Click to enlarge page
Of the 24 million people with schizophrenia worldwide, fewer than half receive appropriate care. Even patients with access to the best available treatments suffer from significant functional and social deficits and multiple unmet needs. Fewer than one-third of treated patients are relatively symptom-free and functional. Even when treated, patients experience high rates of relapse and adherence problems and only minimal improvement in negative and cognitive symptoms, which are significant contributors to functional impairment. New medications for schizophrenia are needed that target different receptors/mechanisms of action. The focus has increasingly shifted toward developing an ideal “medication” for schizophrenia to not only reduce symptoms but also improve functioning and quality of life. Researchers developing such agents must overcome many challenges, including the need for novel designs to test agents with new mechanisms of action, new outcome measures to assess different symptomatic targets (eg, cognitive impairment), globalization, an increased focus on personalized medicine, and new diagnostic formulations coming in the DSM-5. The article concludes with an overview of the efficacy and safety of 4 recently introduced antipsychotics and agents under investigation—asenapine, iloperidone, lurasidone, and sertindole—to give clinicians a better understanding of the expanding treatment options available for patients with schizophrenia and the opportunity to find an agent that is best for each specific patient in terms of enhancing efficacy and minimizing side effects. The article concludes with practical guidance for clinicians on how to integrate new antipsychotics into their clinical practice to achieve best outcomes.
Financial disclosure: Dr Potkin has grant, advisory board, and speakers bureau financial relationships with the following companies: Bioline, Bristol-Myers Squibb, Cortex, Dainippon Sumitomo/Sepracor, Elan, Eli Lilly, Forest, Janssen, Merck/Schering Plough, Minster, Novartis, Otsuka, Pfizer/Wyeth, Solvay, and Roche. Dr Harvey is a consultant for Eli Lilly, Solvay, Shire, Dainippon Sumitomo Pharma America/Sepracor, and Merck and has received grant/research support from AstraZeneca. Dr Kalali has grant, advisory board, and speakers bureau financial relationships with the following companies: AstraZeneca, Merck, Novartis, Pfizer, and Takeda. He is also on the board of directors of Cypress Biosciences. Dr Preda has nothing to disclose.
Financial support for this evidence–based peer-reviewed Academic Highlights was provided by a grant from Dainippon Sumitomo Pharma America, Inc/Sepracor. Project management was provided by Healthcare Global Village, Inc. The faculty acknowledges Ruth Ross, Project Manager, Healthcare Global Village, for editorial assistance in developing the manuscript. The opinions expressed herein are those of the faculty and do not necessarily reflect the views of Healthcare Global Village, Inc, the publisher, or the commercial supporter.
Notice of correction 1/16/2012: In the section “New Antipsychotics and Investigational Agents,” the affinity (pKi) of asenapine for all receptors and the affinity of iloperidone for α1 in Table 1 have been corrected, as has text related to the corrected values.
Of the 24 million people with schizophrenia worldwide, fewer than 50% receive appropriate care.1 Even patients with access to the best available treatments suffer from significant functional and social deficits.2 This article is based on a teleconference that discussed issues related to the development of new treatments for schizophrenia.
SCHIZOPHRENIA TREATMENT
Disability
Adrian Preda, MD, discussed the status of schizophrenia treatment. Although the prevalence of schizophrenia is only 1% in the general population, according to World Health Organization (WHO) data, it is the third leading cause of disability among men and women 15-44 years of age worldwide.1 Thus, schizophrenia is a chronic and disabling condition, and available treatments are not doing an adequate job of helping patients regain functioning.
Patients also need better access to treatment. However, even in developed countries with access to the best treatments, rates of disability and functional impairment are high. A recent study compared 2 samples of patients with schizophrenia in rural Sweden and New York City.2 Despite differences in health care systems, demography, and culture, the majority of patients in both countries were unemployed or disabled and were unable to maintain a primary relationship.
Use of Polypharmacy
Although evidence-based guidelines generally recommend antipsychotic monotherapy for schizophrenia,3 the majority of patients with schizophrenia (eg, 70% in the United States and 50% in the European Union) are being treated with more than 1 antipsychotic.4,5 A 2003 study found that, of the 35% of patients with schizophrenia treated with more than 1 antipsychotic at an academic hospital in New York, 50% were receiving a combination of first- and second-generation antipsychotics.6 Many patients were also receiving mood stabilizing, antidepressant, anxiolytic, hypnotic, and anticholinergic medications. This suggests that, even when patients with schizophrenia are treated with 2 or more antipsychotics, enough residual symptoms remain to cause patients and clinicians to feel that additional treatment is needed. While there is no evidence of enhanced efficacy for treatment with more than 1 antipsychotic, polypharmacy often reflects psychiatrists’ attempts to achieve adequate efficacy. Use of anticholinergics suggests problems with motor side effects. There is clearly a need to develop new agents that will be effective and well tolerated as monotherapy.
Quality of Life
Schizophrenia researchers are increasingly focusing not just on efficacy for symptoms, but also on improvements in quality of life (QOL). In treating a serious illness such as schizophrenia, QOL refers to the minimal requirements for someone to have a meaningful life.
A number of scales have been developed to assess what appear to be important components of QOL7,8 (eg, material comforts; health; relationships with parents and siblings; having and rearing children; relationships with spouse or significant other; close friends; helping and supporting others; and participating in organizations and public affairs). The Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) found only modest improvements on QOL measures over 18 months, with no significant differences in QOL outcomes between perphenazine and 4 second-generation antipsychotics.9,10 Small improvements in QOL were found after 6 and 12 months of treatment, with ratings appearing to plateau before 18 months. These results suggest that patients’ perception of QOL did not improve much, even when the severity of their symptoms as measured by the Positive and Negative Syndrome Scale (PANSS)11 decreased.
Although schizophrenia research has historically focused on decreasing positive symptoms, recent research has shown that much of the functional impairment and poor QOL experienced by patients with schizophrenia are associated with negative and cognitive symptoms.12,13 Unfortunately, currently available treatments are not very effective in targeting negative and cognitive symptoms.
Summary
Dr Preda summarized key unmet needs of patients with schizophrenia:
- Fewer than one-third of treated patients are relatively symptom-free and functional.
- Even when treated, patients have high rates of relapse.
- Patients have high rates of adherence problems, in part because current treatments are only partially effective and have troublesome side effects.
- Current treatments produce only limited improvement in negative and cognitive symptoms, which contribute significantly to functional impairment and poor QOL.
