This work may not be copied, distributed, displayed, published, reproduced, transmitted, modified, posted, sold, licensed, or used for commercial purposes. By downloading this file, you are agreeing to the publisher’s Terms & Conditions.
Objective: To characterize subgroups of subjects with schizophrenia from the Ziprasidone Observational Study of Cardiac Outcomes (ZODIAC) trial who either completed or attempted suicide and those who did not.
Method: The ZODIAC, conducted between February 2002 and March 2007, was an open-label, randomized, large simple trial of patients with schizophrenia (N = 18,154) followed up for 1 year by unblinded investigators providing usual care in 18 countries; the primary outcome measure was nonsuicide mortality. Every report on a completed or attempted suicide was independently adjudicated using a predefined algorithm. Primary analysis for the current report examined the association between completed or attempted suicides and the baseline variables using descriptive statistics and multivariate logistic regression models. Usage of “hard” or “soft” methods for attempted or completed suicide and distribution of suicide-related events by geographical region were also summarized.
Results: Overall incidences of subjects who either completed (35/18,154) or attempted (108/18,154) suicide were low, as were rates per person-time on assigned treatment analysis (0.24 for completed and 0.74 for attempted suicides per 100 person-years of exposure). The highest suicide-related mortality was seen among subjects recently diagnosed with schizophrenia. Among all potential baseline risk factors for completed suicide examined, the variables most associated with completed suicide were history of suicide attempts (OR = 2.6; 95% CI, 1.33-5.12) and usage of antidepressant medication (OR = 3.5; 95% CI, 0.84-14.85). History of > 5 hospitalizations in the past (OR = 2.1; 95% CI, 1.35-3.31) and history of suicide attempts (OR = 5.0; 95% CI, 3.21-7.76) were the variables most associated with attempted suicide among potential baseline risk factors for suicide attempts.
Conclusions: Our results, obtained in a large prospective randomized study, confirm current clinical understanding regarding completed or attempted suicide in schizophrenia and the associated risk factors.
Trial Registration: ClinicalTrials.gov identifier: NCT00418171
Completed and Attempted Suicides Among 18,154 Subjects With Schizophrenia Included in a Large Simple Trial

ABSTRACT
Objective: To characterize subgroups of subjects with schizophrenia from the Ziprasidone Observational Study of Cardiac Outcomes (ZODIAC) trial who either completed or attempted suicide and those who did not.
Method: The ZODIAC, conducted between February 2002 and March 2007, was an open-label, randomized, large simple trial of patients with schizophrenia (N = 18,154) followed up for 1 year by unblinded investigators providing usual care in 18 countries; the primary outcome measure was nonsuicide mortality. Every report on a completed or attempted suicide was independently adjudicated using a predefined algorithm. Primary analysis for the current report examined the association between completed or attempted suicides and the baseline variables using descriptive statistics and multivariate logistic regression models. Usage of “hard” or “soft” methods for attempted or completed suicide and distribution of suicide-related events by geographical region were also summarized.
Results: Overall incidences of subjects who either completed (35/18,154) or attempted (108/18,154) suicide were low, as were rates per person-time on assigned treatment analysis (0.24 for completed and 0.74 for attempted suicides per 100 person-years of exposure). The highest suicide-related mortality was seen among subjects recently diagnosed with schizophrenia. Among all potential baseline risk factors for completed suicide examined, the variables most associated with completed suicide were history of suicide attempts (OR = 2.6; 95% CI, 1.33-5.12) and usage of antidepressant medication (OR = 3.5; 95% CI, 0.84-14.85). History of > 5 hospitalizations in the past (OR = 2.1; 95% CI, 1.35-3.31) and history of suicide attempts (OR = 5.0; 95% CI, 3.21-7.76) were the variables most associated with attempted suicide among potential baseline risk factors for suicide attempts.
Conclusions: Our results, obtained in a large prospective randomized study, confirm current clinical understanding regarding completed or attempted suicide in schizophrenia and the associated risk factors.
Trial Registration: ClinicalTrials.gov identifier: NCT00418171
J Clin Psychiatry 2014;75(3):e184-e190
© Copyright 2014 Physicians Postgraduate Press, Inc.
Submitted: May 2, 2013; accepted October 30, 2013 (doi:10.4088/JCP.13m08563).
Corresponding author: W. Wolfgang Fleischhacker, MD, Department of Psychiatry and Psychotherapy, Medical University Innsbruck, Innsbruck, Austria 6020 ([email protected]).
Suicide is a major cause of death among patients with schizophrenia,1 with lifetime prevalence estimates over 10 times higher than the general population2 and a relatively higher suicide risk found in younger age groups of patients.3,4 The risk is particularly high in the first episode of the illness and decreases with time after the first hospital admission.5 Different studies, using different populations and methods, have yielded varying estimates of the suicide rate in schizophrenia.6,7 While earlier research has suggested that up to 13% of patients with schizophrenia commit suicide,2 more recent studies that take into account the variable suicide risk during the life span (a higher risk close to illness onset and thereafter a declining risk) have reported a lifetime suicide mortality of 4%-5%.6-9 Nevertheless, this figure remains significantly higher than estimates for the general population, with the mortality gap between the 2 populations increasing over the last decades.9,10
A number of risk factors for suicide in patients with schizophrenia have been identified, including young age, male gender, higher level of education, greater number of prior suicide attempts, presence of depressive symptoms, active hallucinations and delusions, and insight.6,7
Approximately 20%-40% of patients with schizophrenia attempt suicide in their lifetime.11-13 Although past suicide attempts are identified as one of the strongest risk factors for completed suicide,14,15 epidemiologic data are sparse. Contrary to completed suicide, which was found to be a more common cause of excess mortality in males versus females with schizophrenia,3 suicide attempts were observed at the same proportion in both sexes.16 Typically, the attempts are serious and require medical attention.
The Ziprasidone Observational Study of Cardiac Outcomes (ZODIAC) was an international, multicenter, randomized, large simple trial designed to examine the risk of nonsuicide mortality associated with 2 second-generation antipsychotics, ziprasidone and olanzapine, in the context of usual clinical treatment in the year after initiation of assigned treatment.17,18 Large simple trials (pragmatic or practical trials) intend to provide generalizable answers to simple clinical questions without the risk of channeling bias.19 In the primary analysis of the ZODIAC study,18 the incidence of nonsuicide mortality within 1 year of initiating pharmacotherapy was 0.91% for ziprasidone and 0.90% for olanzapine; the relative risk was 1.02 (95% CI, 0.76-1.39).
