This work may not be copied, distributed, displayed, published, reproduced, transmitted, modified, posted, sold, licensed, or used for commercial purposes. By downloading this file, you are agreeing to the publisher’s Terms & Conditions.
Objective: Transcranial magnetic stimulation (TMS) is an effective and safe acute treatment for patients not benefiting from antidepressant pharmacotherapy. Few studies have examined its longer term durability. This study assessed the long-term effectiveness of TMS in naturalistic clinical practice settings following acute treatment.
Method: Adult patients with a primary diagnosis of unipolar, nonpsychotic major depressive disorder (DSM-IV clinical criteria), who did not benefit from antidepressant medication, received TMS treatment in 42 clinical practices. Two hundred fifty-seven patients completed a course of acute TMS treatment and consented to follow-up over 52 weeks. Assessments were obtained at 3, 6, 9, and 12 months. The study was conducted between March 2010 and August 2012.
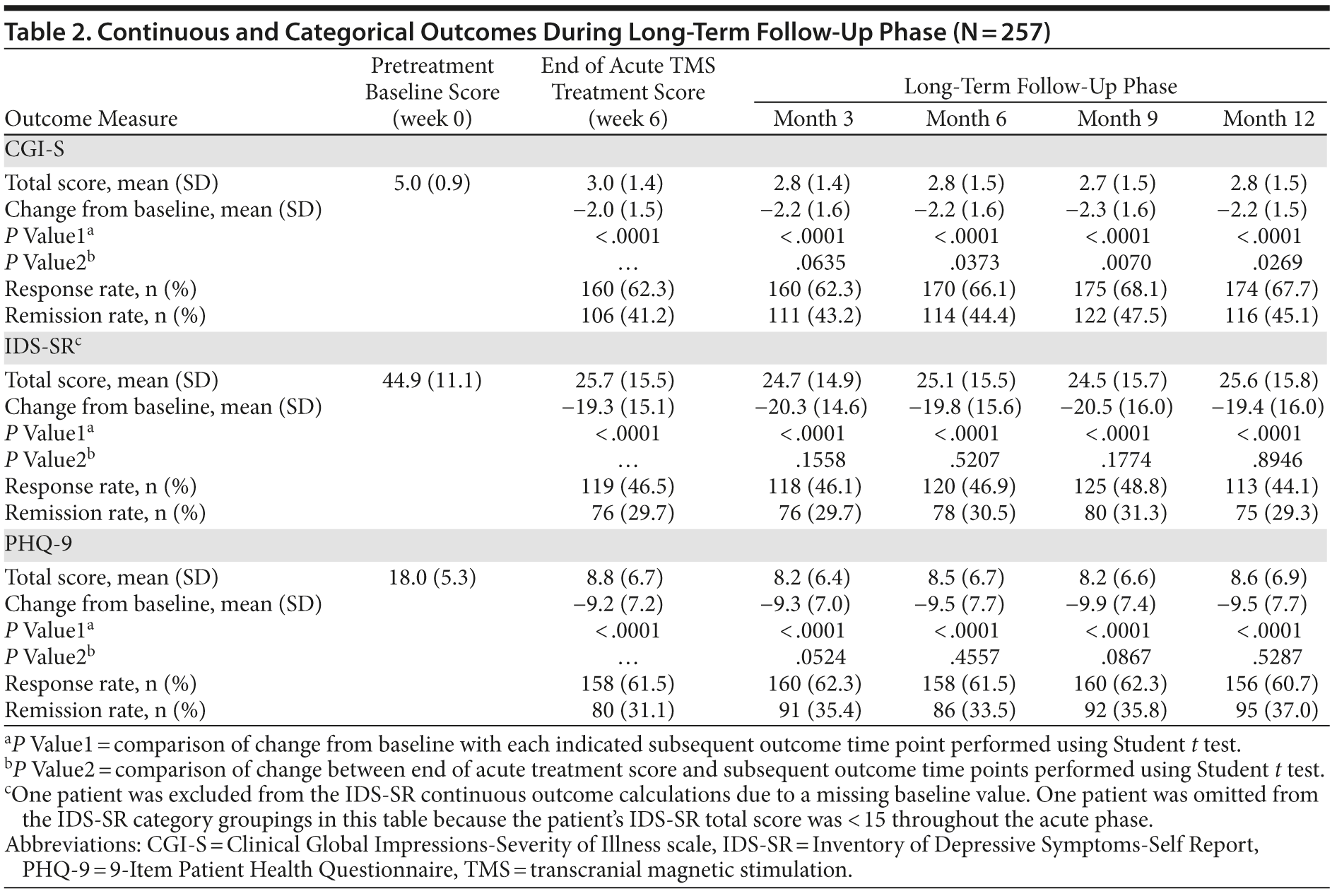
Results: Compared with pre-TMS baseline, there was a statistically significant reduction in mean total scores on the Clinical Global Impressions-Severity of Illness scale (primary outcome), 9-Item Patient Health Questionnaire, and Inventory of Depressive Symptoms-Self Report (IDS-SR) at the end of acute treatment (all P < .0001), which was sustained throughout follow-up (all P < .0001). The proportion of patients who achieved remission at the conclusion of acute treatment remained similar at conclusion of the long-term follow-up. Among 120 patients who met IDS-SR response or remission criteria at the end of acute treatment, 75 (62.5%) continued to meet response criteria throughout long-term follow-up. After the first month, when the majority of acute TMS tapering was completed, 93 patients (36.2%) received reintroduction of TMS. In this group, the mean (SD) number of TMS treatment days was 16.2 (21.1).
