Objective: To characterize the prevalence of metabolic syndrome (MetS) among Dutch patients with bipolar disorder (BD), identify factors associated with MetS, and estimate the rate of pharmacologic treatment of MetS in this population.
Methods: A cross-sectional analysis of medical records of adult patients (≥ 18 years) with BD receiving psychotropic drug treatment was performed in a Dutch psychiatric outpatient setting. MetS was determined according to National Cholesterol Education Program Adult Treatment Panel III-Adapted (NCEP ATP III-A) criteria. Data collection via review of medical records from 2015 was conducted in 2016.
Results: The prevalence of MetS in the sample (N = 71) was 42.3%. Multivariate analysis showed sex and body mass index were independently associated with MetS. Males had an almost 8-fold higher risk of MetS than females (OR = 7.7, 95% CI, 1.5-39.6), and with every point of increase on BMI, the risk of MetS increased by 40% (OR = 1.4, 95% CI, 1.2-1.8). Of the patients with hyperglycemia, hypercholesterolemia, and hypertension, 81%, 72.4%, and 65.0%, respectively, did not receive drug treatment for these conditions.
Conclusions: The study found a high prevalence rate of MetS and substantial undertreatment of the components of MetS in patients with BD in a Dutch outpatient clinic. Follow-up investigation in larger samples is warranted.
Metabolic Syndrome in Dutch Patients With Bipolar Disorder:
A Cross-Sectional Study
ABSTRACT
Objective: To characterize the prevalence of metabolic syndrome (MetS) among Dutch patients with bipolar disorder (BD), identify factors associated with MetS, and estimate the rate of pharmacologic treatment of MetS in this population.
Methods: A cross-sectional analysis of medical records of adult patients (≥ 18 years) with BD receiving psychotropic drug treatment was performed in a Dutch psychiatric outpatient setting. MetS was determined according to National Cholesterol Education Program Adult Treatment Panel III-Adapted (NCEP ATP III-A) criteria. Data collection via review of medical records from 2015 was conducted in 2016.
Results: The prevalence of MetS in the sample (N = 71) was 42.3%. Multivariate analysis showed sex and body mass index were independently associated with MetS. Males had an almost 8-fold higher risk of MetS than females (OR = 7.7, 95% CI, 1.5–39.6), and with every point of increase on BMI, the risk of MetS increased by 40% (OR = 1.4, 95% CI, 1.2–1.8). Of the patients with hyperglycemia, hypercholesterolemia, and hypertension, 81%, 72.4%, and 65.0%, respectively, did not receive drug treatment for these conditions.
Conclusions: The study found a high prevalence rate of MetS and substantial undertreatment of the components of MetS in patients with BD in a Dutch outpatient clinic. Follow-up investigation in larger samples is warranted.
Prim Care Companion CNS Disord 2018;20(6):18m02366
To cite: de Jong M, Belleflamme J, Dale C, et al. Metabolic syndrome in Dutch patients with bipolar disorder: a cross-sectional study. Prim Care Companion CNS Disord. 2018;20(6):18m02366.
To share: https://doi.org/10.4088/PCC.18m02366
© Copyright 2018 Physicians Postgraduate Press, Inc.
aPsyQ Department of Mood Disorders, Mondriaan, Institute of Mental Health, Maastricht, The Netherlands
bDepartment of Psychiatry and Psychology, School for Mental Health and Neuroscience, Maastricht University Medical Center, Maastricht, the Netherlands
cDepression Clinical and Research Program, Massachusetts General Hospital, Harvard Medical School, Boston, Massachusetts
dMaster Advanced Nursing Practice, Zuyd University of Applied Sciences, Heerlen, The Netherlands
eDepartment of Psychiatry, Massachusetts General Hospital, Harvard Medical School, Charlestown, Massachusetts
fTranslational Neuromodeling Unit, Institute for Biomedical Engineering, University of Zurich and ETH Zurich, Zurich, Switzerland
gInstitute for Complementary and Integrative Medicine, University Hospital Zurich and University of Zurich, Zurich, Switzerland
*Corresponding author: Marasha de Jong, MD, PsyQ Department of Mood Disorders, Mondriaan, Institute of Mental Health, Oranjeplein 10, 6224 KD Maastricht, The Netherlands ([email protected]).
Bipolar disorder (BD) is a severe mental illness with a lifetime prevalence of 2.4% worldwide.1 Despite its low prevalence, BD is among the top 20 leading causes of disability worldwide due to its early onset, disabling character, and chronicity.2 Over the last decades, we have learned that severe mental illness affects not only mental health but also physical health, with a reduced life expectancy ranging from 13–30 years.3 Approximately 70% of this excess mortality is accounted for by physical illness, with cardiovascular disease (CVD) being the leading cause of death.4 Most research on medical comorbidities of severe mental illness has focused on schizophrenia, leaving physical health risks in BD relatively underreported.5 A recent meta-analysis by Correll at al6 on CVD risk in severe mental illness demonstrated a transdiagnostic elevated risk for CVD-related mortality. Similarly, a meta-analysis by Vancampfort et al7 examining type 2 diabetes mellitus risk in severe mental illness demonstrated a consistently elevated risk in all diagnostic subgroups with no significant diagnostic subgroup difference, indicating that BD has similar physical comorbidity risk as schizophrenia. Studies8,9 on mortality rates in BD report reduced life expectancies of 9–20 years, which are comparable to other severe mental illnesses.
