Abstract
Objective: This post hoc analysis assessed the benefit-risk of aripiprazole once-monthly 400 mg (AOM 400) as acute and long-term maintenance treatment for patients diagnosed with schizophrenia, based on number needed to treat (NNT), number needed to harm (NNH), and likelihood to be helped or harmed (LHH).
Methods: Data were derived from 2 studies that evaluated efficacy and safety/tolerability of AOM 400 versus placebo in an acute or maintenance setting. Main efficacy outcomes were ≥30% reduction in Positive and Negative Syndrome Scale (PANSS) total score (acute treatment) and patients free from impending relapse (maintenance treatment); main safety/tolerability outcome was incidence of patient discontinuation due to treatment-emergent adverse events (TEAEs). NNT, NNH, and LHH were calculated using standard methods.
Results: In the acute study, 60/167 AOM 400 recipients (37.0%) versus 24/168 placebo recipients (14.4%) had a ≥30% reduction in PANSS total score at week 10 (NNT=5 [95% confidence interval (CI), 4–8]). Corresponding values for avoidance of impending relapse in the maintenance study (median observation time: 113 days) were 242/269 (90.0%) versus 81/134 (60.4%) (NNT=4 [95% CI, 3–5]). Discontinuations due to TEAEs were lower with AOM 400 versus placebo in both studies, resulting in negative NNH values. Calculated LHH values were 200 (acute treatment) and 250 (maintenance treatment), indicating a higher likelihood to be helped than harmed with AOM 400.
Conclusion: Data demonstrate a favorable benefit–risk profile for AOM 400 versus placebo in the acute and maintenance treatment of schizophrenia.
Trial Registration: ClinicalTrials.gov identifiers: NCT01663532 and NCT00705783.
J Clin Psychiatry 2026;87(2):26m16359
Author affiliations are listed at the end of this article.
The natural course of schizophrenia is characterized by alternating periods of acute psychotic symptoms, stability, and relapse.1 Accordingly, management goals differ by symptom state, focusing on the reduction of acute symptoms during psychotic episodes and the prevention of relapse through continued maintenance therapy.2
Antipsychotics are an important component in the treatment of schizophrenia,3 with demonstrated efficacy in managing acute psychotic episodes and for reducing the risk of relapse during maintenance therapy.2 In selecting an antipsychotic drug for a given patient, clinicians must consider a range of factors, including the individual’s clinical characteristics, treatment history, comorbidities, and preferences and the benefits and risks of the treatment in question.3 While clinical trial data provide an insight into the efficacy, safety, and tolerability of a treatment relative to placebo or other therapeutic options, they typically do not provide a full picture of a treatment’s benefit–risk profile. For instance, a statistically significant difference in efficacy outcomes between a medication treatment versus placebo does not indicate the magnitude or clinical relevance of that difference4,5 or how it balances against potential risks.
Number needed to treat (NNT) and number needed to harm (NNH) are metrics of effect size that are used to quantify the clinical relevance of clinical trial results, aiding in decisions regarding treatment.4–6 NNT and NNH quantify how many patients would need to be treated with 1 intervention (eg, a medication) instead of the comparator (eg, another medication or placebo) to expect to encounter 1 additional outcome of interest.5,7 Although NNT and NNH are calculated in the same way, NNT is used when discussing the potential benefit of one intervention versus another, while NNH is used when discussing the potential adverse outcomes of one intervention versus another.7 The ratio of these 2 metrics, known as the likelihood to be helped or harmed (LHH), provides an overall estimate of the treatment’s benefit–risk ratio.5,6 Generally speaking, a treatment is considered useful when it demonstrates a low NNT and a high NNH relative to the comparator, indicating a greater likelihood of benefit than harm.5 Under usual clinical circumstances, a single-digit NNT value (ie, <10) suggests that the intervention is reasonably efficacious, and a double-digit or higher NNH value suggests that the intervention is potentially well tolerated.5
Aripiprazole monohydrate is available as a once-a-month injection (aripiprazole once-monthly 400 mg [AOM 400]).8 It has also been developed as a once-every-2-months injection (aripiprazole 2-month ready-to-use 960 mg [Ari 2MRTU 960]),9,10 which has shown similar efficacy and safety to AOM 400 in people diagnosed with schizophrenia.11,12 Regulatory approval of AOM 400 in 2013 was supported by 2 randomized, double-blind, placebo-controlled studies that demonstrated a significant improvement in symptoms and functioning in acute schizophrenia and a significant delay in the time to impending relapse during maintenance treatment.13,14 To date, a comprehensive analysis of the benefit–risk profile focusing on AOM 400 has not been performed using data derived from these trials. Accordingly, this post hoc analysis estimated NNT, NNH, and LHH values for AOM 400, to generate benefit–risk information that may be informative for clinicians in the process of clinical decision-making.
