Objective: The natural course of prescription opioid use disorder has not been examined in longitudinal studies. The current study examined correlates of opioid abstinence over time after completion of a treatment trial for prescription opioid dependence.
Methods: The multisite Prescription Opioid Addiction Treatment Study examined different durations of buprenorphine-naloxone treatment and different intensities of counseling to treat prescription opioid dependence, as assessed by DSM-IV; following the clinical trial, a longitudinal study was conducted from March 2009-January 2013. At 18, 30, and 42 months after treatment entry, telephone interviews were conducted (N = 375). In this exploratory, naturalistic study, logistic regression analyses examined the association between treatment modality (including formal treatment and mutual help) and opioid abstinence rates at the follow-up assessments.
Results: At the 3 follow-up assessments, approximately half of the participants reported engaging in current substance use disorder treatment (47%-50%). The most common treatments were buprenorphine maintenance (27%-35%) and mutual-help group attendance (27%-30%), followed by outpatient counseling (18%-23%) and methadone maintenance (4%). In adjusted analyses, current opioid agonist treatment showed the strongest association with current opioid abstinence (odds ratios [ORs] = 5.4, 4.6, and 2.8 at the 3 assessments), followed by current mutual-help attendance (ORs = 2.2, 2.7, and 1.9); current outpatient counseling was not significantly associated with abstinence in the adjusted models.
Conclusions: While opioid agonist treatment was most strongly associated with opioid abstinence among patients with prescription opioid dependence over time, mutual-help group attendance was independently associated with opioid abstinence. Clinicians should consider recommending both of these interventions to patients with opioid use disorder.
Trial Registration: ClinicalTrials.gov identifier: NCT00316277‘ ‹

CME Background
Articles are selected for credit designation based on an assessment of the educational needs of CME participants, with the purpose of providing readers with a curriculum of CME articles on a variety of topics throughout each volume. Activities are planned using a process that links identified needs with desired results.
To obtain credit, read the article, correctly answer the questions in the Posttest, and complete the Evaluation. A $10 processing fee will apply.
CME Objective
After studying this article, you should be able to:
- Consider recommending both opioid agonist treatment and mutual-help group attendance to improve opioid abstinence in patients with opioid use disorder
The CME Institute of Physicians Postgraduate Press, Inc., is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Credit Designation
The CME Institute of Physicians Postgraduate Press, Inc., designates this journal-based CME activity for a maximum of 1 AMA PRA Category 1 Credit™. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Note: The American Academy of Physician Assistants (AAPA) accepts certificates of participation for educational activities certified for AMA PRA Category 1 Credit™ from organizations accredited by ACCME or a recognized state medical society. Physician assistants may receive a maximum of 1 hour of Category I credit for completing this program.
Release, Expiration, and Review Dates
This educational activity was published in March 2019 and is eligible for AMA PRA Category 1 Credit™ through April 30, 2021. The latest review of this material was February 2019.
Financial Disclosure
All individuals in a position to influence the content of this activity were asked to complete a statement regarding all relevant personal financial relationships between themselves or their spouse/partner and any commercial interest. The CME Institute has resolved any conflicts of interest that were identified. In the past year, Marlene P. Freeman, MD, Editor in Chief, has received research funding from JayMac and Sage; has been a member of the advisory boards for Otsuka, Alkermes, and Sunovion; has been a member of the Independent Data Safety and Monitoring Committee for Janssen; and, as a Massachusetts General Hospital (MGH) employee, works with the MGH National Pregnancy Registry, which is sponsored by Teva, Alkermes, Otsuka, Actavis, and Sunovion, and works with the MGH Clinical Trials Network and Institute, which receives research funding from multiple pharmaceutical companies and the National Institute of Mental Health. No member of the CME Institute staff reported any relevant personal financial relationships. Faculty financial disclosure appears at the end of the article.
ABSTRACT
Objective: The natural course of prescription opioid use disorder has not been examined in longitudinal studies. The current study examined correlates of opioid abstinence over time after completion of a treatment trial for prescription opioid dependence.
Methods: The multisite Prescription Opioid Addiction Treatment Study examined different durations of buprenorphine-naloxone treatment and different intensities of counseling to treat prescription opioid dependence, as assessed by DSM-IV; following the clinical trial, a longitudinal study was conducted from March 2009-January 2013. At 18, 30, and 42 months after treatment entry, telephone interviews were conducted (N = 375). In this exploratory, naturalistic study, logistic regression analyses examined the association between treatment modality (including formal treatment and mutual help) and opioid abstinence rates at the follow-up assessments.
Results: At the 3 follow-up assessments, approximately half of the participants reported engaging in current substance use disorder treatment (47%-50%). The most common treatments were buprenorphine maintenance (27%-35%) and mutual-help group attendance (27%-30%), followed by outpatient counseling (18%-23%) and methadone maintenance (4%). In adjusted analyses, current opioid agonist treatment showed the strongest association with current opioid abstinence (odds ratios [ORs] = 5.4, 4.6, and 2.8 at the 3 assessments), followed by current mutual-help attendance (ORs = 2.2, 2.7, and 1.9); current outpatient counseling was not significantly associated with abstinence in the adjusted models.
