by the shoulders after explaining the procedure. Patients with normal postural reflexes are able to maintain balance by stepping backward in 1 or 2 steps. Patients with impaired postural reflexes, as seen in Parkinson’s disease and parkinsonian diseases, are prone to fall if not caught by the examiner or take multiple steps backward. Initially, a positive pull test may be the only sign of balance impairment, without evidence of gait changes or falls (Bronte-Stewart et al, 2002; Smithson et al, 1998).
The clinical history and examination revealed the following: cognitive changes, REM sleep behavior disorder, postural instability, possible orthostatic hypotension per patient report, and mild extrapyramidal features. Given these findings, if the patient did have an early progressive neurodegenerative condition, what condition would be most likely?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Normal pressure hydrocephalus | 0% |
| B. Frontotemporal dementia | 0% |
| C. Lewy body dementia | 70% |
| D. Alzheimer’s disease | 30% |
| E. Progressive supranuclear palsy | 0% |
IMPRESSION at first visit
The treating physician felt that, although the patient does not meet criteria for dementia, he may be in the prodromal stages of a Lewy body dementia. In addition to dementia, features commonly observed in Lewy body dementia include visual hallucinations, parkinsonism, dysautonomia, cognitive fluctuations, REM sleep behavior disorder, and neuroleptic sensitivity (McKeith et al, 2005). Whereas the dementia in Alzheimer’s disease typically initially presents with memory loss as the predominant feature, initial cognitive deficits in Lewy body dementia are usually in visuospatial function (Salmon et al, 1996). REM sleep disorder may be an independent, idiopathic condition but could precede other symptoms of Lewy body dementia or other dementias by many years (Iranzo et al, 2006). The treating physician’s overall impression at the first visit was as follows:
The patient is a 66-year-old male who presents for a cognitive evaluation. The results of the cognitive testing in conjunction with the clinical history do not suggest a clear diagnosis. On the basis of the patient’s clinical history, he may meet criteria for early dementia versus mild cognitive impairment; however, the patient’s cognitive testing was essentially within normal limits. It is noted that his baseline cognition is at a very high level, given his educational and professional attainment. Further testing will be initiated. The patient has a recent onset of REM sleep behavior disorder, what is described as light-headedness if he stands up too quickly, some difficulties parking straight, and mild changes with gait stability. This brings into question whether the patient may have the prodrome of a possible Lewy body dementia.
What tests should be ordered at this time?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Structural brain scan, TSH, vitamin B12, and RPR | 0% |
| B. Structural brain scan, CBC, CMP, TSH, vitamin B12 | 10% |
| C. Neuropsychological testing | 0% |
| D. PET scan | 0% |
| E. B and C | 70% |
| F. B, C, and D | 20% |
The treating physician ordered formal neuropsychological testing, a noncontrast magnetic resonance image (MRI) scan of the brain, CMP, CBC, vitamin B12, and TSH (answer: E). One clinician present at the case conference suggested ordering an erythrocyte sedimentation rate test in addition to these tests to screen for vasculitis or other autoimmune processes. Most in the case conference group agreed. Those who thought a PET scan should be ordered felt that it would be interesting to see if there was objective evidence of a dementia process. Current dementia guidelines do not recommend routine fluorodeoxyglucose PET scans in dementia evaluations (Knopman et al, 2001). It is useful for distinguishing clinically ambiguous cases of Alzheimer’s disease versus frontotemporal dementia and is reimbursable under Medicare for this purpose. The treating physician was not considering frontotemporal dementia in the differential diagnosis and did not feel that it was clinically indicated. Guidelines for a routine dementia workup include CBC, CMP, vitamin B12, and TSH tests and structural brain imaging with either MRI or computed tomography (Knopman et al, 2001). Neuropsychological testing is generally indicated in clinically ambiguous cases.
Should a cholinesterase inhibitor be started at this time?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Yes | 10% |
| B. No | 90% |
Most attendees at the case conference did not think a cholinesterase inhibitor was indicated at this time, but for different reasons. Some felt that the REM sleep behavior issues should be pharmacologically addressed as a higher priority, and others agreed that the highest priority should be to address the irritability and anxiety first. Others felt that in this case, since the diagnosis was most likely mild cognitive impairment, a cholinesterase inhibitor was not necessarily indicated (cholinesterase inhibitor treatment is not FDA approved for this purpose). Several studies have investigated cholinesterase inhibitors in treatment of mild cognitive impairment, but they did not show significant evidence to support their use (Petersen et al, 2005; Salloway et al, 2004; Doody et al, 2009; Mayor, 2005; Feldman et al, 2007). A donepezil study showed a lower rate of progression to Alzheimer’s disease during the first year, but this finding was not sustained over the 3-year duration (Petersen et al, 2005). Despite the paucity of data, cholinesterase inhibitors are often initiated for mild cognitive impairment in clinical practice. In this case, the treating physician, after discussing the data with the patient, did not initiate a cholinesterase inhibitor.