A prespecified statistical analysis plan in the ZODIAC study defined completed and attempted suicides as 1 of the secondary outcomes.17,18 Suicide is the leading cause of premature death in schizophrenia.1 Thus, identifying individuals with schizophrenia who are at high risk for completed suicide or suicide attempts is an important public health, clinical, and research priority. A greater understanding and timely recognition of key risk factors for suicidal events would help focus clinical priorities and resources on highly vulnerable patients during critical time periods. Therefore, the aim of the present analysis was to characterize the following subgroups in the ZODIAC study: subjects who completed or attempted suicide and those who did not. An additional aim was to replicate findings on risk factors for completed or attempted suicide previously reported in smaller studies.
METHOD
Full details on the ZODIAC study design and methodology are presented elsewhere17,18 and briefly summarized here. The ZODIAC was a large simple trial, ie, the study was conducted with a minimal modification of routine medical care, except for random allocation of subjects in 1:1 fashion to either ziprasidone or olanzapine. Because the trial was aligned with routine medical care, neither the physician nor the subject was blind to treatment allocation; medication regimens and dosing could be changed based on patients’ responses to the assigned treatment, and use of concomitant medications, including other antipsychotics, was permitted. The trial was conducted between February 2002 and March 2007. It was registered on ClinicalTrials.gov (identifier: NCT00418171).

- Completed or attempted suicide occurs frequently in patients with schizophrenia.
- Data suggest an association between completed suicides and a number of risk factors, including baseline usage of antidepressant medications and history of suicide attempts.
- A greater understanding and recognition of key risk factors for suicidal events in patients with schizophrenia would help focusing clinical priorities and resources on a highly vulnerable population during critical time periods.
Newly treated subjects with schizophrenia as well as those receiving continuing treatment were eligible if their treating psychiatrist was ready to initiate a new antipsychotic medication and would consider using either ziprasidone or olanzapine as appropriate. Furthermore, they had to be diagnosed as having schizophrenia according to the treating physician’s clinical judgment, to be inpatients or outpatients ≥ 18 years of age, and to provide signed and dated informed consent. Subjects were excluded if they were pregnant or lactating, participating in any other studies involving investigational products concomitantly or within 30 days before entry in the study, had a progressive fatal disease or a life expectancy that would prohibit participation in a 1-year research study, or were previously enrolled in this study and randomized to a study medication.
Subjects from 18 countries in Europe, Asia, and North and South America were included in the study. After the subjects signed informed consent, information on demographics, severity of schizophrenia as determined on the Clinical Global Impressions-Severity of Illness scale (CGI-S),20 cardiac risk factors, and prior medication use was collected on the baseline questionnaire.17 Each participant was to be followed for 1 year, regardless of how long he or she remained on randomized treatment. End point information on the participant’s vital status, continued use of the assigned study drug as indicated on study questionnaires, and whether the participant was hospitalized was obtained through follow-up with the treating physician or another designated member of the medical care team. Medical records and other documentation, where applicable, were obtained for end point screening and coding purposes. In the United States, the National Death Index21 was also searched for information on vital status for participants lost to follow-up. In the event that the participant could not be contacted, his or her alternative contact was used.
The primary outcome measure was nonsuicide mortality; secondary end points included sudden death, suicide, all-cause and cardiovascular mortality, all-cause hospitalization, and hospitalization for arrhythmia (including arrhythmia reported during hospitalization for other reasons), diabetic ketoacidosis, and myocardial infarction. Discontinuation of randomized treatment as recorded on study questionnaires was also a secondary end point.
A scientific steering committee (SSC), data safety monitoring board (DSMB), and end point committee conducted the study oversight. The SSC and DSMB role was to safeguard scientific integrity of the study, participants’ interests, and monitor overall study conduct; full details are described in Strom et al.17
The chair of the end point committee, blinded to treatment allocation, reviewed all fatal events and hospitalizations for classification into 1 of 3 categories: a “potential study end point,” “end point with insufficient data,” or “not a potential study end point.” All potential study end points were placed into 1 of 6 categories (myocardial infarction, arrhythmia, sudden death, diabetic ketoacidosis, suicide, or fatal event with other cause) for detailed review and end point determination by end point committee experts in the fields of psychiatry, cardiology, and endocrinology from the regions participating in the study. Two expert coders, blind to treatment allocation, reviewed anonymized records (eg, medical records, laboratory data, hospital discharge or admission notes, and death certificates) specific to each event. A final end point labeling with level of certainty (“definite,” “possible,” “no indication of end point,” and “insufficient data to determine diagnosis”) was assigned for each event according to prespecified algorithms and expert consensus.
The suicide mortality end point algorithm was based on the history of suicide attempt or known suicidal tendencies and description of the event (eg, suicide note, documentation of method used). Events were coded as follows:
- “Suicide, definite” if there were previously documented suicide attempts or a suicide note was discovered, and an accidental cause of death could be excluded.
- “Suicide, possible” if there was suicidal ideation or previous suicide attempt but no suicide note found, and an accidental cause of death could be excluded.
- “Suicide, insufficient data” if it was a fatal event without clear documentation to decide either way.
Likewise, suicide attempts were coded as follows:
- “Suicide attempt, definite” if there was a nonfatal event with documentation of past attempts or a suicide note was discovered, and an accident could be excluded.
- “Suicide attempt, possible” if there was a nonfatal event with suicidal ideation but no suicide note was discovered or documented, or there had been a previous attempt but no suicide note was discovered or documented. In either case, an accident could be excluded.
All events and methods used for attempted or completed suicide, recorded in the adverse events forms and case notes, were reviewed by one of the authors (W.W.F.) and classified as a “hard” or “soft” method. Hard methods were those involving hanging or any sort of trauma; soft methods included poisoning or drug overdose. This procedure also included adjudication as to whether or not identified cases met criteria for completed suicides or suicide attempts. Case notes as well as information available from all sources were used to make a distinction between suicide attempt and nonsuicidal self-injury.
