Objective: In a sample of children and adolescents with first-episode psychosis, we investigated whether multiple treatment failure (MTF, defined as the initiation of a third trial of novel antipsychotic due to nonadherence, adverse effects, or insufficient response) was associated with comorbid autism spectrum disorders.
Methods: Data were from the electronic health records of 638 children (51% male) aged from 10 to 17 years with first-episode psychosis (per ICD-10 criteria) from January 1, 2008, to November 1, 2014, referred to mental health services in South London, United Kingdom; data were extracted using the Clinical Record Interactive Search (CRIS) system. The effect of autism spectrum disorder comorbidity on the development of MTF during a 5-year period was modeled using Cox regression.
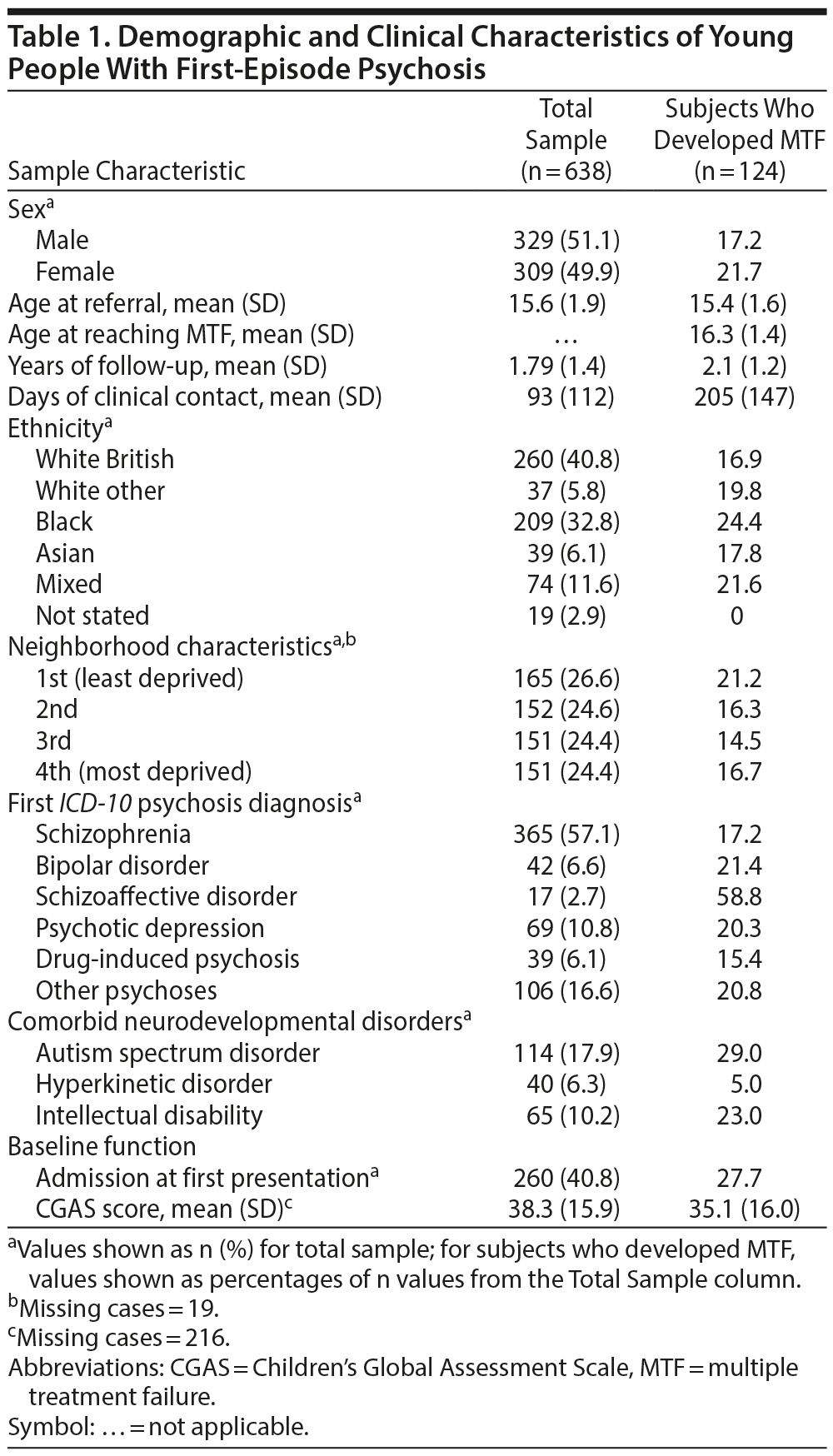
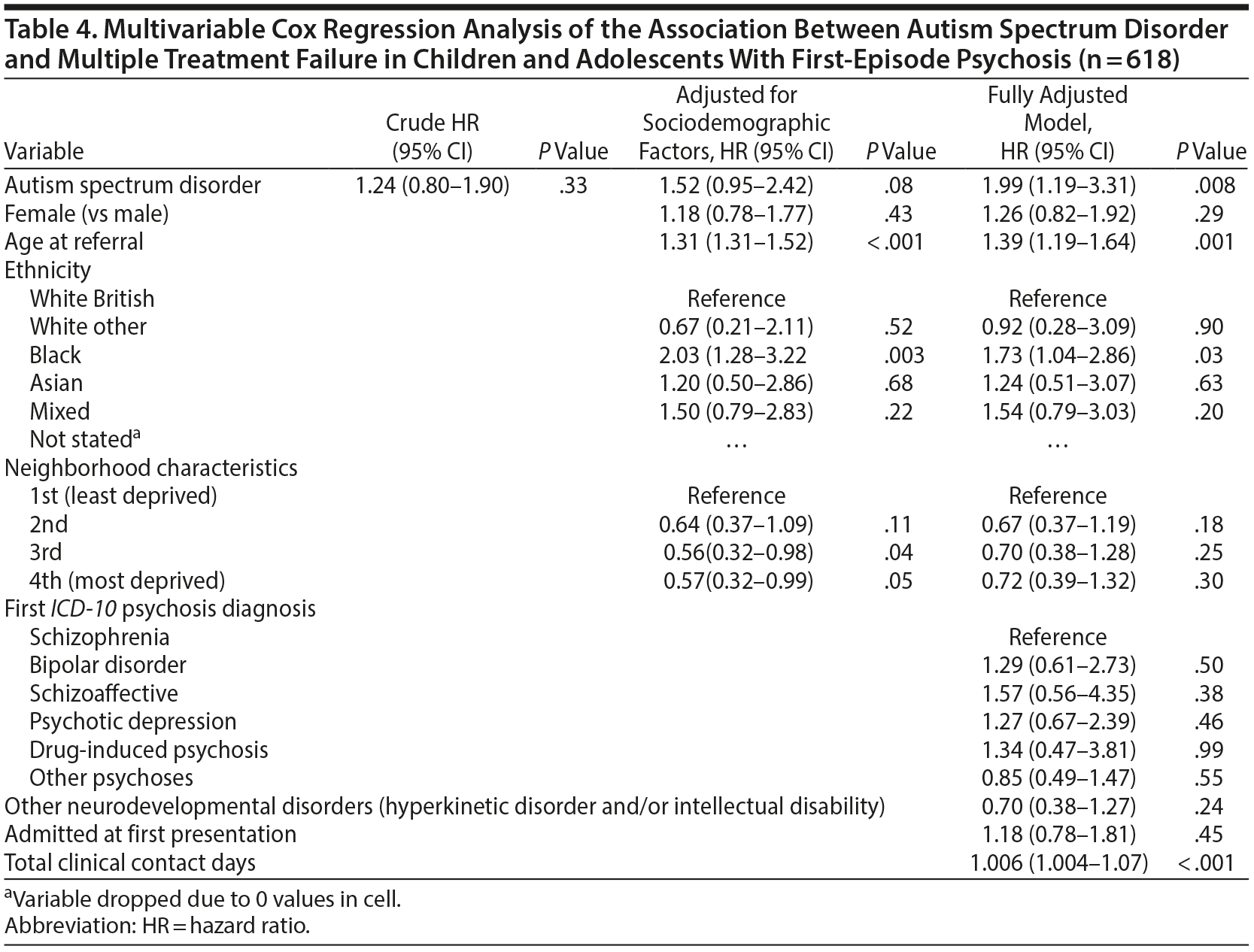
Results: There were 124 cases of MTF prior to the age of 18 (19.4% of the sample). Comorbid autism spectrum disorders were significantly associated with MTF (adjusted hazard ratio = 1.99; 95% CI, 1.19-3.31; P = .008) after controlling for a range of potential confounders. Other factors significantly associated with MTF included higher age at first presentation (P = .001), black ethnicity (P = .03), and frequency of clinical contact (P < .001). No significant association between other comorbid neurodevelopmental disorders (hyperkinetic disorder or intellectual disability) and MTF was found.
