ABSTRACT
Background: Because randomized clinical trials in bipolar disorder include restricted study populations, the possibilities for generalizing to real-world bipolar patients are limited. Naturalistic long-term data can add valuable information about the diversity of treatment and outcome in bipolar disorder.
Method: After discharge from a psychiatric community hospital, 300 consecutively admitted patients with ICD-10 bipolar I (n = 158) and II (n = 142) disorder were followed up naturalistically for 4 years. Patients were assessed with regard to time to relapse, relapse polarity in relation to index episode, prophylactic effects of prescribed medication, prescribing behaviors, and medication adherence. Drugs were chosen by the treating psychiatrists on the basis of clinical judgment. Prescribed medications included lithium, carbamazepine, valproate, lamotrigine, antidepressants, and atypical antipsychotics, all of which were compared as a single mood stabilizer or in combination with at least 2 prophylactic agents. The study was conducted from 2000 through 2008.
Results: 204 of 300 patients (68%) relapsed within 4 years, with a mean of 208 days (SD = 356.2) until the next affective episode. Relapses correlated in a statistically significant manner with the index episode (χ24 = 57.48, P = .000; bipolar I: χ24 = 20.19, P = .000; bipolar II: χ24 = 106.82, P = .000). A Kaplan survival analysis showed that lithium in monotherapy statistically significantly delayed time to the next affective relapse (P = .002). Survival (time to relapse) was also statistically significantly reduced when prophylactic medication was changed by the psychiatrist (P = .000) or stopped by the patient (P = .001). In general, no differences in tested parameters were seen between the bipolar I and II groups.
Conclusions: Our data confirm a high risk of relapse in a naturalistic setting. Lithium seems to offer some advantage over other medication over the long-term treatment of bipolar I and II disorder. Patients tend to relapse with the same polarity as their index episode; this emphasizes the importance of the polarity concept. Changing of medications by the psychiatrist and stopping of medication by the patient appear to be risk factors for an earlier affective relapse.
Trial Registration: ClinicalTrials.gov identifier: NCT01792128
J Clin Psychiatry 2014;75(3):254-263
© Copyright 2014 Physicians Postgraduate Press, Inc.
Submitted: May 28, 2013; accepted November 7, 2013 (doi:10.4088/JCP.13m08601).
Corresponding author: Benedikt L. Amann, MD, PhD, FIDMAG Research Foundation, CIBERSAM, Dr Antoni Pujadas 38, 08830 Sant Boi de Llobregat, Spain ([email protected]).
It has been estimated that the prevalence of bipolar disorder, characterized by manic, depressive, or mixed episodes, is around 1%-2%, but growing evidence indicates that this may be a substantial underestimation.1 Treatment strategies of this complex and dynamic disease include pharmacologic drugs2 and psychosocial interventions, such as cognitive-behavioral, psychoeducational, interpersonal, and family-therapy interventions.3 However, despite the fact that pharmacologic treatment options have notably increased in the last 2 decades,2 more than half of all bipolar patients relapse within 2 years, with over 90% of bipolar patients having experienced at least 1 additional affective episode during their lifetime.4
These high rates of relapse might result from the severity of the disease or the lack of efficacy of current pharmacologic treatment options. However, it is possible that current treatment guidelines for bipolar disorder contribute, as they are based mainly on experts’ opinions and the results of randomized controlled trials (RCTs), which are the most reliable source of evidence from a methodological point of view but lack external validity. The rigid study designs and strict inclusion and especially exclusion criteria limit the possibilities of making generalizations to the real-world bipolar patient. For example, RCTs exclude bipolar patients with highly prevalent psychiatric or physical comorbidities, such as anxiety disorder, substance abuse or dependency, personality disorders, posttraumatic stress disorder, diabetes, or hypertension. This exclusion represents an important and clinically relevant selection bias that potentially disconnects recommendations of treatment guidelines based on RCTs from real-world bipolar patients. Furthermore, treatment strategies in bipolar patients are complex, with polypharmacy being the rule rather than the exception.5 The use of polypharmacy produces effects and interactions of various pharmacologic combinations, which are clearly understudied or simply as yet unknown. Further, it should be taken into account that the major study results of RCTs are derived from bipolar I patients, implying that the results are not necessarily transferable to bipolar II patients.
Observational, naturalistic, and prospective studies are therefore needed as a complementary information source to RCTs in bipolar disorder. Limitations of previous naturalistic prospective long-term studies in bipolar disorder include a focus on isolated outcome criteria, such as the evaluation of general relapse rates6-20 or the effect of lithium,21-30 quetiapine,31 or olanzapine.32 However, few studies evaluated a more global view of treatment effects on the long-term outcome of this disorder.33-35
The objective of this naturalistic, prospective study was to investigate a sample of 300 bipolar I and II patients with respect to time to the next affective relapse and its correlation to the index episode. Moreover, our aims were to compare the preventive effect of different drugs in monotherapy and in combination therapy and to evaluate the influence of existing prescribing habits in a naturalistic setting in Austria and of medication adherence behavior on long-term outcome.
METHOD
Study Design
This prospective, naturalistic, observational study was conducted in the County Hospital, Neunkirchen, Austria, from 2000 through 2008. The hospital is the only psychiatric center in a catchment area of 200,000 habitants. All diagnoses were made following ICD-10 criteria based on a detailed clinical interview and review of case notes carried out by 2 independent psychiatrists. For additional psychiatric comorbidities, the Mini-International Neuropsychiatric Interview36 was used. This instrument is validated for DSM-IV and ICD-10.

- This naturalistic study of 300 bipolar I and II patients suggests a high relapse rate within 1 year, which occurs in spite of the use of pharmacologic strategies developed on the basis of randomized controlled trials (RCTs).
- The diagnosis of bipolar I versus bipolar II disorder did not predict differences regarding response to treatment. Reducing dose and stopping medication are the strongest predictors for relapse to an affective episode.
- The authors propose unification of the results of RCTs and naturalistic studies in bipolar treatment guidelines.
At admission, demographic and clinical variables were collected for the whole sample, including physical comorbidities. Exclusion criteria were limited to the presence of neurologic diseases. During their admissions, all patients were informed informally about their diagnosis, possible risk factors, and the importance of pharmacologic treatment.
After discharge, patients typically remitted clinically and were treated by their own psychiatrist. They were subsequently evaluated in person at least once per year in the outpatient clinic of the hospital (78%) or via telephone or personal contact with their own psychiatrist (22%). No affective rating scales were applied. At follow-up visits, the psychiatrists in charge of the study made their evaluation on the basis of the web-based interview with regard to affective relapses, medications taken, and changes in medication or adherence. Medication adherence was evaluated at every visit by standard questions of our web-based interview. Relapses were defined as a deterioration or change of the affective state needing an explicit pharmacologic intervention and/or rehospitalization. In the event of a severe mood episode, patients continued the study and received additional follow-up. Patients who died a natural death or committed suicide were dropped from the study. All data were directly entered into the self-developed web-based interview.
The study was approved by the ethical committee of the University of Vienna, Austria, and all patients provided written informed consent for participation in a long-term clinical study. The trial was registered at ClinicalTrials.gov with registration number NCT01792128.
Subjects
In total, 515 bipolar I and II patients were consecutively admitted to the local psychiatric hospital in Neunkirchen, Austria, from 2000 through 2004. Of 515 admitted patients, 366 (71.1%) agreed to participate and signed the informed consent form at discharge. Of those, 66 patients (18.0%) did not enter the follow-up period after discharge from the hospital due to the following reasons: 1 patient (0.3%) moved to another country, 23 (6.3%) received a different diagnosis when they were rediagnosed, and 31 (8.4%) could not be contacted. Furthermore, 5 (1.4%) died a natural death, and 6 (1.6%) died from suicide. Thus, the final analysis included data from 300 bipolar I (n = 158) and II (n = 142) patients during 4-year follow-up, with data from at least 2 evaluations. Bipolar patients entered the prospective follow-up in a hypomanic or manic index episode, in a depressive index episode with or without psychotic symptoms, or with “other syndromes” as index episode. “Other syndromes” were defined as an anxiety syndrome, a mixed episode, or rapid cycling.