Conclusions: TMS demonstrates a statistically and clinically meaningful durability of acute benefit over 12 months of follow-up. This was observed under a pragmatic regimen of continuation antidepressant medication and access to TMS retreatment for symptom recurrence.
Trial Registration: ClinicalTrials.gov identifier: NCT01114477
A Multisite, Naturalistic, Observational Study of Transcranial Magnetic Stimulation for Patients With Pharmacoresistant Major Depressive Disorder: Durability of Benefit Over a 1-Year Follow-Up Period

ABSTRACT
Objective: Transcranial magnetic stimulation (TMS) is an effective and safe acute treatment for patients not benefiting from antidepressant pharmacotherapy. Few studies have examined its longer term durability. This study assessed the long-term effectiveness of TMS in naturalistic clinical practice settings following acute treatment.
Method: Adult patients with a primary diagnosis of unipolar, nonpsychotic major depressive disorder (DSM-IV clinical criteria), who did not benefit from antidepressant medication, received TMS treatment in 42 clinical practices. Two hundred fifty-seven patients completed a course of acute TMS treatment and consented to follow-up over 52 weeks. Assessments were obtained at 3, 6, 9, and 12 months. The study was conducted between March 2010 and August 2012.
Results: Compared with pre-TMS baseline, there was a statistically significant reduction in mean total scores on the Clinical Global Impressions-Severity of Illness scale (primary outcome), 9-Item Patient Health Questionnaire, and Inventory of Depressive Symptoms-Self Report (IDS-SR) at the end of acute treatment (all P < .0001), which was sustained throughout follow-up (all P < .0001). The proportion of patients who achieved remission at the conclusion of acute treatment remained similar at conclusion of the long-term follow-up. Among 120 patients who met IDS-SR response or remission criteria at the end of acute treatment, 75 (62.5%) continued to meet response criteria throughout long-term follow-up. After the first month, when the majority of acute TMS tapering was completed, 93 patients (36.2%) received reintroduction of TMS. In this group, the mean (SD) number of TMS treatment days was 16.2 (21.1).
Conclusions: TMS demonstrates a statistically and clinically meaningful durability of acute benefit over 12 months of follow-up. This was observed under a pragmatic regimen of continuation antidepressant medication and access to TMS retreatment for symptom recurrence.
Trial Registration: ClinicalTrials.gov identifier: NCT01114477
J Clin Psychiatry 2014;75(12):1394-1401
© Copyright 2014 Physicians Postgraduate Press, Inc.
Submitted: December 31, 2013; accepted May 13, 2014.
Online ahead of print: September 16, 2014 (doi:10.4088/JCP.13m08977).
Corresponding author: David L. Dunner, MD, 7525 SE 24th St, Ste 400, Center for Anxiety and Depression, Mercer Island, WA 98040 ([email protected]).
Major depressive disorder (MDD) is a prevalent and profoundly debilitating disease.1 Although a variety of antidepressant medications are available, nearly two-thirds of patients do not benefit from initial pharmacotherapy and remain symptomatic and functionally impaired.2,3 Moreover, the likelihood of achieving remission diminishes with each successive treatment attempt, as verified in the Sequenced Treatment Alternatives to Relieve Depression (STAR*D) study.4-11
Transcranial magnetic stimulation (TMS) is a safe, effective, and well-tolerated alternative to pharmacotherapy for MDD.12-18 Cumulative data in the last decade support that benefit of acute TMS appears sustained over periods of weeks to months.12,18-24 Upon symptom reemergence, reintroduction of TMS often results in restoration of acute benefit.12,21,22,25 Nevertheless, definitive conclusions regarding durability of benefit from these studies are limited by the use of different treatment parameters and durations of acute treatment.