Conclusions: While opioid agonist treatment was most strongly associated with opioid abstinence among patients with prescription opioid dependence over time, mutual-help group attendance was independently associated with opioid abstinence. Clinicians should consider recommending both of these interventions to patients with opioid use disorder.
Trial Registration: ClinicalTrials.gov identifier: NCT00316277
J Clin Psychiatry 2019;80(2):18m12292
To cite: Weiss RD, Griffin ML, Marcovitz DE, et al. Correlates of opioid abstinence in a 42-month posttreatment naturalistic follow-up study of prescription opioid dependence. J Clin Psychiatry. 2019;80(2):18m12292.
To share: https://doi.org/10.4088/JCP.18m12292
© Copyright 2019 Physicians Postgraduate Press, Inc.
aDivision of Alcohol and Drug Abuse, McLean Hospital, Belmont, Massachusetts
bDepartment of Psychiatry, Harvard Medical School, Boston, Massachusetts
cDepartment of Psychiatry and Behavioral Sciences, Vanderbilt University Medical Center, Nashville, Tennessee
dDepartment of Biostatistics, Harvard T. H. Chan School of Public Health, Boston, Massachusetts
eLaboratory for Psychiatric Biostatistics, McLean Hospital, Belmont, Massachusetts
fDepartment of Psychiatry, Yale University School of Medicine, New Haven, Connecticut
*Corresponding author: Roger D. Weiss, MD, McLean Hospital, 115 Mill St, Belmont, MA 02478 ([email protected]).
Over 11 million people reported past-year nonmedical use of opioid analgesics in 2016, more than 12 times the number of those using heroin in the same time period.1 Research suggests that nonmedical prescription opioid use is associated with elevated risk for multiple comorbidities, including additional substance use disorders and other psychiatric disorders.2,3 However, most studies of opioid use disorder treatment have included primarily those dependent upon heroin; relatively little treatment outcome research has focused on those dependent either exclusively or primarily upon prescription opioids. The Prescription Opioid Addiction Treatment Study (POATS), carried out as part of the National Drug Abuse Treatment Clinical Trials Network, is the largest treatment outcome study yet conducted for those dependent upon prescription opioids.4 POATS, which included 653 participants at 10 US sites, examined different lengths of buprenorphine-naloxone (bup-nx) treatment and different levels of intensity of counseling in the treatment of patients who were dependent either exclusively or primarily on prescription opioids. The main trial showed far higher rates of “successful” opioid use outcomes (abstinence or near-abstinence from illicit opioids) among study participants while they were stabilized on bup-nx (49%) than they achieved after tapering from bup-nx (7%-9%).
Long-term follow-up of drug-dependent participants can generate important data about treatment response and recovery trajectories. Following completion of the POATS trial, we therefore conducted a 42-month naturalistic long-term follow-up study of this population (N = 375); telephone interviews were conducted 18, 30, and 42 months following initial randomization into the treatment trial. The most striking finding in the follow-up study was the high rates of self-reported opioid abstinence, both among participants receiving opioid agonist treatment (80%) and, even more surprising, among those not receiving agonist treatment (50%) at month 42.5 Neither baseline sociodemographic or clinical characteristics nor response to treatment in the main trial were associated with opioid abstinence at month 425; the only outcome predictor was the use of heroin prior to trial entry; participants with a history of any heroin use (regular users were excluded) were more likely to have opioid dependence at month 42. We found in the follow-up study that opioid agonist treatment was associated with a higher rate of opioid abstinence5,6; this finding was also reported by Hser et al7 in a long-term follow- up of a comparative trial of buprenorphine and methadone. However, the association between opioid abstinence and psychosocial treatment received during the follow-up period was not investigated. The aim of the current exploratory study was to examine the natural history of treatment for prescription opioid use disorder and its association with opioid abstinence after completion of the randomized controlled treatment trial. Specifically, we examined the roles of agonist treatment, formal behavioral treatment, and mutual-help (sometimes called self-help) meeting attendance on opioid abstinence in the POATS long-term follow-up study. Understanding the association between ongoing treatment and long-term outcomes can provide critical information on the optimal long-term management of opioid use disorder.

- While buprenorphine is clearly a highly effective treatment for opioid use disorder, it is not known what concomitant psychosocial treatment approaches would improve treatment outcomes in patients receiving buprenorphine.
- In patients with opioid use disorder, recommending attendance at mutual-help groups such as Alcoholics Anonymous or Narcotics Anonymous in addition to buprenorphine therapy can be helpful.
METHODS
Description of the POATS Treatment Trial and Outcomes
The main POATS trial was conducted from 2006 to 2009 at 10 sites in the United States as part of the National Drug Abuse Treatment Clinical Trials Network. Study participants were individuals aged at least 18 years who met DSM-IV8 criteria for current opioid dependence based either entirely or primarily on opioid analgesics. Those who reported heroin use on > 4 of the previous 30 days, those with a lifetime history of opioid dependence due to heroin alone, and those who had ever injected heroin were excluded, as were those who required ongoing opioid use for pain management. For complete details, see Weiss and colleagues.4
POATS employed a 2-phase, adaptive treatment research design; in the first phase, participants received a 4-week bup-nx taper and 8 weeks of follow-up and were randomized to either standard medical management (SMM) or SMM plus individual opioid drug counseling (ODC). In the second phase, for those who relapsed to opioid use during Phase 1, participants received 12 weeks of bup-nx stabilization, another 4-week taper, and 8 weeks of post-taper follow-up. Again, they were randomized to SMM alone or SMM plus ODC, to investigate whether the addition of counseling to bup-nx and SMM improved opioid use outcomes. In Phase 1, only 7% of participants had successful opioid use outcomes (abstinence or near-abstinence). In Phase 2, 49% of participants had successful outcomes during their last 4 weeks on bup-nx stabilization.4 When participants were tapered after 12 weeks of bup-nx stabilization and followed for 8 weeks post-taper, the success rate dropped to 9%. Adding individual counseling to bup-nx and medical management meetings did not improve outcomes.