Database
The web-based database was specially designed for this study with the objective of uncomplicated data entry via the Internet. All 9 participating psychiatrists were instructed in the use of the database and personally entered corresponding data of their patients, thus avoiding errors that often occur during transcription of medical record to study protocol and eventually data entry into the database. The principal investigator of the study (C.S.) and the statistician (B.K.) regularly checked the accuracy of entered data.
Statistics
Baseline data were reported in a descriptive way. Comparisons across different treatment groups were performed on baseline data, using 1-way analyses of variance and multivariate analyses of variance with post hoc comparisons or χ2 tests. Mean survival time was calculated using Kaplan-Meier and Cox survival analyses. For all analyses, the level of significance after Bonferroni α error correction was set at .002. All tests were 2-tailed. SPSS for Windows 13.0 (IBM; Armonk, New York) was used.
RESULTS
Demographic Data Following Bipolar I and II Diagnosis
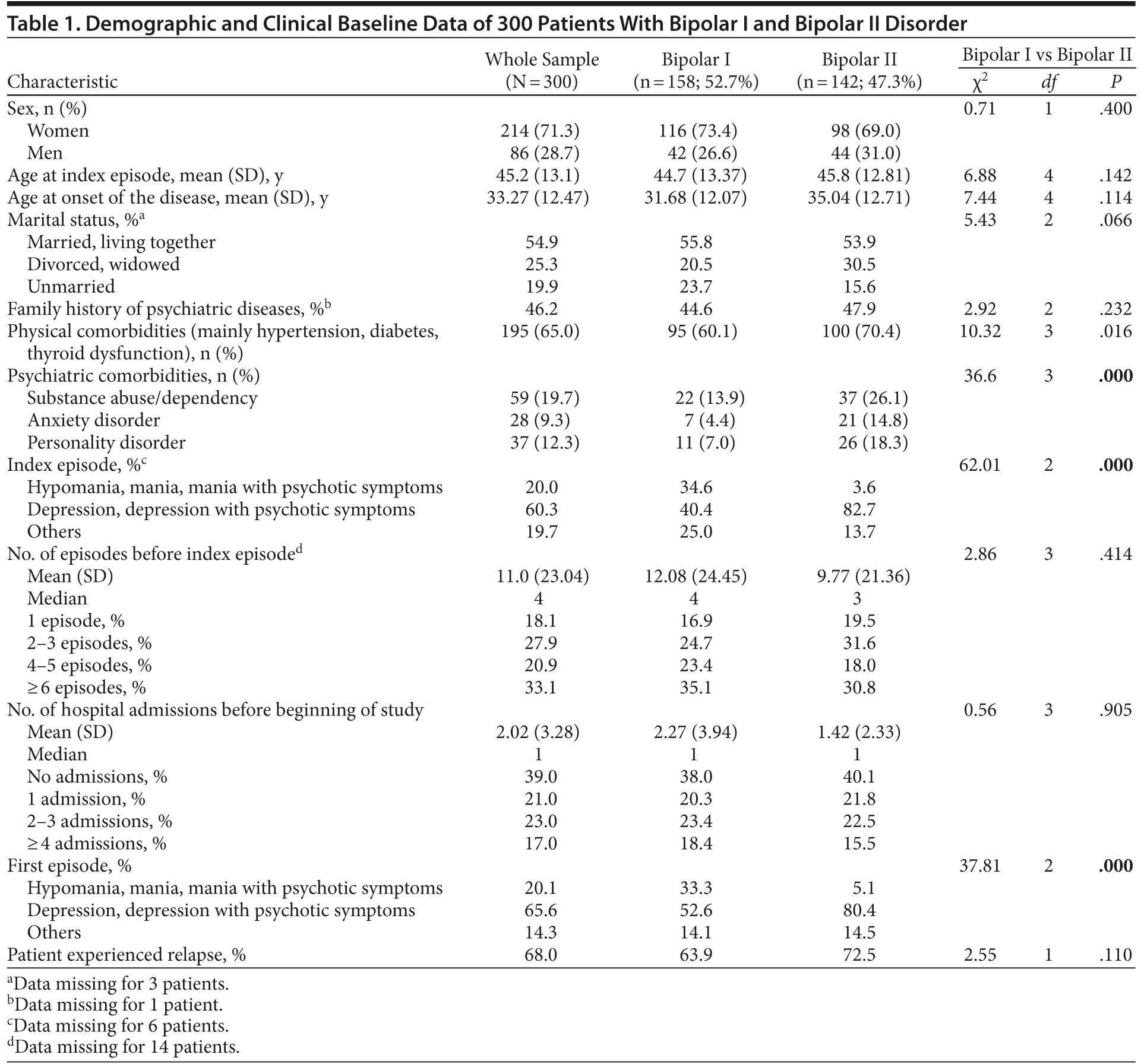
Demographic and clinical data are highlighted in Table 1. No statistical differences were found between bipolar I and bipolar II disorder with respect to sex (χ21 = 0.71, P = .4), age at inclusion (χ24 = 6.88, P = .142), age at onset of the disease (χ24 = 7.44, P = .114), marital status (χ22 = 5.43, P = .066), types of psychiatric comorbidities (χ23 = 1.464, P = .691), family history of psychiatric diseases (χ22 = 2.92, P = .232), number of admissions prior to start of study (χ23 = 0.56, P = .905), or number of affective episodes before index episode (χ23 = 2.86, P = .414). In contrast, the findings for type of first episode and index episode were statistically significant, with more manic or hypomanic episodes in the bipolar I group and more depressive episodes in the bipolar II group (χ22 = 37.81, P = .000, and χ22 = 62.01, P = .000, respectively). Furthermore, psychiatric comorbidities were significantly more frequent in the bipolar II group (χ23 = 36.6, P = .000).
Time to Relapse and Correlation With Index Episode
Three hundred bipolar I and II patients entered the prospective follow-up with the following index episodes: 20.0% presented with a hypomanic/manic episode (bipolar I: 34.6%, bipolar II: 3.6%); 60.3%, with a depressive episode with or without psychotic symptoms (bipolar I: 40.4%, bipolar II: 82.7%); and 19.7%, with “other syndromes” (bipolar I: 25%, bipolar II: 13.7%). Two hundred four patients (68.0%) had an affective relapse, as defined above, within the observation period of 4 years, with a mean of 208 days (SD = 356.2) until the next affective episode. No statistical differences were found between patients who suffered from an affective relapse within the observation period and those who did not with respect to sex (χ21 = 0.17, P = .895), age at inclusion (χ24 = 2.39, P = .664), age at onset of the disease (χ24 = 6.34, P = .175), marital status (χ22 = 3.73, P = .155), frequency of psychiatric comorbidities (χ26 = 11.33, P = .079), family history of psychiatric diseases (χ22 = 3.64, P = .162), number of admissions prior to start of study (χ23 = 7.16, P = .067), number of affective episodes before index episode (χ23 = 2.11, P = .550), polarity of first episode (χ22 = 2.92, P = .233), or polarity of index episode (χ22 = 3.17, P = .205).
Of the 204 patients with at least 1 relapse, 49.5% (n = 101) had a bipolar I diagnosis and 50.5% (n = 103) had a bipolar II diagnosis. Bipolar II patients showed a statistical tendency to relapse more frequently than bipolar I patients (P = .002; after α correction, P > .05). Relapses were diagnosed in 20.5% as a hypomanic/manic episode (bipolar I: 38.4%, bipolar II: 2.1%), in 60.0% as a depressive episode (bipolar I: 39.4%, bipolar II: 81.3%), and in 19.5% as “other syndromes” (bipolar I: 22.2%, bipolar II: 16.7%).