Our group has recently reported on acute benefit among a large cohort of patients (N = 307) treated with TMS from a multisite, naturalistic, observational study in routine clinical practice settings.26,27 The majority of the patients in the acute study participated in a 12-month, long-term follow-up phase. The current report describes their outcomes.
METHOD
Study Subjects
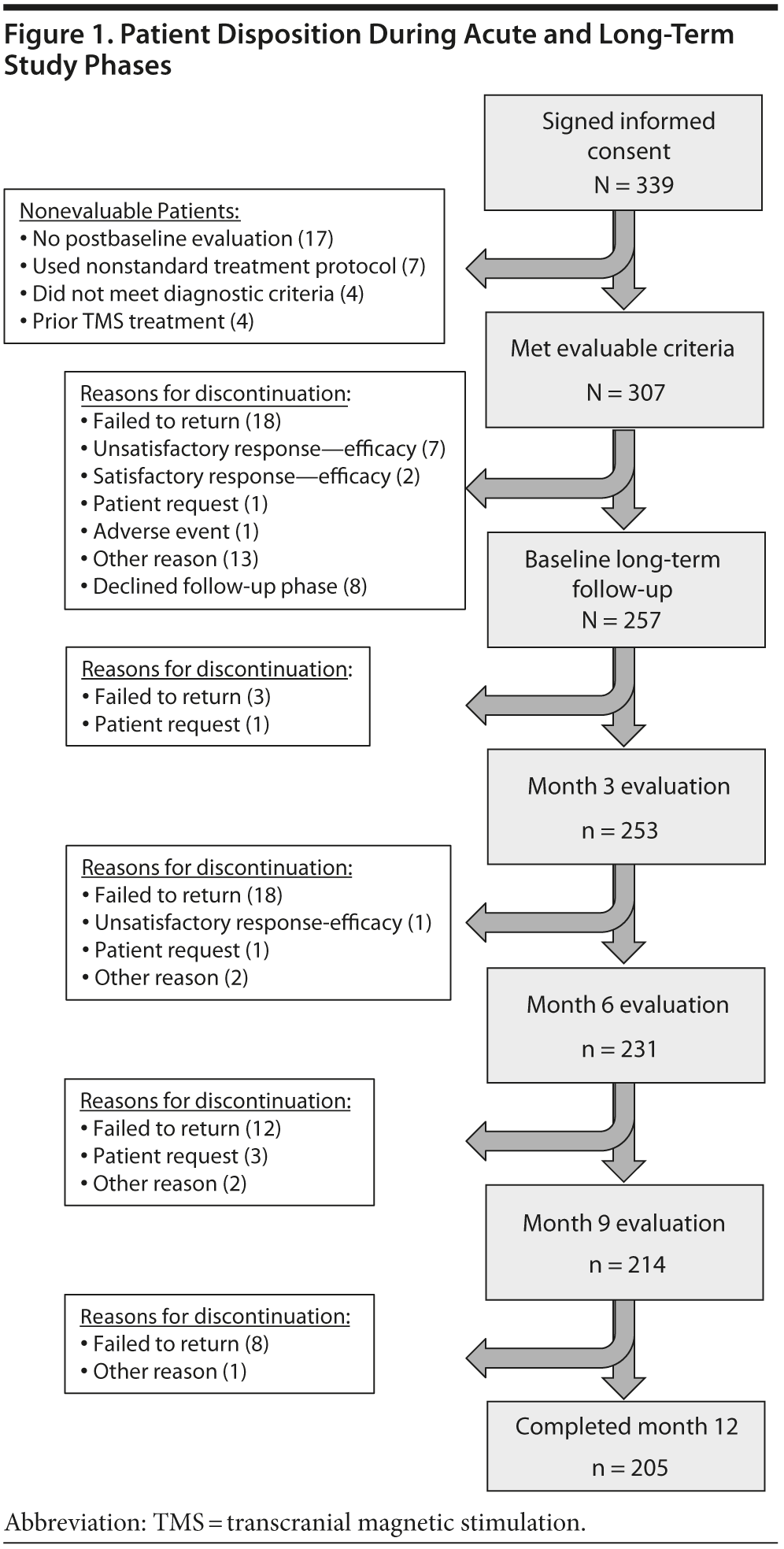
The study participants in this report (N = 257, Table 1) are a subset of those patients who participated in the acute efficacy TMS treatment outcomes study by Carpenter et al.26 Briefly, patients were eligible to participate and considered evaluable if (1) their primary clinical diagnosis was major depressive episode (single or recurrent episode without psychotic features, consistent with DSM-IV criteria), (2) they did not have medical conditions that would preclude the safe use of TMS therapy, (3) they had not received past treatment with TMS for depression, (4) they met standardized criteria for failure to receive clinical benefit from antidepressant medication treatment in the current illness episode, (5) they had a baseline and at least 1 postbaseline rating, (6) their attending clinician determined that TMS represented the most appropriate clinical treatment option, and (7) the attending clinician treating the patient with TMS intended to initiate treatment using the currently labeled TMS treatment parameters. These patients completed their acute treatment and then, regardless of outcome, agreed to enroll in a 12-month long-term follow-up phase. Disposition of patients during the long-term study is shown in Figure 1.
