Objective: This study was designed to evaluate the efficacy and safety of reduced B vitamins as monotherapy in adults with major depressive disorder (MDD) who were also positive for at least 1 methylenetetrahydrofolate reductase (MTHFR) polymorphism associated with depression and further test the hypothesis that reduced (metabolized) B vitamins will lower homocysteine in a majority of clinically responding patients.
Methods: 330 adult patients with MDD (DSM-5) and positive for either MTHFR C677T or A1298C polymorphism were enrolled in a trial conducted between August 1, 2014, and April 3, 2015. 160 patients received placebo, while 170 received a capsule containing a combination of reduced B vitamins. Plasma homocysteine levels were measured at baseline and week 8. The Montgomery-Asberg Depression Rating Scale (MADRS) was used to evaluate efficacy for MDD.
Results: 159 of 170 vitamin-treated patients and 123 of 160 placebo-treated patients were completers. Of the active treatment group, 131 (82.4%) showed a reduction in homocysteine (for a mean in this subgroup of 25%, P < .001), while 28 (17.6%) showed no significant change. Placebo patients demonstrated a small elevation in homocysteine. Active-treatment patients demonstrated, on average, a 12-point reduction on the MADRS by week 8, and 42% achieved full remission (P < .001). No side effect was significantly different between groups. No patients experienced mania.
Conclusions: A combination of reduced B vitamins and micronutrients, when used in the treatment of MDD in patients with MTHFR polymorphism, resulted in a separation from placebo by week 2, and 42% of the treatment arm achieved remission by week 8. Further, clinical improvement correlated with a significant reduction in homocysteine levels in a majority of responders. These results support the homocysteine theory of depression and the safety and therapeutic benefit of reduced B vitamins as monotherapy for MDD, particularly in patients with MTHFR polymorphism.
Trial registration: ClinicalTrials.gov identifier: NCT02709668‘ ‹’ ‹
This work may not be copied, distributed, displayed, published, reproduced, transmitted, modified, posted, sold, licensed, or used for commercial purposes. By downloading this file, you are agreeing to the publisher’s Terms & Conditions.
Correlation of Clinical Response With Homocysteine Reduction During Therapy With Reduced B Vitamins in Patients With MDD Who Are Positive for MTHFR C677T or A1298C Polymorphism:
A Randomized, Double-Blind, Placebo-Controlled Study

ABSTRACT
Objective: This study was designed to evaluate the efficacy and safety of reduced B vitamins as monotherapy in adults with major depressive disorder (MDD) who were also positive for at least 1 methylenetetrahydrofolate reductase (MTHFR) polymorphism associated with depression and further test the hypothesis that reduced (metabolized) B vitamins will lower homocysteine in a majority of clinically responding patients.
Methods: 330 adult patients with MDD (DSM-5) and positive for either MTHFR C677T or A1298C polymorphism were enrolled in a trial conducted between August 1, 2014, and April 3, 2015. 160 patients received placebo, while 170 received a capsule containing a combination of reduced B vitamins. Plasma homocysteine levels were measured at baseline and week 8. The Montgomery-Asberg Depression Rating Scale (MADRS) was used to evaluate efficacy for MDD.
Results: 159 of 170 vitamin-treated patients and 123 of 160 placebo-treated patients were completers. Of the active treatment group, 131 (82.4%) showed a reduction in homocysteine (for a mean in this subgroup of 25%, P < .001), while 28 (17.6%) showed no significant change. Placebo patients demonstrated a small elevation in homocysteine. Active-treatment patients demonstrated, on average, a 12-point reduction on the MADRS by week 8, and 42% achieved full remission (P < .001). No side effect was significantly different between groups. No patients experienced mania.
Conclusions: A combination of reduced B vitamins and micronutrients, when used in the treatment of MDD in patients with MTHFR polymorphism, resulted in a separation from placebo by week 2, and 42% of the treatment arm achieved remission by week 8. Further, clinical improvement correlated with a significant reduction in homocysteine levels in a majority of responders. These results support the homocysteine theory of depression and the safety and therapeutic benefit of reduced B vitamins as monotherapy for MDD, particularly in patients with MTHFR polymorphism.