Conclusions: Children with first-episode psychosis and comorbid autism spectrum disorders at first presentation are less likely to have a beneficial response to antipsychotics.
This work may not be copied, distributed, displayed, published, reproduced, transmitted, modified, posted, sold, licensed, or used for commercial purposes. By downloading this file, you are agreeing to the publisher’s Terms & Conditions.

The Association Between Comorbid Autism Spectrum Disorders and Antipsychotic Treatment Failure in Early-Onset Psychosis:
A Historical Cohort Study Using Electronic Health Records

ABSTRACT
Objective: In a sample of children and adolescents with first-episode psychosis, we investigated whether multiple treatment failure (MTF, defined as the initiation of a third trial of novel antipsychotic due to nonadherence, adverse effects, or insufficient response) was associated with comorbid autism spectrum disorders.
Methods: Data were from the electronic health records of 638 children (51% male) aged from 10 to 17 years with first-episode psychosis (per ICD-10 criteria) from January 1, 2008, to November 1, 2014, referred to mental health services in South London, United Kingdom; data were extracted using the Clinical Record Interactive Search (CRIS) system. The effect of autism spectrum disorder comorbidity on the development of MTF during a 5-year period was modeled using Cox regression.
Results: There were 124 cases of MTF prior to the age of 18 (19.4% of the sample). Comorbid autism spectrum disorders were significantly associated with MTF (adjusted hazard ratio = 1.99; 95% CI, 1.19-3.31; P = .008) after controlling for a range of potential confounders. Other factors significantly associated with MTF included higher age at first presentation (P = .001), black ethnicity (P = .03), and frequency of clinical contact (P < .001). No significant association between other comorbid neurodevelopmental disorders (hyperkinetic disorder or intellectual disability) and MTF was found.
Conclusions: Children with first-episode psychosis and comorbid autism spectrum disorders at first presentation are less likely to have a beneficial response to antipsychotics.
J Clin Psychiatry 2017;78(9):e1233-e1241
https://doi.org/10.4088/JCP.16m11422
© Copyright 2017 Physicians Postgraduate Press, Inc.
aInstitute of Psychiatry, Psychology, and Neuroscience, King’s College London, United Kingdom
bNIHR South London and Maudsley NHS Foundation Trust Biomedical Research Centre, London, United Kingdom
cUniversity of Exeter Medical School, Exeter, United Kingdom
dDepartment of Child and Adolescent Psychiatry, Hospital General Universitario Gregorio Mara×±ón, IiSGM, School of Medicine, Universidad Complutense, CIBERSAM, Madrid, Spain
*Corresponding author: Johnny M. Downs, MSc, MRCPsych, Department of Psychological Medicine, IOPPN Biomedical Research Centre Nucleus, Ground Floor, Mapother House, PO BOX 92, King’s College London, De Crespigny Park, Denmark Hill, London SE5 8AF ([email protected]).
Nearly a fifth of individuals diagnosed with a psychotic disorder experience their first episode before they reach 18 years of age.1 Relative to adults with first-episode psychosis, children appear to have a significantly worse symptomatic and functional recovery,2 hence early-onset psychosis (EOP) may represent a more severe form of the disorder. Comparisons between first-episode psychosis in adult and child cohorts show children have poorer premorbid functioning or adjustment,1,3 greater cognitive deficits,4 more primary negative symptoms at first presentation,2 and—albeit less consistently replicated—longer durations of untreated psychosis.5 It is these factors that appear to be the most consistent predictors of poor clinical and functional improvement in EOP samples at follow-up.6
Premorbid difficulty is a broad construct, often retrospectively ascertained, that encompasses childhood history of developmental milestone delays, poor sociability, poor peer relationships, limited scholastic performance, and problems with adaptation to school and socio-sexual development.7,8 Specific neurodevelopmental conditions, such as autism spectrum disorders (ASD), which, by definition, represent the extreme manifestations of poor premorbid difficulties,9 elevate the risk of developing psychosis.10-12 While premorbid difficulties have been associated with poor outcomes in both early-onset13,14 and adult-onset psychosis,15,16 the mechanism of how they affect psychosis prognosis is unclear. One possibility is that premorbid function is associated with lower responsiveness to antipsychotic treatment, with recent evidence showing poorer premorbid function is a predictor of adult treatment-resistant schizophrenia.17
The effect of ASD on treatment effectiveness has not been examined in EOP samples. This represents an important gap in the evidence, as work in nonpsychotic conditions suggests that psychopharmacologic effectiveness is lower in populations with coexisting ASD.18 Furthermore, children and adolescents with mixed ASD-psychotic profiles are not uncommon to clinical practice.12 Recent studies show that ASD may be present in 30%-50% of children diagnosed with severe psychotic disorders.19
We conducted a longitudinal study that aimed to investigate whether comorbid ASD was associated with a pragmatic measure of poor antipsychotic treatment response in a large historical clinical cohort of children and adolescents with first-episode psychosis. We predicted that patients with comorbid ASD would be more likely to experience treatment failure. We also expected that this association would remain after taking account of potential confounders, including psychotic disorder category, and additional markers of premorbid neurodevelopmental difficulties such as co-occurring hyperkinetic disorder and intellectual disability.
METHODS
This study used data extracted from the electronic mental health records of an open cohort of children and adolescents referred to South London and Maudsley NHS Foundation Trust (SLaM) Child and Adolescent Mental Health services (CAMHS), with a first episode of any psychotic disorder between January 1, 2008, and November 1, 2014. CAMHS comprised inpatient, outpatients and early intervention for psychosis services. Over this period, SLaM provided all aspects of specialist mental health care to a catchment population of approximately 250,000 children resident within 4 London boroughs (Lambeth, Southwark, Lewisham, and Croydon). In addition to the district services, SLaM provided specialist inpatient and outpatient mental health assessment and treatment services for young people from outside the local district. Each borough had a dedicated multidisciplinary service for children, which accepted referrals for school-aged children (4-18 years; exceptionally, cases are accepted below this age) with suspected or previously confirmed neurodevelopmental disorders, displaying emotional or behavioral difficulties. Children were referred from primary care, child health, and educational and social care services and typically underwent a multidisciplinary assessment by CAMHS clinicians.

- The presentation of children and adolescents with autism spectrum disorder (ASD) and psychosis is not uncommon in clinical practice, but little is known about how well dually affected children respond to antipsychotic treatments.