Institutional review board (IRB) approval was obtained at all sites. The cost of all treatment sessions and associated direct clinical care was borne by the patient or the patient’s insurer. Study psychiatrists were provided a modest financial remuneration by the sponsor on a contracted basis for related document preparation and rating scale completion. Patients were provided a modest remuneration for completion of study-specific rating scales. All compensation amounts were reviewed by the site IRB. After a complete description of the procedures, written informed consent was obtained from all subjects. The study was registered at ClinicalTrials.gov (identifier: NCT01114477).
Study Locations, TMS Device, and Clinical Treatment Parameters
Forty-two sites participated in this study. Thirty-two (76%) were in private clinical practices, 7 (17%) were in academic medical centers, and 3 (7%) were in nonacademic institutional settings. The distribution of practice types participating in this study mirrors those currently offering TMS therapy in the United States. The study was conducted between March 2010 and August 2012.

- Transcranial magnetic stimulation (TMS) is effective for treatment-resistant major depressive disorder in real-world practice settings.
- TMS demonstrates a durability of benefit in long-term follow-up that compares favorably to that of current alternatives.
- Prior acute response to TMS is associated with an increased likelihood of benefit upon reintroduction for symptom recurrence.
All treatments were delivered using the NeuroStar TMS Therapy System (Neuronetics, Inc; Malvern, Pennsylvania). The standard treatment protocol described in the product user manual specifies stimulation at 120% of motor threshold, pulse frequency of 10 pulses per second, and a cycle of 4 seconds on (active stimulation) and 26 seconds off (no stimulation), for 75 stimulation cycles, resulting in 3,000 pulses per treatment session. While clinicians initiated treatment with left-sided high-frequency stimulation, the default treatment protocol could be modified for tolerability or logistical reasons. Among the 307 patients initially enrolled, 280 patients (91.2%) received treatment over the left dorsolateral prefrontal cortex only throughout their acute phase treatment. The most common variations observed later in treatment included an increase in session pulse number to 5,000, simple reductions in magnetic field intensity early in treatment to improve tolerability, and the addition of right-sided low-frequency treatment at 1 pulse per second. None of these variations were associated with a significant difference in clinical outcome.
Outcome Measures
Efficacy measures included the Clinician-Reported Clinical Global Impressions-Severity of Illness scale (CGI-S),28,29 the patient-reported Inventory of Depressive Symptoms-Self Report (IDS-SR), and the 9-Item Patient Health Questionnaire (PHQ-9).30,31 The Quick Inventory of Depressive Symptoms-Self Report 16 Item version (QIDS-SR) was derived from the IDS-SR items.32 The outcome measures in this report represent secondary outcomes from the original main study design and statistical analysis plan.
Safety was assessed by summary analysis of medically serious, device-related adverse events or device malfunctions during the study.
Statistical Analysis
Categorical outcomes of response and remission followed conventions commonly used in prior published studies. For the CGI-S, response was defined as an endpoint rating of 3 or less (ie, “mildly ill” or better), while remission on that scale was defined as an endpoint rating of 2 (“borderline mentally ill”) or 1 (“normal/not at all ill”).29 For the PHQ-9, response was defined as an endpoint score less than 10, while remission was defined as an endpoint score less than 5.31 For the IDS-SR, response was defined as a ≥ 50% reduction at endpoint from the baseline rating, while remission was defined as an endpoint score < 15.33 For the QIDS-SR, remission was defined as an endpoint score < 6, and a score of ≥ 11 was used to define relapse.4 The remaining patients not allocated to these categories were classified as nonresponders (for the PHQ-9 and IDS-SR: a decrease < 25% at endpoint compared with the baseline rating; for the CGI-S, an end of acute treatment score of ≥ 4 and ≤ 1 point decrement from baseline or an endpoint score < 4 and no change or worsening compared with baseline score). Partial responders based on the PHQ-9 and IDS-SR required a decrease > 25% but < 50% at endpoint compared with baseline rating; for the CGI-S, these were all patients not allocated to any other CGI-S categories.