What pharmacologic treatment should be initiated, if any?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. None at this time | 10% |
| B. Start a cholinesterase inhibitor | 10% |
| C. Start an SSRI | 30% |
| D. Melatonin | 50% |
| E. Clonazepam | 0% |
Half of the group present at the case conference wished to address the REM sleep behavior disorder initially with melatonin. Others felt that an SSRI would be useful in treating the patient’s irritability, apathy, and anxiety. The treating physician opted to monitor symptoms, and no pharmacologic agents were initiated. Some data suggest efficacy of melatonin for treatment of REM sleep behavior disorder, although clonazepam continues to be the most useful agent (Kunz and Mahlberg, 2010).
Given the issues with driving, what should be done at this time?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. No need to discuss driving at this time; the patient does not have dementia | 0% |
| B. Driving must stop; report to the Department of Motor Vehicles if patient refuses | 0% |
| C. Advocate for wife to monitor; suggest formal driving evaluation | 100% |
| D. Restrict driving to within 5 miles from home | 0% |
The treating physician advocated that his wife monitor driving for safety concerns. It was recommended, but not required, that the patient have a formal driving assessment.
The Treating Physician’s Plan at the First Visit
(1) Order formal neuropsychological testing to help assess the patient’s relative strengths and weaknesses and to see if the patient has a cognitive pattern consistent with an early progressive neurodegenerative condition, and if so, which one.
(2) Order an MRI of the brain to rule out intracranial pathology.
(3) Order CBC, CMP, vitamin B12, and TSH tests as part of the cognitive workup.
(4) Hold off on initiation of a cholinesterase inhibitor at this time and discuss further after the testing has been completed.
(5) The patient has had some increased irritability, anxiety, and social withdrawal. We will continue to monitor the mood and consider an antidepressant in the future if indicated.
(6) The possibility of treating the REM sleep behavior disorder was discussed, but since episodes are relatively infrequent, we decided to hold off at the current time.
(7) The importance of optimizing physical, social, and mental activities was discussed.
(8) The issue of driving was discussed. The patient has had some minor issues with parking, and, therefore, it is recommended at this time that he should have a formal driving evaluation. The patient was provided several possibilities wherein testing could be done. His wife was advised to continue to monitor the patient’s driving.
(9) The possibility of participating in clinical research was briefly discussed. The patient and his wife appeared amenable, and we will discuss this further in the future.
(10) The patient will return for a follow-up visit 2 weeks after the neuropsychological testing has been completed.
Formal Neuropsychological Test Analysis and Conclusions
Scores on almost all of the tests given in this evaluation were within normal limits for his age group. However, a closer examination of the scores reveals a pattern of cognitive strengths and weaknesses, which is consistent with the presence of a mild cognitive impairment; although, from strictly a test-based standpoint, the scores may not be low enough to meet formal criteria for this disorder. Of specific concern were his difficulties on both the immediate and delayed recall of a spoken word list, a task which is sensitive to the presence of mild cognitive impairment.
His performance on an IQ test is of note. This test revealed an IQ in the average range, which is less than expected given his stellar occupational and educational attainment. Examination of the individual subtests that contributed to this summary score revealed a pattern of relative strengths on measures that tapped his overlearned, rote knowledge and abilities and relative weaknesses on measures that required novel problem solving or the mental manipulation of information. Thus, this “average” score is misleading.
It is important to stress that although he displayed the previously mentioned pattern of strengths and weaknesses, even his low scores were generally within normal limits for his age group. However, as mentioned, this is judged to be a decrease from higher premorbid abilities.
Diagnostically, when considered in the context of the information contained in his chart, the results of this evaluation are most consistent with the presence of a multidomain mild cognitive impairment, with deficits in memory, executive functioning, and speed of information processing. This pattern of cognitive deficits could be due to either a Lewy body process or an Alzheimer’s disease process; the pattern is not specific enough.
Laboratory Values
The CBC, CMP, TSH, and vitamin B12 tests were all normal.
Noncontrast MRI Brain Scan
Unremarkable study.