Metabolic syndrome (MetS) comprises various interrelated metabolic risk factors for CVD and type 2 diabetes mellitus, including central obesity, hypertension, dyslipidemia, and glucose metabolism abnormalities.10 Individuals with MetS have a 2-fold increased risk for CVD and a 5-fold increased risk for type 2 diabetes mellitus over the next 5 to 10 years.11,12 In addition to physical burden, MetS is also associated with adverse clinical psychiatric outcomes.13 The etiology of MetS is still not fully understood; however, central adiposity and insulin resistance are thought to play a key role.14
MetS is highly prevalent in BD. A meta-analysis15 reported a 2-fold risk of MetS in BD compared to the general population, with an overall prevalence rate of 37.3%. Obesity, older age, and antipsychotic treatment have been consistently associated with higher rates of MetS in BD.15,16 The exact relationship between BD and MetS remains unknown; in addition to unhealthy lifestyles17 and metabolic side effects of psychotropic medications, neuroendocrine factors, immunoinflammation, and genetics may also contribute.18 Several studies19,20 indicate that metabolic risk factors often remain undetected and undertreated in individuals with BD. Only recently has BD been cited by the American Heart Association21 as a condition that predisposes for early heart disease.
To our knowledge, no data exist on prevalence rates and factors associated with MetS in patients with BD in the Netherlands. Identification of MetS in BD should be encouraged because regular monitoring, preventive measures, and treatment for cardiovascular risk factors can greatly increase quality of life and life expectancy. Our aim in the current study was to elucidate the prevalence rate and factors associated with MetS in a Dutch population of patients with BD. We hypothesized that MetS would be highly prevalent in our sample. On the basis of the literature, we expected comparable prevalence rates between men and women and higher prevalence with increasing age and body mass index (BMI). Also, we hypothesized that patients treated with second-generation antipsychotics (SGAs) would demonstrate higher rates of MetS. Because many patients with BD receive limited medical care due to factors including limited health care access and nonadherence, we also examined rates of pharmacologic treatment for MetS in our sample.
METHODS
Setting and Participants
This cross-sectional medical records study was carried out at Mondriaan Institute of Mental Health, PsyQ Department of Mood Disorders, Maastricht, the Netherlands, at the outpatient clinic for mood disorders. The study was evaluated by the Medical Ethical Committee of Maastricht University Medical Center (MUMC), which determined that informed consent was not required for this retrospective study involving anonymous processing of clinical data. Data collection via review of medical records from 2015 was conducted in 2016.
Inclusion criteria were (1) age ≥ 18 years; (2) met criteria for bipolar disorder type I, bipolar disorder type II, or bipolar disorder not otherwise specified (NOS) as defined by DSM-IV criteria; (3) receiving psychopharmacologic treatment for BD; and (4) underwent full metabolic screening in 2015 (including waist circumference and blood pressure measurement and a blood test for fasting triglycerides, high-density lipoprotein cholesterol, and glucose). Screening for metabolic syndrome was introduced as standard care in 2015 per the Dutch Multidisciplinary Guideline for Bipolar Disorders.22 All outpatient clinic patients participated in this yearly metabolic screening; those who fulfilled the eligibility criteria were included in this sample.
Study Variables and Data Collection
Study variables included metabolic syndrome and its criteria, demographic and clinical characteristics, and psychotropic drug treatment.
Metabolic syndrome definition. We defined MetS according to the modified National Cholesterol Education Program Adult Treatment Panel III-Adapted (NCEP ATP III-A) criteria.11 According to these criteria, a diagnosis of MetS requires the presence of ≥ 3 of the following categorical risk factors:
- Abdominal obesity (waist circumference: men ≥ 102 cm, women ≥ 88 cm)
- High triglycerides (≥ 1.7 mmol/L [150 mg/dL]) or on lipid-lowering drug treatment
- Low high-density lipoprotein (HDL) cholesterol (men < 1.0 mmol/L [40 mg/dL], women < 1.3 mmol/L [50 mg/dL]) or receiving lipid-lowering drug treatment
- High blood pressure (≥ 130/85 mm Hg) or receiving drug treatment for high blood pressure
- High fasting glucose (≥ 5.6 mmol/L [100 mg/dL]) or receiving glucose-lowering drug treatment.
Waist circumference and blood pressure were measured by the nurse or nurse practitioner during the standard yearly outpatient metabolic screening. Waist circumference was measured midway between the superior border of the iliac crest and the inferior margin of the ribs. Blood pressure was measured seated after 15 minutes of rest. A blood specimen was drawn as part of the yearly metabolic screening. The level of fasting triglycerides, HDL cholesterol, and glucose were analyzed in the laboratory of MUMC. Patients had to confirm fasting at least 9 hours prior to the blood test. The use of cholesterol-lowering medication, antihypertensive medication, or glucose-lowering medication was reported in the electronic medical records by the treating psychiatrist.
Demographic and clinical characteristics and psychotropic drug treatment. All relevant demographic and clinical characteristics were entered in the medical records during the intake and updated throughout treatment by the treating mental health care professional. Illness onset was determined as the year the patient was first diagnosed with bipolar disorder. Psychotropic drug treatment was entered into the medical records by the treating psychiatrist.
Data Collection
Data were collected and entered in SPSS software, version 22 and checked for correctness by double entry of a random sample. The collection date of all relevant demographic and clinical variables was set at the date of metabolic screening.
Statistical Analysis
Categorical variables were expressed in absolute numbers and percentages and continuous variables as mean and standard deviation (SD). The association of MetS with the different clinical and demographic variables was assessed with the independent t test for continuous variables and with the χ2 test and Fisher exact test for categorical variables. Effect size was expressed by the odds ratio (OR) for categorical variables and Cohen d for continuous variables. Significance level was set at α = .05 (2-tailed). Variables with P values < .2 in the univariate analysis were included in a multivariate binary logistic regression analysis (enter method) to estimate the independent influence of each variable of interest on MetS, while controlling for the other variables that were associated with MetS in the univariate analysis. All statistical analyses were performed with SPSS.
RESULTS
Seventy-one patients (mean age of 52.5 years [SD = 10.0], 60.6% female) with BD received outpatient psychopharmacologic treatment for BD and underwent a full metabolic screening in 2015. Table 1 provides demographic and clinical characteristics. The majority of the sample was diagnosed with bipolar I disorder (78.9%), and the mean duration of illness was 14.8 years (SD = 10.2).