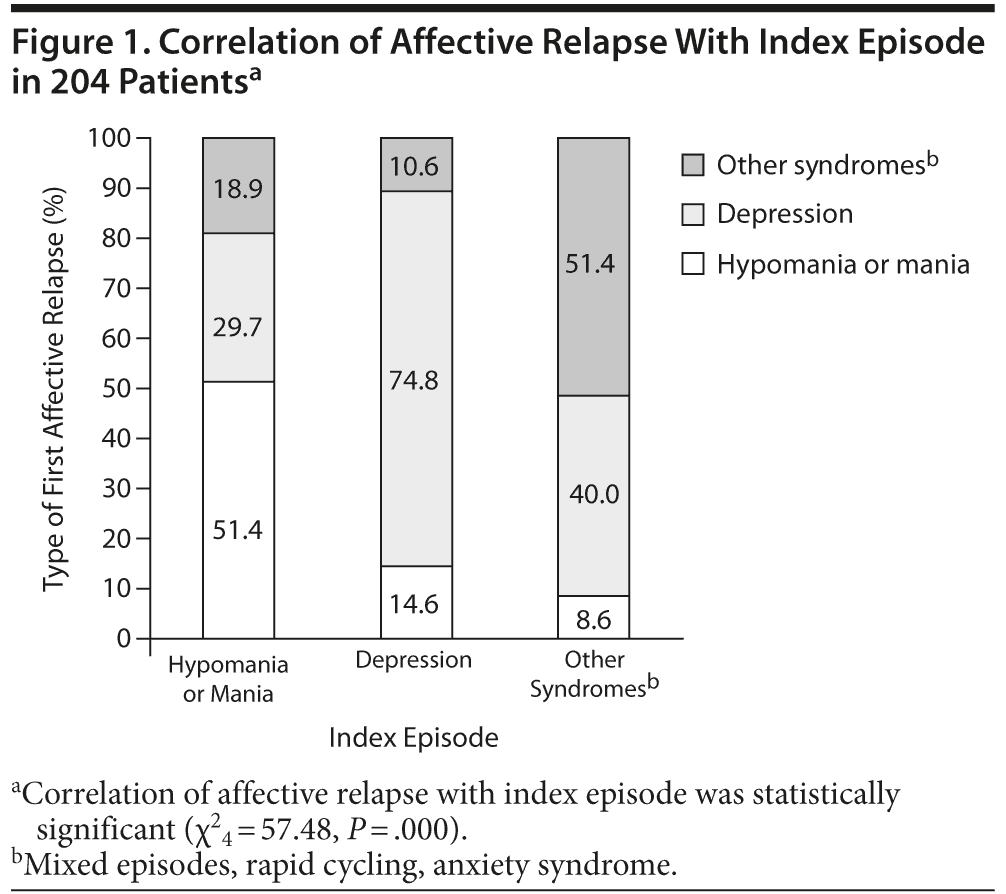
First affective relapses correlated statistically significantly with the corresponding index episode (χ24 = 57.48, P = .000; bipolar I: χ24 = 20.19, P = .000; bipolar II: χ24 = 106.82, P = .000). Of all patients with a hypomanic/manic index episode, 51.4% relapsed with a hypomanic/manic episode (bipolar I: 48.6%, bipolar II: 100%). This effect was even more pronounced when the index episode was depression, with 74.8% of depressed patients experiencing as first relapse a further depressive episode (bipolar I: 48.8%, bipolar II: 87.8%). When “other syndromes” were present as the index episode, 51.4% of those patients developed the same episode (bipolar I: 52.2%, bipolar II: 50.0%), in this case, mixed episodes, anxiety, or rapid cycling (Figure 1).
Medication
Fifty-eight patients (19.3%) in our sample received monotherapy (bipolar I: 19.6%, bipolar II: 19.0%). Of those, 9.3% (n = 28) were prescribed antidepressants (bipolar I: 7.0%, bipolar II: 12.0%), 5.7% (n = 17) lithium (bipolar I: 7.6%, bipolar II: 3.5%), 2.3% (n = 7) atypical antipsychotic drugs (bipolar I: 3.8%, bipolar II: 0.7%), and 2.0% (n = 6) anticonvulsants (bipolar I: 1.3%, bipolar II: 2.8%). It is important to note that the monotherapy groups were not included in the following analyses comparing different drugs and time to relapse due to a lack of statistical power.
A combination strategy was used in 242 bipolar patients (80.7%), with 70.0% (n = 210) receiving antidepressants (mainly selective serotonin reuptake inhibitors: 81.5%). Here, bipolar II patients (85.2%) received antidepressants significantly more often than bipolar I patients (56.7%) (χ21 = 29.71, P = .000). One hundred thirty-four (44.7%) received atypical antipsychotics (bipolar I: 51.3%, bipolar II: 37.3%), which was not statistically significant (χ21 = 5.88, P = .015). One hundred thirty-six (45.3%) were treated with lithium, which was more frequently used in bipolar I patients (57.6%) than in bipolar II patients (31.7%) (χ21 = 20.25, P = .000). Anticonvulsants were used in 36.3% of patients (n = 109), but no differences were observed between bipolar I (36.7%) and II (35.9%) patients (χ21 = 0.04, P = .851).
For our analysis of different drug combinations and time to relapse, we included treatment groups with only 1 mood stabilizer. However, additional drugs, such as antidepressants or general medicine, were allowed. The groups consisted of lithium (n = 49; 16.3%; bipolar I: 18.4%, bipolar II: 14.1%), atypical antipsychotics (n = 45, 15.0%; bipolar I: 12.7%, bipolar II: 17.6%), and anticonvulsants (n = 41; 13.7%, bipolar I: 7.6%, bipolar II: 20.4%). The only statistically significant difference in the comparison of the bipolar I and II groups was found in the anticonvulsant group, with bipolar II patients receiving anticonvulsants more frequently than bipolar I patients (χ21 = 10.43, P = .001).
Those 3 groups then were compared to groups receiving combination therapy with more than 1 prophylactic mood stabilizer, including lithium with anticonvulsants (n = 39), lithium with antipsychotics (n = 65), and antipsychotics with anticonvulsants (n = 44).
Single Mood Stabilizer Versus Combined Mood Stabilizers in Relapse Prophylaxis
With regard to preventive medication response, we first compared 4 groups receiving only 1 mood stabilizer versus 1 group receiving various mood stabilizers. All 5 groups were comparable with respect to basic demographic data, as no differences were found in sex (χ24 = 01.17, P = .884), marital status (χ28 = 6.898, P = .548), age at index episode (χ216 = 13.05, P = .669), age at onset of the disease (χ216 = 20.51, P = .198), diagnosis (χ24 = 8.171, P = .086), psychiatric comorbidities (χ224 = 22.28, P = .562), family history of psychiatric diseases (χ28 = 3.77, P = .878), first episode (χ28 = 14.51, P = .069), number of admissions prior to start of study (χ212 = 9.94, P = .621), or number of affective episodes before index episode (χ212 = 23.62, P = .023).