PRESENTING ILLNESS UPDATE
The patient returned for follow-up 6 weeks after his initial appointment. The REM sleep behavior disorder increased in frequency. He has had some episodes of vocalizations as well. The patient continues to drive and, per his wife, has had 1 minor incident where he hit the curb when turning around. The patient denied this. His wife notes that he may have some slowing down of his processing speed. His cognitive changes have not significantly affected his day-to-day activities in any way. He continues to have anxiety and irritability.
Given the results of the neuropsychological testing, in conjunction with the clinical history, what diagnosis should be given?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Lewy body dementia | 10% |
| B. Alzheimer’s disease | 0% |
| C. Multiple domain amnestic mild cognitive impairment | 90% |
| D. Depression | 0% |
| E. Cognitively normal | 0% |
| F. Not enough data to give a diagnosis | 0% |
The majority of the group agreed with the interpretation of the neuropsychological testing. Although all test scores were in the normal range for age-matched controls, there was a relative weakness in certain areas, and given his occupational and educational attainment, his scores were expected to be higher. Even though his scores were within normal range, they most likely represent a decline from his baseline.
What pharmacologic treatment should be initiated?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. None at this time | 0% |
| B. Start a cholinesterase inhibitor | 10% |
| C. Start an SSRI | 40% |
| D. Melatonin | 50% |
| E. Clonazepam | 0% |
The group continued to be split between treating the irritability/anxiety with an SSRI versus the REM sleep behavior. The treating physician initiated citalopram 10 mg for 1 week, and then 20 mg thereafter. Once the citalopram was stable for 2 weeks, then melatonin 3 mg qhs would be initiated.
Given the new incident with driving, what action should be taken?
Your colleagues who attended the Banner Alzheimer’s Institute Case Conference answered as follows:
| A. Report to Department of Motor Vehicles; patient should not drive | 0% |
| B. Restrict driving to daytime, within 2 miles | 0% |
| C. Formal driving evaluation | 100% |
| D. Take a driving safety class | 0% |
The treating physician strongly recommended that the patient have a formal driving evaluation. The patient had this evaluation through a private company for safety and liability concerns. He passed this test without difficulty.
PRESENTING ILLNESS UPDATE
We received a phone call that citalopram had made REM sleep behavior disorder “more bizarre.” Melatonin was started 2 weeks after citalopram with no obvious benefit. Melatonin was increased to 6 mg with no obvious benefit and discontinued. Although benzodiazepines are generally not recommended in patients with cognitive disorders, clonazepam 0.5 mg qhs was initiated with no cognitive or other adverse events. Clonazepam 0.75 mg qhs was found to be an optimal dose, controlling the REM sleep behaviors by “98%” per the patient’s wife. One year later, the patient continues to be followed in the Memory Disorders Clinic at the Banner Alzheimer’s Institute. Although there has been subtle cognitive worsening, he continues to meet criteria for a diagnosis of mild cognitive impairment.
Disclosure of off-label usage
The authors have determined that, to the best of their knowledge, clonazepam is not approved by the US Food and Drug Administration for the treatment of REM sleep behavior disorder, and donepezil is not approved for the treatment of MCI.
FINANCIAL DISCLOSURE
Dr Yaari is a consultant for Amedisys Home Health. Dr Tariot is a consultant for Acadia, AC Immune, Allergan, Eisai, Epix, Forest, Genentech, MedAvante, Memory Pharmaceuticals, Myriad, Novartis, Sanofi-Aventis, Schering-Plough, and Worldwide Clinical Trials; has received consulting fees and grant/research support from Abbott, AstraZeneca, Avid, Baxter, Bristol-Myers Squibb, GlaxoSmithKline, Elan, Eli Lilly, Medivation, Merck, Pfizer, Toyama, and Wyeth; has received educational fees from Alzheimer’s Foundation of America; has received other research support only from Janssen and GE; has received other research support from National Institute on Aging, National Institute of Mental Health, Alzheimer’s Association, Arizona Department of Health Services, and Institute for Mental Health Research; is a stock shareholder in MedAvante and Adamas; and holds a patent for “Biomarkers of Alzheimer’s Disease.” Drs Fleisher, Burke, and Seward and Mss Brand and Dougherty have no personal affiliations or financial relationships with any commercial interest to disclose relative to the activity.
Disclaimer
The opinions expressed are those of the authors, not of Banner Health or Physicians Postgraduate Press.
Clinical Points
- REM sleep behavior disorder may be idiopathic or may be a harbinger of mild cognitive impairment (MCI) or other neurodegenerative disorders in older adults.
- The diagnosis of MCI can be subjective.
- Although cholinesterase inhibitors are widely used for treatment of MCI, there are insufficient data to warrant their use in MCI.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
REFERENCES
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. Fourth Edition. Arlington, VA: American Psychiatric Association; 2000.