- The risk of ineffective antipsychotic treatment was double in young people dually affected by ASD and psychosis.
- If a young person presents with psychosis and ASD, consider his or her elevated risk of not responding to first-line antipsychotic medication, and possibly schedule more regular reviews in the early stages of treatment.
The sample data were extracted using the Clinical Record Interactive Search (CRIS) system, which provided access to a deidentified record database containing the electronic mental health records of over 34,400 child and adolescent cases held at the UK National Institute for Health Research (NIHR) Biomedical Research Centre (BRC) for Mental Health.20 Approval was granted by the Oxfordshire Ethics Committee (ref: 08/H0606/71 + 5).
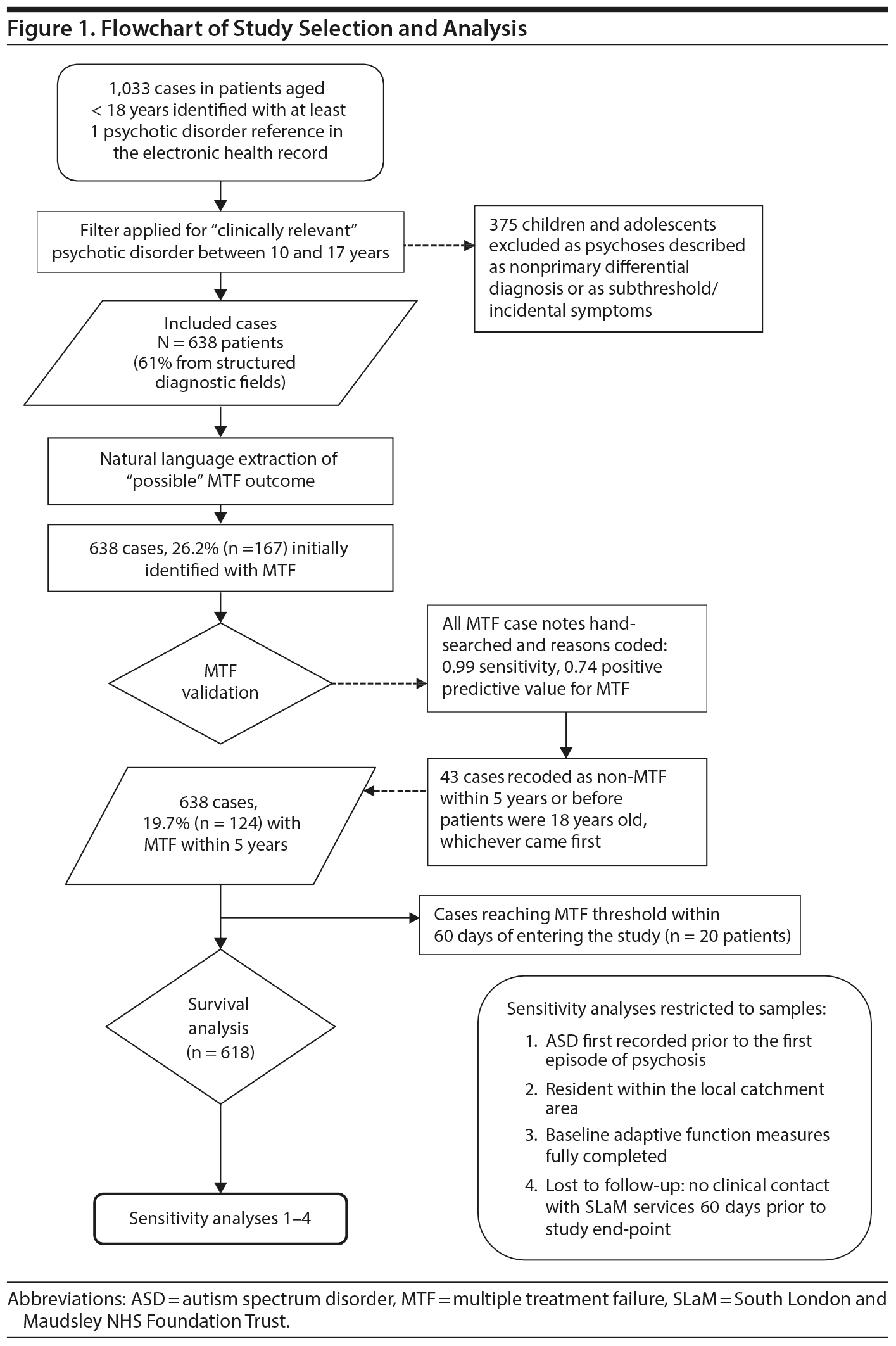
Figure 1 shows the flowchart for inclusion in the study. All patients aged between 10 and 17 years who had presented to SLaM services were screened for ICD-10 diagnoses within clinician-recorded structured or unstructured free text fields. Those with structured data recorded were included if they had at least 1 psychosis diagnosis (ICD-10 codes F20-F29, F30-31, F32.3, F33.3, F1x.5). Missing structured diagnostic data were supplemented by GATE (Generalized Architecture for Text Engineering), a natural language processing tool that codes “free text” diagnostic data.21 GATE extracted all CAMHS records with any free text diagnosis of “schizophrenia,” “schizoaffective disorder,” “bipolar disorder,” “depression with psychosis symptoms,” “acute and transient psychosis,” “delusional disorder,” “induced delusional disorder,” “drug-induced psychosis,” and “psychoses not otherwise specified (NOS).” These were filtered for any clinician-recorded mention of antipsychotic treatment after the psychosis diagnosis. This process reduced the inclusion of children with nonpsychotic indications for antipsychotic use, psychoses as differential diagnoses, and subthreshold/incidental psychotic symptoms. Of the 1,033 cases identified with at least 1 psychotic disorder recorded, only 638 individuals with a “clinically relevant psychotic disorder” were included (see Figure 1). The earliest recorded psychosis diagnosis was coded as the first diagnosis. A hand-searched review of a random sample of 100 records revealed this identification process provided a 0.98 positive predictive value (PPV) for psychotic disorder diagnosis.
For each participant, the study entry date was the accepted referral date to CAMHS for first-episode psychosis. Baseline exposure data (ie, clinical and sociodemographic data) were drawn from all notes entered within 60 days of study entry. The follow-up period ran from 60 days after their accepted referral date to whichever came first, the date of their 18th birthday, date of death, or the end of the 5-year observation period. Frequency of clinical contact during the follow-up period was determined through the days each person had received face-to-face contact as recorded in structured fields. Multiple events on a single day were counted as 1 day of clinical contact, while clinical contact with outpatient services during an inpatient admission was not counted.