Statistical Analysis
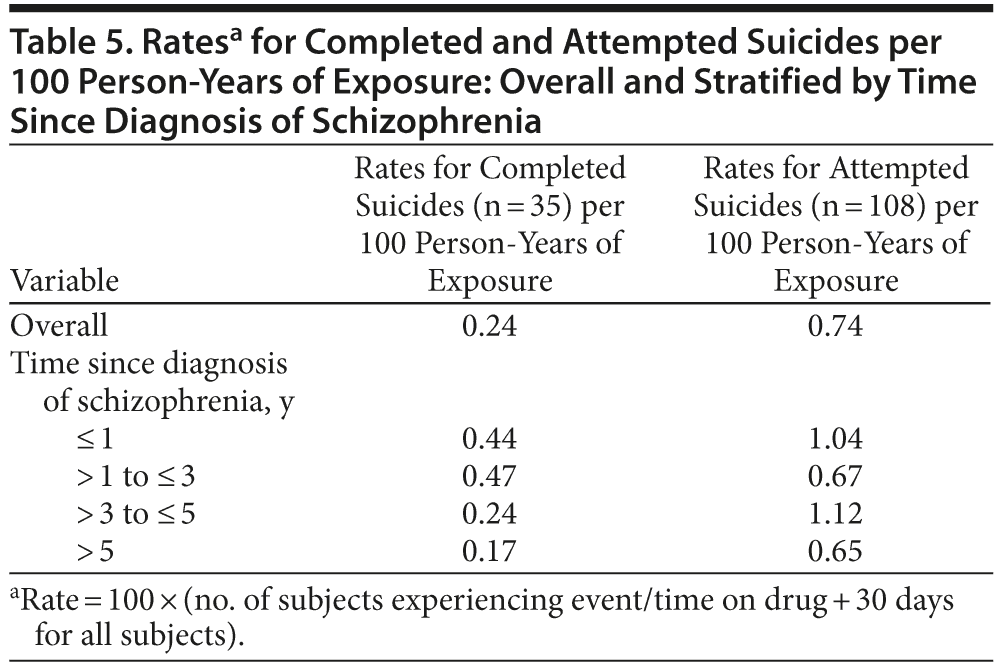
Baseline demographic and illness characteristics were summarized using descriptive statistics for the subgroups of subjects who completed (yes/no) or attempted suicide (yes/no). The methods used by subjects in completing or attempting suicide were summarized using descriptive statistics. Estimates of suicide (completed or attempted) rates per 100 person-years of exposure, stratified by time since diagnosis of schizophrenia (≤ 1 year, > 1 to ≤ 3 years, > 3 to ≤ 5 years, and > 5 years), were also obtained.
A pool of baseline variables that are potential risk factors for completed or attempted suicide were identified by fitting separate logistic regression models (with treatment as a covariate in the model). Variables that were significantly associated (P < .05) with suicide (completed or attempted) from this analysis were then used to build a multivariate logistic regression model. The multivariate regression model (with treatment as a covariate) was used to describe the association between these baseline variables and each of the 2 response variables of completed or attempted suicide.
RESULTS
Completed or Attempted Suicide Incidence
There were 35 completed suicides among 18,154 subjects in the ZODIAC study, identified using a narrowly defined end point classifications of definite (29 cases [82.9%] of all completed suicides) or possible (6 cases [17.1%] of all completed suicides) cases combined, yielding an incidence of 0.19% for suicide-related mortality.18 When a broadly defined end point (which combines definite, possible, and insufficient data cases) was applied, 42 cases were identified, yielding a suicide-related mortality incidence of 0.23%.18 Furthermore, 108 subjects attempted suicide, resulting in an incidence of 0.59%. In the person-time on assigned treatment analysis, the event rates per 100 person-years were 0.24 and 0.74 for completed and attempted suicides, respectively. Multiple suicide attempts occurred for 3 subjects during the study course.
Baseline Demographic and Illness Characteristics
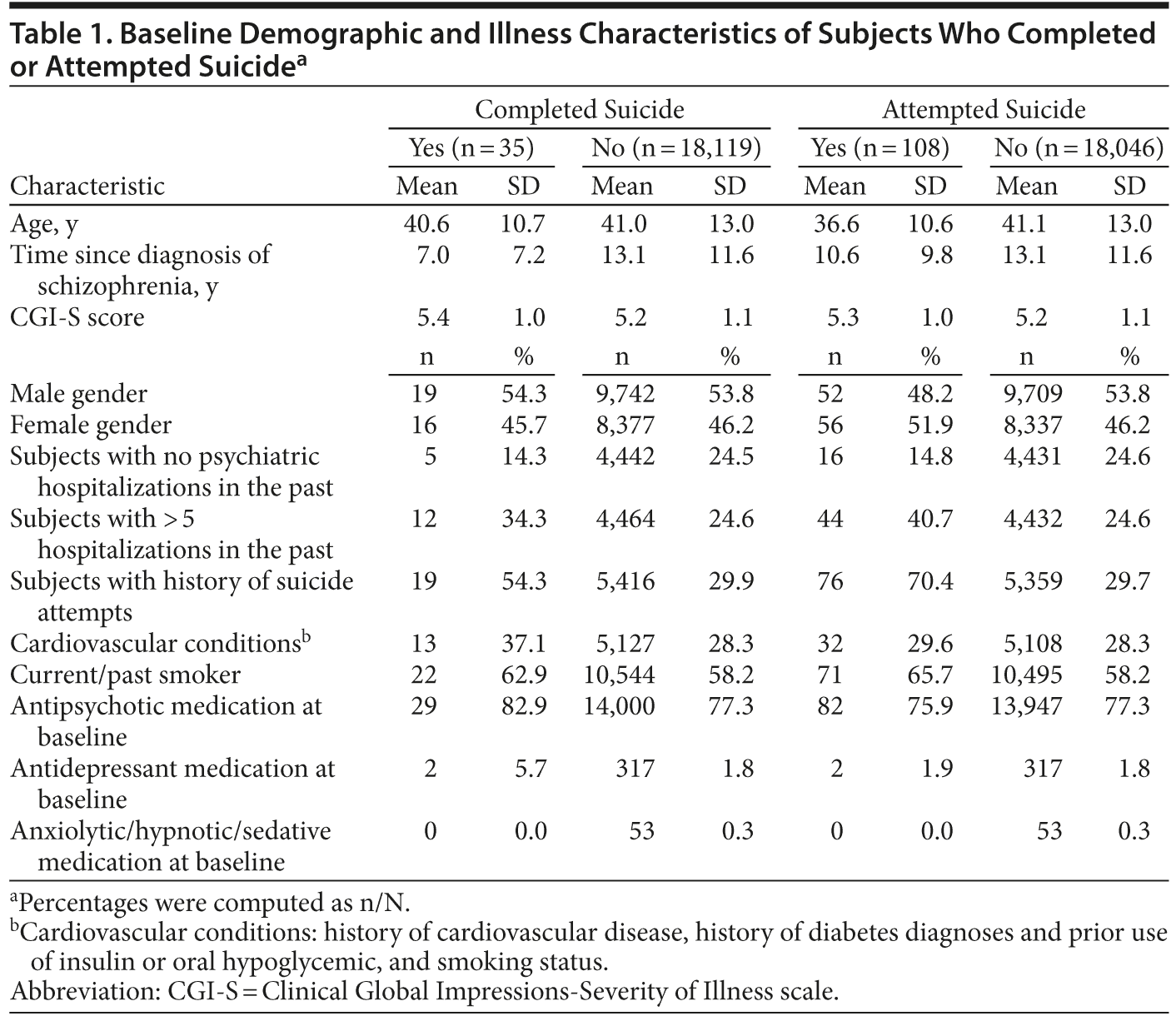