METHODS
Study Overviews
This was an analysis of data from 2 studies of AOM 400 for the acute or maintenance treatment of patients diagnosed with schizophrenia.13,14 The protocols of both studies were approved by local institutional review boards or independent ethics committees, as appropriate.13,14
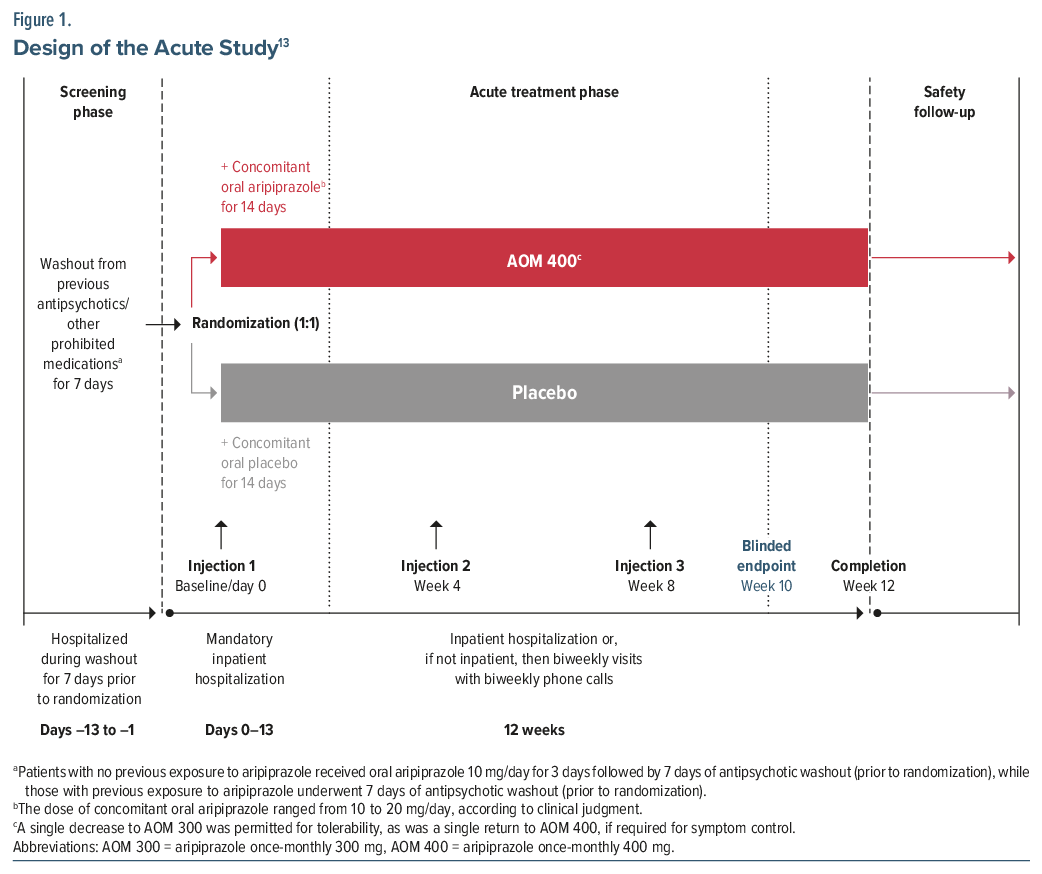
Acute use of AOM 400. The use of AOM 400 in an acute setting was evaluated in a 12-week, randomized, double-blind, placebo-controlled study conducted at 41 sites in the United States and Europe (NCT01663532).13 Following screening and after establishing tolerability to aripiprazole, patients entered a 7-day washout period followed by a 12-week acute treatment phase in which they were randomized (1:1) to AOM 400 or placebo (Figure 1).13
Eligibility criteria for the trial included age 18–65 years; a diagnosis of schizophrenia per the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision (DSM-IV-TR), confirmed using the Mini-International Neuropsychiatric Interview; and an acute psychotic episode at screening and baseline characterized by (1) an acute exacerbation of psychotic symptoms with significant deterioration in clinical or functional status and a Positive and Negative Syndrome Scale (PANSS) total score of ≥80 and (2) a score of >4 on each of the PANSS items of conceptual disorganization, hallucinatory behavior, suspiciousness/persecution, and unusual thought content.13 All participants provided written informed consent.
The primary efficacy outcome was the change from baseline to week 10 in PANSS total score.13 Secondary efficacy outcomes included change from baseline to week 10 in Clinical Global Impressions—Severity of Illness (CGI-S) scale score,13 PANSS responder rate at week 10 (ie, a ≥30% reduction in PANSS total score),13 and Clinical Global Impressions—Improvement (CGI-I) score at end point. Safety/tolerability variables included treatment-emergent adverse events (TEAEs).13
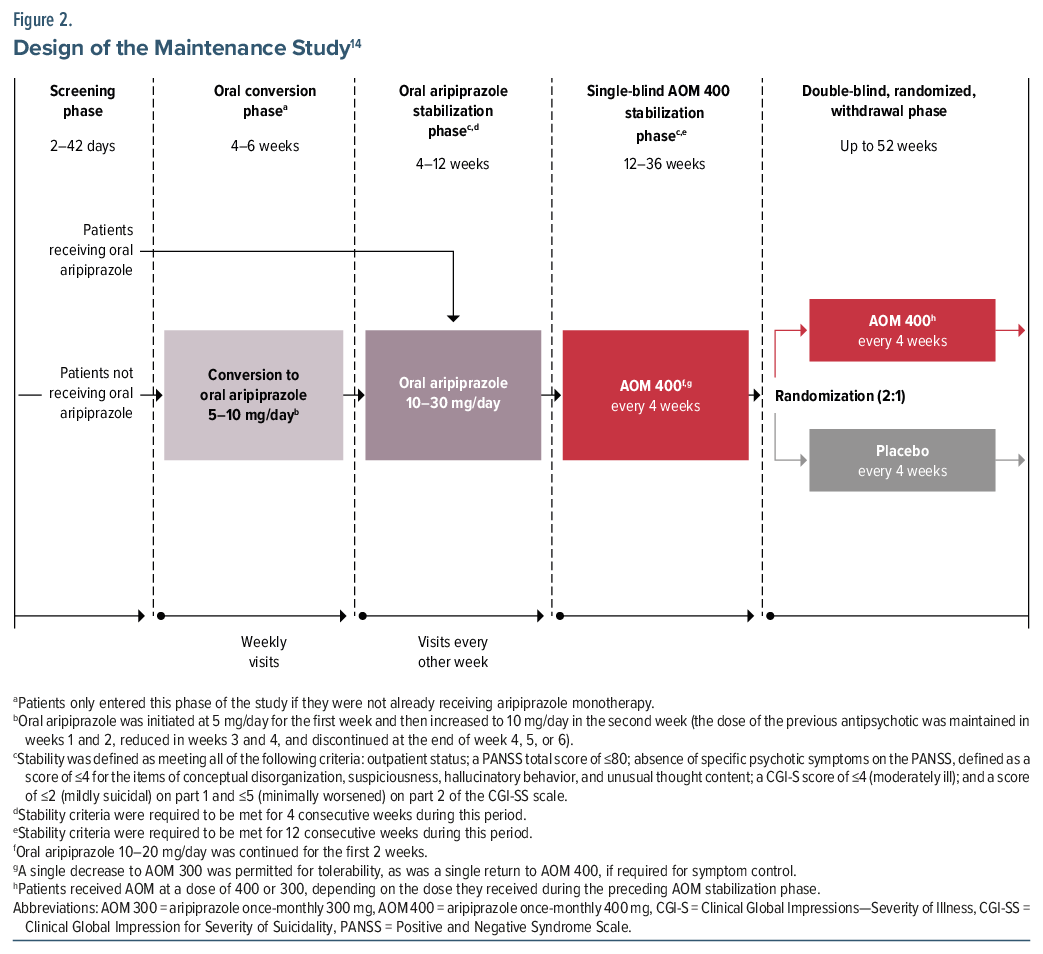