In contrast to standard definitions in adults for treatment response, no established minimum antipsychotic therapeutic dose thresholds or treatment periods exist for children and adolescents with psychosis22; similarly, no standard criteria for poor antipsychotic response or refractory disorder23 appeared suitable to a retrospective cohort study of EOP using electronic health records.24,25 Therefore, we created a proxy based on the antipsychotic effectiveness literature,26-28 which we termed “multiple treatment failure” (MTF). We defined MTF as the initiation of a third trial of a novel antipsychotic due to insufficient response, intolerable adverse effects, or nonadherence to prior antipsychotic treatment. A previously validated GATE application was used to identify novel regular antipsychotic prescription trials as a replacement or adjunctive treatment to the previous trial; this excluded antipsychotic medication prescribed on an “as required basis”29,30 or switching preparations—eg, oral to depot administration. The date of MTF was determined when a third novel antipsychotic medication was started within a 5-year follow-up period. Three master’s level graduates in Early Intervention Psychosis studies (S.L., H.D., N.S.), performed further validation under joint supervision from consultants in child and adolescent (J.M.D.) and adult (L.P.-C.) psychiatry. Each rater, blinded to MTF status, hand-searched 100 cases from the sample, which included all 167 individuals (55-56 per rater) for whom MTF was initially identified and a random selection of non-MTF individuals (44 or 45 per rater). The GATE identification process provided > 0.99 sensitivity for MTF (ie, no false negatives) and 0.74 PPV. False positives largely occurred when antipsychotic medications were used for nonpsychotic indications. These cases (n = 43 subjects, 6.7% of the total sample) were subsequently recoded as non-MTF. Raters also manually coded the reasons for treatment failure for each novel antipsychotic trial in the MTF group and coded the predominant reason. Consistent with previous literature,31 reasons were defined as insufficient response, intolerable adverse effects, nonadherence, “other,” and “reasons not identified.” For 15 randomly selected cases, first and second treatment failure reasons were coded by 2 raters independently. Percentage agreement ranged from 0.67 to 0.87. κ Coefficients indicated agreement from moderate for adverse effects at first treatment failure (κ = 0.33) to substantial for insufficient response at second treatment failure (κ = 0.71). Within the MTF group, those cases identified as having the same reason for antipsychotic discontinuation/switch at first and second trials were grouped into 4 MTF “persistent reason” groups. A “variability in reasons” subgroup (ie, when reasons were different at each antipsychotic trial) was also created.
Clinician-recorded ASD comorbidity (ICD-10 F84.0, F84.1, F84.5-9) was extracted from the clinical record at any time point during the observation period using free text and structured fields.29 Compared with expert consensus, there is a high specificity for ASD diagnoses by clinicians working at a district level.32 Patients were included in the ASD group if they fulfilled ICD-10 criteria for pervasive developmental disorder after direct clinical observation and a full psychiatric and developmental history from at least 1 informant, typically the mother. The Autism Diagnostic Observation Schedule (ADOS)33 was administered by experienced ADOS-trained clinicians when the diagnosis was not clear (52 cases). The final diagnosis was based on best clinical judgment considering all the available information34 by NHS clinicians certified to administer the Autism Diagnostic Interview35 and research-certified to administer the ADOS. Additional validation of ASD diagnosis data extraction was carried out by a hand search of the 100 randomly selected cases. The data extraction methodology was found to have a high sensitivity (0.82) and PPV (0.86).
A number of demographic and clinical variables were extracted at baseline (ie, within 60 days of study entry). Demographic variables included sex, age at referral for first-episode psychosis, ethnicity (categories defined by the UK Office for National Statistics), and index of neighborhood deprivation for the main caregiver residence.36 The first clinically recorded ICD-10 psychosis diagnoses were grouped into schizophrenia, schizoaffective disorder, bipolar disorder, depression with psychotic symptoms, drug-induced psychosis, and other psychoses. Other neurodevelopmental disorder comorbidities extracted included hyperkinetic disorders (ICD-10 F90) and intellectual disability (ICD-10 F70-9). Inpatient admission status and adaptive function—ascertained using the Children’s Global Assessment Scale (CGAS)37 within 60 days of study entry—were also extracted.
To compare demographics, clinical characteristics of individuals, and MTF outcomes with and without comorbid ASD, crude analyses were conducted using χ2 for categorical variables and the Student independent t test for continuous variables.
To examine the prospective association between baseline demographics, clinical exposures, and MTF outcome, we excluded children who had MTF within the 60-day baseline period (n = 20). After checking proportional hazards assumptions, we used a Cox regression to model the association between ASD comorbidity and MTF. The first model examined the crude effect of ASD alone on MTF. Subsequent models were constructed adding potential sociodemographic and clinical confounders. Fully adjusted survival hazards and separate survival curves were plotted to compare the risk of MTF between children with and without ASD comorbidity.
To account for the potential effect of diagnostic re-classification of psychosis to ASD explaining any association between ASD and MTF, we conducted a sensitivity analyses by removing the sample of children with ASD first recorded 30 days after the first psychosis diagnosis date (n = 48). Three additional sensitivity analyses were conducted: (1) to restrict the analyses to children with complete adaptive function measures (CGAS) at first presentation (n = 394; see Figure 1), as this could be a potential confounder for any ASD-MTF association; (2) to test whether being resident within the local catchment area (as opposed to children referred from outside the 4 local districts) had an effect on the association between ASD and MTF, as families residing outside the local catchment area can receive additional non-SLaM mental health services not captured within the SLaM health record system; and (3) to test whether being potentially lost to follow-up by SLaM services (n = 295, defined as no clinical contact within 60 days of the study end point) had an effect on the association between ASD and MTF.
RESULTS
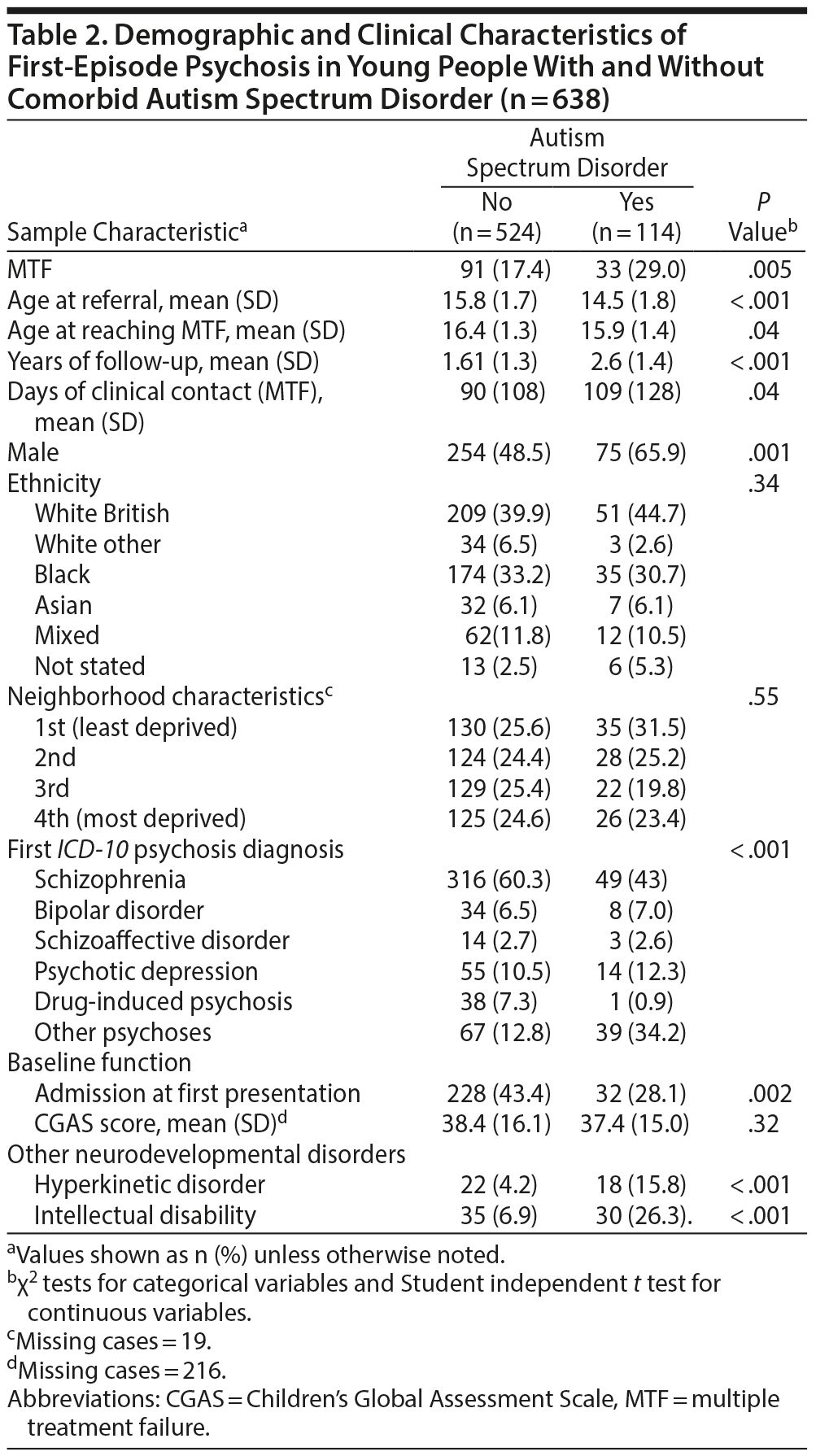
We identified 638 young people (329 male) aged from 10 to 17 years with a clinically relevant psychosis diagnosis (Figure 1). The mean follow-up period was 1.79 years (SD = 1.4, range 0.1-5). Of those, 124 (19.4%) developed MTF during the follow-up period, at a mean age of 16.3 years (SD = 1.4). Table 1 provides further information on the sociodemographic and clinical characteristics of the total sample and the subsample eventually developing MTF. Characteristics of comorbid ASD (n = 114) versus non-ASD (n = 524) subsamples are provided in Table 2. Twenty-nine percent of the sample with comorbid ASD developed MTF compared to 17% of the non-ASD sample (P < .01), and the sample with comorbid ASD reached MTF at an earlier age (P < .05).