Baseline demographic and illness characteristics are presented in Table 1. Some differences were observed between the subjects who completed suicide and those who did not. Compared with the subjects who did not complete suicide, those who did appeared to have a shorter illness duration, a higher number of past suicide attempts and of subjects with > 5 previous hospitalizations, and more frequent baseline usage of antipsychotic or antidepressant medications. Furthermore, subjects who completed suicide had more cardiovascular conditions at baseline and were more likely to be present or past smokers. The age and sex distributions, as well as disease severity assessed by CGI-S ratings, were similar between the 2 subgroups.
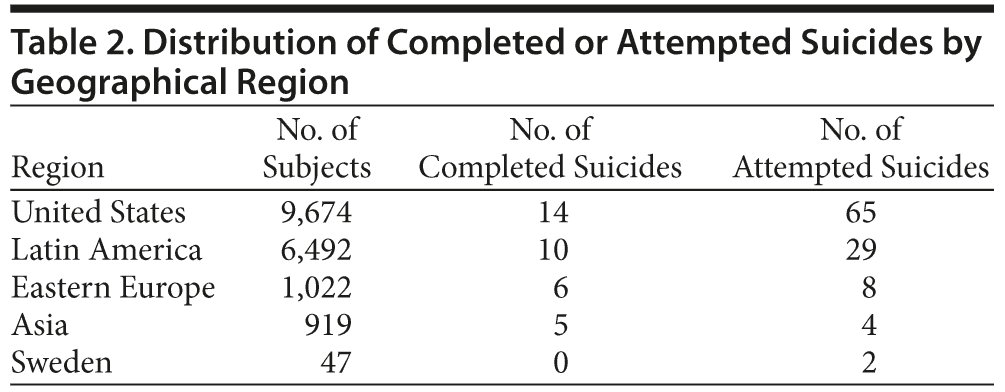
Differences in baseline characteristics were also observed between the subjects who attempted suicide versus those who did not. The subjects who attempted suicide were younger and more frequently female, they had a shorter illness duration, and more of them were present or past smokers at baseline. They had more frequent previous hospitalizations (ie, > 5 previous hospitalizations) and more frequent previous suicide attempts. However, the 2 groups were similar in terms of illness severity as assessed by CGI-S ratings, frequency of cardiovascular disorders, and baseline use of antipsychotic or antidepressant medications. Distribution of completed and attempted suicides across geographical regions is summarized in Table 2. No further statistical analyses were performed due to the uneven distribution of these geographical regions in the study.
Baseline characteristics of the ziprasidone and olanzapine treatment groups as well as results of the ZODIAC study primary analysis are reported in full detail elsewhere.17,18 Overall mortality rates as well as mortality rates due to completed suicide (using both narrow and broad definition) were comparable in both treatment groups.18
Analysis of Predictors of Completed or Attempted Suicide
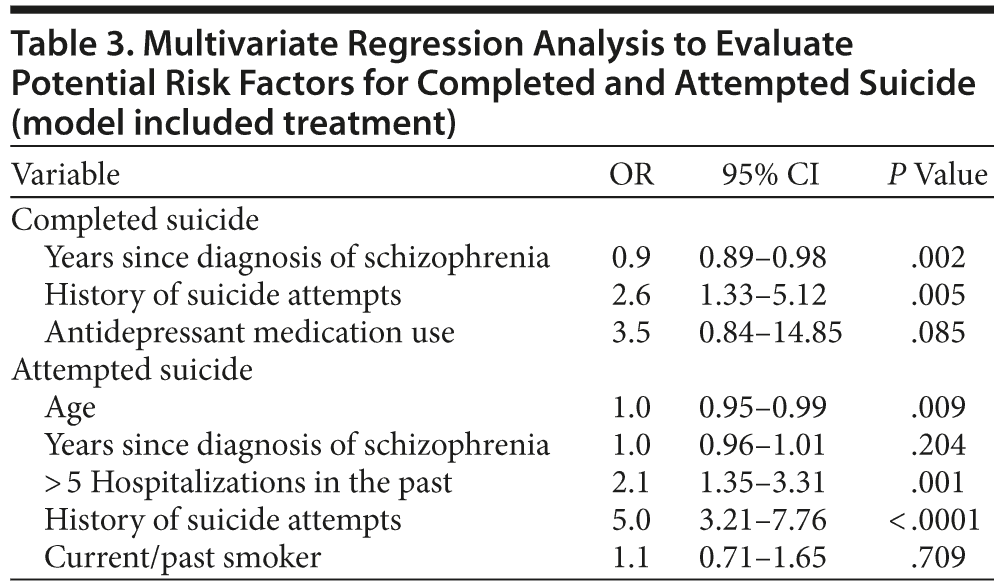
Among all potential baseline risk factors for completed suicide examined, the variables most associated with completed suicide were history of suicide attempts (OR = 2.6; 95% CI, 1.33−5.12) and usage of antidepressant medication (OR = 3.5; 95% CI, 0.84-14.85) (Table 3). History of > 5 hospitalizations in the past (OR = 2.1; 95% CI, 1.35-3.31) and history of suicide attempts (OR = 5.0; 95% CI, 3.21-7.76) were the variables most associated with attempted suicide among potential baseline risk factors for suicide attempts.