Maintenance use of AOM 400. The use of AOM 400 in a maintenance setting was evaluated in a multiphase trial conducted at 108 sites in North America, South America, Europe, and Asia (NCT00705783).14 Following screening, the trial was conducted in 4 phases (Figure 2): (1) conversion to oral aripiprazole (applicable only to patients receiving other antipsychotic[s]), (2) stabilization on oral aripiprazole, (3) single-blind stabilization on AOM 400, and (4) randomization (2:1) to continue AOM 400 or switch to placebo for 52 weeks of maintenance treatment.14 To proceed from the oral aripiprazole stabilization phase to the AOM 400 stabilization phase and from the AOM 400 stabilization phase to the double-blind, maintenance phase, patients were required to meet defined stability criteria, as follows: outpatient status; a PANSS total score of ≤80; absence of specific psychotic symptoms on the PANSS, defined as a score of ≤4 for the items of conceptual disorganization, suspiciousness, hallucinatory behavior, and unusual thought content; a CGI-S score of ≤4 (ie, moderately ill); and a score of ≤2 (ie, mildly suicidal) on part 1 and ≤5 (minimally worsened) on part 2 of the Clinical Global Impression for Severity of Suicidality (CGI-SS) scale.14
Eligibility criteria for the trial included age 18–60 years, schizophrenia per DSM-IV-TR criteria that was diagnosed ≥3 years prior to the study, and a history of symptom exacerbation or relapse when not receiving antipsychotic treatment.14 All participants provided informed written consent.
The primary efficacy end point was the time from randomization to AOM 400 or placebo in the maintenance phase to exacerbation of psychotic symptoms/impending relapse, defined as meeting any or all of the following criteria: clinical worsening (ie, a CGI-I score of ≥5 combined with an increase to a score of >4 on any of the PANSS items of conceptual disorganization, hallucinatory behavior, suspiciousness, or unusual thought content, with either a ≥2-point increase from randomization in that item or a ≥4-point increase from randomization in the combined score of all 4 items); hospitalization due to worsening psychotic symptoms; a risk of suicide (ie, a score of 4 or 5 on part 1 or a score of 6 or 7 on part 2 of the CGI-SS); and/or violent behavior leading to clinically significant self-injury, injury to another person, or destruction of property.14 Secondary end points included the proportion of responders (ie, those who met all stability criteria at the last visit in the maintenance phase) and the proportion of patients who achieved remission (ie, those with a score of ≤3 for each of the PANSS items of delusions, unusual thought content, hallucinatory behavior, conceptual disorganization, mannerisms/posturing, blunted affect, social withdrawal, and lack of spontaneity, maintained for a period of 6 months in the maintenance phase). Safety/tolerability outcomes included adverse events.14