Details on the antipsychotic treatment pathways for the 124 children who developed MTF are provided in Table 3 and in Supplementary eTable 1. The largest proportion (47%) switched their first antipsychotic due to intolerable side effects, while 21% showed insufficient response. After the second antipsychotic trial, nearly one-third of MTF children had an insufficient response (Table 3).
Supplementary eTable 1 provides information regarding treatment failure pathways. The 3 main patterns of discontinuation were the combination of insufficient response and adverse events over time (n = 28, 32.6%), persistent adverse effect (n = 18, 21%), and persistent insufficient response (n = 13, 15.1%) trajectories, with significant differences in the reasons for MTF between ASD and non-ASD groups (P = .05). Children with ASD showed higher rates of the “persistent insufficient response” or the “insufficient response-adverse effect” trajectory but lower rates of adherence-related reasons relative to those without ASD (Supplementary eTable 1).
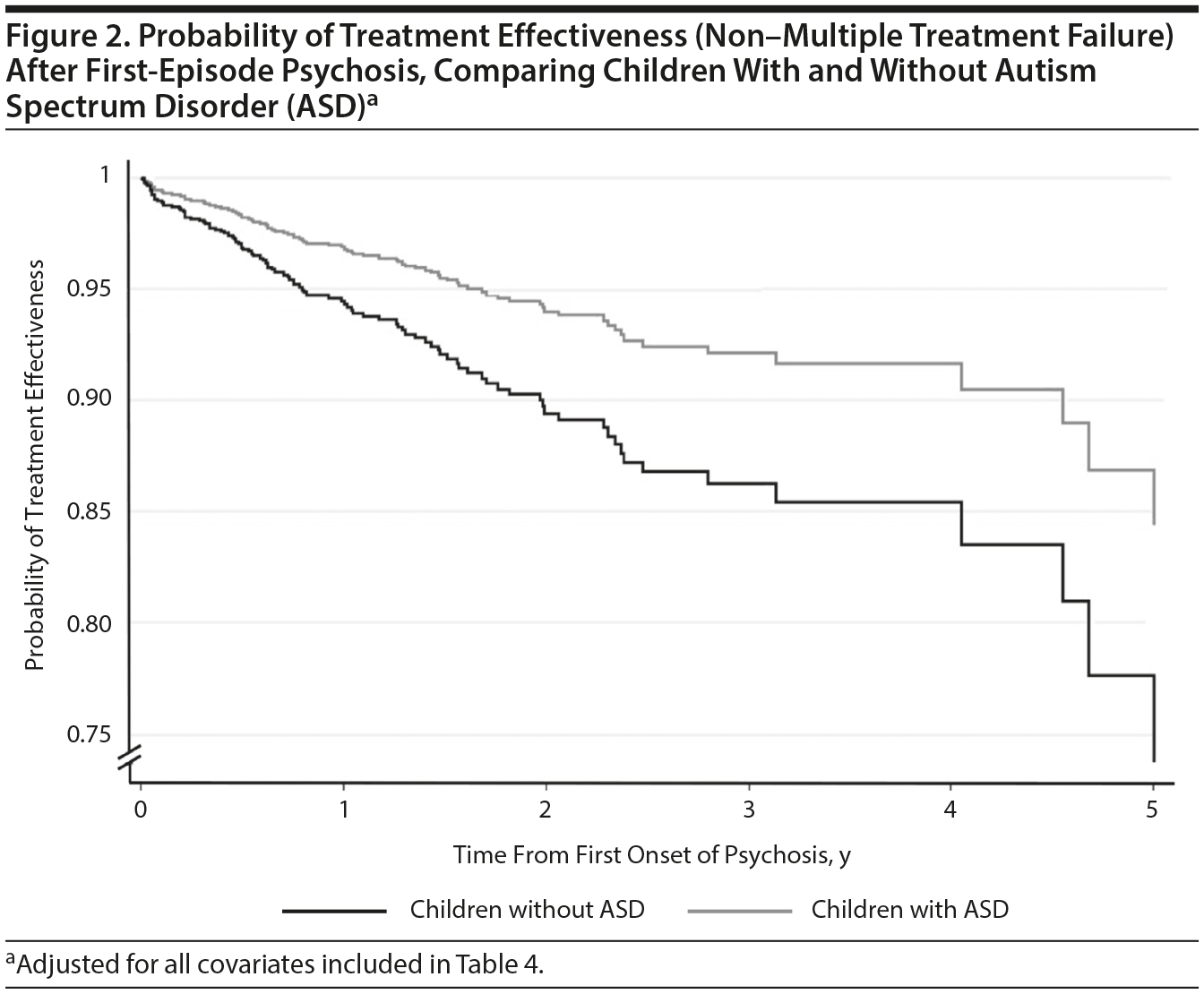
Cox regression models are displayed in Table 4 and graphically represented in Figure 2. Comorbid ASD was associated with an increased risk of reaching MTF during the follow-up period (adjusted hazard ratio [aHR] = 1.99; 95% CI, 1.19-3.31; P = .008). This was after adjusting for potential confounders including sociodemographic factors, comorbid hyperkinetic disorder or intellectual disability, and, as a marker of psychosis severity, admission status at presentation and clinical contact during the follow-up. Age at first referral, black ethnicity, and frequency of clinical contact during the follow-up period were also positively associated with MTF (see Table 4).
From the sensitivity analyses conducted, we found no change in the direction of the effect of ASD on MTF, although the restriction in sample size meant loss of statistical power. Findings were as follows: (1) the subsample of children with ASD diagnosis recorded prior to their psychosis diagnosis (excluding 48 children with comorbid ASD): aHR = 1.48; 95% CI, 0.81-2.73; P = .2; (2) children with complete CGAS information (n = 394): aHR = 1.98; 95% CI, 1.06-3.67; P = .03; (3) children resident exclusively within the local catchment area (n = 329): aHR = 1.51; 95% CI, 0.69-3.28; P = .30; (4) children with no clinical contact recorded within 60 days of the study end date (n = 295): aHR = 2.71; 95% CI, 1.14-6.39; P = .02.