Methods Used in Completed or Attempted Suicides and Associated Gender Distribution
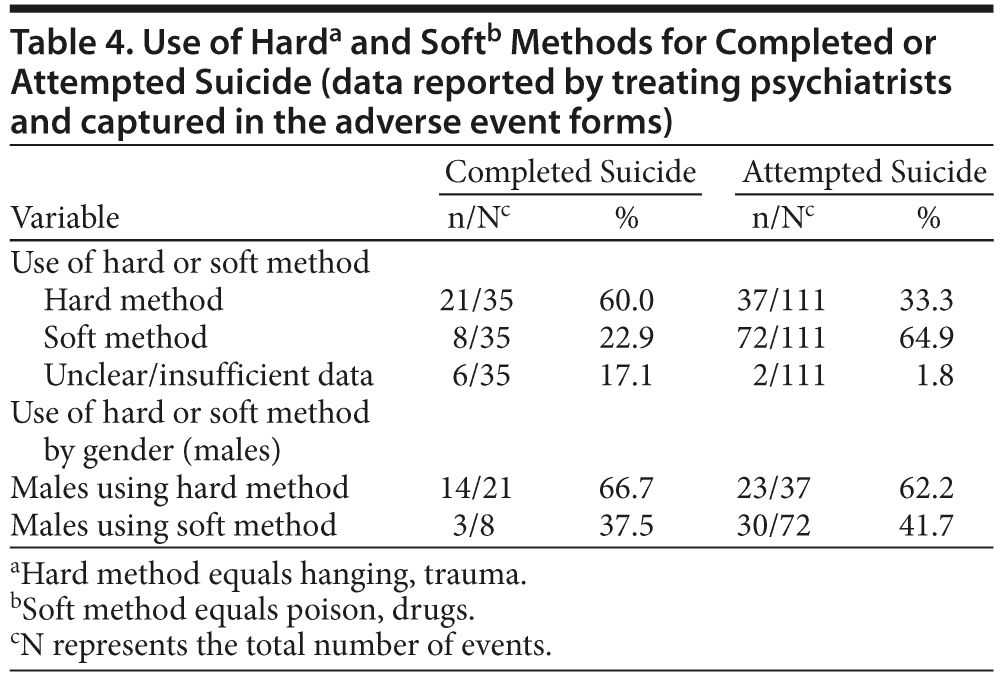
The use of hard methods was more frequent in completed suicides, as well as in male subjects who completed or attempted suicide, as seen by the descriptive summary of these data in Table 4. In contrast, soft methods were more frequent in attempted suicides and in female subjects who completed or attempted suicide (Table 4).
Person-Time on Assigned Treatment Analysis
The estimated rates of completed suicides per 100 person-years of exposure were highest in subjects diagnosed with schizophrenia for ≤ 3 years ago, ie, in subjects early in the illness course (Table 5).
The analyses performed on the 2 medication groups in the ZODIAC trial (ziprasidone, n = 9,077; olanzapine, n = 9,077) yielded similar results as the ones obtained in the overall analysis, without any between-treatment group differences (results not shown).
DISCUSSION
Previously published results of the ZODIAC trial primary analysis18 showed that, in this large simple trial that prospectively followed up > 18,000 subjects diagnosed with schizophrenia for 1 year, suicide-related mortality was low (0.19%, or 0.23% when a broadly defined end point was applied). The respective incidence for suicide attempts was 0.54%. In our person-time on assigned treatment analysis, rates of completed and attempted suicides calculated per 100 person-years of exposure remained low (0.24 and 0.74, respectively), with the highest suicide-related mortality among subjects recently diagnosed with schizophrenia (≤ 3 years ago), ie, in subjects early in their illness course. The highest rates of attempted suicides were seen in subjects diagnosed with schizophrenia > 3 and ≤ 5 years ago. We further expanded the primary study analyses by performing an exploratory logistic regression analysis, which showed that, in our cohort, history of suicide attempts, high CGI-S score at baseline, baseline presence of cardiovascular disorders, and the baseline use of antipsychotic or antidepressant medication were all associated with an increased risk for completed suicide (see Table 2) during 1 year from baseline. History of previous suicide attempts, > 5 hospitalizations in the past, and current/past smoking (see Table 3) were associated with an increased risk for suicide attempt. In our cohort, hard methods were more frequently used among completers and male subjects (both completers and attempters); soft methods were more frequent among attempters and female subjects (both completers and attempters).
Data from the ZODIAC study are similar to that reported in the Sertindole Cohort Prospective Study (SCoP), a prospective study focused on cardiac outcomes among 9,809 subjects with schizophrenia randomized to sertindole or risperidone within the clinical practice setting (0.21 and 0.28 per 100 person-years of exposure for sertindole and risperidone, respectively).22 However, the rates reported in the SCoP and ZODIAC trials were low when compared with pooled results22 of 5 studies commonly cited in the literature,3,23-26 yielding an average suicide mortality rate among persons with schizophrenia of 0.49 per 100 subject-years in a total population of 26,991 subjects. It is possible that patient characteristics (ie, more chronic disease, proportion of female participants) and factors associated with clinical study participation (ie, increased attention or social support) may have impacted the results. However, in ZODIAC, the proportion of female participants was similar to that in other studies in schizophrenia. Further, the trial designs of ZODIAC and SCoP mimicked usual clinical care after randomization, which may have minimized the effect of nonspecific trial participation-related factors. Timing of the studies also challenges comparisons between the 5 seminal trials,3,23-26 SCoP,22 and the ZODIAC trial.
Our results were obtained in > 18,000 subjects participating in the ZODIAC trial, which is the largest sample of subjects with schizophrenia to date in which completed or attempted suicides were prospectively studied. Our cohort was recruited internationally, representing several regions with diverse levels of development and psychiatric care. Furthermore, the analyses presented here are based on predefined secondary outcomes of the ZODIAC trial. In addition to the conventional method of registering serious adverse events and deaths throughout the study, we also applied an independent and blinded adjudication on a case-by-case basis to elucidate and verify each reported case of a potential completed or attempted suicide. The statistical analyses took into account the numbers of cases obtained through standard reporting of completed or attempted suicides as well as those obtained in the most conservative approach to adjudication. Nevertheless, the results obtained in either approach were similar.