Trial registration: ClinicalTrials.gov identifier: NCT02709668
J Clin Psychiatry 2016;77(5):668-671
dx.doi.org/10.4088/JCP.15m10166
© Copyright 2016 Physicians Postgraduate Press, Inc.
aThe Mech Center: Outpatient and Research Clinic, Plano, Texas
bHigh Point Regional Hospital—UNC Health Care, High Point, North Carolina
*Corresponding author: Andrew Farah, MD, 320 Boulevard, High Point, NC 27262 ([email protected]).
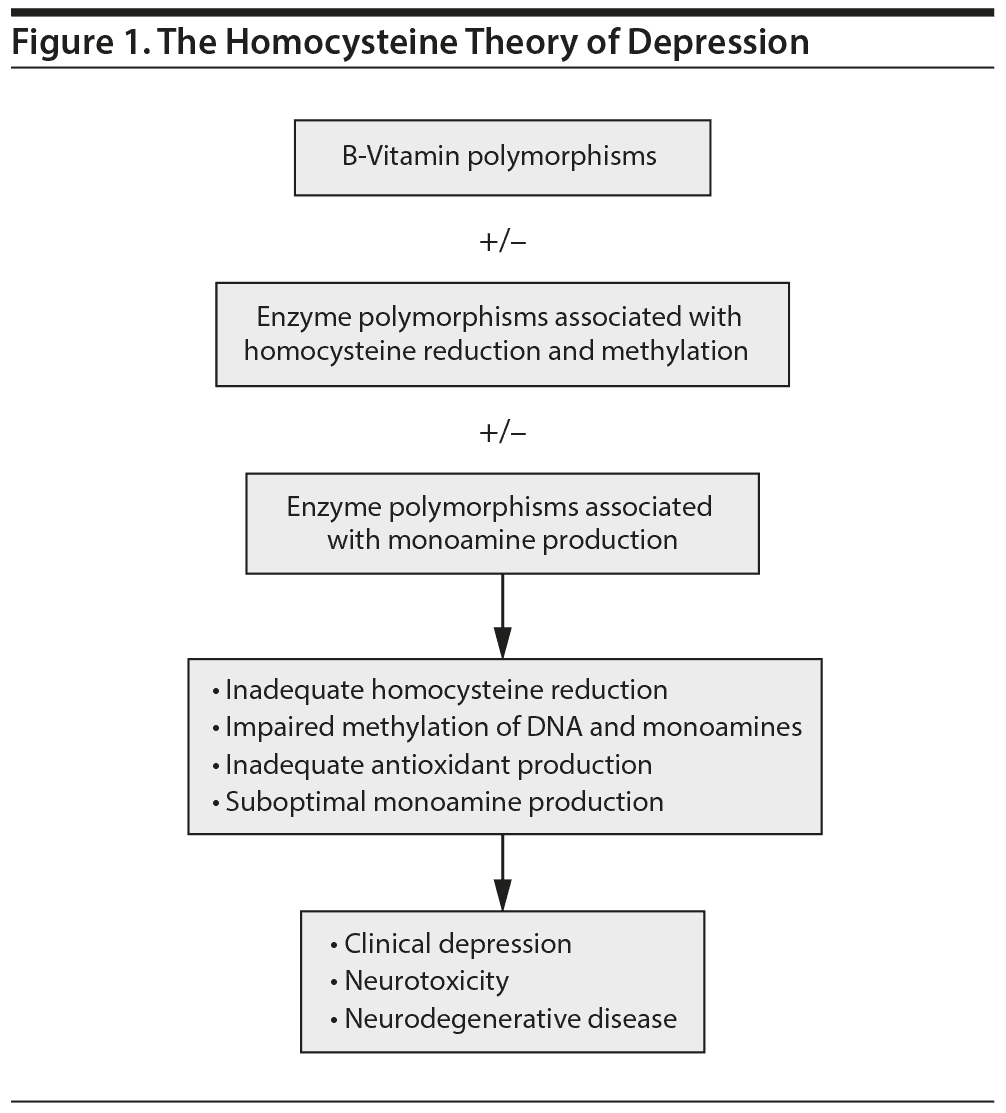
Homocysteine metabolism, or the carbon-1 cycle, plays a key role in the synthesis of monoamines by providing methyl group donation for the production of norepinephrine and dopamine. Reduced, or metabolized, B vitamins are necessary coenzymes in the carbon-1 cycle and in various other enzymatic steps involved in monoamine synthesis, such as the generation of tetrahydrobiopterin, a cofactor for both tryptophan hydroxylase and tyrosine hydroxylase. In general, B vitamins enter the body as prodrugs and must be metabolized to their active "coenzyme" forms. Impaired B-vitamin metabolism will result in a deficiency of coenzymes, a subsequent rise in homocysteine, and less than optimal monoamine production.1-3
Various studies have correlated impaired B-vitamin metabolism with elevated homocysteine levels and a heightened risk of depressive disorders.4-8 Mutations, or even seemingly minor variations in the genes coding for enzymes necessary for B-vitamin metabolism, can lead to inadequate coenzyme production and lower than optimal levels of serotonin, norepinephrine, and dopamine.3 The most common of these polymorphisms are the methylenetetrahydrofolate reductase (MTHFR) variants, of which there are at least 40, although they often coexist in the presence of other less studied polymorphisms involved in metabolizing other B vitamins, such as B12 and B6.
In summary, the homocysteine theory of depression argues that these genetic variants result in a deficiency of metabolized B vitamins, and thus, inadequate coenzymes for homocysteine reduction, elevated homocysteine levels, and suboptimal monoamine production, which can manifest clinically as depression8 (Figure 1). The earliest elaborations of homocysteine’s role in depression also listed inflammatory mechanisms and vascular damage as contributory. Further, new evidence has also supported this genetic predisposition as a contributor to depression, as MTHFR polymorphisms are prevalent in those who have depressive disorders after childhood trauma at a higher rate than in those who experience similar traumas and do not suffer depression.9