NNT, NNH, and LHH Analysis
NNT and NNH values were calculated as (1 /[fAOM 400 − fplacebo], where f represents the frequency of the specified outcome in each treatment group), and were rounded up to the nearest whole number.6 Corresponding 95% confidence intervals (CIs) for statistically significant NNT and NNH values were reported; if the 95% CI included “infinity,” NNT or NNH values were not statistically significant and thus denoted as “NS” instead of showing the 95% CI containing 2 discontinuous domains ranging to infinity and to negative infinity.5,7
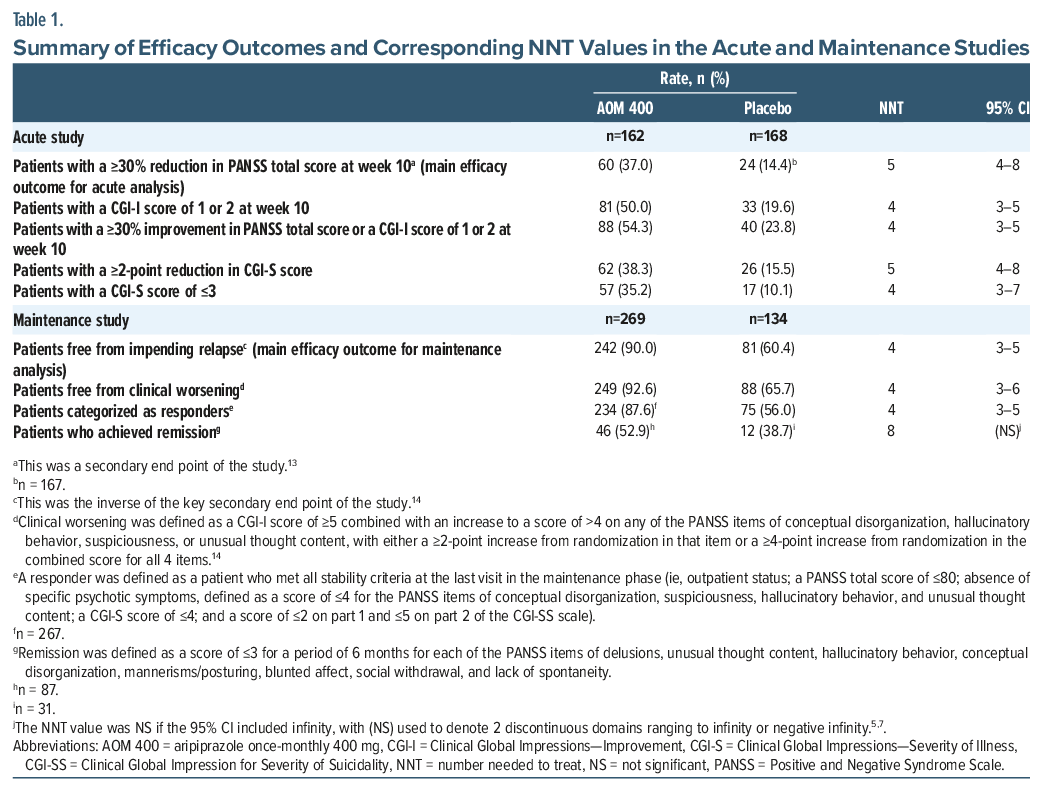
For the study assessing acute use of AOM 400, NNT values were calculated for various efficacy end points at week 10, including a ≥30% reduction in PANSS total score (representing the main efficacy outcome for acute treatment in the current analysis); a CGI-I score of 1 or 2; a ≥30% reduction in PANSS total score or a CGI-I score of 1 or 2; a ≥2-point reduction in CGI-S score; and a CGI-S score of ≤3. For the study assessing maintenance use of AOM 400, NNT values were calculated for the following efficacy end points: patients free from impending relapse (representing the main efficacy outcome for maintenance treatment in the current analysis); patients free from clinical worsening; patients categorized as responders; and patients achieving remission.
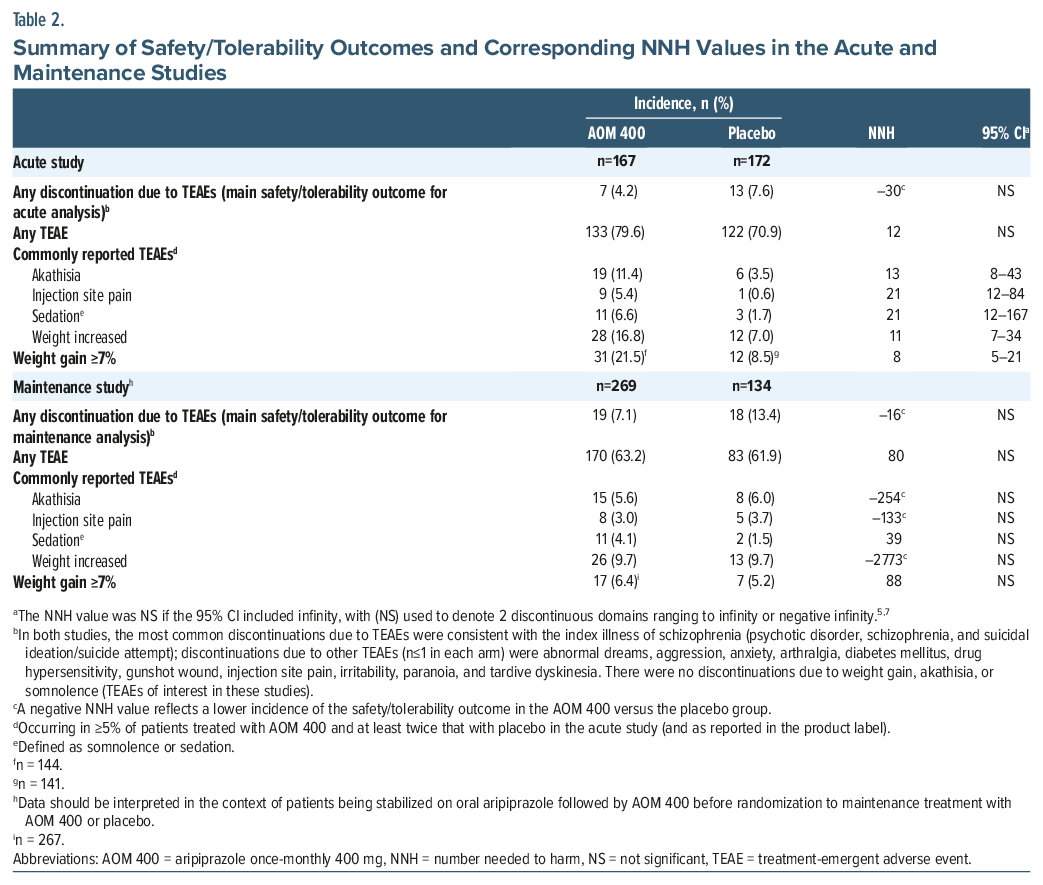
For both studies, NNH values were calculated for the end points of discontinuation due to TEAEs (representing the main safety/tolerability outcome in the current analysis). NNH values were also calculated for commonly reported TEAEs in the acute study, based on the product label.8 This includes TEAEs with an incidence of ≥5% in the AOM 400 group and at least twice that for placebo, namely increased weight, akathisia, injection site pain, and sedation (here defined as somnolence or sedation), as well as the NNH value for the categorical outcome of weight gain of ≥7% from baseline.