Our findings are also validated by the fact that we could replicate a number of results described in earlier, smaller studies. Similar to other authors,5,27-31 we found the highest suicide rates among subjects early in their course of illness. History of prior suicide attempts, identified in the literature as a risk factor for completed suicide,27,32-36 was also replicated in our analysis. Presence of active psychotic or depressive symptoms at baseline has been identified as a risk factor for completed suicide in schizophrenia.6,7 However, as none of the symptom-specific psychiatric rating scales was employed at the baseline assessment in ZODIAC, it can only be speculated that baseline use of antidepressants identified in our study may represent a proxy for the presence of active depressive symptoms, while baseline use of antipsychotics may be a proxy for active psychotic symptoms. Lack of any baseline symptom-specific assessments in ZODIAC, therefore, limits any further conclusions.
Well recognized risk factors for suicide attempts such as female gender, history of suicide attempts, a large number of prior psychiatric hospitalizations, or heavy smoking have also been previously documented4,32,37-39 and replicated in our analysis. The relationships between hard methods, completed suicide, and male gender are well recognized in subjects with schizophrenia.40,41 An additional value of the ZODIAC trial is the fact that it is among a few trials that prospectively studied hard outcomes (in this case completed and attempted suicide) in a population with a high risk for such outcomes (ie, subjects with schizophrenia).42
There were several limitations to our study. The ZODIAC was a large simple trial, and, consequently, only limited clinically relevant information was gathered at a few time points during the 1-year follow-up. Subsequently, only a finite number of baseline variables could be included into the exploratory logistic regression analysis. As such, other clinically relevant factors associated with increased risk for a completed or attempted suicide (eg, concurrent substance use disorders or presence of active psychotic or depressive symptoms at baseline) were not evaluable. In addition, we did not systematically collect information on subjects’ medication status at the time of completed or attempted suicide. Thus, we could neither shed light on potential relationships that could have existed between the current medication and suicidality nor elucidate a potential protective effect ziprasidone or olanzapine may have against suicidality behavior. Therefore, the potential relationship between ongoing treatment with either ziprasidone or olanzapine and suicidality in subjects with schizophrenia needs further investigation in future studies. An analysis based on geographic region was not performed due to overrepresentation of subjects from some regions. While the ZODIAC employed naturalistic follow-up,17 randomization was used, which may have affected the generalizability of the study population. A low overall number of observed events (35 completed suicides and 108 suicide attempts) could have impacted the risk factor analyses and should be taken into account when results are interpreted. Further, the ratio between attempted versus completed suicides during the 1-year study period was 3:1, which may suggest possible underreporting of suicide attempts. However, as previously reported in a Pfizer-sponsored analysis of suicidality events within randomized clinical studies of ziprasidone, which were mostly of short-term (up to 6 weeks) duration, no completed suicides and only 2 suicide attempts were reported among 1,122 ziprasidone-treated subjects with schizophrenia.43 Furthermore, baseline usage of antidepressants reported in the ZODIAC was low: only 2 of the 35 completers and 2 of the 108 attempters were treated with antidepressants. This finding may be at odds with literature showing that history of past and present depressive disorders had a strong association with suicide in schizophrenia,44 while a depressive disorder was suggested to act as a trigger for suicidal behavior in vulnerable patients with schizophrenia.45 Our article focused on evaluating potential risk factors for completed and attempted suicides. Accordingly, a pool of potential risk factors was identified and analyzed using separate logistic regression models, and the resulting subset of risk factors was included in a multivariate model describing their relationship with the odds of completing or attempting suicide. The results of this statistical analysis are presented in Table 3, along with P values. With regard to the data presented in Tables 1, 2, 4, and 5, the intent was to provide only a descriptive summary. Formal inferential analysis of the data presented in these tables was not the focus of the analysis and, hence, out of the scope for this article.
In summary, our results, obtained in a large, prospective randomized study in subjects with schizophrenia, confirm and expand the knowledge about completed or attempted suicide in schizophrenia as well as the risk factors associated with such events.
Drug names: olanzapine (Zyprexa and others), risperidone (Risperdal and others), ziprasidone (Geodon and others).
Author affiliations: Department of Psychiatry and Psychotherapy, Medical University Innsbruck, Innsbruck, Austria (Dr Fleischhacker); Zucker Hillside Hospital, Glen Oaks (Dr Kane); Pfizer Inc, New York (Drs Geier, Karayal, Kolluri, Eng, and Reynolds), New York; and Department of Biostatistics and Epidemiology and Center for Clinical Epidemiology and Biostatistics, University of Pennsylvania School of Medicine, Philadelphia (Dr Strom).
Author contributions: All authors contributed substantially to the conception and design, or acquisition of data, or analysis and interpretation of the data. All authors were involved in drafting and revising the manuscript for important intellectual content, and all authors approved the final version to be published.
Potential conflicts of interest: Dr Fleischhacker has received research grants from Alkermes, Janssen, Otsuka, and Reckitt-Benckiser; has received consulting honoraria from Amgen, Bristol-Myers Squibb, Endo, Janssen, Lundbeck, MedAvante, Otsuka, Pfizer, Richter, Roche, Sunovion, and Vanda; has received speaker honoraria from AstraZeneca, Endo, Janssen, Lundbeck, Otsuka, Richter, and Roche; and owns MedAvante stocks. Dr Kane has served as a consultant to Pfizer in connection with the scientific oversight of the study and has served as a consultant and speaker to or has received honoraria from Abbott, Alkermes, Amgen, AstraZeneca, Bristol-Myers Squibb, Boehringer Ingelheim, Cephalon, Dainippon Sumitomo, Eisai, Eli Lilly, Forrest, Genetech, Intracellular Therapeutics, Janssen, Johnson & Johnson, Lundbeck, Merck, Myriad, Novartis, Otsuka, Pfizer, Proteus Biomedical, Roche, Rules Based Medicine, Sunovion, Takeda, Targacept, Vanda, and Wyeth; and is a MedAvante shareholder. Drs Geier, Karayal, Kolluri and Reynolds are employees and shareholders of Pfizer, the maker and manufacturer of ziprasidone, as was Dr Eng at the time the study was conducted. Dr Strom is now an employee of Rutgers University, and was an employee of the University of Pennsylvania, which received financial support from Pfizer in connection with the scientific oversight of the study and the development of this article. He also served as consultant to Pfizer on topics not related to this study. In the past 12 months, he has consulted for Amgen, Bristol-Myers Squibb, Endo, GlaxoSmithKline, Lundbeck, Novartis, Roche, and Sanofi on topics unrelated to this study.