B vitamins in various forms have been used as monotherapy and adjunctive therapy in major depressive disorder (MDD) since the 1960s.10-12 Yet, due to the variety of preparations utilized, the diversity of study populations and methodologies, and the lack of clarification regarding the exact role of B vitamins in treatment (as adjunctive therapy or monotherapy), no consensus exists regarding current treatment recommendations. Further, it is only in the last decade that the therapeutic emphasis has been on ingesting metabolized B vitamins, as the issue in the vast majority of depressed patients is not dietary intake, but their inability to optimally metabolize dietary or supplemental vitamins that is problematic. Thus, it is possible that many prior studies were unsuccessful because patients were given high doses of vitamins that, for genetic reasons, they simply could not metabolize effectively to the active coenzymes needed. This historical perspective cast doubt on the therapeutic role of B vitamins and raised legitimate safety concerns regarding administering ineffective, high doses of vitamins.13,14
Our study utilized a capsule containing a combination of all metabolized vitamins and micronutrients necessary for homocysteine reduction and monoamine production, and this formulation is regulated by the US Food and Drug Administration with the United States Pharmacopeia and indicated for folate deficiencies associated with elevated homocysteine in the central nervous system (CNS).15 We also utilized MTHFR polymorphism as evidence of impaired folate metabolism, and further, as a marker for the likely presence of other B-vitamin polymorphisms in MDD patients. Thus, utilizing a combination of B vitamins in reduced form addressed all possible polymorphisms contributing to inadequate monoamine production.

- Since the synthesis of the first tricyclic antidepressant in the late 1950s, medications for depression have primarily blocked the reuptake of monoamines rather than address the inability to produce adequate monoamines, possibly the genetic basis of depression.