Longitudinal Follow-Up Study Procedures
Institutional review boards at each site approved the longitudinal follow-up study, which was registered at ClinicalTrials.gov (identifier: NCT00316277). Written informed consent was obtained after the procedures had been fully explained. Trained research assistants conducted telephone interviews at the lead site (McLean Hospital) from March 2009 to January 2013. Assessments were targeted for 18, 30, and 42 months after participants’ initial entry into the treatment study. Assessments (45-60 minutes) covered the past 12 months; $75 was awarded for each assessment, similar to other substance use treatment studies.5,9 For more detail, see Weiss and colleagues.5
Longitudinal Follow-Up Study Measures
Follow-up interviews included a selection of questionnaires used in the POATS trial; additional items assessed participants’ substance use and treatments received since completion of the treatment study. For the current exploratory study, a subset of the longitudinal measures was analyzed, including only those described below. DSM-IV diagnoses of opioid dependence used the Composite International Diagnostic Interview (CIDI) Section L.10 Items from the Addiction Severity Index11 assessed days using opioids over the past 30 days. A questionnaire designed specifically for this follow-up study assessed current utilization of pharmacotherapy, formal behavioral treatment, and mutual-help group attendance for SUD. For the analyses reported, “outpatient counseling” refers to outpatient counseling and/or intensive outpatient/day hospital treatment; the term “agonist treatment” refers to both full agonist (ie, methadone) and partial agonist (ie, buprenorphine) treatment.
Statistical Analysis
For this exploratory analysis, we analyzed the associations between current treatment utilization (dichotomous measures of opioid agonist treatment, mutual-help attendance, and outpatient counseling) and opioid use at the 18-, 30-, and 42-month follow-up assessments in a longitudinal study conducted after participation in the POATS treatment trial. Bivariate associations for categorical variables were assessed with χ2 tests. To examine the adjusted associations between the most common treatments and the binary outcome of opioid abstinence in the past 30 days, generalized linear models (eg, logistic regression) were estimated, while also adjusting for site and initial treatment condition (since not all follow-up participants entered the second phase of the main trial, we adjusted for initial treatment condition only). SPSS v.2012 was used for all analyses.
RESULTS
The follow-up study enrolled 375 (57%) of the 653 original POATS participants, with 338 providing data.5 Follow-up study retention was strong: 94% of the month 18 participants continued to month 30, and 93% of month 30 participants continued to month 42. Overall, follow-up participants and nonparticipants had similar sociodemographic and clinical characteristics at baseline, as well as similar rates of successful treatment outcomes in the main trial. The best predictor of participation in the follow-up study was length of time elapsed since completion of the treatment study: nonparticipants had been out of contact with study staff for twice as many months on average as participants.13
Natural Course of SUD Treatment Utilization
At the 3 follow-up assessments, most participants reported engaging in some form of SUD treatment at some time during the past year (61%-66%); fewer reported current treatment involvement (47%-50%). Because study treatment was limited to the main trial, treatments occurring during follow-up were self-initiated. Buprenorphine maintenance was the most common current treatment (27%-35%). Current use of other SUD medications during follow-up was less common: 4% received methadone maintenance and < 1% used other SUD medications. Other common current treatments were mutual-help group attendance (27%-30%) and outpatient counseling (18%-23%).