LHH values were calculated by dividing the NNH for specific safety/tolerability outcomes by the NNT for the main efficacy outcomes (ie, a ≥30% reduction in PANSS total score in the acute study and patients free from impending relapse in the maintenance study). In cases where the NNH value was negative (ie, when the incidence of the safety/tolerability outcome was lower with AOM 400 than with placebo), an NNH value of 1,000 was imputed for the purposes of calculating the LHH, consistent with previous studies.15,16 An LHH value of >1 indicates that the likelihood of benefit exceeds the likelihood of harm.5
Analyses were performed using R statistical software (version 4.4.2).
RESULTS
The analysis populations comprised 339 patients randomized to 12 weeks of treatment with either AOM 400 (n = 167) or placebo (n = 172) in the acute study and 403 patients randomized to 52 weeks of treatment with either AOM 400 (n = 269) or placebo (n = 134) in the long-term maintenance study. The demographic and clinical characteristics of included patients at baseline are shown in Supplementary Table 1. Although it was intended that patients receive 52 weeks of treatment with AOM 400 or placebo in the maintenance study, it was terminated early due to a positive interim analysis (conducted after 64 events) that showed that the primary end point had been met with no identified safety issues.14 Of note, the median time between randomization and the termination date in the 52-week randomized phase was 113 days.
A summary of efficacy outcomes and corresponding NNT values in the acute and maintenance studies is shown in Table 1 and Supplementary Table 2. For the main efficacy outcome in the acute study, 60 patients (37.0%) treated with AOM 400 and 24 patients (14.4%) treated with placebo had a ≥30% reduction in PANSS total score at week 10. The corresponding NNT value was 5 (95% CI, 4–8). For the main efficacy outcome in the maintenance study, 242 patients (90.0%) treated with AOM 400 and 81 patients (60.4%) treated with placebo were free from impending relapse, with a corresponding NNT value of 4 (95% CI, 3–5).
A summary of safety/tolerability outcomes and corresponding NNH values in the acute and maintenance studies is shown in Table 2. For the main safety/tolerability outcome, 7 patients (4.2%) treated with AOM 400 and 13 patients (7.6%) treated with placebo in the acute study discontinued treatment because of a TEAE; corresponding numbers in the maintenance study were 19 (7.1%) and 18 (13.4%), respectively. Since the incidence of discontinuation due to any TEAE in both studies was lower with AOM 400 versus placebo, NNH values were negative. NNH values for commonly reported TEAEs in the acute study were all double-or triple-digit numbers, except for weight gain of ≥7%, which had an NNH value of 8 (95% CI, 5–21).
LHH values for specific safety/tolerability outcomes relative to the main efficacy outcome in each study are shown in Table 3. All LHH values exceeded 1.