DISCUSSION
This is the first longitudinal study to examine the association of comorbid ASD and poor antipsychotic treatment outcomes in children with first-episode psychosis. Using electronic health record data from community and inpatient CAMH services, we found that 19% of children developed MTF before the age of 18 years. We found that ASD comorbidity was associated with a 2-fold increased risk of MTF, after adjustment for potential sociodemographic and clinical confounders including sex, ethnicity, age at first referral, psychosis subcategory, and illness severity. Among children with MTF, most cases did not show a consistent mechanism of discontinuation over time, but, of note, 28% of those with comorbid ASD compared to 11% of non-ASD children had a persistently insufficient response to antipsychotics. These findings suggest that the effect of developmental delays and poor premorbid adjustment on antipsychotic treatment failure found in adult studies of first-episode psychosis38,39 are applicable to children with EOP.
Our findings may be explained by specific neurobiological profiles related to psychosis-ASD comorbidity. Certainly, pharmacologic treatments for nonpsychotic disorders in ASD appear to have reduced effectiveness.18 For example, children with ASD tend to respond less favorably to methylphenidate or antidepressants and experience adverse effects to these agents more often, and with greater severity, than their peers without ASD.18,40 ASD-psychosis subgroups may have a reduced dopamine synthesis capacity and diminished response to dopamine receptor-blocking antipsychotics.41,42 These theoretical mechanisms cannot be explored within the data available in this study, but our findings support further investigation into interventions that target alternative non-dopaminergic pathways in children with ASD-psychosis comorbidity.
We found other predictive factors that were significantly associated with MTF, including black ethnicity, older age at referral (proxy for age at first episode), and frequency of clinical contact. Children of black ethnicity were twice as likely as those of white British ethnicity to develop MTF, which is consistent with a number of studies in adults.43,44 Clinical contact with services was positively associated with a risk for MTF. This finding is in keeping with other research, in early-onset psychosis samples, that indicates symptom severity and increased service use are associated with a more complicated illness course.6 Male sex was not associated with an increased risk of MTF, which suggests that it is not a prognostic marker for treatment effectiveness, although it is a risk factor for psychosis in adolescence. These results accord with a number of studies examining demographic predictors for poor social functioning and treatment resistance, in early-onset25,45 and adult cohorts.46
Our findings suggest that most young people with EOP do not develop treatment failure via a consistent mechanism of discontinuation. Nearly 60% of the MTF group had different reasons for the discontinuation of each trial of novel antipsychotic. In cross-section, we found patterns of discontinuation similar to those in other early-onset studies. The Treatment of Early-Onset Schizophrenia Spectrum Study (TEOSS) found 39.2% of the discontinuers experienced an insufficient response and 36% reported adverse effects.47 Similarly, we found nearly 32% of children with MTF had switched from the first antipsychotic trial due to intolerable adverse effects and 32% showed insufficient response.
This study has a number of strengths. We studied one of the largest child and adolescent samples presenting with their first episode of psychosis, which permitted us sufficient power and precision to estimate the strength of the association between ASD and MTF while taking account of a number of potential confounders. It was a first-episode sample; hence, participants shared a common starting point in their illness course, which reduced the confounding effects of illness duration and unknown treatment exposures typically found in other early- and adult-onset schizophrenia cohort studies. Importantly, our findings can be readily generalized to clinical practice. The sample included the whole clinical population of 4 south London boroughs that were accessing “real world” inpatient and outpatient CAMH services.
Some limitations should be considered when interpreting the results of this study. As with all health record databases, there is some risk that not all clinical details are available for participants throughout the study duration. However, we would expect our data to be representative of children with psychoses living in urban and suburban areas since SLaM is a near-monopoly provider of specialist mental health care for its geographic catchment. We drew on complete electronic clinical records for over 600 cases, providing the statistical power to control for a range of potential confounders. The findings were also robust to a series of sensitivity analyses. An additional limitation that may affect our findings is diagnostic overshadowing, in which a diagnosis of psychosis may decrease the likelihood of giving additional psychiatric diagnoses. Hence, the association between ASD and MTF may be an underestimate. Another possible explanation for the observed association between ASD and increased risk of MTF could be that of misdiagnosis, in which the association found between ASD and MTF could be explained by a subgroup within comorbid ASD that better fit a “multidimensional impairment” phenotype, which we were unable to ascertain from the clinical record. Multidimensionally impaired children, first described by Kumra et al,48 present with early transient autistic features, postpsychotic cognitive decline, and psychotic symptoms that are less likely to be amenable to antipsychotic treatment.48,49 Another potential limitation is that individual reasons for each discontinuation of treatment were likely to be multifactorial. By rating treatment failure to 1 of 4 potential categories at each point of discontinuation/failure, we may have underestimated the contribution of other underlying reasons. Nonetheless, inclusion of this additional information is likely to further support our findings of the heterogeneity that underlies recurrent treatment discontinuation.
Our results are consistent with the evidence that shows psychotic illness experienced by children and adults with ASD may be different from non-ASD samples,10,12 as we found diagnostic profiles in children with ASD comorbidity had lower rates of ICD-10 schizophrenia and higher rates of psychosis not otherwise specified. Although there are risks of diagnostic misclassification between psychotic illness and ASD within our clinical sample, we believe the availability of detailed professional observations of children’s behavior within the free text records has provided a greater clarity in the diagnostic validation of these complex symptoms, which are not always feasible using structured assessments.50
Our findings provide evidence at arguably the most sensitive point in psychosis development that may help guide early detection of those children and adolescents at risk of not responding to first-line antipsychotic medications. Our findings may help delineate a subgroup of first-episode patients with EOP—ie, those with comorbid ASD—who have nearly double the risk for eventual development of MTF. This may explain why some children with premorbid difficulties and EOP are at increased risk for adverse social, educational, and occupational functioning.6,51 Furthermore, given the size of our sample, the longitudinal nature of the analyses, and comprehensive review of the psychotic symptoms within the electronic health care record, we believe our findings provide further support for the atypical diagnostic distribution for psychotic illness in ASD previously described in both adult and child populations.10,12 Further work could focus on identifying reliable predictors of response to non-dopaminergic treatments and adjunctive non-pharmacologic interventions, which would enable stratified or individualized treatment in specific patient subgroups, such as those children with psychosis and comorbid ASD. This work could help direct finite resources to improve outcomes for those most in need and reduce the current heterogeneity of therapeutic response.52
Submitted: December 21, 2016; accepted April 12, 2017.
Published online: November 7, 2017.
Potential conflicts of interest: Dr Hayes and Mr Shetty have received research funding from Roche, Pfizer, Johnson & Johnson, and Lundbeck. Prof Arango has been a consultant to, or received honoraria/grants from, Abbot, AMGEN, AstraZeneca, Bristol-Myers Squibb, Caja Navarra, Otsuka, Pfizer, Roche, Servier, Shire, Schering-Plough, and Takeda. Mss Lechler and Sears, Mr Dean, and Drs Downs, Patel, Simonoff, Hotopf, Ford, Diaz-Caneja, MacCabe, and Pina-Camacho have no conflicts to disclose.