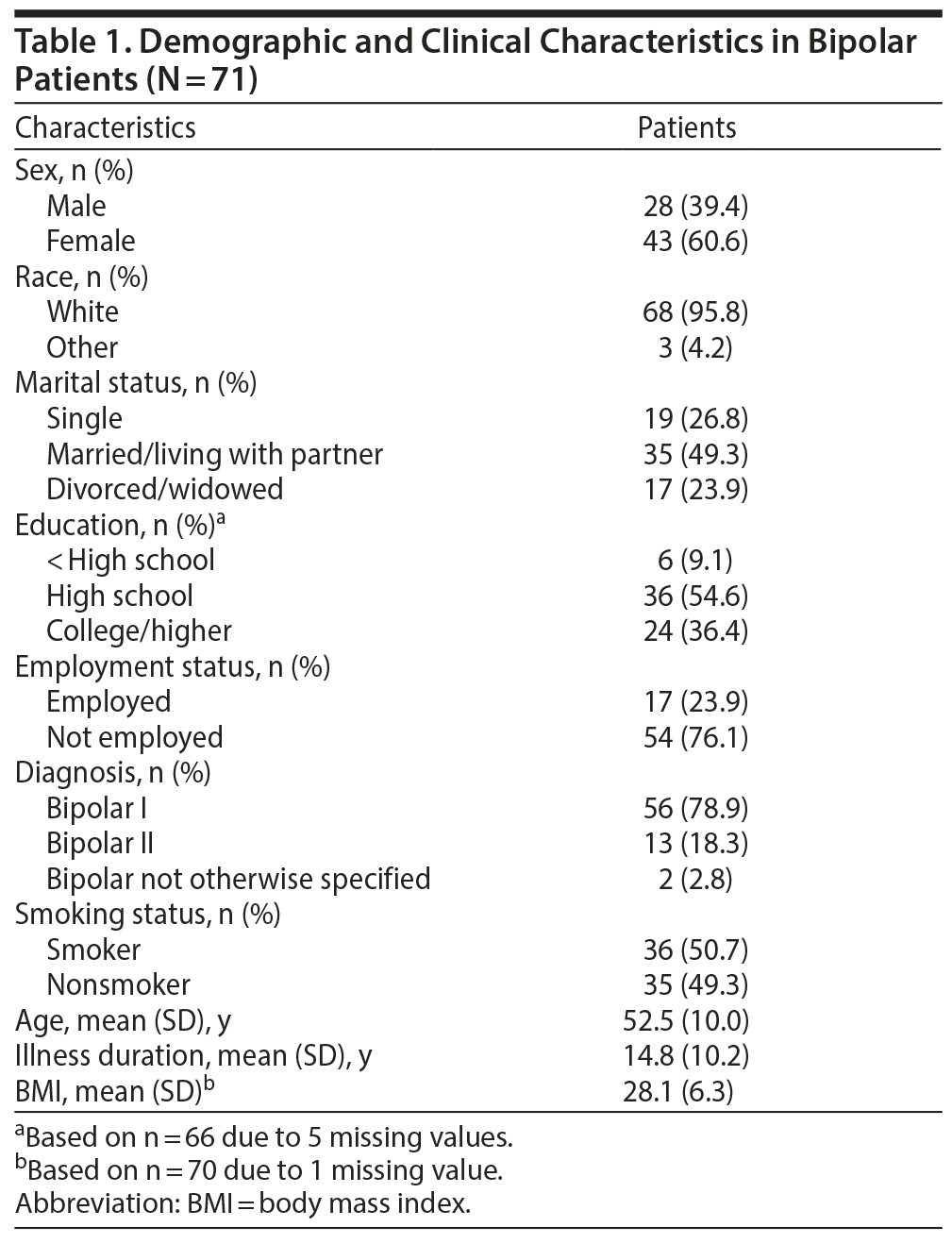
Table 2 provides an overview of the different psychotropic drug regimens for BD. A high percentage of the sample was taking mood stabilizers (93%), of which lithium (57.7%) and valproic acid (33.8%) were the most common. Second-generation antipsychotics (SGAs) were prescribed in 38.0% of the sample, with quetiapine being the most frequent (22.5%). Thirty-one percent of the sample took a combination of a mood stabilizer and an SGA.