Durability outcomes were examined in several ways. For continuous variables, an analysis of covariance model examined the change from baseline in scores during long-term follow-up. Antidepressant treatment record (ATR) group status (0-1 failures of adequate antidepressant treatment versus ≥ 2 failures in the current episode) and site were between-subject factors, and baseline score was a covariate. For all continuous efficacy outcome measures, within-group testing was performed using the Student t test for normally distributed data and the Wilcoxon signed rank test otherwise. Normality testing used the Shapiro-Wilk statistic. All tests were 2-sided, at the 5% level of significance. All analyses were conducted in a last-observation-carried-forward (LOCF) manner for the intent-to-treat (ITT) population (N = 257). Analyses were repeated in the completer sample (N = 205). There were no substantive differences in results with these 2 analyses, so only the ITT population analyses are reported. Categorical outcomes (responders and remitters) were calculated for the overall population.
We examined durability of the outcome among the patients based on their end of acute treatment outcome. For this analysis, categorical outcome defined by the IDS-SR total score at the end of acute treatment (nonresponder, partial responder, responder, or remitter) was used. First, the within-group changes (from end of acute score with each subsequent time point in the long-term follow-up) were examined using Student t tests. Then, a mixed model with repeated measures (MMRM) analysis was conducted under the missing at random framework (using Proc Mixed in SAS Version 9.2). The model included baseline symptom score as a fixed-effect covariate, acute phase endpoint outcome category as the grouping variable, and time (study visit at 3, 6, 9, and 12 months) as a repeated measure. Additionally, the group-by-time interaction term was included in the model to allow for an examination of the clinical trajectory of each acute phase group and to identify any group effect at the follow-up time points. An unstructured covariance matrix was used (SAS Institute, Inc, Cary, North Carolina).
To characterize durability of benefit based on individual patients’ clinical trajectories over time, we examined the probability of acute responders and remitters maintaining either a sustained response or remission by IDS-SR criteria at all follow-up time points. For this analysis, sustained response was defined as serial scores consistently reflecting ≥ 40% improvement relative to baseline, and the criterion for sustained remission was serial IDS-SR scores < 16. These criteria were used to allow for minor fluctuations in symptom scores over extended follow-up. These classifications required the patient to meet the stated criterion at every observation during long-term follow-up. An exploratory logistic regression analysis was performed with data from acute responders or remitters to determine whether any pretreatment characteristics were associated with a durable, sustained response during long-term follow-up. Candidate variables examined were age (continuous, and categorically grouped as < 55 or ≥ 55 years; this age stratification was chosen to be compatible with prior published analyses of this TMS device26,34), gender (categorical), baseline IDS-SR score (continuous), a history of prior hospitalization for depression (categorical), and level of antidepressant resistance (categorical: ATR = 0 or 1 or ATR ≥ 2).
The proportion of patients in remission who experienced symptomatic relapse (defined as a QIDS-SR total score ≥ 11 at any observation time point during long-term follow-up as noted above) was examined. A QIDS-SR total score < 6 at entry into long-term follow-up defined remission. In this analysis, we used the QIDS-SR to permit a direct comparison of clinical significance with other published benchmarks, which used the same outcome measures to define remission and relapse.4
Finally, all patients were permitted access to clinician-directed treatment as usual. We examined the number and pattern of use of psychiatric medications and the incidence of reintroduction TMS to determine whether variations in concomitant treatment contributed to durability. Concomitant psychiatric medications (number and type of medication) intended to treat the patient’s depression were recorded (start and stop dates designated medication change events) from patient report and medical record documentation and verified during monitoring visits. All TMS treatments were recorded as stored in the device’s electronic software database.
RESULTS
Demographic and Clinical Characteristics of the Long-Term Follow-Up Population
Two hundred fifty-seven patients agreed to enroll in the long-term follow-up phase. Two hundred five patients completed outcome evaluations through 12 months (Figure 1).
There were no medically serious, device-related adverse events or device malfunctions during this study. No suicide attempts or suicides were reported during the long-term follow-up phase.