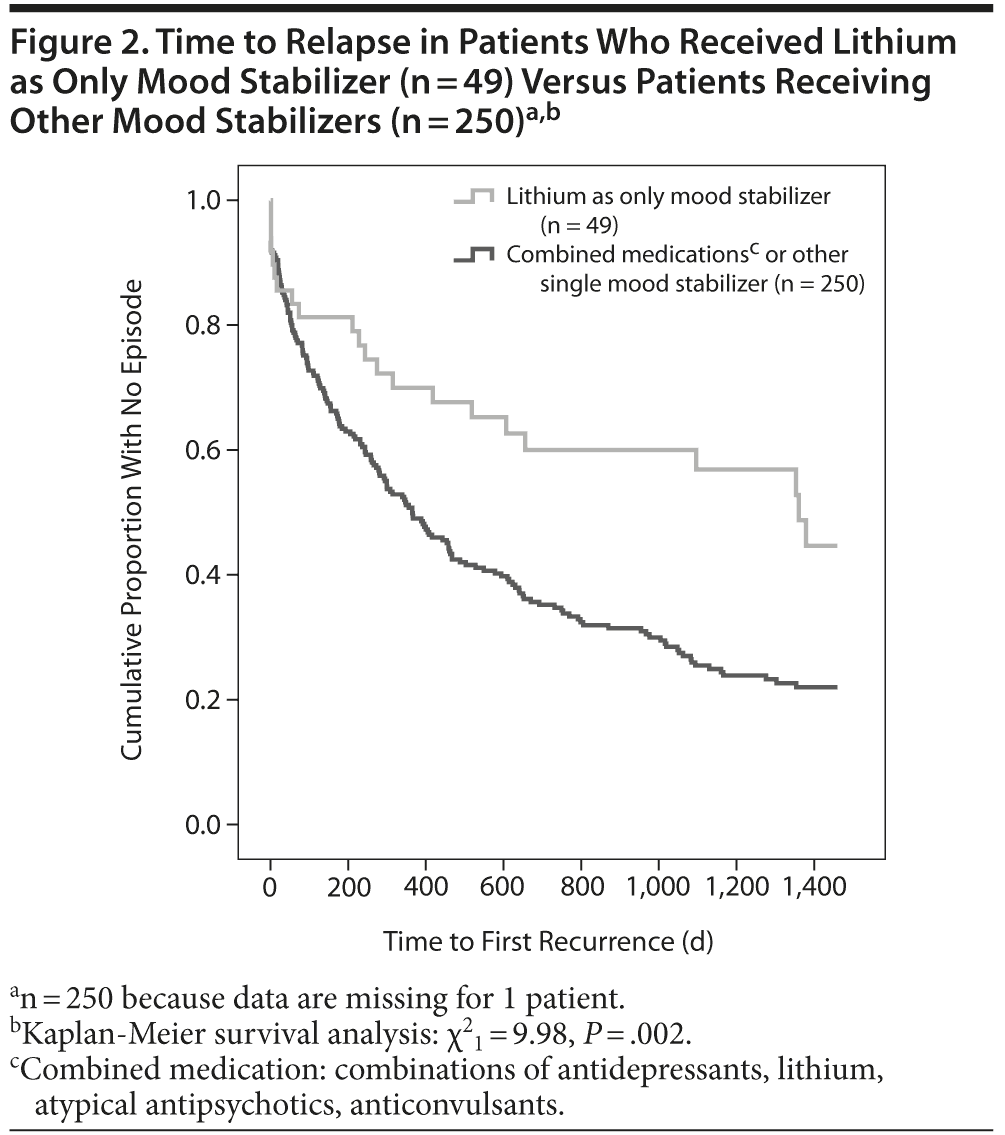
Carbamazepine (n = 16; χ21 = 1.13, P = .288), lamotrigine (n = 19; χ21 = 3.8, P = .051), and atypical antipsychotics (n = 45; χ21 = 0.24, P = .628), all used alone as mood stabilizer, revealed no statistically significant advantage in relapse prevention when compared to combined prophylactic drugs; results did not change when the covariable “medication stopped” was controlled for (carbamazepine: f1 = 1.07, P = .301; atypical antipsychotics: f1 = 0.08, P = .775; lamotrigine: f1 = 1.07, P = .301). Only lithium in monotherapy showed a statistically significant relapse prevention effect (n = 49; χ21 = 9.98, P = .002; Figure 2). This effect continued when the covariable “medication stopped” was controlled for (f1 = 12.33, P = .001). Within the anticonvulsants group (n = 41), the valproate sample (n = 6) was not included due to a lack of statistical power.
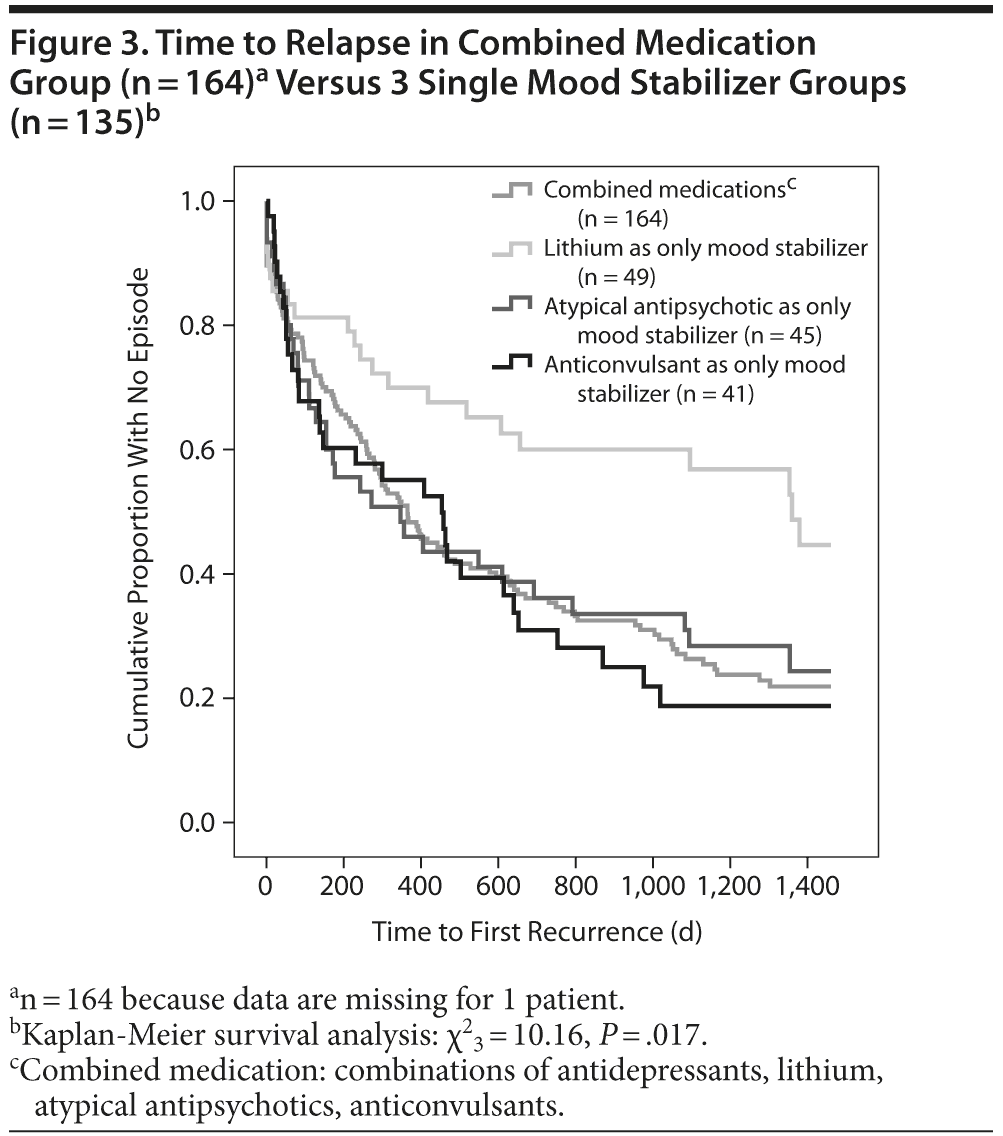
Lithium also seems to be superior to other mood stabilizers used in monotherapy and the combined prophylactic medication group with regard to days until first affective relapse: lithium (mean = 940.3, SD = 90.8) versus atypical antipsychotics (mean = 605.5, SD = 89.7) versus anticonvulsants (mean = 548.7, SD = 84.3) and combined prophylactic medication (mean = 598.3, SD = 44.7) (χ23 = 10.16, P = .017; Figure 3).