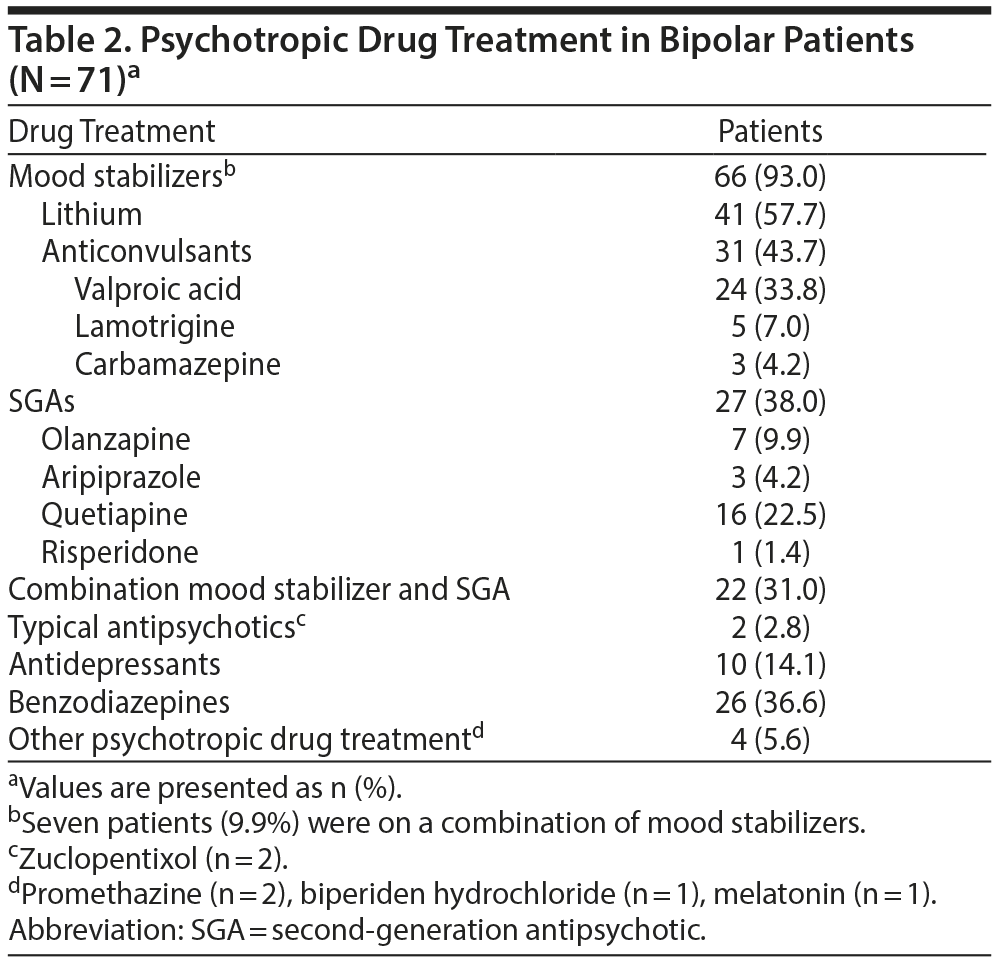
Prevalence of Metabolic Syndrome
The prevalence of MetS in the patients with BD was 42.3%. MetS was present in 71.4% of males and 23.3% of females. Central obesity was present in 52.1% of the sample; 63.4% had hypertension, 26.8% had low HDL cholesterol, 40.8% had hypertriglyceridemia, and 29.6% had hyperglycemia. The prevalence of categorical diabetes mellitus including type I and type II (glucose ≥ 7.0 mmol/L [126 mg/dL]) was 7.1%. MetS was significantly more prevalent in men than in women (χ21 = 16.13, P < .01, OR = 8.3). Likewise, men were significantly more likely than women to have hypertension (χ21 = 4.60, P = .03, OR = 3.2), low HDL cholesterol (χ21 = 6.11, P = .01, OR = 3.9), and hypertriglyceridemia (χ21 = 10.51, P < .01, OR = 5.2) (Table 3).
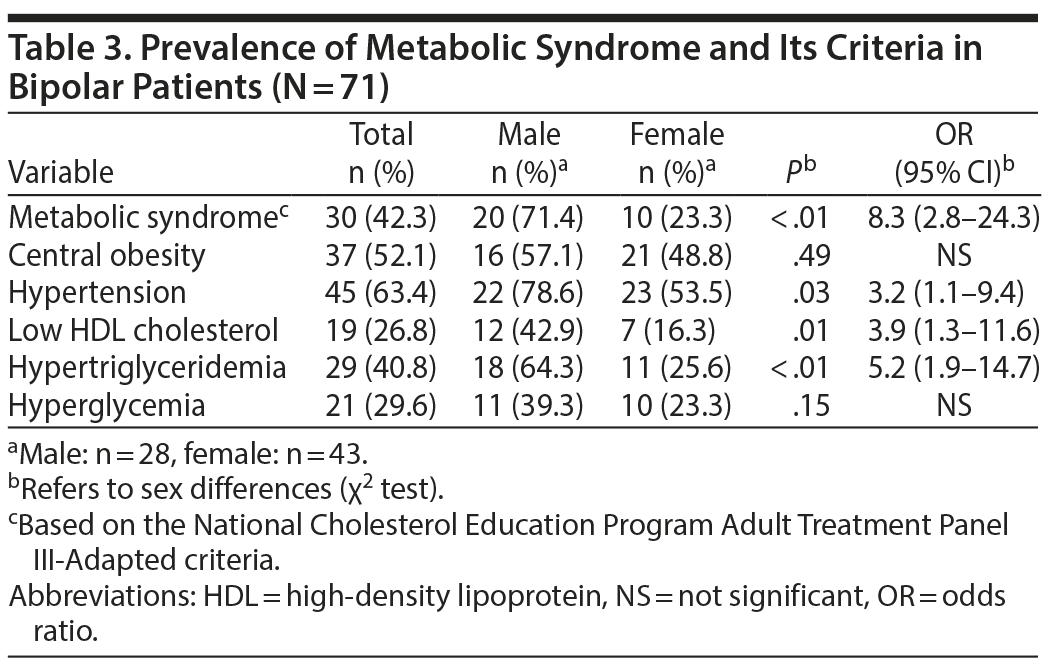
Factors Associated With Metabolic Syndrome
Univariate analysis showed that MetS was more prevalent in patients who were male (χ21 = 16.1, P < .01, OR = 8.3), had a lower level of education (χ21 = 4.9, P = .04, OR = 5.9), were older (t69 = 2.3, P = .02, d = 0.57), had a longer duration of illness (t69 = 2.1, P = .04, d = 0.48), and had a higher BMI (t68 = 5.7, P < .01, d = 1.4). In multivariate analysis, sex and BMI were the only factors that kept their significant association with MetS when controlling for confounders. The association between the presence of MetS and the use of anticonvulsants was marginally significant in univariate analysis but not in multivariate analysis (Table 4).