Persistence of Acute Benefit During Long-Term Follow-Up
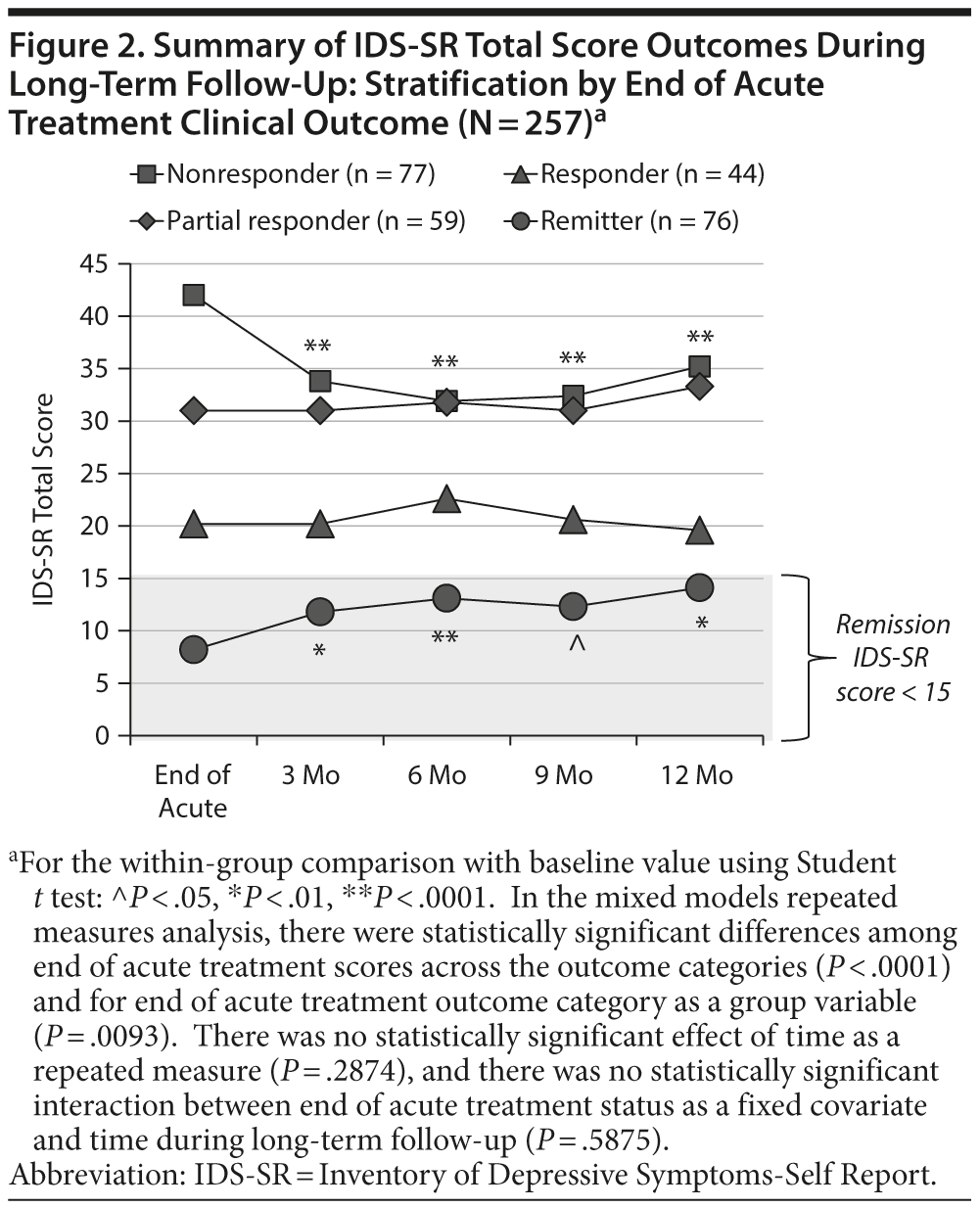
Continuous and categorical outcomes for the entire patient population at the end of acute treatment and during follow-up are summarized in Table 2. Figure 2 shows the mean IDS-SR score for each of the 4 end-of-acute-treatment categories across the long-term time points. For nonresponder and remitter groups, there was a statistically significant difference in scores at all time points of the long-term follow-up phase compared with the end of acute treatment observation. For nonresponders, there was a modest but continued improvement, while for remitters, the pattern was reversed. The mean IDS-SR scores, however, remained below the remission threshold of < 15 at all time points for remitters. For the MMRM analysis, there were statistically significant differences among end of acute treatment score across outcome categories (P < .0001) and for end of acute treatment outcome category as a group variable (P = .0093). There was no significant effect of time as a repeated measure (P = .2874), and there was no significant effect of the interaction between end of acute treatment group by time during long-term follow-up (P = .5875), consistent with the view that the end of acute treatment categories remained stable over long-term follow-up. Similar results were observed with the CGI-S and PHQ-9.