Different Combination Mood Stabilizer Strategies in Relapse Prophylaxis
Using Kaplan-Meier analysis, we found no statistical advantage in relapse prevention for carbamazepine, valproate, lamotrigine, lithium, or atypical antipsychotics in combination with other prophylactic drugs. Carbamazepine in combination did not differ statistically significantly from other prophylactic agents (n = 36; χ21 = 3.63, P = .057), which did not change when we controlled for the covariable “medication stopped” (f1 = 1.31, P = .254). Atypical antipsychotics, also in combination, did not differ statistically significantly from other combinations of prophylactic drugs (n = 134; χ21 = 0.08, P = .782). Again, this result did not change after controlling for the covariable “medication stopped” (f1 = 0.08, P = .781). The same was true for valproate in combination with other prophylactic treatment (n = 35; χ21 = 1.18, P = .278) and also when the covariable “medication stopped” was controlled for (f1 = 1.36, P = .245), for lamotrigine (n = 38; χ21 = 5.43, P = .020) and when the covariable “medication stopped” was controlled for (f1 = 3.26, P = .072), and, finally, for lithium in combination with other drugs (n = 136; χ21 = 3.77, P = .052), a result that was also maintained when the covariable “medication stopped” was controlled for (f1 = 5.35, P = .021).
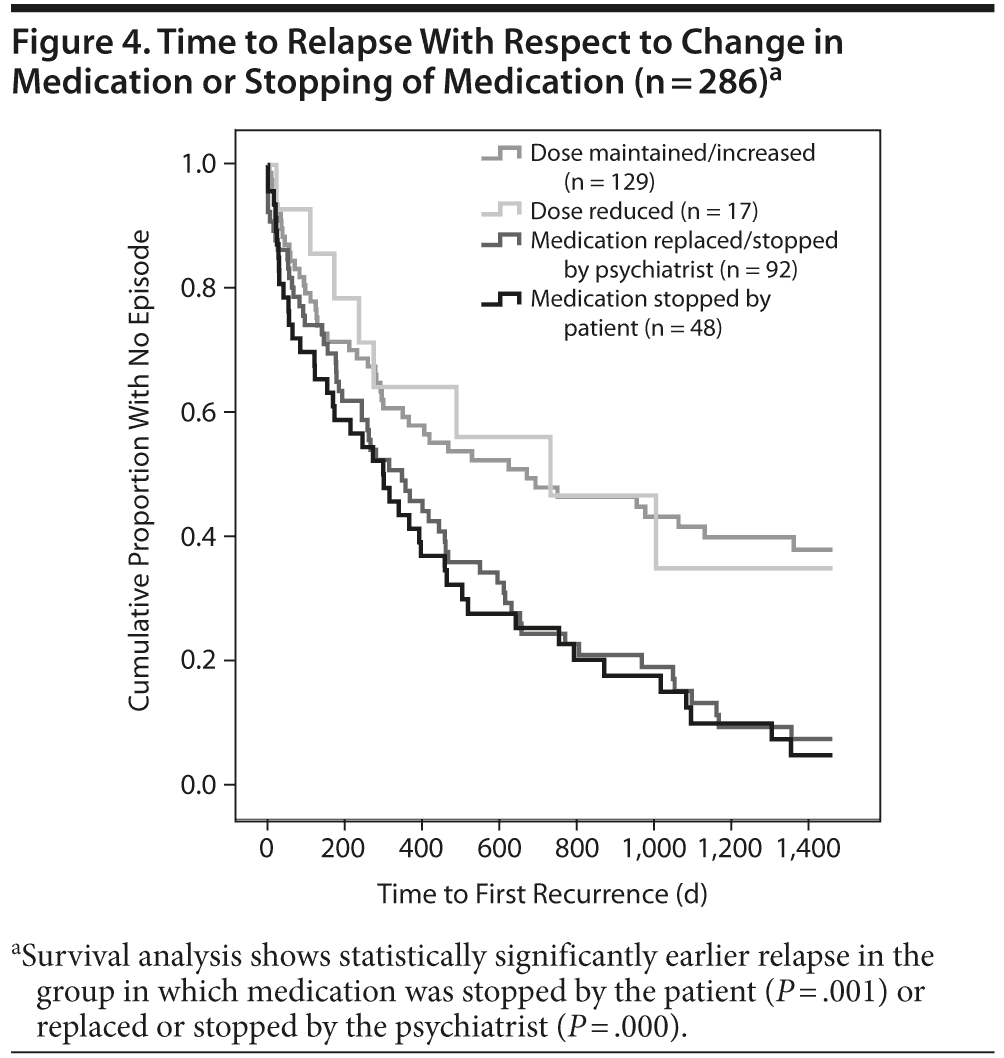
Time to Relapse With Respect to Change in Medication or Stopping of Medication
We also tested whether changes in medication such as reducing, maintaining, or increasing the dose or stopping medication by the psychiatrist or by the patient influenced the time to relapse. Changes in medication had a negative influence on general time to the next relapse (χ23 = 19.65, P = .000). As can be gathered from Figure 4, bipolar patients who stopped their medication (n = 48) or whose medication was replaced or stopped by the psychiatrist (n = 92) relapsed statistically significantly earlier (stopped by patients: χ21 = 10.97, P = .001; stopped by psychiatrist: χ21 = 13.47, P = .000, respectively) than the group of patients whose medication was maintained (n = 129; χ21 = 0.15, P = .618) or only reduced by the psychiatrist (n = 17; χ21 = 0.69, P = .405). No statistical differences in this respect were found when bipolar I or II patients were independently analyzed (χ23 = 9.51, P = .023).
Using a Cox regression model, further variables were taken into account: sex (W1 = 0.5, P = .48), age at onset (W1 = 0.95, P = .330), current age at index episode (W1 = 0.89, P = .518), polarity of first episode (W1 = 0.41, P = .521), index episode (W1 = 0.89, P = .345), episodes before index episode (W1 = 0.64, P = .423), and number of hospitalizations before index (W1 = 1.59, P = .208) revealed no statistically significant influence on our results. The only important parameter influencing relapse was whether patients did or did not take medication (W1 = 11.29, P = .001).
DISCUSSION
The aim of our study was to add real-world data of a large, representative sample of bipolar I and II patients in order to complement results of controlled clinical trials. Our study yielded some remarkable findings, such as a general high relapse rate of affective episodes: 68% of 300 bipolar I and II patients suffered at least from 1 affective relapse within 4 years, with a mean of 208 days until the next episode. Relapse rates in previous naturalistic prospective studies differ substantially, even though our results support some of those data with similar relapse rates. The European Mania in Bipolar Longitudinal Evaluation of Medication (EMBLEM) study12 indicates, for instance, that 66% of 771 bipolar I patients experienced a manic phase and 34% experienced a mixed episode within the 2-year follow-up. Another study by Silverstone and colleagues29 studied 120 bipolar I patients during 2 years and also found that two-thirds of the patients suffered from an affective recurrence in the observation period. Similar results were proposed by a 10-year follow-up of 120 bipolar I patients in which the authors compared mixed with nonmixed affective relapses.14 Even though only 37% suffered from additional mixed episodes, 63% relapsed with nonmixed episodes within the observation period. A recent Australian trial32 studied the outcome of 239 bipolar I or schizoaffective patients during a 2-year follow-up with either a conventional mood stabilizer or olanzapine with or without a conventional mood stabilizer. Independent of treatment modality, relapse rates were similar for both the cohort receiving conventional mood stabilizer alone (65%) and the cohort receiving olanzapine with or without a conventional mood stabilizer (61%). A somewhat lower relapse rate of 56% was found in 152 bipolar I and II patients on the German site of the Stanley Foundation Bipolar Network, who were followed up during 2.5 years.10 In the Systematic Treatment Enhancement Program for Bipolar Disorder (STEP-BD) trial, 1,469 bipolar patients were followed up during 24 months, with 48.5% experiencing recurrences; more than twice as many developed depressive episodes as developed hypomanic, manic, or mixed episodes.4 A similar relapse rate of 44% with at least 1 recurrence within 5-year follow-up was described in a sample of 52 adolescents with bipolar affective illness.8 Forty percent of 73 manic patients relapsed within a 1.7-year follow-up study,6 which was higher than the relapse rate in unipolar depressed patients. Even lower relapse rates were found in a Spanish naturalistic study,16 in which 87.6% of 473 bipolar patients remained in clinical remission after 1-year follow-up. The main focus of this trial was evaluating functioning and not relapse rates. However, lower relapse rates such as in the latter study16 or rates of 40%-50% such as in the aforementioned studies4,6,8 might simply be explained by the shorter observation periods, which varied between 1 and 2 years, clearly less than in our 4-year study. Looking at our relapse results at the 2-year level, we find percentages of relapse similar to those in other studies.