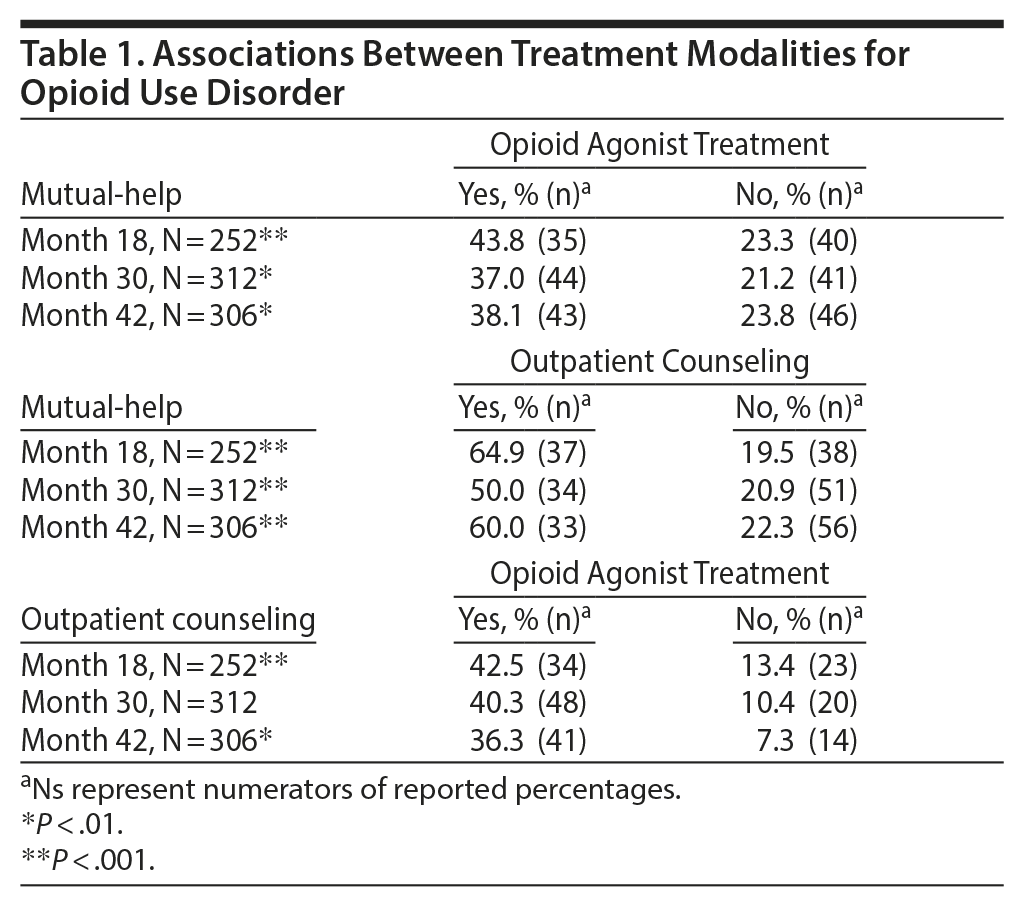
We examined the associations between the most common treatments and found that current utilization of one was associated with current utilization of the others (Table 1). Participants reporting current opioid agonist treatment were more likely to report mutual-help attendance at each follow-up time, when compared to those not currently receiving agonist treatment. For example, at month 18, patients receiving opioid agonist treatment were significantly more likely to be receiving mutual-help treatment than those not receiving opioid agonist treatment (43.8% vs 23.3%). Similarly, current opioid agonist treatment was associated with current outpatient counseling at each follow-up assessment. Finally, participants reporting mutual-help attendance were more likely to report outpatient counseling as well.
Which Patient-Initiated Treatments Were Correlated With Opioid Abstinence?
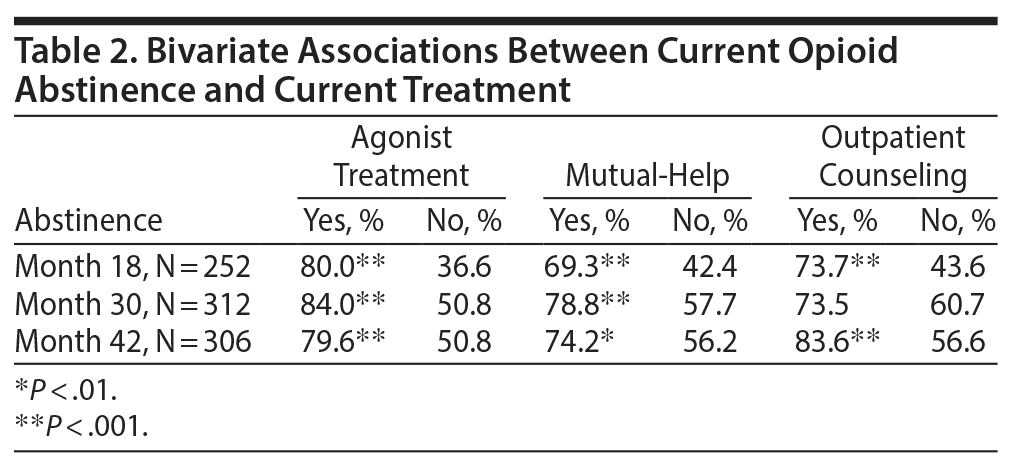
Current abstinence from opioids was associated with receiving current treatment in bivariate analysis (Table 2). At months 18, 30, and 42, agonist treatment was associated with abstinence; mutual-help attendance was also associated with abstinence at each follow-up time. However, in bivariate analyses, participants receiving outpatient counseling were more likely to be abstinent at months 18 and 42 only.