- The majority of depressed individuals have genetic polymorphisms that convey less optimal vitamin metabolism. Since metabolized B vitamins act as coenzymes in critical steps of monoamine synthesis and methylation, reduced B vitamins may be used as antidepressant therapy.
- Our study demonstrated significant efficacy of reduced B vitamin monotherapy in patients who had known MTHFR polymorphisms associated with impaired folate metabolism.
Further, the study was also designed to test the homocysteine theory of depression, ie, circumventing the patient’s genetic inability to metabolize B vitamins by administering the metabolized forms in an optimal preparation for CNS availability would lower homocysteine, increase monoamine production, and facilitate clinical improvement.
METHODS
Patients
Patients were recruited using print advertisements and referrals from other professionals in the Dallas, Texas, area. The trial was conducted between August 1, 2014, and April 3, 2015, and is registered at ClinicalTrials.gov (NCT02709668). Ages ranged from 18 to 67 years, with a mean age of 32 years. The study population comprised 42% male and 58% female patients. All patients met the DSM-5 criteria for MDD without psychotic features, and those with comorbid attention-deficit/hyperactivity disorder or generalized anxiety disorder were not excluded. However, exclusion criteria included active substance abuse or dependence, dementia, current psychotic symptoms, suicidality requiring hospital care, and bipolar disorder. Past treatment failures or lack of response to current therapy did not exclude patients. All patients consented to the study voluntarily and understood the possibility of receiving a placebo agent. All patients were instructed to phone immediately if manic symptoms, suicidal intent, or new-onset suicidal thoughts occurred. The study was approved through our local institutional review board.
Study Design
All clinic and referred patients were screened for MTHFR polymorphisms, and 330 who were positive for one of the two C677T polymorphisms or one of the A1298C polymorphisms associated with MDD chose to participate in the trial. Patients were randomly assigned to treatment group. Baseline homocysteine levels were drawn and repeated at 8 weeks with either reduced B vitamins or placebo. The Montgomery-Asberg Depression Rating Scale (MADRS) was administered at baseline, week 2, and week 8.16 Patients who were taking ineffective antidepressants were tapered off to prevent withdrawal symptoms and were drug-free for 2 weeks before starting the study. The active treatment group received EnLyte, a gelatin capsule provided by Jaymac Pharmaceuticals, which also provided an identical placebo gelatin capsule.
The active ingredients in the gelatin capsule dispensed to the treatment group included 3 forms of B9 (1 mg of citrated folic acid, 2.5 mg of folinic acid, and 7 mg of l-methylfolate magnesium), 25 μg of thiamine pyrophosphate, 25 μg of flavin adenine dinucleotide, 25 μg of pyridoxal 5′ -phosphate, 50 μg of adenosylcobalamin, 25 μg of thiamine pyrophospahate, 25 μg of nicotinamide adenine dinucleotide (NADH), 500 μg of trimethyl glycine, 1.5 mg of AminoFerr (Viva Pharmaceutical), 24 mg of magnesium ascorbate, 1 mg of zinc ascorbate, 1 mg of l-threonic acid magnesium, and 20 mg of Sharp PS Gold (phosphatidylserine-omega-3 conjugated) (Enzymotec Ltd).
Patients could withdraw at any time. Of the 170 vitamin therapy patients, 159 patients completed the 8-week trial versus 123 of the 160 placebo-assigned patients. Of the active treatment patients who withdrew from the study, none did so because of side effects, but most commonly due to a geographic move or for nonspecific reasons. The main reasons for placebo dropout were lack of improvement and patient request for a nonstudy, active agent to hasten recovery.