- New drugs need to be developed to target different receptors or mechanisms of action with potential to improve functional outcomes.
Dr Preda concluded that we should no longer think about an ideal antipsychotic, but should redirect our thinking toward an ideal drug for schizophrenia that will not only reduce symptoms but also help restore functioning and improve QOL.
FACTORS AFFECTING DRUG DEVELOPMENT
Amir H. Kalali, MD, discussed factors affecting new drug development.
Methodological Factors
Due to the way studies are powered statistically, even superior treatments can fail to show superiority by chance. Other factors affecting clinical trial success are study design, choice of outcome measures, conduct of the study, types of patients enrolled, and personnel performing assessments.
Programs to develop psychiatric drugs can be high risk compared with other therapeutic areas, if careful attention is not paid to methodology. In interpreting clinical trial results, one must distinguish negative from failed trials. A negative trial is one in which the agent under investigation does not separate from placebo, while a comparator compound (ie, an agent with known efficacy for the indication) does. A negative trial indicates that the investigational medication does not work, at least at the doses tested. A failed trial is one in which the agent under investigation and the comparator medication that is known to work both do not separate from placebo. A failed trial provides no meaningful scientific information about whether the new drug works or not.
Outcome measures in psychiatric trials are subjective. Thus, clinicians’ ratings at baseline, when certain levels of severity are needed for study entry, are subject to both conscious and unconscious inflation compared with patient ratings.14 Clinician ratings may then decrease rapidly after baseline, creating an elevated placebo response that is an artifact of the higher scores assigned at baseline. This makes it more difficult to distinguish drug from placebo.
While historically a problem in depression trials, lack of separation from placebo has not traditionally been a major problem in schizophrenia trials. However, a recent study found higher rates of placebo response and lower rates of drug response (even for previously established, comparator drugs) in more recent studies compared with earlier trials.15 In addition, in some global studies, sites in the United States showed no separation from placebo, while sites outside the United States did show separation.16 The causes of this phenomenon remain a subject of research and debate. High failure rates in psychiatric drug trials have led to increasingly large and expensive phase 3 programs prior to approval.
Globalization
A major trend in clinical research is globalization, driven by the desire to bring new treatments to patients as expeditiously as possible and to test drugs in more representative and ethnically diverse populations. Clinical trials in the United States and Western Europe have slower patient recruitment. In addition, while historically most drugs have been approved based on US and Western European data, these drugs are used all over the world. Studies in less developed countries provide better access to patients who have not been treated with multiple drugs or may even be drug-naive. Patient retention rates are also higher outside the United States, improving follow-up; this may reflect the more limited treatment options in those settings.
Regulatory Requirements and Public Distrust
Drug development, particularly in the United States, faces a number of other challenges, including increased US Food and Drug Administration (FDA) scrutiny, imposition of black box warnings for certain agents, and requirements for new suicidality assessments. High levels of media coverage, coupled with a general decline of trust in scientific research, have been fueled by examples of scientific misconduct in academia and lack of transparency in data dissemination by the pharmaceutical industry.
Market Complications
Pharmaceutical companies also face challenges in bringing new drugs to market. In an era when generic antidepressants and antipsychotics are available, and regulators, third party payers, and patients are increasingly demanding, a new agent has to stand out as clearly differentiated from available drugs for that indication to proceed through development to marketing.
Summary
For drug development to be sustainable and meet patients’ needs, it must become more efficient and demonstrate value. Drug developers need to focus on personalized medicine, in which “one size does not fit all.” Globalization will help to increase efficiency and diversity of data. The future publication of the fifth edition of the Diagnostic and Statistical Manual of Mental Disorders17 will affect diagnosis and types of patients enrolled in clinical trials. Future psychiatric drug development is likely to involve novel designs to test agents with new mechanisms of action, new outcome measures to assess different symptomatic targets, novel methods of administering outcome measures, and increasing patient self-assessment.
NEW TARGETS FOR DRUG DEVELOPMENT
Philip D. Harvey, PhD, discussed cognitive impairment and negative symptoms as new targets for drug development. In 2003, the National Institute of Mental Health launched the Measurement and Treatment Research to Improve Cognition in Schizophrenia (NIMH-MATRICS) initiative. Its goals were to identify domains of cognitive impairment, suggest molecular targets that may modulate cognition and candidate drugs likely to affect those targets, and work with the FDA to develop clinical trial designs that could lead to approval of agents for this indication.18,19 The FDA subsequently recommended that studies of treatments for cognitive impairment (1) use a consensus-based cognitive performance measure along with a co-primary measure to assess functioning, (2) enroll clinically stable patients to ensure that the agent is not exerting an effect by improving psychotic symptoms, and (3) be of extended duration (eg, approximately 6 months for registration trials), although appropriate duration is still under discussion.
Consensus Cognitive Battery
In keeping with FDA recommendations, the NIMH-MATRICS initiative developed the MATRICS Consensus Cognitive Battery (MCCB),20,21 which takes about 1 hour to administer to someone with schizophrenia. The MCCB assesses processing speed, attention and vigilance, working memory, verbal learning, visual learning, reasoning and problem-solving, and social cognition, producing a score for overall global neuropsychological performance.22
Co-Primary Outcome Measures
Based on the FDA’s recommendations, the NIMH-MATRICS initiative investigated performance-based assessments of functional skills and structured interviews that gather information about functioning.23
The University of California, San Diego Performance-Based Skills Assessment (UPSA),24 which assesses communication, finance, planning and comprehension, transportation, and home maintenance, appears to be the most psychometrically valid of these types of measures. The UPSA has been shown to be very sensitive to the skills required to live independently, and its psychometric characteristics indicate that it is likely to be as sensitive to change as the MCCB.25 A short version, the UPSA-Brief, consists of the communication and financial subscales and takes only 10-15 minutes to administer. It has adequate psychometric properties, predicts residential independence, is sensitive to change, and correlates well with the full UPSA.26,27
Two functional interviews have been assessed: the CGI Cogs28 and the Schizophrenia Cognition Rating Scale (SCoRS).29 They are rated by the interviewer, the patient, and an informant who knows the patient well and generate item scores and global ratings. However, the MATRICS Co-Primary and Translation Study25 found that these interviews, particularly if the patient is the sole informant, correlate poorly with scores on the MCCB, suggesting that performance-based co-primary measures may be more suitable for use in clinical trials unless there is a definite plan to include informants who know the patients well.