Funding/support: This study was supported in full by funding from Pfizer, Inc, New York, New York.
Previous presentations: Partial results were previously presented at the International Congress on Schizophrenia Research (ICOSR); March 28-April 1, 2009; San Diego, California ▪ 163rd American Psychiatric Association Annual Meeting; May 22-26, 2010; New Orleans, Louisiana.
Acknowledgments: The authors acknowledge the efforts of all investigators and subjects participating in the ZODIAC; Vito Calamia, MS, Pfizer, Inc, New York, New York, for statistical analyses performed in connection with this study; Milana Zivkov, MD, consultant, Trogir, Croatia, in providing editorial support, revising the paper based on author feedback and styling the paper for journal submission; and Mark Olfson, MD, Columbia University, New York, New York, for critical review of the paper. Mr Calamia is an employee and shareholder of Pfizer, the maker and manufacturer of ziprasidone. Dr Zivkov was a paid consultant to Pfizer, Inc, New York, New York, in connection with the development of this article. Dr Olfson has no conflicts of interest to report.
REFERENCES
1. Carlborg A, Winnerbפck K, Jönsson EG, et al. Suicide in schizophrenia. Expert Rev Neurother. 2010;10(7):1153-1164. PubMed doi:10.1586/ern.10.82
2. Baxter D, Appleby L. Case register study of suicide risk in mental disorders. Br J Psychiatry. 1999;175(10):322-326. PubMed doi:10.1192/bjp.175.4.322
3. Osby U, Correia N, Brandt L, et al. Mortality and causes of death in schizophrenia in Stockholm county, Sweden. Schizophr Res. 2000;45(1-2):21-28. PubMed doi:10.1016/S0920-9964(99)00191-7
4. Barak Y, Baruch Y, Achiron A, et al. Suicide attempts of schizophrenia patients: a case-controlled study in tertiary care. J Psychiatr Res. 2008;42(10):822-826. PubMed doi:10.1016/j.jpsychires.2007.09.002
5. Heilפ H, Haukka J, Suvisaari J, et al. Mortality among patients with schizophrenia and reduced psychiatric hospital care. Psychol Med. 2005;35(5):725-732. PubMed doi:10.1017/S0033291704004118
6. Hor K, Taylor M. Suicide and schizophrenia: a systematic review of rates and risk factors. J Psychopharmacol. 2010;24(suppl):81-90. PubMed doi:10.1177/1359786810385490
7. Gómez-Durán EL, Martin-Fumadó C, Hurtado-Ru×z G. Clinical and epidemiological aspects of suicide in patients with schizophrenia. Actas Esp Psiquiatr. 2012;40(6):333-345. PubMed
8. Caldwell CB, Gottesman II. Schizophrenics kill themselves too: a review of risk factors for suicide. Schizophr Bull. 1990;16(4):571-589. PubMed doi:10.1093/schbul/16.4.571
9. Palmer BA, Pankratz VS, Bostwick JM. The lifetime risk of suicide in schizophrenia: a reexamination. Arch Gen Psychiatry. 2005;62(3):247-253. PubMed doi:10.1001/archpsyc.62.3.247
10. Saha S, Chant D, McGrath J. A systematic review of mortality in schizophrenia: is the differential mortality gap worsening over time? Arch Gen Psychiatry. 2007;64(10):1123-1131. PubMed doi:10.1001/archpsyc.64.10.1123
11. Planansky K, Johnston R. The occurrence and characteristics of suicidal preoccupation and acts in schizophrenia. Acta Psychiatr Scand. 1971;47(4):473-483. PubMed doi:10.1111/j.1600-0447.1971.tb03704.x
12. Landmark J, Cernovsky ZZ, Merskey H. Correlates of suicide attempts and ideation in schizophrenia. Br J Psychiatry. 1987;151:18-20. PubMed doi:10.1192/bjp.151.1.18
13. Drake RE, Gates C, Cotton PG. Suicide among schizophrenics: a comparison of attempters and completed suicides. Br J Psychiatry. 1986;149:784-787. PubMed doi:10.1192/bjp.149.6.784
14. Harris EC, Barraclough B. Excess mortality of mental disorder. Br J Psychiatry. 1998;173:11-53. PubMed doi:10.1192/bjp.173.1.11
15. Carlborg A, Jokinen J, Nordström AL, et al. Attempted suicide predicts suicide risk in schizophrenia spectrum psychosis. Nord J Psychiatry. 2010;64(1):68-72. PubMed doi:10.3109/08039480903274431
16. Harkavy-Friedman JM, Restifo K, Malaspina D, et al. Suicidal behavior in schizophrenia: characteristics of individuals who had and had not attempted suicide. Am J Psychiatry. 1999;156(8):1276-1278. PubMed
17. Strom BL, Faich GA, Reynolds RF, et al. The Ziprasidone Observational Study of Cardiac Outcomes (ZODIAC): design and baseline subject characteristics. J Clin Psychiatry. 2008;69(1):114-121. PubMed doi:10.4088/JCP.v69n0115
18. Strom BL, Eng SM, Faich G, et al. Comparative mortality associated with ziprasidone and olanzapine in real-world use among 18,154 patients with schizophrenia: the Ziprasidone Observational Study of Cardiac Outcomes (ZODIAC). Am J Psychiatry. 2011;168(2):193-201. PubMed doi:10.1176/appi.ajp.2010.08040484
19. March JS, Silva SG, Compton S, et al. The case for practical clinical trials in psychiatry. Am J Psychiatry. 2005;162(5):836-846. PubMed doi:10.1176/appi.ajp.162.5.836
20. Guy W. ECDEU Assessment Manual for Psychopharmacology. US Dept Health, Education, and Welfare publication (ADM) 76-338. Rockville, MD: National Institute of Mental Health; 1976:218-222.