RESULTS
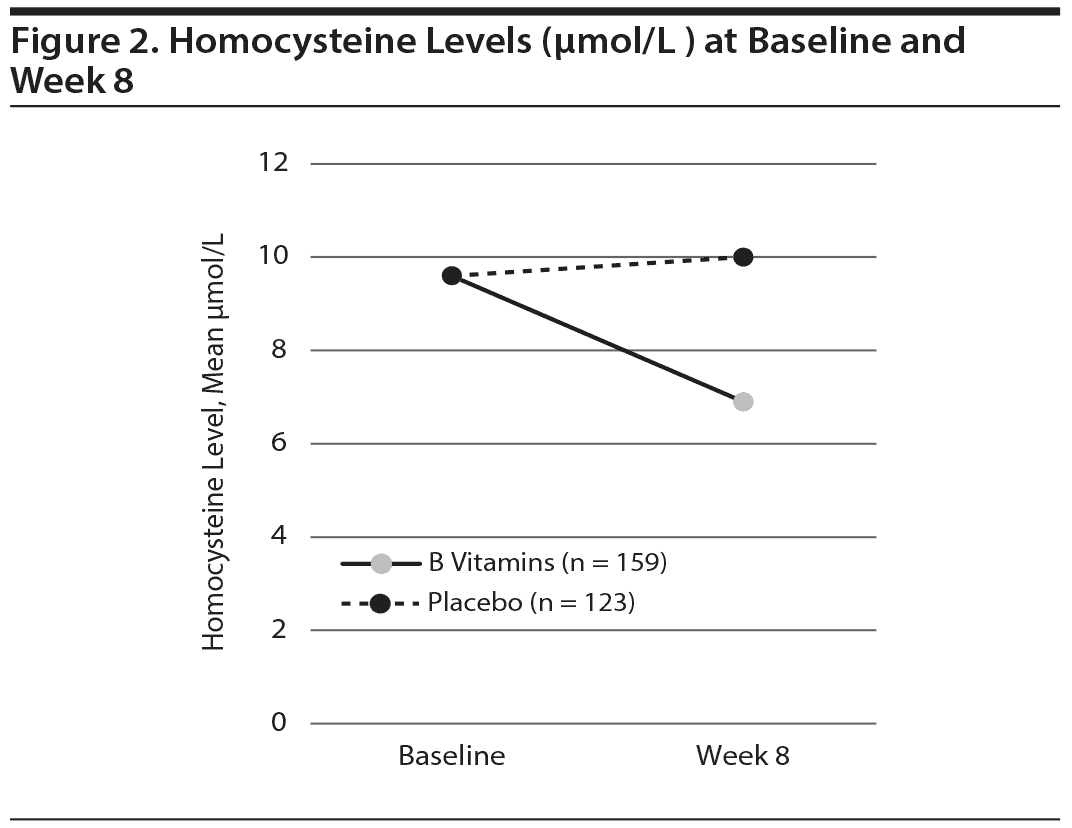
Of the 159 active-treatment patients who completed the 8-week trial, 131 (82.4%) demonstrated a reduction in homocysteine levels (from 9.6 at baseline to 7.2 µmol/L at week 8 on average, P < .001, Cohen d effect size of -0.88). For 28 active-treatment patients (17.6%), no statistically significant change in homocysteine was noted. The 123 placebo-assigned patients who completed the study demonstrated a slight increase in homocysteine from baseline, rising on average 0.4 µmol/L (Figure 2). The mean homocysteine reduction for the 159 active-treatment patients was 19.9%.