Persistent Negative Symptoms
Approximately 30% to 60% of patients with schizophrenia continue to experience negative symptoms of at least moderate severity after positive symptoms have improved.30 Such persistent negative symptoms, in particular social avoidance and anhedonia, are associated with disability, impairment, and poorer functional outcomes, especially in social functioning.13 The FDA is considering allowing studies of adjunctive pharmacotherapy and psychosocial interventions for negative symptoms using a similar research design as for cognitive symptoms.
Pharmacologic Mechanisms
The MATRICS initiative identified a number of agents that might have beneficial cognitive effects in combination with antipsychotics.31 These included dopamine D1 agonists, nicotinic partial agonists, muscarinic agonists, cholinesterase inhibitors, agents that target the glutamatergic system, agents targeting serotonin-1A and -7 receptor subtypes, and norepinephrine agonists and transport modulators. Research data from studies of these agents have started to emerge.
Clinical Research on Cognition
Cognitive deficits in schizophrenia are likely not due to a single impairment in the way that psychotic symptoms seem to be correlated with D2 dysfunction. If so, a single treatment may not be adequate. Research on treatment of cognitive impairment in schizophrenia has focused on (1) newer and investigational antipsychotics with novel mechanisms that might have better efficacy for cognition dysfunction, (2) possible adjunctive agents, and (3) psychosocial interventions such as cognitive remediation.
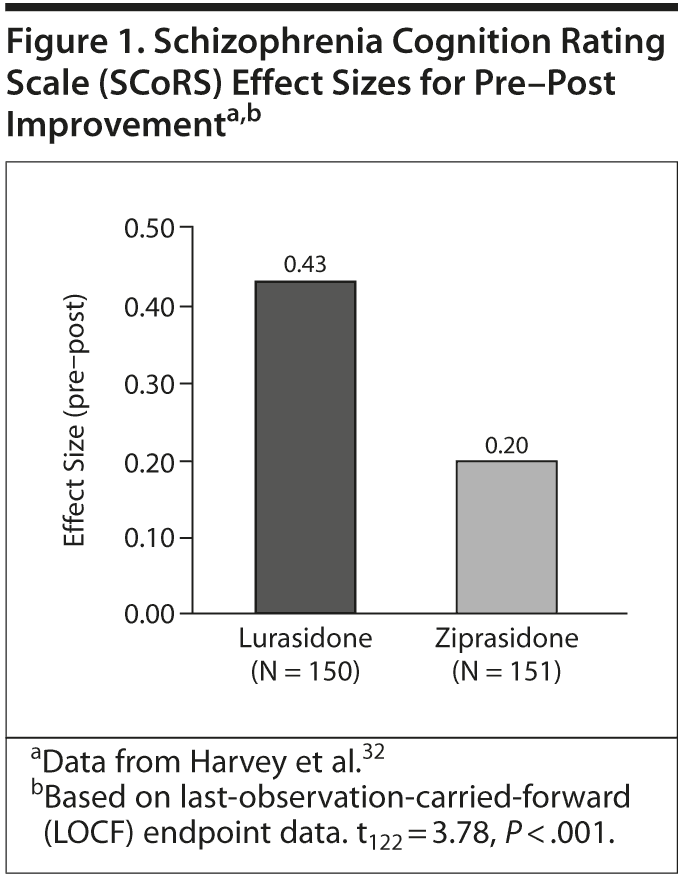
Antipsychotics. Antipsychotic treatments typically produce only modest improvements on the MCCB. For example, in a short-term randomized trial that compared lurasidone and ziprasidone in stable outpatients with schizophrenia or schizoaffective disorder,32 the average improvement from baseline on a subset battery from the MCCB was 0.15 standard deviations for lurasidone and 0.10 for ziprasidone. However, after 3 weeks, lurasidone but not ziprasidone was associated with significant within-group improvement from baseline on this measure. This same study reinforced the potential importance of including a co-primary measure. In this study, an interview-based co-primary measure, the ScoRS, that used information from the clinician, an informant, and the patient, showed considerably greater sensitivity to cognitive change than the MCCB performance-based measures. As shown in Figure 1, there was a significant within-group improvement from baseline in ratings on the ScoRS in the lurasidone group, but not in the ziprasidone group, and the difference between the change scores for the 2 agents was close to statistically significant (P = .058). This improvement was approximately 3 times as large as the change in the MCCB. These preliminary findings need to be interpreted with caution, given that the study was short-term (while the FDA suggests 6 months for study of cognitive effects), that evaluating cognitive change was not a primary goal of the study, and that it used a fixed-dose design. Further long-term studies involving measures such as the UPSA, MCCB, and SCoRS are needed to establish the relative sensitivity of these measures to cognitive enhancement.
Adjunctive agents. Studies of adjunctive agents suggested by the MATRICS initiative have found either no benefit or only very modest effects. However, some of these agents do produce effects in the brains of normal subjects that can be detected with neuroimaging33 and a few have shown efficacy in individuals with schizotypal personality disorder.34 Research is needed to investigate why compounds that improve cognition in normal subjects and those with personality disorders do not seem to benefit patients with schizophrenia.
Nevertheless, this research has established appropriate study designs for testing agents for cognitive effects. For example, a large randomized, double-blind, 16-week study, the Cognitive and Negative Symptoms in Schizophrenia Trial (CONSIST),30 evaluated effects of glycine and d-cycloserine versus placebo on negative and cognitive symptoms in 157 clinically stable patients with schizophrenia or schizoaffective disorder. While this was a negative study (neither glycine nor d-cycloserine was more effective than placebo for negative or cognitive symptoms), approximately 80% of patients in all 3 groups completed the study, indicating that this is a viable research design.
Cognitive remediation. Research on psychosocial interventions for cognitive remediation is promising, with several different remediation methods producing improvements from baseline in cognitive performance in patients with schizophrenia.35 Cognitive remediation has been found to produce improvements in psychophysiological measures and increases in levels of brain-derived neurotrophic factor (BDNF),36 which has been found to promote neurogenesis and to play an important role in learning and memory in animal studies.37,38 Cognitive remediation may also have promise for use in conjunction with potential pharmacologic cognitive enhancers.
Summary
Several studies have identified feasible research designs for evaluating potential cognitive enhancers in schizophrenia. Although studies with a number of different agents have had only modest success, given promising findings concerning psychosocial cognitive remediation, it is hoped that promising agents will be identified in the future. Persistent negative symptoms are also an appropriate target for interventions.