Funding/support: This article represents independent research partly funded by the National Institute for Health Research (NIHR) Biomedical Research Centre at South London and Maudsley NHS Foundation Trust and King’s College London. Dr Downs is supported by a Medical Research Council (MRC) Clinical Research Training Fellowship (MR/L017105/1). Mr Dean and Ms Lechler received salary support from the Foundation of Professional Services to Adolescents, UK. Dr Hayes was funded by an MRC Population Health Scientist Fellowship (MR/J01219X/1). Dr Patel was funded by an MRC CRTF (MR/K002813/1). Professors Hotopf and Simonoff are NIHR Senior Investigators. Professor Arrango, Dr Pina-Camacho, and Dr Diaz-Caneja received honoraria or grants from Instituto de Salud Carlos III, co-financed by funds from the ERDF, European Commission, CIBERSAM, Madrid Regional Government, EU Structural Funds and Seventh Framework Program, Fundación Alicia Koplowitz, and Fundación Mutua Madrileña. Other funders include Guy’s and St Thomas’ Charity and the Maudsley Charity (BRC-2011-10035).
Role of the sponsor: No funders had any role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; and preparation, review, or approval of the manuscript.
Disclaimer: The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health.
Previous presentation: This study was presented at the 16th International ESCAP Congress, Madrid, Spain, June 20-24, 2015, and the 5th Biennial Schizophrenia International Research Conference. Florence, Italy, April 2-6, 2016.
Acknowledgment: The authors appreciate the technical support from informatics personnel in the Biomedical Research Centre.
Additional information: The ethical approval to access CRIS data (Oxfordshire Research Ethics Committee C 08/H0606/71+5) requires the data to be stored behind an NHS firewall with access governed by a patient-led oversight committee. For this reason, the data cannot be made available in the manuscript, Supporting Information files, or a public repository. However, subject to approval from the oversight committee, data access for research purposes is possible by contacting Prof Robert Stewart ([email protected]), who is the academic lead for the CRIS data resource.
Supplementary material: See accompanying pages.
REFERENCES
1. Schimmelmann BG, Conus P, Cotton S, et al. Pre-treatment, baseline, and outcome differences between early-onset and adult-onset psychosis in an epidemiological cohort of 636 first-episode patients. Schizophr Res. 2007;95(1-3):1-8. PubMed doi:10.1016/j.schres.2007.06.004
2. Ballageer T, Malla A, Manchanda R, et al. Is adolescent-onset first-episode psychosis different from adult onset? J Am Acad Child Adolesc Psychiatry. 2005;44(8):782-789. PubMed doi:10.1097/01.chi.0000164591.55942.ea
3. Hui CL, Li AW, Leung CM, et al. Comparing illness presentation, treatment and functioning between patients with adolescent- and adult-onset psychosis. Psychiatry Res. 2014;220(3):797-802. PubMed doi:10.1016/j.psychres.2014.08.046
4. Rajji TK, Ismail Z, Mulsant BH. Age at onset and cognition in schizophrenia: meta-analysis. Br J Psychiatry. 2009;195(4):286-293. PubMed doi:10.1192/bjp.bp.108.060723
5. Stentebjerg-Olesen M, Pagsberg AK, Fink-Jensen A, et al. Clinical characteristics and predictors of outcome of schizophrenia-spectrum psychosis in children and adolescents: a systematic review. J Child Adolesc Psychopharmacol. 2016;26(5):410-427. PubMed doi:10.1089/cap.2015.0097
6. Díaz-Caneja CM, Pina-Camacho L, Rodríguez-Quiroga A, et al. Predictors of outcome in early-onset psychosis: a systematic review. NPJ Schizophr. 2015;1:14005. PubMed doi:10.1038/npjschz.2014.5
7. Cannon-Spoor HE, Potkin SG, Wyatt RJ. Measurement of premorbid adjustment in chronic schizophrenia. Schizophr Bull. 1982;8(3):470-484. PubMed doi:10.1093/schbul/8.3.470
8. Del Rey-Mejías Á, Fraguas D, Díaz-Caneja CM, et al. Functional deterioration from the premorbid period to 2 years after the first episode of psychosis in early-onset psychosis. Eur Child Adolesc Psychiatry. 2015;24(12):1447-1459. PubMed doi:10.1007/s00787-015-0693-5
9. Sporn AL, Addington AM, Gogtay N, et al. Pervasive developmental disorder and childhood-onset schizophrenia: comorbid disorder or a phenotypic variant of a very early onset illness? Biol Psychiatry. 2004;55(10):989-994. PubMed doi:10.1016/j.biopsych.2004.01.019
10. Larson FV, Wagner AP, Jones PB, et al. Psychosis in autism: comparison of the features of both conditions in a dually affected cohort. Br J Psychiatry. 2017;210(4):269-275. PubMed doi:10.1192/bjp.bp.116.187682
11. Khandaker GM, Stochl J, Zammit S, et al. A population-based longitudinal study of childhood neurodevelopmental disorders, IQ and subsequent risk of psychotic experiences in adolescence. Psychol Med. 2014;44(15):3229-3238. PubMed doi:10.1017/S0033291714000750
12. Pina-Camacho L, Parellada M, Kyriakopoulos M. Autism spectrum disorder and schizophrenia: boundaries and uncertainties. BJ Psych Adv. 2016;22(5):316-324. doi:10.1192/apt.bp.115.014720
13. Hassan GAM, Taha GRA. Long term functioning in early onset psychosis: two years prospective follow-up study. Behav Brain Funct. 2011;7:28. PubMed doi:10.1186/1744-9081-7-28
14. Fleischhaker C, Schulz E, Tepper K, et al. Long-term course of adolescent schizophrenia. Schizophr Bull. 2005;31(3):769-780. PubMed doi:10.1093/schbul/sbi014
15. Starling J, Dossetor D. Pervasive developmental disorders and psychosis. Curr Psychiatry Rep. 2009;11(3):190-196. PubMed doi:10.1007/s11920-009-0030-0
16. Sheitman BB, Kraus JE, Bodfish JW, et al. Are the negative symptoms of schizophrenia consistent with an autistic spectrum illness? Schizophr Res. 2004;69(1):119-120. PubMed doi:10.1016/S0920-9964(03)00177-4
17. Frank J, Lang M, Witt SH, et al. Identification of increased genetic risk scores for schizophrenia in treatment-resistant patients. Mol Psychiatry. 2015;20(2):150-151. PubMed doi:10.1038/mp.2014.56
18. Politte LC, Henry CA, McDougle CJ. Psychopharmacological interventions in autism spectrum disorder. Harv Rev Psychiatry. 2014;22(2):76-92. PubMed doi:10.1097/HRP.0000000000000030
19. Rapoport J, Chavez A, Greenstein D, et al. Autism spectrum disorders and childhood-onset schizophrenia: clinical and biological contributions to a relation revisited. J Am Acad Child Adolesc Psychiatry. 2009;48(1):10-18. PubMed doi:10.1097/CHI.0b013e31818b1c63
20. Perera G, Broadbent M, Callard F, et al. Cohort profile of the South London and Maudsley NHS Foundation Trust Biomedical Research Centre (SLaM BRC) Case Register: current status and recent enhancement of an electronic mental health record-derived data resource. BMJ Open. 2016;6(3):e008721. PubMed doi:10.1136/bmjopen-2015-008721
21. Cunningham H. GATE, a General Architecture for Text Engineering. Comput Hum. 2002;36(2):223-254. doi:10.1023/A:1014348124664
22. National Collaborating Centre for Mental Health (UK). Psychosis and Schizophrenia in Children and Young People: Recognition and Management. Leicester, UK: British Psychological Society; 2013.