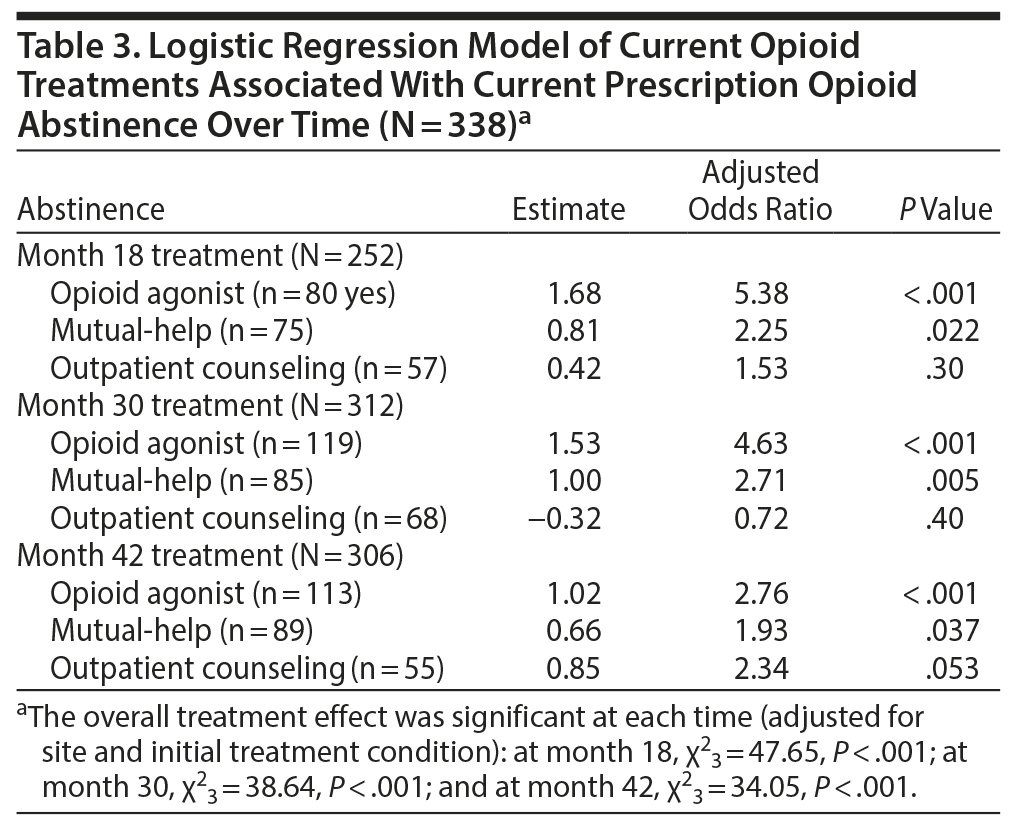
Multivariable logistic regression models examined the association among the 3 most commonly utilized treatments and opioid abstinence at months 18, 30, and 42 (Table 3). Overall, opioid agonist treatment showed the strongest association with opioid abstinence, with odds ratios over time of 5.4, 4.6, and 2.8 at months 18, 30, and 42, respectively. This association was statistically significant at each follow-up time point, even after controlling for the effects of the other 2 treatment types, mutual-help attendance and outpatient counseling. That is, participants in opioid agonist treatment at month 18 had more than 5 times the odds of being opioid abstinent than those not receiving this treatment, and they had almost 3 times the odds of being abstinent at month 42.
The association between opioid abstinence and mutual-help group attendance was also significant. Participants who attended mutual-help groups had more than 2 times the odds of being opioid abstinent at month 18, almost 3 times the odds at month 30, and almost twice the odds at month 42 compared to those not attending mutual-help groups, even after controlling for the effects of opioid agonist treatment and outpatient counseling. Outpatient treatment, however, was not associated with opioid abstinence at any follow-up time in the models controlling for opioid agonist treatment and mutual-help attendance. Interactions among the 3 treatments were examined, but none were significant at any follow-up time.
DISCUSSION
In the Prescription Opioid Addiction Treatment Study, participants receiving opioid agonist treatment in the 3.5-year naturalistic follow-up phase of the trial reported significantly higher rates of abstinence from opioids than those not receiving agonist treatment.5 In the current study, bivariate analysis showed that not only was agonist treatment associated with opioid abstinence, but mutual-help attendance and outpatient counseling were also associated with abstinence. Multivariable logistic regression models confirmed the association between opioid abstinence and both agonist treatment and mutual-help attendance at months 18, 30, and 42. However, outpatient counseling was not associated with opioid abstinence in the adjusted models at any of the follow-up time points. Overall, these findings suggest that continued receipt of treatment, optimally opioid agonist treatment plus mutual help, is robustly associated with abstinence from illicit opioids.
The strong association between opioid agonist treatment and opioid abstinence over time contributes further to the literature supporting the efficacy of pharmacotherapy for opioid use disorder. Across timepoints, participants currently receiving opioid agonist treatment had between 2.8 and 5.4 times higher odds of being abstinent from opioids. This benefit underscores the critical importance of access to opioid agonist therapies, particularly against the backdrop of the continued rise of opioid overdose deaths.14
The lack of a significant interaction effect between receipt of agonist treatment and mutual-help attendance on opioid use in our follow-up study suggests that while receipt of agonist treatment and attendance at mutual-help meetings were both helpful, neither detracted from nor enhanced the abstinence benefit of the other. Rather, the benefits were independent and additive.
The literature supports mutual help, especially Alcoholics Anonymous (AA), as a useful treatment adjunct for those with alcohol use disorder,15 but the literature on the impact of mutual help for patients with opioid use disorder is less robust, especially beyond 6-month follow-up.16,17 We are aware of 1 longitudinal study examining mutual help in patients with opioid use disorder,18 which found greater abstinence among those attending mutual-help meetings at year 4 follow-up. However, most participants were heroin users (77%), and agonist medication status was not reported.
Short-term cohort studies of patients receiving outpatient buprenorphine therapy have noted an association between mutual-help attendance and improved abstinence,19,20 treatment retention,21,22 or both.23 Our findings are consistent with these studies and replicate the results for the first time in a longitudinal follow-up of prescription opioid users. These findings suggest that mutual-help attendance is associated with higher odds of abstinence, independent of opioid agonist treatment.