NEW ANTIPSYCHOTICS AND INVESTIGATIONAL AGENTS
Steven G. Potkin, MD, first discussed whether remission is a realistic goal for patients with schizophrenia and what receptor profiles might play a role in supporting remission.
Is Remission a Realistic Goal?
The longest double-blind medication study in schizophrenia to date compared 2 doses of ziprasidone with haloperidol.39 Patients showing improvement after 40 weeks continued on the same treatment for 3 more years on a double-blind basis. Although outcomes were equivalent in the 3 groups after 40 weeks, treatments diverged over the next 3 years, with ziprasidone 80-160 mg/d producing significantly greater reductions in symptom severity, a higher rate of remission, and improvement in QOL compared with haloperidol. This study suggests that patients who are showing improvement and remain on their atypical antipsychotic regimen may achieve increasing benefits over time to the point where remission becomes a realistic goal.
An Ideal Drug for Schizophrenia
The ideal drug for restoring functioning in patients with schizophrenia would reduce positive, negative, and affective symptoms without causing or exacerbating cognitive deficits, and, if possible, enhance cognition. It would also avoid negative effects (eg, weight gain, metabolic problems) on patients’ health. Thus, the goal of drug discovery for schizophrenia is to develop agents with improved efficacy not only for positive but also for negative, cognitive, and affective symptoms, with as few side effects as possible.
Receptor Profiles
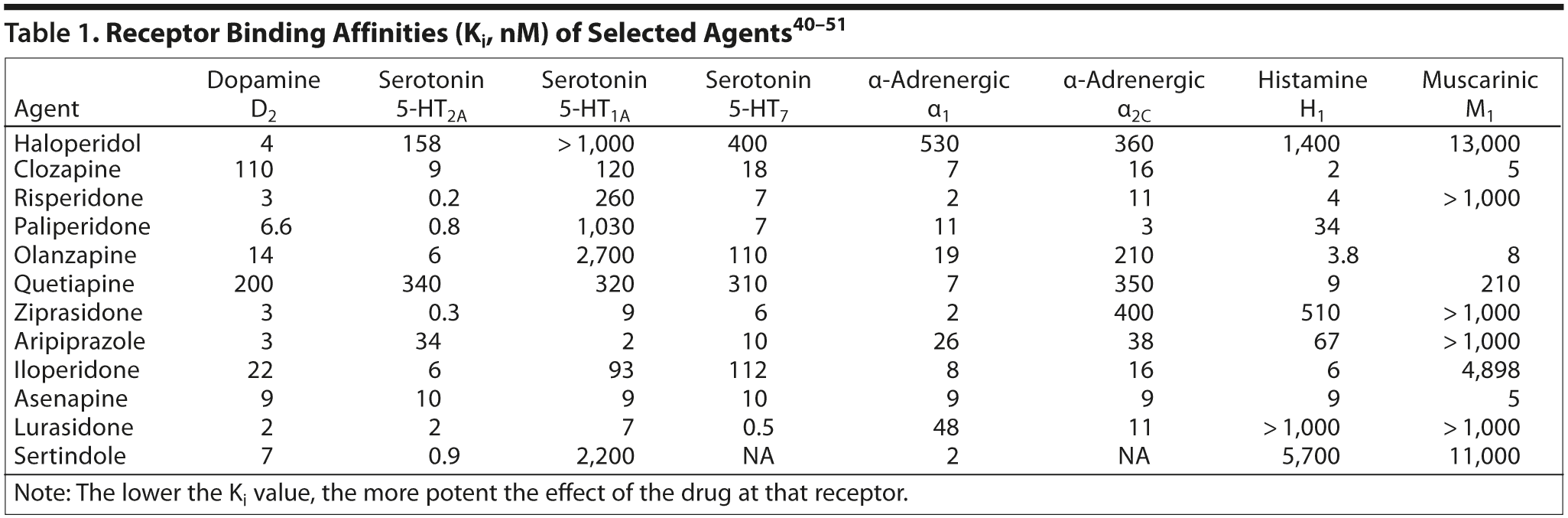
Table 1 shows receptor affinities of 10 currently available antipsychotics and 2 agents not yet approved, lurasidone and sertindole (references 40-51, especially Shahid et al47). Table 2 describes functions associated with several neurotransmitter receptors.52-54 Clinicians can use the information in these tables to gain a better understanding about a drug’s potential clinical profile.
Dopamine D2 receptors. D2 receptor blockade appears to be necessary to treat positive (psychotic) symptoms. The greater an agent’s affinity for D2, the lower the dose needed and the more potent the compound. Thus, haloperidol, risperidone, and paliperidone are quite potent with only a few milligrams needed; clozapine and quetiapine are much less potent so that hundreds of milligrams are needed; while intermediate doses of a drug such as olanzapine are needed. An exception is ziprasidone; while quite potent, doses up to 160 mg are needed for optimal efficacy. Aripiprazole, while quite potent, requires doses of 10-30 mg to occupy > 90% of D2 receptors (needed to produce 70% D2 blockade since it is a partial agonist). In general, antipsychotic medications are dosed to achieve adequate (usually > 70%) D2 receptor blockade. At those doses, other receptors are also affected. By comparing a drug’s affinity for those other receptors to its D2 affinity, clinicians can estimate a drug’s potential clinical profile.
Serotonin receptors. The greater an agent’s affinity for the 5-HT2A receptor, the more it balances D2 receptor blockade and attenuates extrapyramidal symptoms (EPS). Thus, haloperidol, with a much stronger affinity for D2 than 5-HT2A (a pKi value of 4 nM compared with 158 nM), is associated with high rates of EPS, while clozapine, with a much lower affinity for D2 than 5-HT2A (110 vs 9), causes very few, if any, EPS. There are exceptions to this prediction; for example, risperidone can produce EPS, especially at higher doses despite its stronger affinity for 5-HT2A than D2; and olanzapine, quetiapine and iloperidone, with more equal affinities for D2 and 5-HT2A, are associated with only mild EPS.
The 5-HT1A receptor appears to be involved in anxiety, mood regulation, and cognition. It is hypothesized that agents with relatively greater affinity for 5-HT1A may have the potential to improve cognition.
The 5-HT7 receptor is involved in mood, cognition, and perhaps sleep normalization and circadian rhythms. The relative greater affinity for 5-HT7 compared with D2 receptors is most marked for clozapine and lurasidone.