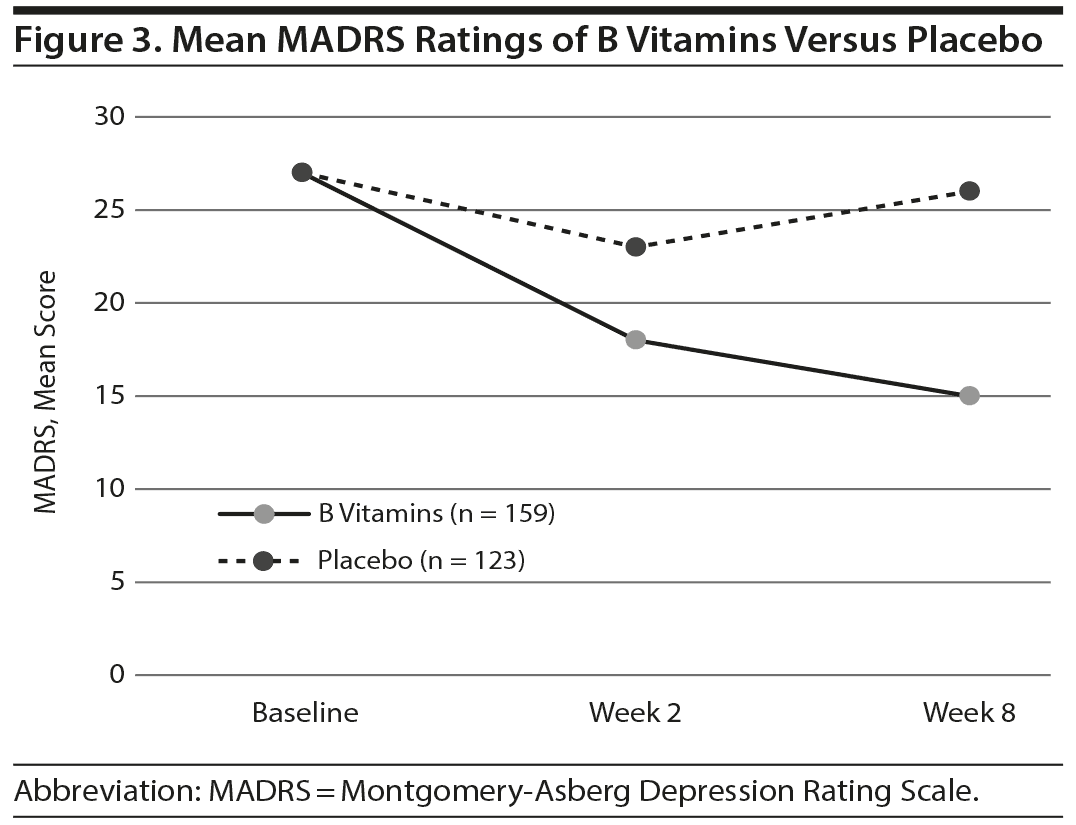
The entry MADRS score was, on average, 27 for participants. In the active treatment group, separation from placebo was seen by week 2 (P < .001), and by week 8, the average drop in MADRS score in the treatment group was 12 points (Figure 3). Further, 67 of the 159 vitamin therapy completers, or 42%, had achieved full remission (MADRS < 7) by week 8 (P < .001). The placebo group displayed on average a 1.3-point drop in MADRS score (Cohen d effect size of −0.81).
Although some patients reported thoughts of death or suicidal ideation prior to entry, no patient in the active treatment group experienced an increase in, or the new onset of, suicidal ideation. Rates of nausea, headache, anxiety, and tremor were all less than 5% in both groups, and no side effect occurred at a rate that was statistically different than placebo. No patient in either group experienced mania, hypomania, or psychosis.
DISCUSSION
Homocysteine is considered elevated if greater than 15 µmol/L, and a principal determinant of homocysteine levels is one’s MTHFR status. The homocysteine theory of depression argues that inadequate monoamine production results from a genetic inability to optimally metabolize homocysteine. Homocysteine metabolism in the CNS can occur via 2 pathways, the first using methyl-B12 and l-methylfolate as coenzymes to allow for the production of methyl donors needed in the CNS, including for monoamine production, and the second, predominately in glial cells, utilizing reduced B6 and resulting in antioxidant production (glutathione).
B vitamins enter the body as prodrugs and must be metabolized to their active forms through enzymatic steps. Due to genetic variants, or polymorphisms in the enzymes responsible for metabolizing B vitamins to their coenzyme forms, many less functional enzymes are possible in our patients. Thus, homocysteine is less optimally reduced, its levels increase, and lower levels of monoamines result. Further, B1, B2, B3, and mineral cofactors are essential in various other steps needed for monoamine synthesis.