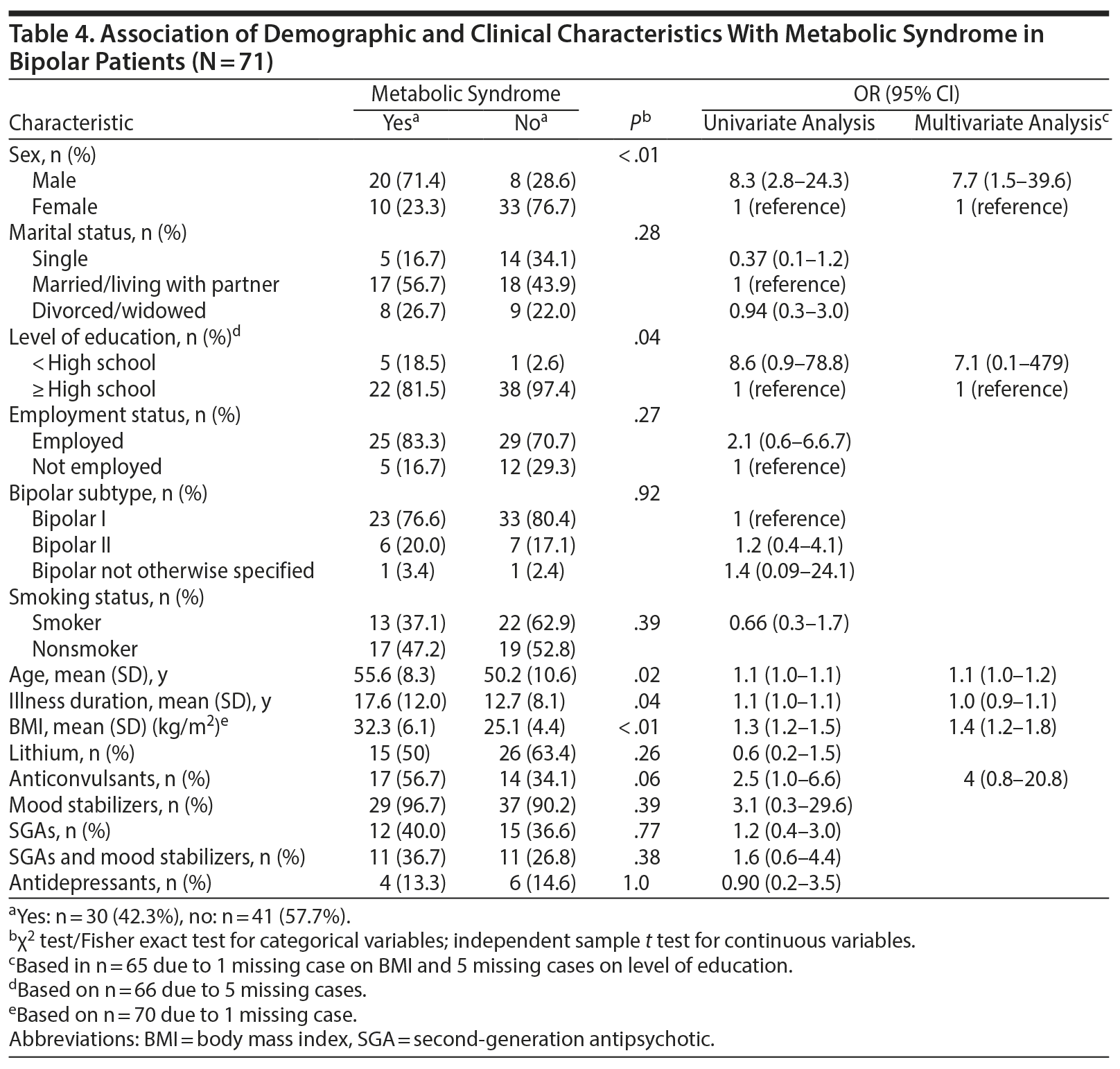
Pharmacologic Treatment of Metabolic Syndrome
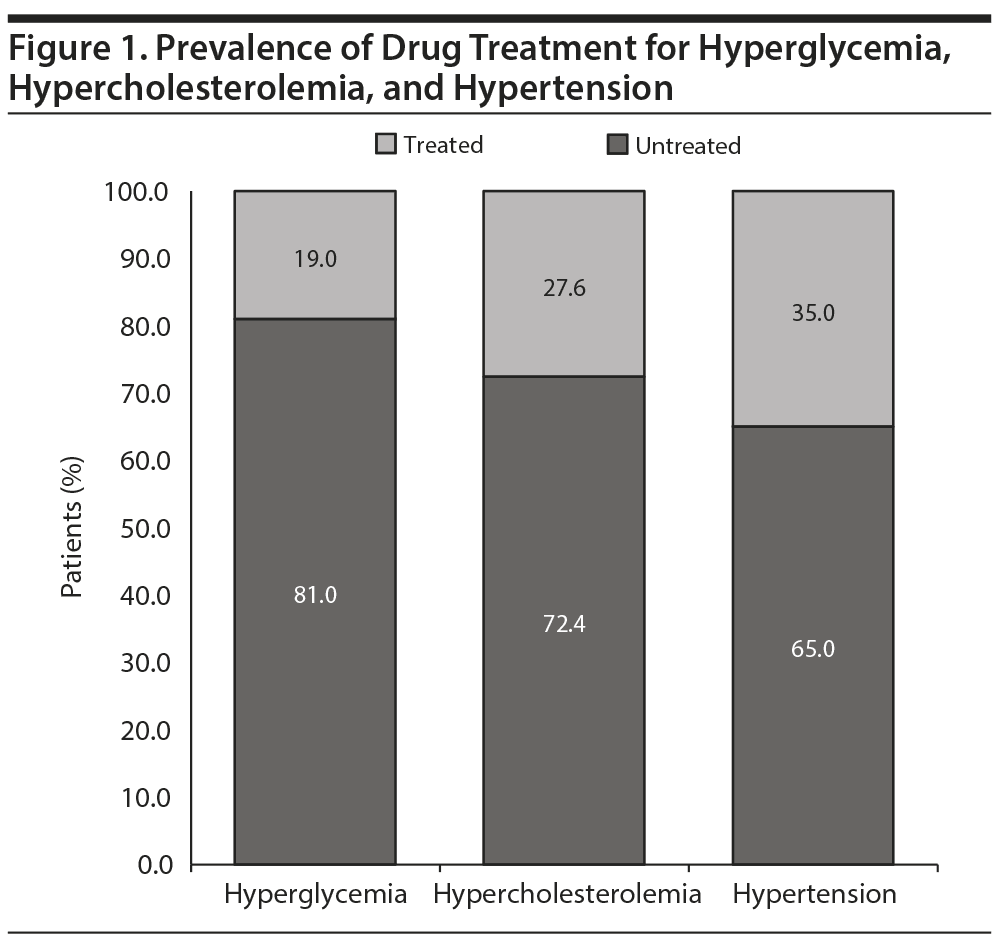
We found that 19.0%, 27.6%, and 35.0% of patients with hyperglycemia, hypercholesterolemia, and hypertension, respectively, were receiving drug treatment for these conditions (Figure 1).