To summarize the results, relapse rates in observational, naturalistic bipolar studies seem to vary between 40% and almost 70%, with the latter observed in our study. It is important to note that relapse rates depend largely on the length of the observation period and the number of prior episodes, as suggested by the results from the Zurich Cohort Study showing that the risk of subsequent recurrence increases with the number of episodes.18 Our results are also in accordance with further data from the same group suggesting that bipolar disorder in general is a disorder with a high relapse rate, with bipolar II disorder presenting with only a slightly higher risk of more relapses than bipolar I disorder.19 With respect to relapse rates, we found a statistical tendency indicating more relapses in bipolar II than bipolar I patients. This is of interest, as bipolar II patients in our sample suffered from more depressive episodes, had more psychiatric comorbidities, and received more antidepressants than the bipolar I group. We could not identify further clinical or demographic characteristics for the group suffering from relapses within the observation period when compared to the group who did not suffer from relapses. Interestingly, from a historical point of view, recurrences in bipolar disorder have always been described as being high during the last 2 centuries. The prescription of modern antidepressants or mood stabilizers20 does not seem to make a big difference here.
The latter finding raises serious doubts about the efficacy of prescribed prophylactic agents recommended by current treatment guidelines in real-world bipolar patients. Specialized bipolar centers seem to be rather the exception, as they are able to improve the outcome of bipolar patients.33 Our study showed relatively low relapse prevention with prophylactic mood-stabilizer agents such as carbamazepine, valproate, lamotrigine, and some atypical antipsychotics, all prescribed as single mood stabilizers or in combination with other prophylactic mood-stabilizer drugs. Lithium still seems to be the most powerful prophylactic agent in the prevention of affective episodes in bipolar disorder, despite the emergence of various treatment alternatives in the last 2 decades, such as anticonvulsants and atypical antipsychotics. When lithium was prescribed as monotherapy or as a prophylactic agent in combination, affective relapses could be statistically significantly delayed in comparison to other mood stabilizers in monotherapy or in combination.
However, taking into account the high number of relapses despite lithium, its effect must be considered as moderate at best. Recent data indicate that lithium use for bipolar disorder has declined over the last decade, having been replaced with use of alternate, commercially promoted medications; some bipolar research groups claim that lithium should be reconsidered as a cornerstone and first-line agent in the long-term treatment of bipolar disorder.37,38 This view is based on a recent review and meta-analysis suggestive of a preventive effect of lithium in mania, even though the effect is less pronounced in depression.39 Our data support this view, as in our sample lithium was prescribed more in the classical bipolar I disorder with manic polarity than in bipolar II disorder with predominantly depressive episodes. However, there were no differences in the effectiveness of lithium in both subtypes. Naturalistic observational studies of lithium proposed a more ambivalent view on its preventive effect.21-30 Markar and Mander,21 for example, suggested “modest benefits” of lithium when compared to results from clinical trials, whereas Goldberg and colleagues22 found that 41% of lithium-treated patients had a “poor outcome”; conversely, this result means that 59% of lithium-treated patients had a favorable illness course. These findings seem like good news when compared to our results suggesting a relapse rate of almost 60% in the lithium group after 4 years of follow-up versus almost 80% among patients treated with other prophylactic agents (Figure 2). However, both of the previous studies21,22 mentioned included only 41 patients, so conclusions must be interpreted cautiously. Another study23 compared 139 bipolar patients with lithium prophylaxis and 42 without and found no significant advantage in one group over the other during a 5-year follow-up period. In a specific lithium clinic, an Italian group evaluated 402 bipolar patients 5 years after they began lithium prophylaxis and found that 38% of patients were still taking lithium and had suffered from at least 1 recurrence of the disorder, and 23% were still taking lithium and had experienced no recurrence.24 Further, 28% of the patients were no longer taking lithium at follow-up, which led the authors to conclude that it is complicated to evaluate the efficacy of lithium taking into account the high dropout rate. However, the same group later evaluated the effect of lithium on bipolar patients with mood-incongruent psychotic symptoms versus bipolar patients without. In that study, a significant effect of lithium on long-term outcome was observed in both groups.25
It is interesting that in our sample lithium in monotherapy was statistically significantly more effective in relapse prevention but lithium in combination with other mood stabilizers showed only a statistical trend. This is also an argument for a cautious reading of our results and might be interpreted to show that more severely ill patients receive more medication but do not necessarily respond better. Furthermore, lithium must be prescribed with great care, as severe side effects are possible. Physicians must be aware that serum lithium level and mood state are independently associated with lithium adverse events.30 Good adherence is of special importance in lithium treatment, as discontinuation is clearly correlated with a rapid onset of further affective episodes.29
We also found that relapses of prior stabilized patients in our sample correlated statistically significantly with the index episode, with a pronounced effect on all affective states but especially in the presence of a depressed index episode. We did not find differences in clinical variables of patients with or without relapse, but bipolar I patients relapsed more frequently with a manic or hypomanic episode, and bipolar II patients relapsed more frequently with depressive or “other syndromes” episodes. This finding adds evidence to existing data indicating that the polarity of episodes over the course of bipolar disorder and the polarity of the index episode may be 2 of the strongest predictors of recurrence to a specific affective episode.40-42 Therefore, predominant polarity, defined as at least twice as many episodes of one pole of the disorder over the other, should be taken into account for an adequate and individualized treatment selection.
As expected, relapses in our study were influenced by changes in prescription by the physician and stopping of medication by the patient. It is not clear whether the correlation of more relapses and medication change by the physician was found because the patient was already in a worse mental state at evaluation that subsequently provoked a relapse or the medication was changed by the psychiatrist for some other reason, such as just lowering the dose or acquiescing to requests by the patient. Furthermore, the relapse could also be related to poor adherence and the subsequent lack of prophylactic effect, rather than the change of medication in itself. The design of our study does not allow for clear conclusions, but we propose that psychiatrists act with caution when replacing or stopping drugs and explain to patients that stopping their medication carries a high risk of relapse. As a matter of fact, our Cox analysis showed clearly that the most protective effect against an affective relapse is that patients be adherent to at least 1 medication.
Strengths of this study include an adequate sample size; the representativeness of our sample with respect to demographic and clinical variables, especially for the bipolar I and II groups; and its global evaluation for medication effects without an a priori hypothesis in favor of a specific drug or class of drugs. Information on patients and relatives and therapists in the field was gathered by members of the hospital department and entered into the web-based database.