Recently, the gelatin capsule EnLyte was created specifically to address homocysteine metabolism. This capsule contains all B vitamins in their most reduced, ie, fully metabolized, forms and all micronutrients associated with optimal homocysteine metabolism, theoretically allowing for adequate monoamine synthesis.15 Although indicated and marketed for methylfolate deficiency in depression, EnLyte also has theoretical utility for neuroprotection.
Although MTHFR polymorphism is the most studied genetic variant resulting in suboptimal vitamin metabolism, a common finding in many patients with depression,17 patients with MDD can present with multiple polymorphisms. A study of nondepressed individuals found that the regulation of homocysteine was, in fact, polygenic for over 50% of the study population.18 The possibility of multiple polymorphisms argues for utilizing a cluster of reduced B vitamins, rather than single vitamins, for clinical benefit.
Our study demonstrated that for individuals with known MTHFR polymorphisms associated with depression, addressing all other possible polymorphisms that may result in inadequate homocysteine metabolism with reduced B vitamins was superior to placebo in lowering homocysteine levels and further resulted in a statistically significant clinical response for the majority of patients and complete remission for 42% of the active treatment group. Patients receiving placebo experienced a slight elevation in plasma homocysteine. We assume that the untreated depression resulted in further and prolonged psychosocial stress, a known cause of homocysteine elevation. No side effect was reported at a greater rate than that in placebo, no patients converted to mania, and no patients reported new-onset suicidal thinking on either reduced vitamin or placebo therapy.
The limitations of our study include the lack of an active comparator agent, such as a selective serotonin reuptake inhibitor or serotonin-norepinephrine reuptake inhibitor. Because it is not practical to measure intracellular homocysteine levels in the CNS, we relied on peripheral homocysteine measures. Patients also understood that they were possibly receiving a natural compound, which may have conveyed a sense of safety not associated with typical active drug versus placebo studies and may have allowed for a minimization of side effect reporting. Further, the appeal of a natural therapy may have conveyed a transference to the agent itself that perhaps enhanced response. Finally, because all patients tested positive for at least 1 MTHFR polymorphism, which confers defective folate metabolism, and because there is pathological synergy associated with the dual presence of C677T and A1298C, many of our patients would be expected to respond robustly to the compound containing 3 forms of highly bioavailable folate. Therefore, we cannot extrapolate these findings to individuals who test for normal variants of C677T and A1298C.
In conclusion, our findings help to confirm the homocysteine basis of MDD. MTHFR polymorphisms have long been established as a risk factor for depression, but they may also be considered markers for the presence of coexisting polymorphisms associated with homocysteine metabolism and suboptimal monoamine production. Addressing the homocysteine theory of depression clinically by circumventing all possible genetic polymorphisms associated with elevated homocysteine levels is, in effect, possibly addressing one of the root causes of depression.
Because higher than normal homocysteine levels are associated with many neurodegenerative disorders and congenital conditions, neuroprotection and prenatal implications of reduced B vitamins are obvious areas of future study.
Submitted: June 12, 2015; accepted December 21, 2015.
Online first: March 29, 2016.
Potential conflicts of interest: Dr Mech has received research support and honoraria from Jaymac Pharmaceuticals. Dr Farah is a consultant for Jaymac Pharmaceuticals.
Funding/support: Jaymac Pharmaceuticals of Sunset, Louisiana, provided the study vitamins (EnLyte capsules) and placebo gelatin capsules and provided payment to The Mech Center for this research.
Role of the sponsor: The funding agency had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; or preparation, review, and approval of the manuscript.
Previous presentation: This study was the topic of a poster presentation at the 2015 NEI Psychopharmacology Congress meeting; November 12-15, 2015; Orlando, Florida.