α-Adrenergic receptors. Drugs with potent effects on the α1 receptor (eg, clozapine, quetiapine, iloperidone) can cause orthostatic hypotension and dizziness and generally need to be titrated up to a therapeutic dose to minimize risk of side effects. The α2C receptor may play a role in cognition. Agents with potent affinity for the α2C receptor relative to D2 receptors include clozapine, paliperidone, and iloperidone.
Histamine (H1) receptors. Agents with strong affinity for H1 (eg, clozapine, olanzapine, quetiapine, iloperidone) can produce weight gain, sedation, and cognitive impairment. In contrast, haloperidone, ziprasidone, aripiprazole, and lurasidone have much stronger affinity for the D2 than the H1 receptor and cause little sedation, weight gain, or cognitive impairment. Cognitive improvement is associated with modulations of the serotonin system and/or cortical increases in dopamine and acetylcholine, changes not observed with haloperidol.
Muscarinic (M1) effects. Agents with potent affinity for M1 (eg, clozapine, olanzapine, quetiapine) can cause dry mouth, blurred vision, and urinary hesitancy and have the potential to impair memory because of their muscarinic receptor blockade.
Half-Lives
Based on their plasma half-lives, all compounds in Table 1, except perhaps asenapine, could be given as once-a-day dosing after any required titration has occurred and steady state levels have been reached. However, many drugs, such as quetiapine immediate release (IR), asenapine, ziprasidone, and iloperidone, have not been studied with once-a day dosing and, therefore, the FDA approval is for twice-a-day dosing for these agents.
Newer Antipsychotics
Dr Potkin then reviewed data on 2 new antipsychotics and 2 investigational agents.
Asenapine was approved in 2009 for the acute treatment of both schizophrenia and manic or mixed episodes of bipolar disorder.55 It is given as a sublingual formulation, with a dose of 5 mg twice a day recommended for treatment of schizophrenia with no titration required.55 It is generally well tolerated, with somnolence the most common side effect due to its high affinity for H1 receptors; however, despite its H1 affinity, little weight gain has been observed (average of only 2 lb in year-long studies). It is associated with mild EPS and very mild akathisia, is relatively lipid neutral, and has a favorable prolactin profile.55 Evidence from short- and long-term studies suggests that asenapine may have a potential role in treating negative symptoms.56
Iloperidone was approved in 2009 for treatment of schizophrenia, with a recommended target dose of 12 mg twice a day.57-60 Its most common side effects are dizziness, headache, and pulse increases related to orthostatic changes due to strong affinity for α1; thus, titration is required. Iloperidone is also associated with some nausea and dry mouth, despite very weak affinity for M1 receptors. It causes virtually no EPS and akathisia. It causes mild weight gain (approximately 6 lb in year-long studies), is lipid neutral, and produces a very modest increase in glucose levels. Iloperidone is associated with an average QTc prolongation of 9 ms, comparable to that seen with some other antipsychotics such as ziprasidone.
Lurasidone has shown efficacy in the treatment of schizophrenia in several placebo-controlled trials, and a New Drug Application (NDA) was filed with the FDA in 2009.61 A 6-week, double-blind, placebo-controlled study found significant treatment response as early as day 3 that continued throughout the study.62 The most common side effects associated with lurasidone in clinical trials were akathisia, nausea, somnolence, and sedation.61,62 Given its strong affinity for 5-HT2A and lack of appreciable affinity for M1 and H1, lurasidone is, as predicted, associated with low rates of parkinsonism and minimal effects on weight and lipids. It also has strong affinity for several receptors (5-HT7, 5-HT1A, and α2C) that may play a role in reducing functional impairment related to cognitive and negative symptoms as well as in mood regulation. The prediction of cognitive effects is supported by animal studies that suggested that lurasidone may be effective in improving learning and memory functioning.63,64
Sertindole is approved in many countries but is not available in the United States. The NDA for sertindole was voluntarily withdrawn in 1998 due to concerns about QTc prolongation. After over 10 years of experience in Europe, where sertindole is used primarily to manage treatment-refractory illness, the NDA was resubmitted to the FDA. It is not currently approved for treatment in the United States. If approved, sertindole is unlikely to be a first-line antipsychotic because of risk of QTc prolongation that can rarely result in the potentially fatal torsades de pointes arrhythmia. It must also be titrated to minimize side effects such as tachycardia and dizziness. Sertindole has been shown to have efficacy in the treatment of schizophrenia and to cause only minimal EPS, perhaps related to its greater affinity for 5-HT2A than D2.65
Personalized Medicine
A double-blind study comparing iloperidone and ziprasidone with placebo produced data concerning genetic markers that may be associated with efficacy. Based on a genome-wide association study, the investigators found 6 genes that predicted treatment response to iloperidone.66 If nothing is known about genotype, a patient has a 48% chance of achieving a response rate greater than 20%. However, when genotype is known, a pattern emerges: patients with 0-2 predictive genes have a ~15% chance of responding, those with 3 have a ~35% chance, those with 4 have a 55%-60% chance, those with 5 have an 80% chance, and those with all 6 predictive genes have more than a 90% chance of responding. In the evaluated population, approximately 25% of patients had 0-2 predictive genes, 25% had 3, 25% had 4, and 25% had 5 or 6, so that about half of the patients have a greater than 50% chance of responding. If these findings are confirmed and genotype information can be collected concerning other antipsychotics, the stage will be set for personalized medicine in which clinicians can use genotyping to help select the most appropriate agent for a specific patient. Similar methods may also help predict which patients would be most at risk for certain side effects such as tardive dyskinesia, QTc prolongation, and rash.
Summary
Over the last decade, the rising placebo response rate in studies of schizophrenia has been making it increasingly difficult to show the value and efficacy of newer agents due to loss of statistical power. The data presented here highlight a number of important take-home points:
- Staying on medication can have accruing benefits, which may result in remission over time.
- Clinical trials of antipsychotics report mean changes, which may obscure individual response in some patients. Clinicians need to keep in mind the variability in individual patients’ responses and remember that one size does not fit all.
- Access to a variety of treatment options gives clinicians the opportunity to find the agent that is best for each specific patient in terms of enhanced efficacy and minimizing side effects.
Despite current challenges in drug development, a number of new agents with new mechanisms of action are, or soon will be, available to treat patients with schizophrenia. It is important that clinicians focus not just on positive but also on negative symptoms and cognitive dysfunction, in order to reduce disability and improve patients’ functioning and QOL.