Limitations of our trial are important and must be taken into account when translating results to clinical work. Some strict methodological aspects of RCTs could not be integrated in the design of a naturalistic, observational study, such as periodic coordinated visits, defined dosages of drugs, pill counts, or blood sampling during all visits. Adherence was checked by questions of our web-based interview, and plasma levels were checked only in some patients and not in a continuous manner. We did not include affective scales and therefore cannot provide information on specific affective symptoms or, for example, subsyndromal symptoms. Our findings cannot be generalized to newer atypical antipsychotics, such as aripiprazole, asenapine, or paliperidone, which were not on the market in Austria at the time. We did not use a more objective and precise way to evaluate prior affective episodes, such as the Life Chart Method, in order to perform pairwise comparisons within a mirror design, which would have added valuable information to our trial.
It is probable that relapse rates observed in naturalistic studies of bipolar patients are higher than previously thought and certainly higher than in RCTs due to the RCT design characteristics mentioned above, such as shorter observation period and restricted study population without comorbidities. More intriguing is that the high relapse rate seems almost independent of the prescribed prophylactic drug, when historical data are compared to current data.20 Our naturalistic trial shows a generally poor response to prophylactic agents, independent of bipolar I or II type, with a certain advantage of lithium in relapse prevention. Furthermore, we propose that new substances should be tested long-term in bipolar disorder by way of long-term naturalistic studies with less restrictive designs, including real-world bipolar patients with psychiatric and physical comorbidities, but also by way of RCTs with an observation period of more than 2 years. Results from both RCTs and naturalistic long-term studies of bipolar patients should be integrated in recommendations of current treatment guidelines.
Drug names: aripiprazole (Abilify), asenapine (Saphris), carbamazepine (Carbatrol, Equetro, and others), lamotrigine (Lamictal and others), lithium (Lithobid and others), olanzapine (Zyprexa), paliperidone (Invega), quetiapine (Seroquel).
Disclosure of off-label usage: The authors have determined that, to the best of their knowledge, no investigational information about pharmaceutical agents that is outside US Food and Drug Administration-approved labeling has been presented in this article.
Author affiliations: Bipolar Center Wiener Neustadt, Wiener Neustadt, Austria (Drs Simhandl and König); and FIDMAG Research Foundation, Germanes Hospitaláries, and CIBERSAM, Barcelona, Spain (Dr Amann).
Author contributions: The authors are fully responsible for the planning of the study, the statistical calculations, and the reporting of this study.
Financial disclosure: Dr Amann is a member of the speakers/advisory boards for Bristol-Myers Squibb and Otsuka. Drs Simhandl and König have no personal affiliations or financial relationships with any commercial interest to disclose relative to the article.
Funding/support: Development of the web-based interview and database was funded by the ×–sterreichische Forschungsförderungsgesellschaft mbH (FFG), which is the Austrian Federal Department for Transport, Innovation, and Technology, Department of Innovations and Telecommunications. Additionally, the study was funded by grant support from the Instituto de Salud Carlos Miguel Servet Research Contract to Dr Amann (CP06/0359).
Role of the sponsor: The sponsors did not influence the study in any way.
Previous presentations: Presented as posters at the Deutsche Gesellschaft für Psychiatrie und Psychotherapie (DGPPN); January 11, 2006; Berlin, Germany; the 5th European Stanley Foundation Conference on Bipolar Disorder; October 5-7, 2006; Barcelona, Spain; and the 7th International Society for Bipolar Disorders (ISBD) conference; June 7-9, 2007; Pittsburgh, Pennsylvania.
Acknowledgments: The authors acknowledge support from the Centro de Investigación Biomédica en Red de Salud Mental (CIBERSAM). They also acknowledge support from the Research Group Industrial Software Engineering of the Technical University of Vienna and their department head Prof DI Dr Thomas Grechenig. Furthermore, they thank Drs Jutta Bilonoha, Elisabeth Denk, Birgit Gasselseder, Jens Mersch, Klaudia Mitterwachauer, Stephanie Schiebel, Christian Wunsch, Ali Zoghlami, and Daniela Renhofer for data entry. The authors acknowledge support from Thomas Simhandl, BSc, for the first version of the web-based interview and thank Jules Angst, MD, for reading and discussing the manuscript with the authors. They also thank Marylou Selo for editing the manuscript. None of the acknowledged individuals report conflict of interest.
REFERENCES
1. Fagiolini A, Forgione R, Maccari M, et al. Prevalence, chronicity, burden and borders of bipolar disorder. J Affect Disord. 2013;148(2-3):161-169. PubMed doi:10.1016/j.jad.2013.02.001
2. Fountoulakis KN, Kasper S, Andreassen O, et al. Efficacy of pharmacotherapy in bipolar disorder: a report by the WPA section on pharmacopsychiatry. Eur Arch Psychiatry Clin Neurosci. 2012;262(suppl 1):1-48. PubMed doi:10.1007/s00406-012-0323-x
3. Miklowitz DJ, Scott J. Psychosocial treatments for bipolar disorder: cost-effectiveness, mediating mechanisms, and future directions. Bipolar Disord. 2009;11(suppl 2):110-122. PubMed doi:10.1111/j.1399-5618.2009.00715.x
4. Perlis RH, Ostacher MJ, Patel JK, et al. Predictors of recurrence in bipolar disorder: primary outcomes from the Systematic Treatment Enhancement Program for Bipolar Disorder (STEP-BD). Am J Psychiatry. 2006;163(2):217-224. PubMed doi:10.1176/appi.ajp.163.2.217
5. Mojtabai R, Olfson M. National trends in psychotropic medication polypharmacy in office-based psychiatry. Arch Gen Psychiatry. 2010;67(1):26-36. PubMed doi:10.1001/archgenpsychiatry.2009.175
6. Harrow M, Goldberg JF, Grossman LS, et al. Outcome in manic disorders: a naturalistic follow-up study. Arch Gen Psychiatry. 1990;47(7):665-671. PubMed doi:10.1001/archpsyc.1990.01810190065009
7. Winokur G, Coryell W, Akiskal HS, et al. Manic-depressive (bipolar) disorder: the course in light of a prospective ten-year follow-up of 131 patients. Acta Psychiatr Scand. 1994;89(2):102-110. PubMed doi:10.1111/j.1600-0447.1994.tb01495.x
8. Strober M, Schmidt-Lackner S, Freeman R, et al. Recovery and relapse in adolescents with bipolar affective illness: a five-year naturalistic, prospective follow-up. J Am Acad Child Adolesc Psychiatry. 1995;34(6):724-731. PubMed doi:10.1097/00004583-199506000-00012
9. Coryell W, Turvey C, Endicott J, et al. Bipolar I affective disorder: predictors of outcome after 15 years. J Affect Disord. 1998;50(2-3):109-116. PubMed doi:10.1016/S0165-0327(98)00043-3
10. Dittmann S, Biedermann NC, Grunze H, et al. The Stanley Foundation Bipolar Network: results of the naturalistic follow-up study after 2.5 years of follow-up in the German centres. Neuropsychobiology. 2002;46(suppl 1):2-9. PubMed doi:10.1159/000068018