Many patients have encountered the traditional view held by some members in 12-step mutual-help organizations, especially NA, that opioid-agonist treatment is at odds with true abstinence.23,24 Indeed, this view is codified in NA official documents25: participants receiving agonist treatment are barred from holding service positions and are not recognized for continuous sober periods. In contrast, the official stance of AA differs significantly and indeed provides affirmation that medication choices are between patient and physician.26 Understanding these nuances is important for treatment professionals. A survey of patients receiving bup-nx found that a considerable percentage (30%) were worried about encountering negative attitudes toward their buprenorphine use; only 33% of participants reported that their providers had discussed disclosing their buprenorphine use.27 Monico and colleagues23 found that NA members receiving buprenorphine used various strategies to deal with this issue, including limiting disclosure of their buprenorphine prescriptions and avoiding particular meetings. Patients in these studies achieved some success in managing contradictory viewpoints, an important reminder for clinicians who would categorically avoid referring their patients to NA meetings due to such concerns. Indeed, in our follow-up study, participants receiving opioid agonist treatment were significantly more likely than their counterparts not receiving opioid agonist treatment to report attending mutual-help meetings at all 3 follow-up periods, although data on specific meeting type (eg, AA vs NA vs non-12-step meetings) were not collected. Notably, some evidence suggests that those with a primary drug use disorder report similarly strong abstinence rates whether they attend AA or NA.28
While many patients manage to reconcile the dissonant viewpoints between NA and opioid agonist treatment, another approach that has received positive reviews from patients and professional treatment staff has been the development of 12-step-based mutual-help recovery groups specifically catering to patients on opioid agonist treatment, such as Methadone Anonymous.29-31 Data from these qualitative studies indicate that patients engaged in such groups have reported improved abstinence from co-occurring cocaine, alcohol, and cannabis use.29
Although we did not solicit information from participants about their reasons for attending mutual-help meetings during follow-up, it is noteworthy that the standard medical management (MM) in the main trial of POATS consistently included a recommendation to attend mutual-help meetings.4 Similarly, in the multisite NIAAA COMBINE trial,32 the study physicians’ recommendation at every visit that participants with alcohol dependence attend AA was thought to increase AA attendance among those who received MM; significantly more patients randomized to placebo plus MM attended AA than did those receiving behavioral therapy alone.33 Based on our POATS follow-up findings, it is possible that regular recommendation of mutual-help attendance by a clinician may have a beneficial effect in opioid use disorder treatment as well, whereas requiring mutual-help attendance as a condition of treatment may not be beneficial.23 Our finding regarding the beneficial effect of mutual-help meetings, coupled with the caveats and special considerations for patients taking buprenorphine who attend NA meetings, suggests that the development and testing of a Twelve Step Facilitation treatment34 for those taking opioid use disorder medications could be timely.
The association between outpatient counseling and abstinence that we found at some follow-up time points, in bivariate analysis, was no longer statistically significant in models adjusted for agonist treatment and mutual-help attendance. Much has been written about the limited empirical data supporting efficacy of behavioral treatment (other than contingency management) offered concomitantly with opioid agonist treatment to improve overall treatment outcomes,35 with the main POATS trial itself underscoring this challenge.4 There have been exceptions to this finding in studies of subsets of opioid use disorder patients.36,37 The fact that increased psychosocial services have generally been considered to enhance the efficacy of methadone maintenance treatment38 suggests that perhaps we have yet to design studies of bup-nx treatment that accurately pair specific at-risk patients with optimal behavioral treatments. For example, patients with high levels of psychiatric symptoms in methadone maintenance treatment have been found to benefit from psychotherapy,39 but no comparable study has focused on those receiving bup-nx.
The POATS follow-up study has several limitations (for details, see Weiss and colleagues5) including participation by only 57% of the original randomized sample and telephone-based contact with main-trial participants precluding the opportunity for collection of urine toxicology data. Other limitations include lack of data regarding participants’ extent of involvement in mutual help, type of mutual-help meetings attended, the temporal association between mutual-help initiation and abstinence, reasons for self-selection in and out of various treatments (eg, agonist treatment, outpatient counseling, and mutual-help groups), dosing of opioid agonist treatment, and frequency of attendance or perceived quality of outpatient counseling services. It is also possible that the associations between treatment participation and outcome may not be causal, but may instead reflect increased motivation and thus inherently better prognosis.
In future studies of opioid use disorder, it would be useful to collect the data on mutual help that has emerged over the past 25 years for alcohol use disorder, including mediators of any positive effects and the degree to which these elements may augment professional services (ie, peer support services integrated within professional treatment systems). Experimental trials might formally assess the impact of referral to mutual-help meetings for those with opioid use disorder, ensuring that the active referral condition excluded the compulsory element that has not been previously effective. Finally, additional longitudinal data about the experiences of patients with opioid use disorder in mutual-help meetings, including the role of 12-step alternatives such as SMART Recovery, could be informative.