Bronte-Stewart HM, Minn AY, Rodrigues K, et al. Postural instability in idiopathic Parkinson’s disease: the role of medication and unilateral pallidotomy. Brain. 2002;125(pt 9):2100-2114. PubMed doi:10.1093/brain/awf207
Crum RM, Anthony JC, Bassett SS, et al. Population-based norms for the Mini-Mental State Examination by age and educational level. JAMA. 1993;269(18):2386-2391. PubMed
Doody RS, Ferris SH, Salloway S, et al. Donepezil treatment of patients with MCI: a 48-week randomized, placebo-controlled trial. Neurology. 2009;72(18):1555-1561. PubMed doi:10.1212/01.wnl.0000344650.95823.03
Feldman HH, Ferris S, Winblad B, et al. Effect of rivastigmine on delay to diagnosis of Alzheimer’s disease from mild cognitive impairment: the InDDEx study. Lancet Neurol. 2007;6(6):501-512. PubMed doi:10.1016/S1474-4422(07)70109-6
Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189-198. doi:10.1016/0022-3956(75)90026-6 PubMed
Gollan TH, Montoya RI, Werner GA. Semantic and letter fluency in Spanish-English bilinguals. Neuropsychology. 2002;16(4):562-576. PubMed doi:10.1037/0894-4105.16.4.562
Iranzo A, Molinuevo JL, Santamar×a J, et al. Rapid-eye-movement sleep behaviour disorder as an early marker for a neurodegenerative disorder: a descriptive study. Lancet Neurol. 2006;5(7):572-577. PubMed doi:10.1016/S1474-4422(06)70476-8
Knopman DS, DeKosky ST, Cummings JL, et al; Report of the Quality Standards Subcommittee of the American Academy of Neurology. Practice parameter: diagnosis of dementia (an evidence-based review). Neurology. 2001;56(9):1143-1153. PubMed
Kunz D, Mahlberg R. A two-part, double-blind, placebo-controlled trial of exogenous melatonin in REM sleep behaviour disorder. J Sleep Res. 2010;19(4):591-596. PubMed doi:10.1111/j.1365-2869.2010.00848.x
Links KA, Merims D, Binns MA, et al. Prevalence of primitive reflexes and parkinsonian signs in dementia. Can J Neurol Sci. 2010;37(5):601-607. PubMed
Mayor S. Regulatory authorities review use of galantamine in mild cognitive impairment. BMJ. 2005;330(7486):276. PubMed doi:10.1136/bmj.330.7486.276-b
McKeith IG, Dickson DW, Lowe J, et al; Consortium on DLB. Diagnosis and management of dementia with Lewy bodies: third report of the DLB Consortium. Neurology. 2005;65(12):1863-1872. PubMed doi:10.1212/01.wnl.0000187889.17253.b1
Mitrushina M, Boone KB, Razani J, et al. Handbook of Normative Data for Neuropsychological Assessment. Second edition. New York, NY: Oxford University Press; 2005.
Mungas D. In-office mental status testing: a practical guide. Geriatrics. 1991;46(7):54-58, 63, 66. PubMed
Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695-699. PubMed doi:10.1111/j.1532-5415.2005.53221.x
Petersen RC, Smith GE, Waring SC, et al. Mild cognitive impairment: clinical characterization and outcome. Arch Neurol. 1999;56(3):303-308. PubMed doi:10.1001/archneur.56.3.303
Petersen RC, Thomas RG, Grundman M, et al; Alzheimer’s Disease Cooperative Study Group. Vitamin E and donepezil for the treatment of mild cognitive impairment. N Engl J Med. 2005;352(23):2379-2388. PubMed doi:10.1056/NEJMoa050151
Salloway S, Ferris S, Kluger A, et al; Donepezil 401 Study Group. Efficacy of donepezil in mild cognitive impairment: a randomized placebo-controlled trial. Neurology. 2004;63(4):651-657. PubMed
Salmon DP, Galasko D, Hansen LA, et al. Neuropsychological deficits associated with diffuse Lewy body disease. Brain Cogn. 1996;31(2):148-165. PubMed doi:10.1006/brcg.1996.0039
Smithson F, Morris ME, Iansek R. Performance on clinical tests of balance in Parkinson’s disease. Phys Ther. 1998;78(6):577-592. PubMed
Voisin T, Touchon J, Vellas B. Mild cognitive impairment: a nosological entity? Curr Opin Neurol. 2003;16(suppl 2):S43-S45. PubMed doi:10.1097/00019052-200312002-00008
Enjoy this premium PDF as part of your membership benefits!