DISCUSSION
To our knowledge, this is the first study examining prevalence rate and factors associated with MetS and its components in BD in the Netherlands. This cross-sectional analysis of medical records of adult outpatients (≥ 18 years) with BD receiving psychotropic drug treatment (N = 71) showed a high prevalence rate of MetS (42.3%) and substantial undertreatment of MetS components, with two-thirds or more of affected patients receiving no drug treatment for these conditions (Figure 1). Sex and BMI were the only factors that were independently associated with the presence of MetS.
The high prevalence rate of MetS supports the general notion that patients with BD are at high cardiovascular risk. Compared to other European countries, the identified prevalence rate in our sample is remarkably high. For example, prevalence rates of MetS in BD in Belgium and France have been estimated to be considerably lower at 18.3 and 18.5%, respectively.19,23 Prevalence rates in the general population are known to vary across different countries due to genetic and environmental differences. The Netherlands has a high prevalence rate of MetS in the general population compared to other European countries. Hu et al24 pooled data from 11 European cohort studies and found an age-standardized prevalence of MetS of 15.7% in men and 14.2% in women. In comparison, the most recent Dutch National Health Examination Survey25 on cardiovascular risk factors in the general population indicated that in the age group 30–70 years, 34% of men and 24% of women have MetS. These higher prevalence rates in the Dutch general population might explain in part the high prevalence rate in this study of Dutch patients with BD compared to other European countries. Also, our sample was relatively older with a narrow age range (mean = 52.5 years, SD = 10, median = 53), which might have contributed to the high prevalence of MetS, given that increasing age is one of the strongest moderators of MetS in the general population.26
Regarding the individual MetS criteria, rates of abdominal obesity, hypertension, and lipid disturbances in our sample agree with other studies. The meta-analysis by Vancampfort et al15 also found that approximately half of patients with bipolar disorder had abdominal obesity, half were hypertensive, and about 40% had abnormal levels of either HDL or triglycerides. The determined prevalence of hyperglycemia in our sample of 29.6% is high compared to the results of the meta-analysis.15
This study demonstrated a significant association between BMI and MetS: with every BMI point increase, the risk of MetS increased by 40% (OR = 1.4, 95% CI, 1.2–1.8). Such an association would be expected because abdominal obesity is one of the criteria for MetS and also plays a central causal role in abnormalities of the other MetS criteria.14 Others have found similar effect sizes for the association between BMI and MetS in BD.19,27
Most studies15,28,29 report no significant difference in MetS between sexes. In our study, however, men were overwhelmingly more likely to have MetS (OR = 7.7, 95% CI, 1.5–39.6) than women. This might be a reflection of the prevalence distribution of sex in the general population of the Netherlands. The Dutch population study25 also showed higher prevalence in men, but the difference in prevalence in the general population was not that extreme, which suggests that BD may render men especially more vulnerable to MetS. However, these findings should be interpreted with caution because of the small sample size and may also reflect sampling bias or statistical “noise.” Larger sample studies investigating the impact of sex on MetS in this population are needed.
This study examined the association between the presence of MetS and the use of different types of psychotropic agents. None of these therapies showed a significant association with MetS. The association between anticonvulsants and MetS did approach significance in univariate analysis but not in multivariate analysis. Mood stabilizers30,31 and antidepressants32 have been associated with weight gain and metabolic disturbances, but, to our knowledge, no significant association has yet been found between MetS and these psychotropic drug groups in patients with BD, which is consistent with our findings. In contrast, SGAs have been consistently associated with MetS in studies19,33 in populations with BD, and it is well established that SGAs cause obesity and metabolic syndrome.33,34 A meta-analysis15 concluded that patients with BD taking antipsychotic medication were at significantly greater risk of MetS compared to antipsychotic-free BD patients (OR = 1.72, 95% CI, 1.24–2.38). Surprisingly, in our sample we found no significant association between the use of SGAs and the presence of MetS. This finding is inconsistent with previous studies and might reflect the small sample size. Also, the risk of metabolic side effects varies considerably between antipsychotics, with clozapine having the highest risk, followed by olanzapine, quetiapine, risperidone, and aripiprazole.35 In our sample, no patients were taking clozapine, and the relative proportion of patients taking olanzapine was only 25.9%, which might in part explain the absence of association between SGAs and MetS. The lack of associations between metabolic syndrome and SGAs could also be due to selective prescribing, ie, patients with metabolic syndrome may be less likely to be prescribed or accept medications associated with weight gain. We did not investigate the association of MetS with the individual SGAs or with first-generation antipsychotics because of the small sample size.