DISCUSSION
The NNT metric is used to describe the potential benefit of 1 intervention versus another, indicating how many patients need to be treated with 1 intervention instead of the comparator to achieve 1 additional positive outcome of interest.4 Among similarly conducted randomized clinical trials with placebo as the comparator, indirect treatment comparisons of NNT are possible.17,18 As mentioned previously, single-digit NNT values versus placebo generally represent clinically useful interventions.5–7 In the current analysis, in an acute treatment setting, the NNT value for a ≥30% reduction in PANSS total score at week 10 was 5 (95% CI, 4–8). This means that, for every 5 patients treated with AOM 400 instead of placebo in the trial, 1 additional treatment response was expected after 10 weeks of treatment. Similar NNT values were observed for the proportions of patients with a ≥2-point reduction in CGI-S score (NNT = 5) and a CGI-S score of ≤3 (NNT = 4). These data are consistent with those found for aripiprazole lauroxil19 and with the findings of a systematic review and network meta-analysis that examined the efficacy and acceptability of available long-acting injectable (LAI) antipsychotics in acutely ill individuals with schizophrenia-spectrum disorders.20 Per the latter analysis, LAI formulations of aripiprazole, olanzapine, risperidone, and paliperidone palmitate all outperformed placebo, with NNT values ranging from 3.2 to 4.7 for mean change in psychopathology rating scale scores.20 In the current analysis, in a maintenance treatment setting, the NNT value for patients free from impending relapse was 4 (95% CI, 3–5), with identical NNT results identified for patients free from clinical worsening and patients categorized as responders. These values are consistent with those for other LAI formulations approved in the United States for the maintenance treatment of schizophrenia, with overlapping 95% CIs.17 In a review of data from registration studies, calculated NNT values for LAI formulations of risperidone and paliperidone palmitate versus placebo ranged from 5 to 7 for the prevention of relapse.17
NNH is used to describe the potential harm of one intervention compared with another, indicating how many patients need to be treated with the intervention instead of the comparator for 1 additional patient to experience a specified adverse outcome. Double-digit or higher NNH values suggest that the intervention is potentially well tolerated5; in the context of a placebo-controlled trial, this indicates that the treatment has no more than a 10% disadvantage versus placebo in terms of potential harms.6 In the current analysis, negative NNH values were calculated for the primary safety/tolerability outcome of discontinuation due to any TEAE in both the acute and maintenance settings; this reflects that TEAE-related discontinuations occurred more frequently in placebo recipients than in AOM 400 recipients. Regarding individual TEAEs in the acute short-term study with AOM 400, NNH values were double-or triple-digit numbers, with the exception of potentially clinically relevant weight gain of ≥7%, which had an NNH value of 8. This is comparable to NNH values of <10 for weight gain of ≥7% in acute short-term studies of LAI formulations of risperidone and olanzapine.17 A possible explanation for the greater weight gain with AOM 400 versus placebo in the acute study may be recovery-related increases in food intake during hospitalization, where patients had consistent access to regular meals.13 This is supported by the finding of a positive correlation between the number of inpatient days and weight gain among patients treated with AOM 400, but not placebo.13 Of note, long-term outpatient treatment with AOM 400 is not associated with notable weight gain in most patients.13 In the maintenance study, mean change in bodyweight from the start to the end of the double-blind, maintenance phase was –0.2 kg in AOM recipients and –0.4 kg in placebo recipients, with a similar incidence of weight gain of ≥7% with both treatments (AOM, 6.4%; placebo, 5.2%).14
Per the literature, a useful psychiatric treatment is one with a low NNT and a high NNH,5,6 although patient-related factors may shift this balance. For instance, if a patient’s priority is to avoid weight gain, they may value a higher NNH over a lower NNT.7 This underscores the need to align treatment decisions with patient preferences and experiences.5,7 While no patient in the acute study discontinued due to weight gain, this was in the context of a controlled clinical trial.13 In a real-world setting, if a patient was particularly sensitive to weight gain and wished to avoid it as much as possible, then its occurrence may result in treatment discontinuation.6
This analysis has several limitations. The analysis was conducted post hoc, and the findings can be considered hypothesis-generating and require prospectively designed studies to confirm. Analyses were descriptive, and determination of NNT and NNH values relied on the use of binary outcomes (eg, response/nonresponse).6,21 Although this approach is considered more clinically intuitive than other measures of effect size (eg, Cohen’s d),4,6,7 it lacks more granular information captured in analyses of continuous data.16,21 Second, calculated NNT and NNH values were derived from highly selected clinical trial populations and may not be generalizable to real-world patients living with schizophrenia.5,15 This extends to discontinuations due to TEAEs, which in clinical trials may have complex causes that do not necessarily reflect real-world tolerability.5,15 In the maintenance study, patients randomized to AOM 400 had been stabilized on AOM 400 for ≥12 weeks in an earlier phase of the study,14 suggesting that the sample was enriched with patients who tolerated AOM 400. Lastly, consistent with the work of others,15,16 when the calculated NNH value was negative, it was imputed as 1,000 to allow for the calculation of LHH.
CONCLUSION
This post hoc analysis of 2 clinical trials supports an overall favorable benefit–risk profile of AOM 400 in the acute and maintenance treatment of patients diagnosed with schizophrenia. Based on calculated NNT and NNH values, patients treated with AOM 400 were 200 times more likely to have a reduction in symptoms than to discontinue due to a TEAE in an acute setting and were 250 times more likely to be free from impending relapse than to discontinue due to a TEAE in a maintenance setting. These data provide insight into the magnitude and clinical relevance of the benefits and risks associated with AOM 400 and may be useful to support informed decision-making in clinical practice. Given its comparable pharmacokinetic, safety, and efficacy outcomes to AOM 400,10–12 Ari 2MRTU 960 is likely to provide a similar benefit–risk profile, but with fewer injections per year.
Article Information
Published Online: May 13, 2026. https://doi.org/10.4088/JCP.26m16359
© 2026 Physicians Postgraduate Press, Inc.
Submitted: February 3, 2026; accepted April 1, 2026.
To Cite: Citrome L, Bell Lynum KS, Zhang Z, et al. Aripiprazole once-monthly for patients diagnosed with schizophrenia: number needed to treat, number needed to harm, and likelihood to be helped or harmed. J Clin Psychiatry 2026;87(2):26m16359.
Author Affiliations: Department of Psychiatry and Behavioral Sciences, New York Medical College, Valhalla, New York (Citrome); Otsuka Pharmaceutical Development & Commercialization Inc., Princeton, New Jersey (Bell Lynum, Zhang, Atkins); Lundbeck LLC, Deerfield, Illinois (Hutson Walker); H. Lundbeck A/S, Valby, Denmark (Yildirim).
