J Clin Psychiatry 2023;84(4):22br14755
To cite: Richardson JL, George N, Greenall AJ, et al. Cardiac malformation risks following maternal first trimester methylphenidate use. J Clin Psychiatry. 2023;84(4):22br14755.
To share: https://doi.org/10.4088/JCP.22br14755
© 2023 Physicians Postgraduate Press, Inc.
aUK Teratology Information Service, Newcastle-upon-Tyne Hospitals NHS Foundation Trust, Newcastle upon Tyne, United Kingdom
bInstitute of Health and Society, Newcastle University, Newcastle-upon-Tyne, United Kingdom
cWomen’s Services, Newcastle Upon Tyne NHS Foundation Trust, Royal Victoria Infirmary, Newcastle-upon-Tyne, United Kingdom
*Corresponding author: Jonathan L. Richardson, PhD, UK Teratology Information Service, The Regional Drug and Therapeutics Centre, 16/17 Framlington Pl, Newcastle upon Tyne, Tyne and Wear, UK NE2 4AB ([email protected]).
Several studies have investigated congenital heart defect (CHD) risks following maternal first trimester (T1) methylphenidate use (tabulated review of the data available from the authors).1–5
Although individually none of the studies provide clear evidence of an increased risk of CHD, a 2020 meta-analysis6 combining data from several studies1–3,5 described a small but statistically significant increased risk (RR = 1.59; 95% CI, 1.02 to 2.49). However, this meta-analysis only combined the crude counts of healthy and affected fetuses from the primary source studies while also including data from overlapping study samples (Pottegård et al,5 Källén et al,3 and Huybrechts et al2 included; among other data, Huybrechts et al2 included an update for the dataset previously described in the other two reports3,5). Consequently, the pooled risk estimate did not consider the impact of measured confounders from the individual studies, and due to the overlapping datasets, the same case could have contributed to the analysis more than once.
The most recent study4 describes nearly 300 additional methylphenidate exposed pregnancies than previously reported from the same data source.2 Although the study identified a non–statistically significant increased risk (aOR = 1.65; 95% CI, 0.89 to 3.05),4 understanding how this finding alters the interpretation of the available evidence as a whole remains difficult.
To provide clarity on CHD risks following in utero methylphenidate exposure, we combined the most recently published risk estimates from each available dataset using a meta-analysis approach.
Methods
A literature review of PubMed/MEDLINE/Embase was conducted in November 2022 searching databases from inception for studies (reviews and abstracts excluded) published in English describing T1 methylphenidate exposed and non–methylphenidate exposed comparator groups and fetal CHD outcomes.
Two authors (J.L.R., N.G.) independently reviewed the literature and extracted the data from studies meeting the inclusion criteria. Where available, adjusted point estimates and their confidence limits were extracted to include the least confounded point estimates. Where point estimates were unavailable, unadjusted odds ratios and confidence limits were calculated from crude CHD risks7 and Haldane-Anscombe correction was used in the case of zero events.8 Non–odds ratio point estimates were converted to odds ratios.9 Where datasets overlapped, data from the most recent publication (and hence the largest sample size) were included in the primary meta-analysis. A sensitivity analysis was performed, excluding unadjusted point estimates from the meta-analysis.
CHD risk estimates and their 95% confidence intervals were pooled in a fixed effects meta-analysis using the generic inverse variance method in RevMan 5.3.10 Heterogeneity was assessed using the I2 statistic.
Results
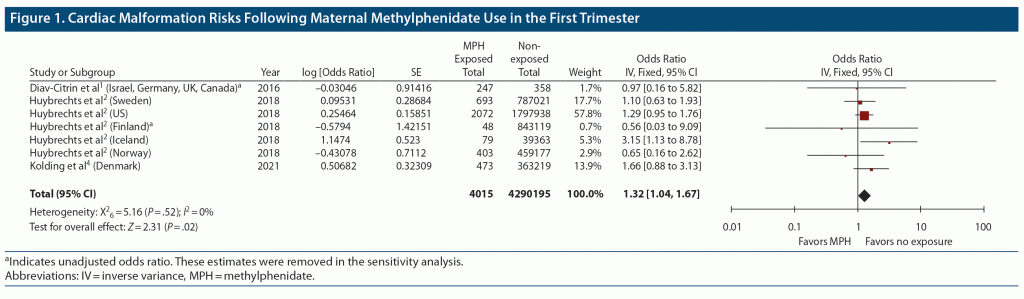
Data were pooled from 7 study samples reported in 3 comparative cohort studies describing 4,015 T1 methylphenidate exposed and approximately 4.29 million non-exposed comparator pregnancies.1,2,4 The pooled odds ratio identified a small but statistically significant increased risk of CHD following T1 methylphenidate exposure (pooled OR = 1.32; 95% CI, 1.04 to 1.67; Figure 1). The sensitivity analysis did not reveal a substantially different risk estimate (pooled OR = 1.34; 95% CI, 1.05 to 1.70).