REFERENCES
1. Bottiglieri T, Laundy M, Crellin R, et al. Homocysteine, folate, methylation, and monoamine metabolism in depression. J Neurol Neurosurg Psychiatry. 2000;69(2):228-232. PubMed doi:10.1136/jnnp.69.2.228
2. Fava M, Borus JS, Alpert JE, et al. Folate, vitamin B12, and homocysteine in major depressive disorder. Am J Psychiatry. 1997;154(3):426-428. PubMed doi:10.1176/ajp.154.3.426
3. Mitchell ES, Conus N, Kaput J. B vitamin polymorphisms and behavior: evidence of associations with neurodevelopment, depression, schizophrenia, bipolar disorder and cognitive decline. Neurosci Biobehav Rev. 2014;47:307-320. PubMed doi:10.1016/j.neubiorev.2014.08.006
4. Carney MW, Chary TK, Laundy M, et al. Red cell folate concentrations in psychiatric patients. J Affect Disord. 1990;19(3):207-213. PubMed doi:10.1016/0165-0327(90)90093-N
5. Penninx BW, Guralnik JM, Ferrucci L, et al. Vitamin B(12) deficiency and depression in physically disabled older women: epidemiologic evidence from the Women’s Health and Aging Study. Am J Psychiatry. 2000;157(5):715-721. PubMed doi:10.1176/appi.ajp.157.5.715
6. Ebly EM, Schaefer JP, Campbell NR, et al. Folate status, vascular disease and cognition in elderly Canadians. Age Ageing. 1998;27(4):485-491. PubMed doi:10.1093/ageing/27.4.485
7. Tiemeier H, van Tuijl HR, Hofman A, et al. Vitamin B12, folate, and homocysteine in depression: the Rotterdam Study. Am J Psychiatry. 2002;159(12):2099-2101. PubMed doi:10.1176/appi.ajp.159.12.2099
8. Folstein M, Liu T, Peter I, et al. The homocysteine hypothesis of depression. Am J Psychiatry. 2007;164(6):861-867. PubMed doi:10.1176/ajp.2007.164.6.861
9. Lok A, Bockting CL, Koeter MW, et al. Interaction between the MTHFR C677T polymorphism and traumatic childhood events predicts depression. Transl Psychiatr. 2013;3(7):e288. PubMed doi:10.1038/tp.2013.60
10. Carney MWP, Sheffield BF. Associations of subnormal serum folate and vitamin B12 values and effects of replacement therapy. J Nerv Ment Dis. 1970;150(5):404-412. PubMed doi:10.1097/00005053-197005000-00009
11. Reynolds EH, Preece JM, Bailey J, et al. Folate deficiency in depressive illness. Br J Psychiatry. 1970;117(538):287-292. PubMed
12. Godfrey PS, Toone BK, Carney MW, et al. Enhancement of recovery from psychiatric illness by methylfolate. Lancet. 1990;336(8712):392-395. PubMed doi:10.1016/0140-6736(90)91942-4
13. Troen AM, Mitchell B, Sorensen B, et al. Unmetabolized folic acid in plasma is associated with reduced natural killer cell cytotoxicity among postmenopausal women. J Nutr. 2006;136(1):189-194. PubMed
14. Reynolds EH. Benefits and risks of folic acid to the nervous system. J Neurol Neurosurg Psychiatry. 2002;72(5):567-571. PubMed doi:10.1136/jnnp.72.5.567
15.EnLyte [package insert]. Sunset, LA: Jaymac Pharmaceuticals; 2014.
16. Montgomery SA, Asberg M. A new depression scale designed to be sensitive to change. Br J Psychiatry. 1979;134(4):382-389. PubMed doi:10.1192/bjp.134.4.382
17. Gilbody S, Lewis S, Lightfoot T. Methylenetetrahydrofolate reductase (MTHFR) genetic polymorphisms and psychiatric disorders: a HuGE review. Am J Epidemiol. 2007;165(1):1-13. PubMed doi:10.1093/aje/kwj347
18. Tsai MY, Bignell M, Yang F, et al. Polygenic influence on plasma homocysteine: association of two prevalent mutations, the 844ins68 of cystathionine β-synthase and A(2756)G of methionine synthase, with lowered plasma homocysteine levels. Atherosclerosis. 2000;149(1):131-137. PubMed doi:10.1016/S0021-9150(99)00297-X
This PDF is free for all visitors!