11. Nolen WA, Luckenbaugh DA, Altshuler LL, et al. Correlates of 1-year prospective outcome in bipolar disorder: results from the Stanley Foundation Bipolar Network. Am J Psychiatry. 2004;161(8):1447-1454. PubMed doi:10.1176/appi.ajp.161.8.1447
12. Azorin JM, Aubrun E, Bertsch J, et al. Mixed states vs pure mania in the French sample of the EMBLEM study: results at baseline and 24 months—European Mania in Bipolar Longitudinal Evaluation of Medication. BMC Psychiatry. 2009;9(1):33. PubMed doi:10.1186/1471-244X-9-33
13. Haro JM, Reed C, Gonzalez-Pinto A, et al; EMBLEM Advisory Board. 2-Year course of bipolar disorder type I patients in outpatient care: factors associated with remission and functional recovery. Eur Neuropsychopharmacol. 2011;21(4):287-293. PubMed doi:10.1016/j.euroneuro.2010.08.001
14. González-Pinto A, Barbeito S, Alonso M, et al. Poor long-term prognosis in mixed bipolar patients: 10-year outcomes in the Vitoria prospective naturalistic study in Spain. J Clin Psychiatry. 2011;72(5):671-676. PubMed doi:10.4088/JCP.09m05483yel
15. Bowden CL, Perlis RH, Thase ME, et al. Aims and results of the NIMH Systematic Treatment Enhancement Program for Bipolar Disorder (STEP-BD). CNS Neurosci Ther. 2012;18(3):243-249. PubMed doi:10.1111/j.1755-5949.2011.00257.x
16. Montoya A, Tohen M, Vieta E, et al. Functioning and symptomatic outcomes in patients with bipolar I disorder in syndromal remission: a 1-year, prospective, observational cohort study. J Affect Disord. 2010;127(1-3):50-57. PubMed doi:10.1016/j.jad.2010.04.026
17. Sagman D, Lee B, Chandresena R, et al. A Canadian naturalistic study of a community-based cohort treated for bipolar disorder. BMC Psychiatry. 2010;10(1):24. PubMed doi:10.1186/1471-244X-10-24
18. Kessing LV, Hansen MG, Andersen PK, et al. The predictive effect of episodes on the risk of recurrence in depressive and bipolar disorders: a life-long perspective. Acta Psychiatr Scand. 2004;109(5):339-344. PubMed doi:10.1046/j.1600-0447.2003.00266.x
19. Angst J, Gamma A, Sellaro R, et al. Recurrence of bipolar disorders and major depression: a life-long perspective. Eur Arch Psychiatry Clin Neurosci. 2003;253(5):236-240. PubMed doi:10.1007/s00406-003-0437-2
20. Angst J, Sellaro R. Historical perspectives and natural history of bipolar disorder. Biol Psychiatry. 2000;48(6):445-457. PubMed doi:10.1016/S0006-3223(00)00909-4
21. Markar HR, Mander AJ. Efficacy of lithium prophylaxis in clinical practice. Br J Psychiatry. 1989;155(4):496-500. PubMed doi:10.1192/bjp.155.4.496
22. Goldberg JF, Harrow M, Leon AC. Lithium treatment of bipolar affective disorders under naturalistic followup conditions. Psychopharmacol Bull. 1996;32(1):47-54. PubMed
23. Coryell W, Winokur G, Solomon D, et al. Lithium and recurrence in a long-term follow-up of bipolar affective disorder. Psychol Med. 1997;27(2):281-289. PubMed doi:10.1017/S0033291796004461
24. Maj M, Pirozzi R, Magliano L, et al. Long-term outcome of lithium prophylaxis in bipolar disorder: a 5-year prospective study of 402 patients at a lithium clinic. Am J Psychiatry. 1998;155(1):30-35. PubMed
25. Maj M, Pirozzi R, Bartoli L, et al. Long-term outcome of lithium prophylaxis in bipolar disorder with mood-incongruent psychotic features: a prospective study. J Affect Disord. 2002;71(1-3):195-198. PubMed doi:10.1016/S0165-0327(01)00350-0
26. Biel MG, Peselow E, Mulcare L, et al. Continuation versus discontinuation of lithium in recurrent bipolar illness: a naturalistic study. Bipolar Disord. 2007;9(5):435-442. PubMed doi:10.1111/j.1399-5618.2007.00389.x
27. Nierenberg AA, Friedman ES, Bowden CL, et al. Lithium Treatment Moderate-Dose Use Study (LiTMUS) for bipolar disorder: a randomized comparative effectiveness trial of optimized personalized treatment with and without lithium. Am J Psychiatry. 2013;170(1):102-110. PubMed doi:10.1176/appi.ajp.2012.12060751
28. Licht RW, Vestergaard P, Rasmussen NA, et al. A lithium clinic for bipolar patients: 2-year outcome of the first 148 patients. Acta Psychiatr Scand. 2001;104(5):387-390. PubMed doi:10.1034/j.1600-0447.2001.00389.x
29. Silverstone T, McPherson H, Hunt N, et al. How effective is lithium in the prevention of relapse in bipolar disorder? a prospective naturalistic follow-up study. Aust N Z J Psychiatry. 1998;32(1):61-66. PubMed doi:10.3109/00048679809062707
30. Wilting I, Heerdink ER, Mersch PP, et al. Association between lithium serum level, mood state, and patient-reported adverse drug reactions during long-term lithium treatment: a naturalistic follow-up study. Bipolar Disord. 2009;11(4):434-440. PubMed doi:10.1111/j.1399-5618.2009.00699.x
31. Altamura AC, Mundo E, Dell’ Osso B, et al. Quetiapine and classical mood stabilizers in the long-term treatment of bipolar disorder: a 4-year follow-up naturalistic study. J Affect Disord. 2008;110(1-2):135-141. PubMed doi:10.1016/j.jad.2008.01.017
32. Kulkarni J, Filia S, Berk L, et al. Treatment and outcomes of an Australian cohort of outpatients with bipolar I or schizoaffective disorder over twenty-four months: implications for clinical practice. BMC Psychiatry. 2012;12(1):228. PubMed doi:10.1186/1471-244X-12-228
33. Born C, Seitz NN, Grunze H, et al. Preliminary results of a fine-grain analysis of mood swings and treatment modalities of bipolar I and II patients using the daily prospective life-chart-methodology. Acta Psychiatr Scand. 2009;120(6):474-480. PubMed doi:10.1111/j.1600-0447.2009.01412.x
34. Post RM, Altshuler LL, Frye MA, et al. Complexity of pharmacologic treatment required for sustained improvement in outpatients with bipolar disorder. J Clin Psychiatry. 2010;71(9):1176-1186, quiz 1252-1253. PubMed doi:10.4088/JCP.08m04811yel
35. Arvilommi P, Suominen K, Mantere O, et al. Maintenance treatment received by patients with bipolar I and II disorders—a naturalistic prospective study. J Affect Disord. 2010;121(1-2):116-126. PubMed doi:10.1016/j.jad.2009.05.005
36. Sheehan DV, Lecrubier Y, Sheehan KH, et al. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry. 1998;59(suppl 20):22-33, quiz 34-57. PubMed
37. Licht RW. Lithium: still a major option in the management of bipolar disorder. CNS Neurosci Ther. 2012;18(3):219-226. PubMed doi:10.1111/j.1755-5949.2011.00260.x
38. Nivoli AM, Murru A, Vieta E. Lithium: still a cornerstone in the long-term treatment in bipolar disorder? Neuropsychobiology. 2010;62(1):27-35. PubMed doi:10.1159/000314307
39. Geddes JR, Burgess S, Hawton K, et al. Long-term lithium therapy for bipolar disorder: systematic review and meta-analysis of randomized controlled trials. Am J Psychiatry. 2004;161(2):217-222. PubMed doi:10.1176/appi.ajp.161.2.217
40. Calabrese JR, Vieta E, El-Mallakh R, et al. Mood state at study entry as predictor of the polarity of relapse in bipolar disorder. Biol Psychiatry. 2004;56(12):957-963. PubMed doi:10.1016/j.biopsych.2004.09.022
41. Vieta E, Berk M, Wang W, et al. Predominant previous polarity as an outcome predictor in a controlled treatment trial for depression in bipolar I disorder patients. J Affect Disord. 2009;119(1-3):22-27. PubMed doi:10.1016/j.jad.2009.02.028
42. Popovic D, Reinares M, Goikolea JM, et al. Polarity index of pharmacological agents used for maintenance treatment of bipolar disorder. Eur Neuropsychopharmacol. 2012;22(5):339-346. PubMed doi:10.1016/j.euroneuro.2011.09.008