Corresponding Author: Karimah S. Bell Lynum, PharmD, Otsuka Pharmaceutical Development & Commercialization Inc., 508 Carnegie Center, Princeton, NJ 08540 ([email protected]).
Author Contributions: Dr Zhang conducted the formal analysis. All authors participated in critical review and editing of drafts. All authors gave final approval of the version to be published and agree to be accountable for all aspects of the work.
Relevant Financial Relationships: Dr Citrome is a consultant to AbbVie/Allergan, Acadia, Adamas, Adheretech, Alkermes, Alumis, Angelini, Astellas, Autobahn, Avanir, Axsome, Biogen, BioXcel, Bristol Myers Squibb, Boehringer Ingelheim, Cadent Therapeutics, Cerevel, Clinilabs, COMPASS, Delpor, Draig Therapeutics, Eisai, Enteris BioPharma, HLS Therapeutics, Idorsia, INmune Bio, Impel, Intra-Cellular Therapies, Janssen, Karuna, Lundbeck, Luye, Lyndra, MapLight, Marvin, Medavante-ProPhase, Merck, Mitsubishi-Tanabe Pharma, Neumora, Neurocrine, Neurelis, Noema, Novartis, Noven, Otsuka, Ovid, Praxis, Recordati, Relmada, Reviva, Sage, Sumitomo/Sunovion, Supernus, Teva, University of Arizona, Vanda, and Wells Fargo; has undertaken one-off ad hoc consulting for individuals/entities conducting marketing, commercial, or scientific scoping research; has been a speaker for AbbVie/Allergan, Acadia, Alkermes, Angelini, Axsome, BioXcel, Bristol Myers Squibb, Eisai, Idorsia, Intra-Cellular Therapies, Janssen, Lundbeck, Neurocrine, Neopharm, Noven, Otsuka, Recordati, Sage, Sunovion, Takeda, Teva, Vanda, and CME activities organized by medical education companies such as Medscape, NACCME, NEI, Vindico, and Universities and Professional Organizations/Societies; owns stocks (small number of shares of common stock) in Bristol Myers Squibb, Eli Lilly, J & J, Merck, Pfizer purchased >10 years ago, and stock options in Reviva; and has received royalties/publishing income from Taylor & Francis (Editor-in-Chief, Current Medical Research and Opinion, from 2022), Wiley (Editor-in-Chief, International Journal of Clinical Practice, through end 2019), UpToDate (reviewer), Springer Healthcare (book), and Elsevier (Topic Editor, Psychiatry, Clinical Therapeutics, through Spring 2025). Dr Bell Lynum was a full-time employee of Otsuka Pharmaceutical Development & Commercialization Inc. at the time of this analysis. Dr Zhang is a full-time employee of Otsuka Pharmaceutical Development & Commercialization Inc. Dr Atkins is a consultant for Otsuka Pharmaceutical Development & Commercialization Inc. Dr Hutson Walker is a full-time employee of Lundbeck LLC, Deerfield, IL, USA. Dr Yildirim is a full-time employee of H. Lundbeck A/S.
Funding/Support: The work described here was sponsored by Otsuka Pharmaceutical Development & Commercialization Inc. and Lundbeck LLC.
Role of the Funders/Sponsors: Otsuka Pharmaceutical Development & Commercialization Inc. and Lundbeck LLC were involved in the design of the analysis; the collection, analysis, and interpretation of data; the writing and reviewing of this article; and the decision to submit the article for publication.
Acknowledgment: Medical writing support was provided by Lyndal Staples, BSc, and editorial support was provided by Sinead Stewart of the Prime Group of Companies (Knutsford, UK) and was funded by Otsuka Pharmaceutical Development & Commercialization Inc. and Lundbeck LLC. The sponsors thank the patients and their families who participated in the studies on which this analysis is based.
ORCID: Leslie Citrome: https://orcid.org/0000-0002-6098-9266; Karimah S. Bell Lynum: https://orcid.org/0000-0002-2045-9578; Zhen Zhang: https://orcid.org/0000-0001-8448-5663; Norman Atkins, Jr: https://orcid.org/0000-0002-0802-6032; Anne M. Hutson Walker: https://orcid.org/0009-0005-9304-1809; Murat Yildirim: https://orcid.org/0000-0001-6192-1047
Supplementary Material: Available at Psychiatrist.com.
Clinical Points
- While clinical trials demonstrate the efficacy, safety, and tolerability of a drug relative to placebo or other therapeutic options, they may not fully convey the drug’s benefit–risk profile. Number needed to treat (NNT), number needed to harm (NNH), and likelihood to be helped or harmed (LHH) help translate trial results into clinically useful measures for benefit–risk decisions.
- A favorable benefit–risk profile was demonstrated for aripiprazole once-monthly 400 mg (AOM 400) versus placebo in schizophrenia. AOM 400 recipients were 200 times more likely to achieve symptom reduction than discontinue due to an adverse event (AE) in an acute setting and 250 times more likely to remain relapse-free than to discontinue due to an AE in a maintenance setting.
- These findings provide clinically useful information, helping to clarify the magnitude and relevance of the benefits and risks of AOM 400 in people diagnosed with schizophrenia. A similar benefit–risk profile is expected for a once-every-2-months long-acting injectable formulation of aripiprazole in a maintenance setting, based on a comparable pharmacokinetic, safety, and efficacy profile to AOM 400.