USING NEW ANTIPSYCHOTICS
The faculty discussed how to integrate new antipsychotics into clinical practice to achieve best outcomes. Only a brief overview of their recommendations is given here, but readers can access detailed recommendations and listen to the faculty discuss these issues online (“Using New Antipsychotics in Your Clinical Practice” at PSYCHIATRIST.COM, Keyword: New Antipsychotics). Limiting use of new antipsychotics only to those patients who have failed to respond to other antipsychotics is likely to produce disappointing results. It also denies patients who have achieved only a partial response a chance for a better overall response with a newer agent. The faculty recommended that clinicians:
- Move beyond the concept of a “good enough response.”
- Treat patients more aggressively to try to prevent relapses and achieve remission.
- Assess cognitive status as well as positive and negative symptoms.
- Consider treatment strategies to promote cognitive enhancement.
- Integrate developing principles of personalized medicine in their practice as they become available.
- Before making a change, simplify and streamline the current treatment regimen, reducing polypharmacy if possible.
- Consider a trial of a different antipsychotic if a patient has had only a partial response or has developed bothersome side effects after several months of treatment.
- Educate patients and family members about potential risks and benefits of switching to a new agent and the value of cognitive remediation, rehabilitation, and family and community support.
- Generally use a slower switch (eg, 4-8 weeks) from one medication to another to promote stability and minimize withdrawal effects, unless a rapid switch is needed because of symptomatic decompensation or serious side effects.
- Assess response (positive, negative, and cognitive symptoms) on a regular basis using short measurement tools (see online report for specific assessment tools at PSYCHIATRIST.COM, Keyword: New Antipsychotics). It may take significantly longer (eg, 6 months or longer) to see an effect on cognition and perhaps negative symptoms than on positive symptoms.
- Decide in advance on the specific treatment goals and when to reasonably expect a response and discontinue the agent if these goals are not met to avoid side effects and other problems that can occur with polypharmacy.
Drug names: aripiprazole (Abilify), asenapine (Saphris), clozapine (Clozaril, FazaClo, and others), cycloserine (Seromycin), haloperidol (Haldol and others), iloperidone (Fanapt), olanzapine (Zyprexa), paliperidone (Invega), quetiapine (Seroquel), risperidone (Risperdal and others), ziprasidone (Geodon).
REFERENCES
1. World Health Organization. World Health Report 2001; Mental Health: New Understanding. www.who.int/whr/2001/en/whr01_en.pdf. Updated 2001. Accessed February 3, 2010.
2. Harvey PD, Helldin L, Bowie CR, et al. Performance-based measurement of functional disability in schizophrenia: a cross-national study in the United States and Sweden. Am J Psychiatry. 2009;166(7):821-827. PubMed doi:10.1176/appi.ajp.2009.09010106
3. Lehman AF, Lieberman JA, Dixon LB, et al; American Psychiatric Association; Steering Committee on Practice Guidelines. Practice guideline for the treatment of patients with schizophrenia, second edition. Am J Psychiatry. 2004;161(suppl):1-56. PubMed
4. Pickar D, Vinik J, Bartko JJ. Pharmacotherapy of schizophrenic patients: preponderance of off-label drug use. PLoS ONE. 2008;3(9):e3150. PubMed doi:10.1371/journal.pone.0003150
5. Pandurangi AK, Dalkilic A. Polypharmacy with second-generation antipsychotics: a review of evidence. J Psychiatr Pract. 2008;14(6):345-367. PubMed doi:10.1097/01.pra.0000341890.05383.45
6. Correll CU, Kane JM, O’ Shea D, et al. Antipsychotic polypharmacy in the treatment of schizophrenia. Schizophr Res. 2003;60(suppl.):37. doi:10.1016/S0920-9964(03)80107-X
7. Flanagan JC. Measurement of quality of life: current state of the art. Arch Phys Med Rehabil. 1982;63(2):56-59. PubMed
8. Lehman AF. A Quality of Life Interview for the chronically mentally ill. Eval Program Plann. 1988;11(1):51-62. doi:10.1016/0149-7189(88)90033-X
9. Rosenheck RA, Leslie DL, Sindelar J, et al; CATIE Study Investigators. Cost-effectiveness of second-generation antipsychotics and perphenazine in a randomized trial of treatment for chronic schizophrenia. Am J Psychiatry. 2006;163(12):2080-2089. PubMed doi:10.1176/appi.ajp.163.12.2080
10. Swartz MS, Perkins DO, Stroup TS, et al; CATIE Investigators. Effects of antipsychotic medications on psychosocial functioning in patients with chronic schizophrenia: findings from the NIMH CATIE study. Am J Psychiatry. 2007;164(3):428-436. PubMed doi:10.1176/appi.ajp.164.3.428
11. Kay SR, Fiszbein A, Opler LA. The Positive and Negative Syndrome Scale (PANSS) for schizophrenia. Schizophr Bull. 1987;13(2):261-276. PubMed
12. Bowie CR, Leung WW, Reichenberg A, et al. Predicting schizophrenia patients’ real-world behavior with specific neuropsychological and functional capacity measures. Biol Psychiatry. 2008;63(5):505-511. PubMed doi:10.1016/j.biopsych.2007.05.022
13. Leifker FR, Bowie CR, Harvey PD. Determinants of everyday outcomes in schizophrenia: the influences of cognitive impairment, functional capacity, and symptoms. Schizophr Res. 2009;115(1):82-87. PubMed doi:10.1016/j.schres.2009.09.004
14. DeBrota D, Demitrack M, Landin R, et al. A comparison between interactive voice response system Y-administered HAM-D and clinician-administered HAM-D in patients with major depressive episode. NCDEU 39th Annual Meeting. Boca Raton, FL; 1999
15. Kemp AS, Schooler NR, Kalali AH, et al. What is causing the reduced drug-placebo difference in recent schizophrenia clinical trials and what can be done about it? Schizophr Bull. 2010;36(3):504-509. PubMed doi:10.1093/schbul/sbn110
16. Kahn RS, Schulz SC, Palazov VD, et al; Study 132 Investigators. Efficacy and tolerability of once-daily extended release quetiapine fumarate in acute schizophrenia: a randomized, double-blind, placebo-controlled study. J Clin Psychiatry. 2007;68(6):832-842. PubMed doi:10.4088/JCP.v68n0603