21. CDC. National Death Index. http://www.cdc.gov/nchs/ndi.htm. Updated February 21, 2013. Accessed December 23, 2013.
22. Crocq MA, Naber D, Lader MH, et al. Suicide attempts in a prospective cohort of patients with schizophrenia treated with sertindole or risperidone. Eur Neuropsychopharmacol. 2010;20(12):829-838. PubMed doi:10.1016/j.euroneuro.2010.09.001
23. Newman SC, Bland RC. Mortality in a cohort of patients with schizophrenia: a record linkage study. Can J Psychiatry. 1991;36(4):239-245. PubMed
24. Allgulander C, Allebeck P, Przybeck TR, et al. Risk of suicide by psychiatric diagnosis in Stockholm County: a longitudinal study of 80,970 psychiatric inpatients. Eur Arch Psychiatry Clin Neurosci. 1992;241(5):323-326. PubMed doi:10.1007/BF02195984
25. Mortensen PB, Juel K. Mortality and causes of death in first admitted schizophrenic patients. Br J Psychiatry. 1993;163:183-189. PubMed doi:10.1192/bjp.163.2.183
26. Sernyak MJ, Desai R, Stolar M, et al. Impact of clozapine on completed suicide. Am J Psychiatry. 2001;158(6):931-937. PubMed doi:10.1176/appi.ajp.158.6.931
27. Besnier N, Gavaudan G, Navez A, et al. Clinical features of suicide occurring in schizophrenia, 1: risk-factors identification [in French]. Encephale. 2009;35(2):176-181. PubMed doi:10.1016/j.encep.2008.02.009
28. Dutta R, Murray RM, Hotopf M, et al. Reassessing the long-term risk of suicide after a first episode of psychosis. Arch Gen Psychiatry. 2010;67(12):1230-1237. PubMed doi:10.1001/archgenpsychiatry.2010.157
29. Osborn D, Levy G, Nazareth I, et al. Suicide and severe mental illnesses: cohort study within the UK general practice research database. Schizophr Res. 2008;99(1-3):134-138. PubMed doi:10.1016/j.schres.2007.11.025
30. Thong JY, Su AH, Chan YH, et al. Suicide in psychiatric patients: case-control study in Singapore. Aust N Z J Psychiatry. 2008;42(6):509-519. PubMed doi:10.1080/00048670802050553
31. Melle I, Johannessen JO, Friis S, et al. Course and predictors of suicidality over the first two years of treatment in first-episode schizophrenia spectrum psychosis. Arch Suicide Res. 2010;14(2):158-170. PubMed doi:10.1080/13811111003704787
32. Carlborg A, Jokinen J, Jönsson EG, et al. Long-term suicide risk in schizophrenia spectrum psychoses: survival analysis by gender. Arch Suicide Res. 2008;12(4):347-351. PubMed doi:10.1080/13811110802325133
33. Kasckow J, Liu N, Haas GL, et al. Case-control study of the relationship of depressive symptoms to suicide in a community-based sample of individuals with schizophrenia in China. Schizophr Res. 2010;122(1-3):226-231. PubMed doi:10.1016/j.schres.2010.02.1056
34. Neuner T, Hübner-Liebermann B, Haen E, et al; AGATE. Completed suicides in 47 psychiatric hospitals in Germany—results from the AGATE-study. Pharmacopsychiatry. 2011;44(7):324-330. PubMed doi:10.1055/s-0031-1284428
35. Pompili M, Lester D, Grispini A, et al. Completed suicide in schizophrenia: evidence from a case-control study. Psychiatry Res. 2009;167(3):251-257. PubMed doi:10.1016/j.psychres.2008.03.018
36. Reutfors J, Brandt L, Jönsson EG, et al. Risk factors for suicide in schizophrenia: findings from a Swedish population-based case-control study. Schizophr Res. 2009;108(1-3):231-237. PubMed doi:10.1016/j.schres.2008.12.023
37. Montross LP, Kasckow J, Golshan S, et al. Suicidal ideation and suicide attempts among middle-aged and older patients with schizophrenia spectrum disorders and concurrent subsyndromal depression. J Nerv Ment Dis. 2008;196(12):884-890. PubMed doi:10.1097/NMD.0b013e31818ec823
38. Uzun O, Tamam L, Ozcüler T, et al. Specific characteristics of suicide attempts in patients with schizophrenia in Turkey. Isr J Psychiatry Relat Sci. 2009;46(3):189-194. PubMed
39. Zhang XY, Al Jurdi RK, Zoghbi AW, et al. Prevalence, demographic and clinical correlates of suicide attempts in Chinese medicated chronic inpatients with schizophrenia. J Psychiatr Res. 2013;47(10):1370-1375. doi:10.1016/j.jpsychires.2013.05.024 PubMed
40. Brecić P, Ostojić D, Vidović D, et al. Characteristics of patients who committed suicide during hospitalization in Psychiatric Hospital “Vrapce” in the period 1996-2006. Coll Antropol. 2009;33(1):233-236. PubMed
41. Chen YY, Lee MB, Chang CM, et al. Methods of suicide in different psychiatric diagnostic groups. J Affect Disord. 2009;118(1-3):196-200. PubMed doi:10.1016/j.jad.2009.05.020
42. Perlis RH. Hard outcomes: clinical trials to reduce suicide. Am J Psychiatry. 2011;168(10):1009-1011. PubMed doi:10.1176/appi.ajp.2011.11081250
43. Karayal ON, Anway SD, Batzar E, et al. Assessments of suicidality in double-blind, placebo-controlled trials of ziprasidone. J Clin Psychiatry. 2011;72(3):367-375. PubMed doi:10.4088/JCP.10m06281gre
44. Harkavy-Friedman JM, Nelson EA, Venarde DF, et al. Suicidal behavior in schizophrenia and schizoaffective disorder: examining the role of depression. Suicide Life Threat Behav. 2004;34(1):66-76. PubMed doi:10.1521/suli.34.1.66.27770
45. Hawton K, Sutton L, Haw C, et al. Schizophrenia and suicide: systematic review of risk factors. Br J Psychiatry. 2005;187(7):9-20. PubMed doi:10.1192/bjp.187.1.9
This PDF is free for all visitors!