This study, using long-term follow-up data from the largest randomized trial of treatment for prescription opioid dependence to date, found that ongoing treatment was strongly associated with odds of opioid abstinence up to 42 months following the trial. Although current opioid agonist treatment had the strongest association with abstinence, mutual-help attendance was also significantly associated with abstinence. Critically, mutual-help attendance was associated with an additive benefit among those receiving opioid agonist treatment and was also associated with abstinence in those not receiving agonist treatment. Adults with prescription opioid dependence appear to benefit from continued medication and mutual-help participation as part of long-term, ongoing care.
Submitted: April 11, 2018; accepted November 12, 2018.
Published online: March 26, 2019.
Disclosure of off-label usage: The authors have determined that, to the best of their knowledge, no investigational information about pharmaceutical agents that is outside US Food and Drug Administration-approved labeling has been presented in this article.
Financial disclosure: Dr Weiss has been a consultant for US World Meds, Braeburn Pharmaceuticals, Daiichi Sankyo, and GW Pharmaceuticals. Drs Griffin, Marcovitz, Hilton, Fitzmaurice, McHugh, and Carroll have no personal affiliations or financial relationships with any commercial interest to disclose relative to the article.
Funding/support: This work was supported by grants from National Institute on Drug Abuse (NIDA) as part of the Cooperative Agreement on the Clinical Trials Network (Grant U10DA015831) and NIDA grants K24DA022288 and K23DA035297.
Role of the sponsor: NIDA had no further role in study design; in the collection, analysis, and interpretation of data; or in the writing of the report. The NIDA Clinical Trials Network Publication Committee reviewed a draft of this manuscript and approved it for submission for publication.
Previous presentation: An earlier version of this paper was presented at the 79th Annual Meeting of the College on Problems of Drug Dependence; June 17-22, 2017; Montreal, Quebec, Canada.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
REFERENCES
1. Substance Abuse and Mental Health Services Administration. 2016 National Survey on Drug Use and Health: Detailed Tables. Rockville, MD: Center for Behavioral Health Statistics and Quality; 2017.
2. Huang B, Dawson DA, Stinson FS, et al. Prevalence, correlates, and comorbidity of nonmedical prescription drug use and drug use disorders in the United States: results of the National Epidemiologic Survey on Alcohol and Related Conditions. J Clin Psychiatry. 2006;67(7):1062-1073. PubMed CrossRef
3. Saha TD, Kerridge BT, Goldstein RB, et al. Nonmedical prescription opioid use and DSM-5 nonmedical prescription opioid use disorder in the United States. J Clin Psychiatry. 2016;77(6):772-780. PubMed CrossRef
4. Weiss RD, Potter JS, Fiellin DA, et al. Adjunctive counseling during brief and extended buprenorphine-naloxone treatment for prescription opioid dependence: a 2-phase randomized controlled trial. Arch Gen Psychiatry. 2011;68(12):1238-1246. PubMed CrossRef
5. Weiss RD, Potter JS, Griffin ML, et al. Long-term outcomes from the National Drug Abuse Treatment Clinical Trials Network Prescription Opioid Addiction Treatment Study. Drug Alcohol Depend. 2015;150:112-119. PubMed CrossRef
6. Weiss RD, Rao V. The Prescription Opioid Addiction Treatment Study: what have we learned. Drug Alcohol Depend. 2017;173(suppl 1):S48-S54. PubMed CrossRef
7. Hser YI, Evans E, Huang D, et al. Long-term outcomes after randomization to buprenorphine/naloxone versus methadone in a multi-site trial. Addiction. 2016;111(4):695-705. PubMed CrossRef
8. American Psychiatric Association. Diagnostic and Statistical Manual for Mental Disorders. Fourth Edition, Text Revision. Washington, DC: American Psychiatric Association; 2000.
9. Festinger DS, Marlowe DB, Croft JR, et al. Do research payments precipitate drug use or coerce participation? Drug Alcohol Depend. 2005;78(3):275-281. PubMed CrossRef
10. World Health Organization. Composite International Diagnostic Interview (CIDI): Core Version 2.1. 1997. WHO website. https://www.hcp.med.harvard.edu/wmhcidi/about-the-who-wmh-cidi/.
11. McLellan AT, Kushner H, Metzger D, et al. The Fifth Edition of the Addiction Severity Index. J Subst Abuse Treat. 1992;9(3):199-213. PubMed CrossRef