23. Correll CU, Kishimoto T, Nielsen J, et al. Quantifying clinical relevance in the treatment of schizophrenia. Clin Ther. 2011;33(12):B16-B39. PubMed doi:10.1016/j.clinthera.2011.11.016
24. Suzuki T, Remington G, Mulsant BH, et al. Defining treatment-resistant schizophrenia and response to antipsychotics: a review and recommendation. Psychiatry Res. 2012;197(1-2):1-6. PubMed doi:10.1016/j.psychres.2012.02.013
25. Schneider C, Papachristou E, Wimberley T, et al. Clozapine use in childhood and adolescent schizophrenia: a nationwide population-based study. Eur Neuropsychopharmacol. 2015;25(6):857-863. PubMed doi:10.1016/j.euroneuro.2015.02.003
26. Lieberman JA, Stroup TS, McEvoy JP, et al; Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) Investigators. Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med. 2005;353(12):1209-1223. PubMed doi:10.1056/NEJMoa051688
27. Patel R, Wilson R, Jackson R, et al. Association of cannabis use with hospital admission and antipsychotic treatment failure in first episode psychosis: an observational study. BMJ Open. 2016;6(3):e009888. PubMed doi:10.1136/bmjopen-2015-009888
28. Kahn RS, Fleischhacker WW, Boter H, et al; EUFEST study group. Effectiveness of antipsychotic drugs in first-episode schizophrenia and schizophreniform disorder: an open randomised clinical trial. Lancet. 2008;371(9618):1085-1097. PubMed doi:10.1016/S0140-6736(08)60486-9
29. Downs J, Hotopf M, Ford T, et al. Clinical predictors of antipsychotic use in children and adolescents with autism spectrum disorders: a historical open cohort study using electronic health records. Eur Child Adolesc Psychiatry. 2016;25(6):649-658. PubMed doi:10.1007/s00787-015-0780-7
30. Hayes RD, Downs J, Chang C-K, et al. The effect of clozapine on premature mortality: an assessment of clinical monitoring and other potential confounders. Schizophr Bull. 2015;41(3):644-655. PubMed doi:10.1093/schbul/sbu120
31. Weiden PJ. Discontinuing and switching antipsychotic medications: understanding the CATIE schizophrenia trial. J Clin Psychiatry. 2007;68(suppl 1):12-19. PubMed
32. Baird G, Simonoff E, Pickles A, et al. Prevalence of disorders of the autism spectrum in a population cohort of children in South Thames: the Special Needs and Autism Project (SNAP). Lancet. 2006;368(9531):210-215. PubMed doi:10.1016/S0140-6736(06)69041-7
33. Lord C, Rutter M, DiLavore P, et al. Autism Diagnostic Observation Schedule (ADOS). Los Angeles, CA: Western Psychological Services; 2000.
34. Volkmar F, Siegel M, Woodbury-Smith M, et al; American Academy of Child and Adolescent Psychiatry (AACAP) Committee on Quality Issues (CQI). Practice parameter for the assessment and treatment of children and adolescents with autism spectrum disorder. J Am Acad Child Adolesc Psychiatry. 2014;53(2):237-257. PubMed doi:10.1016/j.jaac.2013.10.013
35. Lord C, Rutter M, Le Couteur A. Autism Diagnostic Interview-Revised: a revised version of a diagnostic interview for caregivers of individuals with possible pervasive developmental disorders. J Autism Dev Disord. 1994;24(5):659-685. PubMed doi:10.1007/BF02172145
36. McLennan D, Barnes H, Noble M, et al. The English Indices of Deprivation 2010. London, UK: Department for Communities and Local Government; 2011.
37. Shaffer D, Gould MS, Brasic J, et al. A children’s global assessment scale (CGAS). Arch Gen Psychiatry. 1983;40(11):1228-1231. PubMed doi:10.1001/archpsyc.1983.01790100074010
38. Rabinowitz J, Harvey PD, Eerdekens M, et al. Premorbid functioning and treatment response in recent-onset schizophrenia. Br J Psychiatry. 2006;189:31-35. PubMed doi:10.1192/bjp.bp.105.013276
39. Rabinowitz J, Napryeyenko O, Burba B, et al. Premorbid functioning and treatment response in recent-onset schizophrenia: prospective study with risperidone long-acting injectable. J Clin Psychopharmacol. 2011;31(1):75-81. PubMed doi:10.1097/JCP.0b013e31820568c6
40. Research Units on Pediatric Psychopharmacology Autism Network. Randomized, controlled, crossover trial of methylphenidate in pervasive developmental disorders with hyperactivity. Arch Gen Psychiatry. 2005;62(11):1266-1274. PubMed doi:10.1001/archpsyc.62.11.1266
41. Kästner A, Begemann M, Michel TM, et al. Autism beyond diagnostic categories: characterization of autistic phenotypes in schizophrenia. BMC Psychiatry. 2015;15:115. PubMed doi:10.1186/s12888-015-0494-x
42. Demjaha A, Egerton A, Murray RM, et al. Antipsychotic treatment resistance in schizophrenia associated with elevated glutamate levels but normal dopamine function. Biol Psychiatry. 2014;75(5):e11-e13. PubMed doi:10.1016/j.biopsych.2013.06.011
43. Lin K-M, Poland R. Ethnicity, culture, and psychopharmacology. In: Bloom FE, Kupfer DJ, eds. Psychopharmacology. The Fourth Generation of Progress. New York, NY: American College of Neuropsycopharmacology, Raven Press; 2000:1907-1917.
44. McCutcheon R, Beck K, Bloomfield MAP, et al. Treatment resistant or resistant to treatment? antipsychotic plasma levels in patients with poorly controlled psychotic symptoms. J Psychopharmacol. 2015;29(8):892-897. PubMed doi:10.1177/0269881115576688
45. Meng H, Schimmelmann BG, Mohler B, et al. Pretreatment social functioning predicts 1-year outcome in early onset psychosis. Acta Psychiatr Scand. 2006;114(4):249-256. PubMed doi:10.1111/j.1600-0447.2006.00773.x
46. Wimberley T, Støvring H, Sørensen HJ, et al. Predictors of treatment resistance in patients with schizophrenia: a population-based cohort study. Lancet Psychiatry. 2016;3(4):358-366. PubMed doi:10.1016/S2215-0366(15)00575-1
47. Findling RL, Johnson JL, McClellan J, et al. Double-blind maintenance safety and effectiveness findings from the Treatment of Early-Onset Schizophrenia Spectrum (TEOSS) study. J Am Acad Child Adolesc Psychiatry. 2010;49(6):583-594, quiz 632. PubMed
48. Kumra S, Jacobsen LK, Lenane M, et al. “Multidimensionally impaired disorder”: is it a variant of very early-onset schizophrenia? J Am Acad Child Adolesc Psychiatry. 1998;37(1):91-99. PubMed doi:10.1097/00004583-199801000-00022
49. Buitelaar JK, van der Gaag RJ. Diagnostic rules for children with PDD-NOS and multiple complex developmental disorder. J Child Psychol Psychiatry. 1998;39(6):911-919. PubMed doi:10.1017/S0021963098002820
50. Kyriakopoulos M, Stringaris A, Manolesou S, et al. Determination of psychosis-related clinical profiles in children with autism spectrum disorders using latent class analysis. Eur Child Adolesc Psychiatry. 2015;24(3):301-307. PubMed doi:10.1007/s00787-014-0576-1
51. Remschmidt H, Theisen F. Early-onset schizophrenia. Neuropsychobiology. 2012;66(1):63-69. PubMed doi:10.1159/000338548
52. Kane JM, Correll CU. Past and present progress in the pharmacologic treatment of schizophrenia. J Clin Psychiatry. 2010;71(9):1115-1124. PubMed doi:10.4088/JCP.10r06264yel
Editor’s Note: We encourage authors to submit papers for consideration as a part of our Early Career Psychiatrists section. Please contact Erika F. H. Saunders, MD, at [email protected].
This PDF is free for all visitors!