References (21)

- Davidson M, Carpenter WT Jr. Targeted treatment of schizophrenia symptoms as they manifest, or continuous treatment to reduce the risk of psychosis recurrence. Schizophr Bull. 2024;50(1):14–21. doi:10.1093/schbul/sbad145. PubMedCrossRef
- Kahn RS, Sommer IE, Murray RM, et al. Schizophrenia. Nat Rev Dis Primers. 2015;1:15067. PubMed CrossRef
- American Psychiatric Association. Practice guideline for the treatment of patients with schizophrenia. 3rd ed.. American Psychiatric Association; 2020.
- Citrome L. Quantifying clinical relevance. Innov Clin Neurosci. 2014;11(5-6):26–30. PubMed
- Citrome L, Juday T, Frech F, et al. Lemborexant for the treatment of insomnia: direct and indirect comparisons with other hypnotics using number needed to treat, number needed to harm, and likelihood to be helped or harmed. J Clin Psychiatry. 2021;82:20m13795. PubMed CrossRef
- Citrome L, Ketter TA. When does a difference make a difference? Interpretation of number needed to treat, number needed to harm, and likelihood to be helped or harmed. Int J Clin Pract. 2013;67(5):407–411. PubMed CrossRef
- Citrome L, Kantrowitz J. Antipsychotics for the treatment of schizophrenia: likelihood to be helped or harmed, understanding proximal and distal benefits and risks. Expert Rev Neurother. 2008;8(7):1079–1091. PubMed CrossRef
- Abilify Maintena® (aripiprazole). Prescribing information. Otsuka America Pharmaceutical, Inc. https://www.otsuka-us.com/media/static/Abilify-M-PI.pdf. Accessed January 20, 2026
- Abilify Asimtufii (aripiprazole). Prescribing information. Otsuka America Pharmaceutical, Inc. https://www.otsuka-us.com/media/static/Abilify-Asimtufii-PI.pdf. Accessed January 20, 2026
- Harlin M, Yildirim M, Such P, et al. A randomized, open-label, multiple-dose, parallel-arm, pivotal study to evaluate the safety, tolerability, and pharmacokinetics of aripiprazole 2-month long-acting injectable in adults with schizophrenia or bipolar I disorder. CNS Drugs. 2023;37(4):337–350. PubMed CrossRef
- Citrome L, Such P, Yildirim M, et al. Plain language summary of publication: a comparison of once-monthly and once-every-2-months injectable formulations of aripiprazole in people with schizophrenia. Ther Adv Psychopharmacol. 2024;14:20451253241286319. PubMed CrossRef
- Citrome L, Such P, Yildirim M, et al. Safety and efficacy of aripiprazole 2-month ready-to-use 960 mg: secondary analysis of outcomes in adult patients with schizophrenia in a randomized, open-label, parallel-arm, pivotal study. J Clin Psychiatry. 2023;84(5):23m14873. PubMed CrossRef
- Kane JM, Peters-Strickland T, Baker RA, et al. Aripiprazole once-monthly in the acute treatment of schizophrenia: findings from a 12-week, randomized, double-blind, placebo-controlled study. J Clin Psychiatry. 2014;75(11):1254–1260. PubMed CrossRef
- Kane JM, Sanchez R, Perry PP, et al. Aripiprazole intramuscular depot as maintenance treatment in patients with schizophrenia: a 52-week, multicenter, randomized, double-blind, placebo-controlled study. J Clin Psychiatry. 2012;73(5):617–624. PubMed CrossRef
- Citrome L, Sánchez Del Rio M, Dong Y, et al. Benefit-risk assessment of galcanezumab versus placebo for the treatment of episodic and chronic migraine using the metrics of number needed to treat and number needed to harm. Adv Ther. 2021;38(8):4442–4460. PubMed CrossRef
- Vo P, Wen S, Martel MJ, et al. Benefit-risk assessment of erenumab and current migraine prophylactic treatments using the likelihood of being helped or harmed. Cephalalgia. 2019;39(5):608–616. PubMed CrossRef
- Citrome L. Choosing among the long-acting injectable antipsychotics: an evidence-based pragmatic guide. CNS Spectr. 2025;30(1):e73. PubMed CrossRef
- Citrome L, Neugebauer NM, Meli AA, et al. Xanomeline and trospium chloride versus placebo for the treatment of schizophrenia: a post hoc analysis of number needed to treat, number needed to harm, and likelihood to be helped or harmed. Neuropsychiatr Dis Treat. 2025;21:761–773. PubMed CrossRef
- Citrome L, Du Y, Weiden PJ. Assessing effectiveness of aripiprazole lauroxil vs placebo for the treatment of schizophrenia using number needed to treat and number needed to harm. Neuropsychiatr Dis Treat. 2019;15:2639–2646. PubMed CrossRef
- Vita G, Pollini D, Canozzi A, et al. Efficacy and acceptability of long-acting antipsychotics in acutely ill individuals with schizophrenia-spectrum disorders: a systematic review and network meta-analysis. Psychiatry Res. 2024;340:116124. PubMed CrossRef
- Gao K, Yuan C, Wu R, et al. Important clinical features of atypical antipsychotics in acute bipolar depression that inform routine clinical care: a review of pivotal studies with number needed to treat. Neurosci Bull. 2015;31(5):572–588. PubMed CrossRef
This PDF is free for all visitors!