12. SPSS Statistics for Windows, Version 20.0 [computer program]. Armonk, NY: IBM Corp; 2011.
13. Potter JS, Dreifuss JA, Marino EN, et al. The multi-site Prescription Opioid Addiction Treatment Study: 18-month outcomes. J Subst Abuse Treat. 2015;48(1):62-69. PubMed CrossRef
14. Rudd RA, Aleshire N, Zibbell JE, et al. Increases in drug and opioid overdose deaths: United States, 2000-2014. MMWR Morb Mortal Wkly Rep. 2016;64(50-51):1378-1382. PubMed CrossRef
15. Kaskutas LA. Alcoholics anonymous effectiveness: faith meets science. J Addict Dis. 2009;28(2):145-157. PubMed CrossRef
16. Christo G, Franey C. Drug users’ spiritual beliefs, locus of control and the disease concept in relation to Narcotics Anonymous attendance and six-month outcomes. Drug Alcohol Depend. 1995;38(1):51-56. PubMed CrossRef
17. Galanter M. Combining medically assisted treatment and Twelve-Step programming: a perspective and review. Am J Drug Alcohol Abuse. 2018;44(2):151-159. PubMed CrossRef
18. Gossop M, Stewart D, Marsden J. Attendance at Narcotics Anonymous and Alcoholics Anonymous meetings, frequency of attendance and substance use outcomes after residential treatment for drug dependence: a 5-year follow-up study. Addiction. 2008;103(1):119-125. PubMed CrossRef
19. Mintzer IL, Eisenberg M, Terra M, et al. Treating opioid addiction with buprenorphine-naloxone in community-based primary care settings. Ann Fam Med. 2007;5(2):146-150. PubMed CrossRef
20. Parran TV, Adelman CA, Merkin B, et al. Long-term outcomes of office-based buprenorphine/naloxone maintenance therapy. Drug Alcohol Depend. 2010;106(1):56-60. PubMed CrossRef
21. Marcovitz DE, McHugh RK, Volpe J, et al. Predictors of early dropout in outpatient buprenorphine/naloxone treatment. Am J Addict. 2016;25(6):472-477. PubMed CrossRef
22. Stein MD, Cioe P, Friedmann PD. Buprenorphine retention in primary care. J Gen Intern Med. 2005;20(11):1038-1041. PubMed CrossRef
23. Monico LB, Gryczynski J, Mitchell SG, et al. Buprenorphine treatment and 12-step meeting attendance: conflicts, compatibilities, and patient outcomes. J Subst Abuse Treat. 2015;57:89-95. PubMed CrossRef
24. Greenfield BL, Owens MD, Ley D. Opioid use in Albuquerque, New Mexico: a needs assessment of recent changes and treatment availability. Addict Sci Clin Pract. 2014;9(1):10. PubMed CrossRef
25. World Service Board of Trustees Bulletin #29: Regarding methadone and other drug replacement programs. Narcotics Anonymous World Services website. https://www.na.org/?ID=bulletins-bull29. 1996.
26. Alcoholics Anonymous. The AA Member-Medications and Other Drugs. New York, NY: Alcoholics Anonymous World Services Inc; 2011.
27. Suzuki J, Dodds T. Clinician recommendation of 12-step meeting attendance and discussion regarding disclosure of buprenorphine use among patients in office-based opioid treatment. Subst Abus. 2016;37(1):31-34. PubMed CrossRef
28. Kelly JF, Greene MC, Bergman BG. Do drug-dependent patients attending Alcoholics Anonymous rather than Narcotics Anonymous do as well? a prospective, lagged, matching analysis. Alcohol Alcohol. 2014;49(6):645-653. PubMed CrossRef
29. Gilman SM, Galanter M, Dermatis H. Methadone Anonymous: a 12-step program for methadone maintained heroin addicts. Subst Abus. 2001;22(4):247-256. PubMed CrossRef
30. Glickman L, Galanter M, Dermatis H, et al. Recovery and spiritual transformation among peer leaders of a modified Methadone Anonymous group. J Psychoactive Drugs. 2006;38(4):531-533. PubMed CrossRef
31. Ronel N, Gueta K, Abramsohn Y, et al. Can a 12-step program work in methadone maintenance treatment? Int J Offender Ther Comp Criminol. 2011;55(7):1135-1153. PubMed CrossRef
32. Anton RF, O’ Malley SS, Ciraulo DA, et al; COMBINE Study Research Group. Combined pharmacotherapies and behavioral interventions for alcohol dependence: the COMBINE study: a randomized controlled trial. JAMA. 2006;295(17):2003-2017. PubMed CrossRef
33. Weiss RD, O’ malley SS, Hosking JD, et al; COMBINE Study Research Group. Do patients with alcohol dependence respond to placebo? results from the COMBINE Study. J Stud Alcohol Drugs. 2008;69(6):878-884. PubMed CrossRef
34. Nowinski J, Baker S, Carroll K. Twelve Step Facilitation Therapy Manual: A Clinical Research Guide for Therapists Treating Individuals with Alcohol Abuse and Dependence. Vol. 1. Rockville, MD: National Institute on Alcohol Abuse and Alcoholism; 1995.
35. Carroll KM, Weiss RD. The role of behavioral interventions in buprenorphine maintenance treatment: a review. Am J Psychiatry. 2017;174(8):738-747. PubMed CrossRef
36. Moore BA, Fiellin DA, Cutter CJ, et al. Cognitive Behavioral Therapy improves treatment outcomes for prescription opioid users in primary care buprenorphine treatment. J Subst Abuse Treat. 2016;71:54-57. PubMed CrossRef
37. Weiss RD, Griffin ML, Potter JS, et al. Who benefits from additional drug counseling among prescription opioid-dependent patients receiving buprenorphine-naloxone and standard medical management? Drug Alcohol Depend. 2014;140:118-122. PubMed CrossRef
38. McLellan AT, Arndt IO, Metzger DS, et al. The effects of psychosocial services in substance abuse treatment. Addictions Nursing Network. 1993;5(2):38-47. CrossRef
39. Woody GE, McLellan AT, Luborsky L, et al. Severity of psychiatric symptoms as a predictor of benefits from psychotherapy: the Veterans Administration-Penn study. Am J Psychiatry. 1984;141(10):1172-1177. PubMed
This PDF is free for all visitors!