Durability of Acute Response: Probability of Sustained Response or Remission During Long-Term Follow-Up
Among the 44 patients who met IDS-SR responder criteria at entry into long-term follow-up, 25 (56.8%) met criteria for sustained response by IDS-SR criteria at every time point, and, among these 25 patients, 5 patients (ie, 5 of the 44 responders, 11.4%) were sustained remitters at every time point. Among the 76 patients who were IDS-SR remitters at entry into long-term follow-up, 50 (65.8%) met criteria for sustained response by IDS-SR criteria at every time point. Among these, 38 (50.0%) showed sustained remission at every time point. Thus, of the 120 patients who were either responders or remitters after acute treatment, 75 (62.5%) continued to meet response criteria at all time points. None of the candidate pretreatment demographic or clinical variables were statistically significantly related to the durability of outcome. Among the 45 responders or remitters who did not maintain a pattern of sustained response, symptom reemergence tended to occur during the first 6 months of follow-up (months 0-3: n = 17, months 4-6: n = 14, months 7-9: n = 9, months 10-12: n = 5).
Durability of Acute Response:Relapse During Long-Term Follow-Up
Fifty-five (70.5%) of 78 patients who met QIDS-SR full remission criteria (QIDS-SR total score < 6) at entry did not relapse (QIDS-SR score ≤ 11) over the entire 12 months. Among the 23 QIDS-SR remitters who did not maintain a pattern of sustained remission, relapse of illness tended to occur during the first 6 months of follow-up (months 0-3: n = 6, months 4-6: n = 8, months 7-9: n = 6, months 10-12: n = 3).
Use of Concomitant Medications and Incidence of TMS Therapy Reintroduction During Long-Term Follow-Up
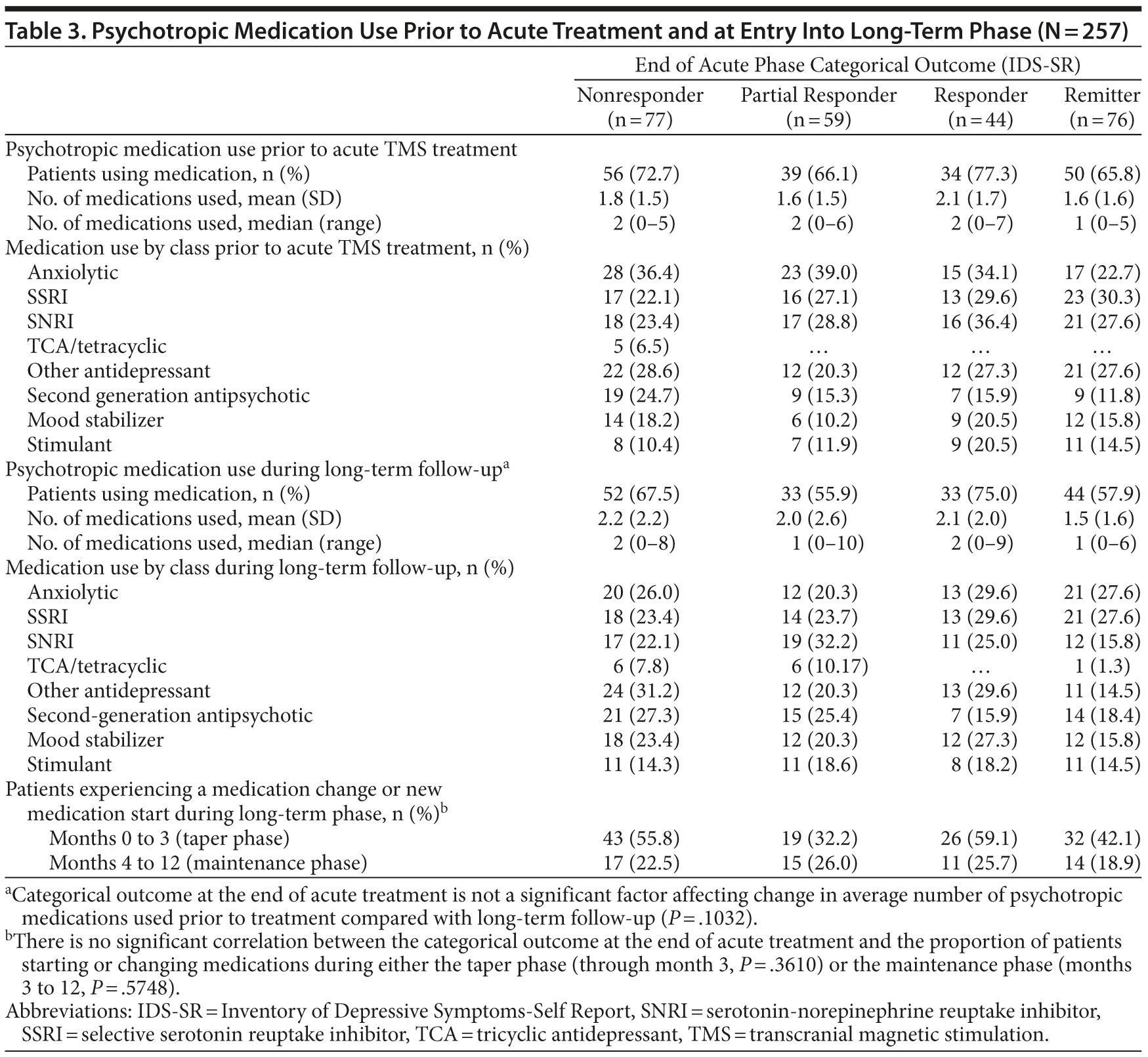
The mean number of psychotropic medications at study entry and during long-term follow-up was similar across outcome categories (Table 3). Categorical outcome at the end of acute treatment did not affect change in the average number of medications used from pretreatment to the follow-up phase. There was no statistically significant relationship between the end of acute treatment categorical outcome and the proportion of patients starting or changing to a new medication during either of these time intervals during long-term follow-up.