17. American Psychiatric Association. DSM-5 development. http://www.dsm5.org/Pages/Default.aspx. Accessed March 4 2010.
18. Marder SR, Fenton W. Measurement and Treatment Research to Improve Cognition in Schizophrenia: NIMH MATRICS initiative to support the development of agents for improving cognition in schizophrenia. Schizophr Res. 2004;72(1):5-9. PubMed doi:10.1016/j.schres.2004.09.010
19. Marder SR, Fenton W, Youens K. Schizophrenia, IX: cognition in schizophrenia—the MATRICS initiative. Am J Psychiatry. 2004;161(1):25. PubMed doi:10.1176/appi.ajp.161.1.25
20. Nuechterlein KH, Green MF, Kern RS, et al. The MATRICS Consensus Cognitive Battery, part 1: test selection, reliability, and validity. Am J Psychiatry. 2008;165(2):203-213. PubMed doi:10.1176/appi.ajp.2007.07010042
21. Kern RS, Nuechterlein KH, Green MF, et al. The MATRICS Consensus Cognitive Battery, part 2: co-norming and standardization. Am J Psychiatry. 2008;165(2):214-220. PubMed doi:10.1176/appi.ajp.2007.07010043
22. MATRICS Consensus Cognitive Battery. www.matricsinc.org. Accessed February 15, 2010.
23. Green MF, Nuechterlein KH, Kern RS, et al. Functional co-primary measures for clinical trials in schizophrenia: results from the MATRICS Psychometric and Standardization Study. Am J Psychiatry. 2008;165(2):221-228. PubMed doi:10.1176/appi.ajp.2007.07010089
24. Patterson TL, Goldman S, McKibbin CL, et al. UCSD Performance-Based Skills Assessment: development of a new measure of everyday functioning for severely mentally ill adults. Schizophr Bull. 2001;27(2):235-245. PubMed
25. Leifker FR, Patterson TL, Bowie CR, et al. Psychometric properties of performance-based measurements of functional capacity: test-retest reliability, practice effects, and potential sensitivity to change. Schizophr Res. 2010;119(1-3):246-252. PubMed
26. Mausbach BT, Harvey PD, Goldman SR, et al. Development of a brief scale of everyday functioning in persons with serious mental illness. Schizophr Bull. 2007;33(6):1364-1372. PubMed doi:10.1093/schbul/sbm014
27. Mausbach BT, Harvey PD, Pulver AE, et al. Relationship of the Brief UCSD Performance-based Skills Assessment (UPSA-B) to multiple indicators of functioning in people with schizophrenia and bipolar disorder. Bipolar Disord. 2010;12(1):45-55. PubMed doi:10.1111/j.1399-5618.2009.00787.x
28. Ventura J, Cienfuegos A, Boxer O, et al. Clinical Global Impression of Cognition in Schizophrenia (CGI-CogS): reliability and validity of a co-primary measure of cognition. Schizophr Res. 2008;106(1):59-69. PubMed doi:10.1016/j.schres.2007.07.025
29. Keefe RS, Poe M, Walker TM, et al. The Schizophrenia Cognition Rating Scale: an interview-based assessment and its relationship to cognition, real-world functioning, and functional capacity. Am J Psychiatry. 2006;163(3):426-432. PubMed doi:10.1176/appi.ajp.163.3.426
30. Buchanan RW, Javitt DC, Marder SR, et al. The Cognitive and Negative Symptoms in Schizophrenia Trial (CONSIST): the efficacy of glutamatergic agents for negative symptoms and cognitive impairments. Am J Psychiatry. 2007;164(10):1593-1602. PubMed doi:10.1176/appi.ajp.2007.06081358
31. Geyer MA, Tamminga CA. Measurement and treatment research to improve cognition in schizophrenia: neuropharmacological aspects. Psychopharmacology (Berl). 2004;174(1):1-2. doi:10.1007/s00213-004-1846-2
32. Harvey PD, Ogasa M, Cucchiaro J, et al. Performance and interview-based assessments of cognitive change in a randomized, double-blind comparison of lurasidone vs ziprasidone. International Congress on Schizophrenia Research Meeting. San Diego, CA; 2009
33. Friedman JI, Carpenter D, Lu J, et al. A pilot study of adjunctive atomoxetine treatment to second-generation antipsychotics for cognitive impairment in schizophrenia. J Clin Psychopharmacol. 2008;28(1):59-63. PubMed doi:10.1097/jcp.0b013e318161318f
34. McClure MM, Barch DM, Romero MJ, et al. The effects of guanfacine on context processing abnormalities in schizotypal personality disorder. Biol Psychiatry. 2007;61(10):1157-1160. PubMed doi:10.1016/j.biopsych.2006.06.034
35. McGurk SR, Mueser KT, Feldman K, et al. Cognitive training for supported employment: 2-3 year outcomes of a randomized controlled trial. Am J Psychiatry. 2007;164(3):437-441. PubMed doi:10.1176/appi.ajp.164.3.437
36. Vinogradov S, Fisher M, Holland C, et al. Is serum brain-derived neurotrophic factor a biomarker for cognitive enhancement in schizophrenia? Biol Psychiatry. 2009;66(6):549-553. PubMed doi:10.1016/j.biopsych.2009.02.017
37. Scharfman H, Goodman J, Macleod A, et al. Increased neurogenesis and the ectopic granule cells after intrahippocampal BDNF infusion in adult rats. Exp Neurol. 2005;192(2):348-356. PubMed doi:10.1016/j.expneurol.2004.11.016
38. Yamada K, Nabeshima T. Brain-derived neurotrophic factor/TrkB signaling in memory processes. J Pharmacol Sci. 2003;91(4):267-270. PubMed doi:10.1254/jphs.91.267
39. Potkin SG, Weiden PJ, Loebel AD, et al. Remission in schizophrenia: 196-week, double-blind treatment with ziprasidone vs haloperidol. Int J Neuropsychopharmacol. 2009;12(9):1233-1248. PubMed doi:10.1017/S1461145709000352
40. Bymaster FP, Calligaro DO, Falcone JF, et al. Radioreceptor binding profile of the atypical antipsychotic olanzapine. Neuropsychopharmacology. 1996;14(2):87-96. PubMed doi:10.1016/0893-133X(94)00129-N
41. Heylen L, Gys L, Meulders G, et al. Comparative in vitro receptor and transporter binding profile and neurotransmitter uptake of paliperidone and risperidone, J&J Pharmaceutical Research & Development, June 8, 2006.