This study reveals considerable pharmacologic undertreatment of MetS components in patients with BD; 81% of patients with hyperglycemia and 72.4% and 65.0% of patients with hypercholesterolemia and hypertension, respectively, received no medications for these conditions. A study19 in a French cohort found similar rates of untreated MetS components. It must be noted that the cutoffs to start pharmacologic treatment clinically and the cutoffs used for MetS are different; for example, hyperglycemia (glucose ≥ 5.6 mmol/L) does not necessarily constitute diabetes (glucose ≥ 7.0 mmol/L), and, hence, hyperglycemia in the range between 5.6 mmol/L and 7.0 mmol/L in regular clinical practice would not lead to treatment with oral antidiabetic medication. However, shortened life expectancy of patients with severe mental illness supports more aggressive (pharmacologic) treatment of MetS in this population, with metformin being the first-choice pharmacologic to counteract metabolic disturbances.36
Our study has several limitations. First, this exploratory study had a small sample size. Second, the cross-sectional design makes it difficult to infer causality of the associations between MetS and the risk factors. Third, different studies define metabolic syndrome with different cutoff points for its components, making it difficult to assess geographical variations in the prevalence of MetS and contextualize our findings on the basis of previous work. Fourth, we did not examine physical activity, dietary habits, and other lifestyle factors, thus limiting inferences about causation of the high prevalence of MetS in the sample. We also did not examine the rate of applied behavioral interventions, which might be even more efficacious than pharmacologic interventions in altering metabolic status in patients with SMI, especially when administered in early stages of pharmacologic treatment37 and including regular physical activity.38 Finally, this study was performed in a Dutch sample from Maastricht, which means the findings may not be generalizable to other regions of the Netherlands or to all populations.
Nonetheless, the results of this study demonstrate a high prevalence of MetS in a sample with BD, and clinicians treating patients with BD should, therefore, be alert for the presence of MetS. Our results stress the importance of regular monitoring and follow-up management of MetS in this population. Further prospective research with larger sample sizes and inclusion of more clinical variables such as lifestyle factors should better characterize the association between BD and MetS.
Submitted: August 2, 2018; accepted September 25, 2018.
Published online: December 6, 2018.
Potential conflicts of interest: Dr Mischoulon has received research support from FisherWallace, Nordic Naturals, Methylation Sciences, PharmoRx Therapeutics, and Blackmores; honoraria for speaking from the Massachusetts General Hospital Psychiatry Academy; and royalties from Lippincott Williams & Wilkins for Natural Medications for Psychiatric Disorders: Considering the Alternatives. Dr Peeters received financial compensation as an independent symposium speaker for Astra-Zeneca, Eli Lilly, Janssen-Cilag, Lundbeck, SCEM, Benecke, Servier, and NVvP. Drs de Jong, Gard, and Gamel and Mss Belleflamme and Dale report no conflicts of interest related to the subject of this article.
Funding/support: The study was funded through PsyQ Department of Mood Disorders, Mondriaan, Institute of Mental Health. No external funding agencies were involved.
Role of the sponsor: The sponsor had no role in the design or conduct of the study.
REFERENCES
1. Merikangas KR, Jin R, He JP, et al. Prevalence and correlates of bipolar spectrum disorder in the World Mental Health Survey Initiative. Arch Gen Psychiatry. 2011;68(3):241–251. PubMed CrossRef
2. Ferrari AJ, Stockings E, Khoo JP, et al. The prevalence and burden of bipolar disorder: findings from the Global Burden of Disease Study 2013. Bipolar Disord. 2016;18(5):440–450. PubMed CrossRef
3. De Hert M, Cohen D, Bobes J, et al. Physical illness in patients with severe mental disorders. II. Barriers to care, monitoring and treatment guidelines, plus recommendations at the system and individual level. World Psychiatry. 2011;10(2):138–151. PubMed CrossRef
4. Walker ER, McGee RE, Druss BG. Mortality in mental disorders and global disease burden implications: a systematic review and meta-analysis. JAMA Psychiatry. 2015;72(4):334–341. PubMed CrossRef
5. Leboyer M, Soreca I, Scott J, et al. Can bipolar disorder be viewed as a multi-system inflammatory disease? J Affect Disord. 2012;141(1):1–10. PubMed CrossRef
6. Correll CU, Solmi M, Veronese N, et al. Prevalence, incidence and mortality from cardiovascular disease in patients with pooled and specific severe mental illness: a large-scale meta-analysis of 3,211,768 patients and 113,383,368 controls. World Psychiatry. 2017;16(2):163–180. PubMed CrossRef
7. Vancampfort D, Correll CU, Galling B, et al. Diabetes mellitus in people with schizophrenia, bipolar disorder and major depressive disorder: a systematic review and large scale meta-analysis. World Psychiatry. 2016;15(2):166–174. PubMed CrossRef
8. Laursen TM, Wahlbeck K, Hällgren J, et al. Life expectancy and death by diseases of the circulatory system in patients with bipolar disorder or schizophrenia in the Nordic countries. PLoS One. 2013;8(6):e67133. PubMed CrossRef
9. Crump C, Sundquist K, Winkleby MA, et al. Comorbidities and mortality in bipolar disorder: a Swedish national cohort study. JAMA Psychiatry. 2013;70(9):931–939. PubMed CrossRef
10. Grundy SM, Cleeman JI, Daniels SR, et al; American Heart Association; National Heart, Lung, and Blood Institute. Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation. 2005;112(17):2735–2752. PubMed CrossRef