Discussion
These findings indicate that maternal T1 methylphenidate use may be associated with a small increased risk of CHD. Given that CHDs occur in approximately 1% of all live born infants,11 the absolute risk following in utero methylphenidate exposure is estimated as 1.3%. This is equivalent to 3 additional infants with a CHD for every 1,000 exposed to methylphenidate.
As this small increased risk is demonstrated through the pooling of observational data, a causal association cannot be confirmed due to unmeasured confounding (eg, recreational drug or alcohol use) that may have influenced the primary source studies. However, maternal T1 methylphenidate use may be a prognostic risk factor for fetal CHD. Additional antenatal screening, with a fetal echocardiogram between 18–22 weeks gestation, could be considered following T1 methylphenidate exposure.
Published online: May 15, 2023.
Author contributions: Richardson, Stephens, and Hodson identified the need for this communication and conceived the study methods. Richarson performed the literature search. Richardson and George performed the initial data extraction and summarized the evidence. Richardson performed the meta-analysis and produced the forest plots. All authors reviewed the summarized evidence and developed a consensus opinion on the interpretation of this evidence through multiple rounds of discussion. Richardson prepared the first draft of the brief report, for which all authors subsequently provided feedback and input for re-drafting.
Relevant financial relationships: All authors and contributors declare no conflicts of interest, no other financial relationships with any organizations that might have an interest in the submitted work in the previous 3 years, and no previous or ongoing relationships or activities that could appear to have influenced the submitted work.
Funding/support: The UK Teratology Information Service is commissioned by the UK Health Security Agency and hosted by Newcastle upon Tyne Hospitals NHS Foundation Trust.
Role of the sponsor: The funder played no role in study design, data interpretation, decision to publish, or preparation of the manuscript.
Acknowledgment: The authors thank Jane Ingram for proofreading the manuscript.
ORCID: Jonathan L. Richardson: https://orcid.org/0000-0003-3494-9559; Nathan George: https://orcid.org/0000-0002-5273-9689; Sally Stephens: https://orcid.org/0000-0003-4567-4781; Kenneth K. Hodson: https://orcid.org/0000-0003-3091-6952
References (11)

- Diav-Citrin O, Shechtman S, Arnon J, et al. Methylphenidate in pregnancy: a multicenter, prospective, comparative, observational study. J Clin Psychiatry. 2016;77(9):1176–1181. PubMed CrossRef
- Huybrechts KF, Bröms G, Christensen LB, et al. Association between methylphenidate and amphetamine use in pregnancy and risk of congenital malformations: a cohort study from the International Pregnancy Safety Study Consortium. JAMA Psychiatry. 2018;75(2):167–175. PubMed CrossRef
- Källén B, Borg N, Reis M. The use of central nervous system active drugs during pregnancy. Pharmaceuticals (Basel). 2013;6(10):1221–1286. PubMed CrossRef
- Kolding L, Ehrenstein V, Pedersen L, et al. Associations between ADHD medication use in pregnancy and severe malformations based on prenatal and postnatal diagnoses: a Danish registry-based study. J Clin Psychiatry. 2021;82(1):20m13458. PubMed CrossRef
- Pottegård A, Hallas J, Andersen JT, et al. First-trimester exposure to methylphenidate: a population-based cohort study. J Clin Psychiatry. 2014;75(1):e88–e93. PubMed CrossRef
- Koren G, Barer Y, Ornoy A. Fetal safety of methylphenidate: a scoping review and meta analysis. Reprod Toxicol. 2020;93:230–234. PubMed CrossRef
- Bland JM, Altman DG. Statistics notes: the odds ratio. BMJ. 2000;320(7247):1468. PubMed CrossRef
- Weber F, Knapp G, Ickstadt K, et al. Zero-cell corrections in random-effects meta-analyses. Res Synth Methods. 2020;11(6):913–919. PubMed CrossRef
- Zhang J, Yu KF. What’s the relative risk? a method of correcting the odds ratio in cohort studies of common outcomes. JAMA. 1998;280(19):1690–1691. PubMed CrossRef
- The Cochrane Collaboration. Review Manager (RevMan) (computer program) v5.3.5. Cochrane Training. 2014. https://training.cochrane.org/online-learning/core-software/revman
- US Centers for Disease Control and Prevention. Data and statistics on congenital heart defects. 2022. https://www.cdc.gov/ncbddd/heartdefects/data.html#:~:text=CHDs%20affect%20nearly%201%25%20of,year%20in%20the%20United%20States
This PDF is free for all visitors!