42. Kalkman HO, Subramanian N, Hoyer D. Extended radioligand binding profile of iloperidone: a broad spectrum dopamine/serotonin/norepinephrine receptor antagonist for the management of psychotic disorders. Neuropsychopharmacology. 2001;25(6):904-914. PubMed doi:10.1016/S0893-133X(01)00285-8
43. Kongsamut S, Roehr JE, Cai J, et al. Iloperidone binding to human and rat dopamine and 5-HT receptors. Eur J Pharmacol. 1996;317(2-3):417-423. PubMed doi:10.1016/S0014-2999(96)00840-0
44. Ma J, Ye N, Cohen BM. Expression of noradrenergic α1, serotoninergic 5HT2a and dopaminergic D2 receptors on neurons activated by typical and atypical antipsychotic drugs. Prog Neuropsychopharmacol Biol Psychiatry. 2006;30(4):647-657. PubMed doi:10.1016/j.pnpbp.2005.11.040
45. Sanchez C, Arnt J, Dragsted N, et al. Neurochemical and in vivo pharmacological profile of sertindole, a limbic-sensitive neuroleptic compounds. Drug Dev Res. 1991;22:239-250. doi:10.1002/ddr.430220306
46. Sánchez C, Arnt J. In-vivo assessment of 5-HT2A and 5-HT2C antagonistic properties of newer antipsychotics. Behav Pharmacol. 2000;11(3-4):291-298. PubMed
47. Shahid M, Walker GB, Zorn SH, et al. Asenapine: a novel psychopharmacologic agent with a unique human receptor signature. J Psychopharmacol. 2009;23(1):65-73. PubMed doi:10.1177/0269881107082944
48. Spina E, Zoccali R. Sertindole: pharmacological and clinical profile and role in the treatment of schizophrenia. Expert Opin Drug Metab Toxicol. 2008;4(5):629-638. PubMed doi:10.1517/17425255.4.5.629
49. Theisen FM, Haberhausen M, Firnges MA, et al. No evidence for binding of clozapine, olanzapine and/or haloperidol to selected receptors involved in body weight regulation. Pharmacogenomics J. 2007;7(4):275-281. PubMed doi:10.1038/sj.tpj.6500418
50. Wood MD, Scott C, Clarke K, et al. Pharmacological profile of antipsychotics at monoamine receptors: atypicality beyond 5-HT2A receptor blockade. CNS Neurol Disord Drug Targets. 2006;5(4):445-452. PubMed doi:10.2174/187152706777950693
51. Ishibashi T, Horisawa T, Tokuda K, et al. Pharmacological profile of lurasidone, a novel antipsychotic agent with potent 5-HT7 and 5-HT1A receptor activity. J Pharmacol Exp Ther.2010;334(1):171-181. PubMed
52. Kim DH, Maneen MJ, Stahl SM. Building a better antipsychotic: receptor targets for the treatment of multiple symptom dimensions of schizophrenia. Neurotherapeutics. 2009;6(1):78-85. PubMed doi:10.1016/j.nurt.2008.10.020
53. Weiden PJ, Preskorn SH, Fahnestock PA, et al; Roadmap Survey. Translating the psychopharmacology of antipsychotics to individualized treatment for severe mental illness: a Roadmap. J Clin Psychiatry. 2007;68(suppl 7):1-48. PubMed
54. Gardner DM, Baldessarini RJ, Waraich P. Modern antipsychotic drugs: a critical overview. CMAJ. 2005;172(13):1703-1711. PubMed
55. Saphris prescribing information. Schering-Plough Corporation. www.spfiles.com/pisaphrisv1.pdf. Updated June 2009. Accessed February 23, 2010.
56. Potkin SG, Cohen M, Panagides J. Efficacy and tolerability of asenapine in acute schizophrenia: a placebo- and risperidone-controlled trial. J Clin Psychiatry. 2007;68(10):1492-1500. PubMed doi:10.4088/JCP.v68n1004
57. Fanapt (iloperidone) prescribing information. Novartis (Vanda) Pharmaceuticals. (www.fanapt.com/index.jsp?irmasrc=FPTWB0013 , Updated July 2009. Accessed March 18, 2010.
58. Potkin SG, Litman RE, Torres R, et al. Efficacy of iloperidone in the treatment of schizophrenia: initial phase 3 studies. J Clin Psychopharmacol. 2008;28(suppl 1):S4-S11. PubMed
59. Cutler AJ, Kalali AH, Weiden PJ, et al. Four-week, double-blind, placebo- and ziprasidone-controlled trial of iloperidone in patients with acute exacerbations of schizophrenia. J Clin Psychopharmacol. 2008;28(suppl 1):S20-S28. PubMed doi:10.1097/JCP.0b013e318169d4ce
60. Weiden PJ, Cutler AJ, Polymeropoulos MH, et al. Safety profile of iloperidone: a pooled analysis of 6-week acute-phase pivotal trials. J Clin Psychopharmacol. 2008;28(suppl 1):S12-S19. PubMed
61. Meyer JM, Loebel AD, Schweizer E. Lurasidone: a new drug in development for schizophrenia. Expert Opin Investig Drugs. 2009;18(11):1715-1726. PubMed doi:10.1517/13543780903286388
62. Nakamura M, Ogasa M, Guarino J, et al. Lurasidone in the treatment of acute schizophrenia: a double-blind, placebo-controlled trial. J Clin Psychiatry. 2009;70(6):829-836. PubMed doi:10.4088/JCP.08m04905
63. Ishiyama T, Tokuda K, Ishibashi T, et al. Lurasidone (SM-13496), a novel atypical antipsychotic drug, reverses MK-801-induced impairment of learning and memory in the rat passive-avoidance test. Eur J Pharmacol. 2007;572(2-3):160-170. PubMed doi:10.1016/j.ejphar.2007.06.058
64. Enomoto T, Ishibashi T, Tokuda K, et al. Lurasidone reverses MK-801-induced impairment of learning and memory in the Morris water maze and radial-arm maze tests in rats. Behav Brain Res. 2008;186(2):197-207. PubMed doi:10.1016/j.bbr.2007.08.012
65. FDA Psychopharmacologic Drugs Advisory Committee Hearings April 7-8, 2009. www.aacap.org/galleries/LegislativeAction/FDA Psychopharm Hearing Summary_Final.pdf. Updated April 2009. Accessed April 22, 2010.
66. Volpi S, Potkin SG, Malhotra AK, et al. Applicability of a genetic signature for enhanced iloperidone efficacy in the treatment of schizophrenia. J Clin Psychiatry. 2009;70(6):801-809. PubMed doi:10.4088/JCP.08m04391
doi:10.4088/JCP.10064ah1yel
© Copyright 2010 Physicians Postgraduate Press, Inc.
This PDF is free for all visitors!