11. Alberti KG, Eckel RH, Grundy SM, et al; International Diabetes Federation Task Force on Epidemiology and Prevention; Hational Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; International Association for the Study of Obesity. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation. 2009;120(16):1640–1645. PubMed CrossRef
12. Grundy SM. Metabolic syndrome update. Trends Cardiovasc Med. 2016;26(4):364–373. PubMed CrossRef
13. Bai YM, Li CT, Tsai SJ, et al. Metabolic syndrome and adverse clinical outcomes in patients with bipolar disorder. BMC Psychiatry. 2016;16(1):448. PubMed CrossRef
14. Eckel RH, Alberti KG, Grundy SM, et al. The metabolic syndrome. Lancet. 2010;375(9710):181–183. PubMed CrossRef
15. Vancampfort D, Vansteelandt K, Correll CU, et al. Metabolic syndrome and metabolic abnormalities in bipolar disorder: a meta-analysis of prevalence rates and moderators. Am J Psychiatry. 2013;170(3):265–274. PubMed CrossRef
16. Grover S, Malhotra N, Chakrabarti S, et al. Metabolic syndrome in bipolar disorders. Indian J Psychol Med. 2012;34(2):110–118. PubMed CrossRef
17. Vancampfort D, Firth J, Schuch FB, et al. Sedentary behavior and physical activity levels in people with schizophrenia, bipolar disorder and major depressive disorder: a global systematic review and meta-analysis. World Psychiatry. 2017;16(3):308–315. PubMed CrossRef
18. de Almeida KM, Moreira CL, Lafer B. Metabolic syndrome and bipolar disorder: what should psychiatrists know? CNS Neurosci Ther. 2012;18(2):160–166. PubMed CrossRef
19. Godin O, Etain B, Henry C, et al; FondaMental Advanced Centers of Expertise in Bipolar Disorders (FACE-BD) Collaborators. Metabolic syndrome in a French cohort of patients with bipolar disorder: results from the FACE-BD cohort. J Clin Psychiatry. 2014;75(10):1078–1085, quiz 1085. PubMed CrossRef
20. Fiedorowicz JG, Palagummi NM, Forman-Hoffman VL, et al. Elevated prevalence of obesity, metabolic syndrome, and cardiovascular risk factors in bipolar disorder. Ann Clin Psychiatry. 2008;20(3):131–137. PubMed CrossRef
21. Goldstein BI, Carnethon MR, Matthews KA, et al; American Heart Association Atherosclerosis; Hypertension and Obesity in Youth Committee of the Council on Cardiovascular Disease in the Young. Major depressive disorder and bipolar disorder predispose youth to accelerated atherosclerosis and early cardiovascular disease: a scientific statement from the American Heart Association. Circulation. 2015;132(10):965–986. PubMed CrossRef
22. Kupka R, Goossens P, van Bendegem M, et al. Multidisciplinaire Richtlijn Bipolaire Stoornissen. Utrecht, The Netherlands: Trimbos-instituut; 2015.
23. van Winkel R, De Hert M, Van Eyck D, et al. Prevalence of diabetes and the metabolic syndrome in a sample of patients with bipolar disorder. Bipolar Disord. 2008;10(2):342–348. PubMed CrossRef
24. Hu G, Qiao Q, Tuomilehto J, et al; DECODE Study Group. Prevalence of the metabolic syndrome and its relation to all-cause and cardiovascular mortality in nondiabetic European men and women. Arch Intern Med. 2004;164(10):1066–1076. PubMed CrossRef
25. Blokstra A, Vissink P, Venmans LMAJ, et al. Measuring the Netherlands. A Monitoring Study of Risk Factors in the General Population, 2009-2010: Rijksinstituut voor Volksgezondheid en Milieu. Bilthoven, The Netherlands: RIVM; 2011.
26. Ford ES, Giles WH, Dietz WH. Prevalence of the metabolic syndrome among US adults: findings from the third National Health and Nutrition Examination Survey. JAMA. 2002;287(3):356–359. PubMed CrossRef
27. Garcia-Portilla MP, Saiz PA, Benabarre A, et al. The prevalence of metabolic syndrome in patients with bipolar disorder. J Affect Disord. 2008;106(1-2):197–201. PubMed CrossRef
28. Czepielewski L, Daruy Filho L, Brietzke E, et al. Bipolar disorder and metabolic syndrome: a systematic review. Rev Bras Psiquiatr. 2013;35(1):88–93. PubMed CrossRef
29. McIntyre RS, Danilewitz M, Liauw SS, et al. Bipolar disorder and metabolic syndrome: an international perspective. J Affect Disord. 2010;126(3):366–387. PubMed CrossRef
30. Verrotti A, D’Egidio C, Mohn A, et al. Weight gain following treatment with valproic acid: pathogenetic mechanisms and clinical implications. Obes Rev. 2011;12(5):e32–e43. PubMed CrossRef
31. McKnight RF, Adida M, Budge K, et al. Lithium toxicity profile: a systematic review and meta-analysis. Lancet. 2012;379(9817):721–728. PubMed CrossRef
32. McIntyre RS, Park KY, Law CW, et al. The association between conventional antidepressants and the metabolic syndrome: a review of the evidence and clinical implications. CNS Drugs. 2010;24(9):741–753. PubMed CrossRef
33. Yumru M, Savas HA, Kurt E, et al. Atypical antipsychotics related metabolic syndrome in bipolar patients. J Affect Disord. 2007;98(3):247–252. PubMed CrossRef
34. De Hert M, Detraux J, van Winkel R, et al. Metabolic and cardiovascular adverse effects associated with antipsychotic drugs. Nat Rev Endocrinol. 2011;8(2):114–126. PubMed CrossRef
35. Rojo LE, Gaspar PA, Silva H, et al. Metabolic syndrome and obesity among users of second generation antipsychotics: a global challenge for modern psychopharmacology. Pharmacol Res. 2015;101:74–85. PubMed CrossRef
36. Mizuno Y, Suzuki T, Nakagawa A, et al. Pharmacological strategies to counteract antipsychotic-induced weight gain and metabolic adverse effects in schizophrenia: a systematic review and meta-analysis. Schizophr Bull. 2014;40(6):1385–1403. PubMed CrossRef
37. Teasdale SB, Ward PB, Rosenbaum S, et al. Solving a weighty problem: systematic review and meta-analysis of nutrition interventions in severe mental illness. Br J Psychiatry. 2017;210(2):110–118. PubMed CrossRef
38. Taylor J, Stubbs B, Hewitt C, et al. The effectiveness of pharmacological and non-pharmacological interventions for improving glycaemic control in adults with severe mental illness: a systematic review and meta-analysis. PLoS One. 2017;12(1):e0168549. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!