Consistent with product labeling, an acute TMS treatment series is usually tapered slowly over several weeks, progressively reducing the number of sessions each week. Inspection of the overall data suggested that the majority of tapering was completed over the first 30 days following the end of acute treatment. From month 2 onward, 93 patients (36.2%) received at least 1 TMS treatment session (data not shown). The mean (SD) number of TMS treatment sessions was 16.2 (21.1).
Patients who had received clinical benefit from TMS were significantly more likely to receive TMS reintroduction and were also significantly more likely to experience subsequent clinical benefit from reintroduction treatment. Specifically, TMS reintroduction was seen in 15/77 (19.5%), 19/59 (32.2%), 27/44 (61.4%), and 32/76 (42.1%) of IDS-SR nonresponders, partial responders, responders, and remitters, respectively (Mantel-Haenszel χ2, P = .0004). Among patients who received TMS reintroduction, 12/15 (80.0%), 15/19 (78.9%), 15/27 (55.6%), and 11/32 (34.4%) of IDS-SR nonresponders, partial responders, responders, and remitters, respectively, experienced later relapse of their illness (Mantel-Haenszel χ2, P = .0004).
DISCUSSION
Acute TMS benefits patients with pharmacoresistant major depression and shows a sustained durability of effect across 12 months of follow-up. Approximately two-thirds of those who were responders to treatment maintained that level of benefit under conditions of clinician-selected, continuation pharmacotherapy with general access to TMS reintroduction as needed. These data are particularly notable because of the difficult-to-treat nature of illness in this population.
It is notable that the findings were congruent regardless of the approach used to define durability of benefit. Symptom levels and rates of response and remission were consistent over time, showing little variation. Approximately two-thirds of responders or remitters maintained their designation over time. Fewer than 30% of initial responders/remitters relapsed within the year. Among responders or remitters, the emergence of illness deterioration was slightly greater during the first 6 months of follow-up compared to later time points.
These results are consistent with the durability of clinical outcome in research study populations reported in 2 studies using the same device and identical treatment protocol for periods of 3 and 6 months.12,18 The report by Mantovani and colleagues18 followed a cohort of patients (N = 50) for 3 months who had remitted (HDRS-24 score ≤ 10) following acute TMS in either an earlier multisite randomized trial14 or an open-label follow-up study,35 in which patients were tapered off TMS and transitioned to maintenance medications. At 3 months, 29 of 50 patients (58.0%) remained in remission (HDRS-24 score < 11), with an overall relapse rate of 13.5% (HDRS-24 score ≥ 20). Janicak and colleagues12 described outcome over 6 months in 99 patients who benefited from acute TMS treatment in a similar manner during either an earlier multisite randomized trial17 or an open-label follow-up study.15 Those patients also were tapered off TMS, transitioning to maintenance medication monotherapy over 3 weeks. During follow-up, the maintenance medication could not be switched or combined with other medications. TMS was reintroduced if patients met protocol-specified criteria for symptom recurrence. Relapse was defined as either the reemergence of the full DSM-IV criteria for MDD at any point or the failure of symptom resolution upon 6 weeks of TMS reintroduction. The researchers found that 10 of 99 patients (10%; Kaplan-Meier survival estimate = 12.9%) met criteria for relapse during the 6-month follow-up, 38 (38.4%) met criteria for symptom worsening, and 32/38 (84.2%) reachieved symptomatic benefit with TMS.
How do these outcomes compare with other treatment options? In a study examining naturalistic outcomes with treatment as usual in treatment-resistant patients, Dunner and colleagues36 found that the majority of patients who achieved response or remission showed only a transient, nonsustained pattern of benefit. After 24 months of follow-up, only 10% of patients reached response criterion in any 3-month interval. Further, for most patients, this effect was not sustained at subsequent time points. These results are similar to the outcomes observed with the Level 3 and Level 4 treatment options used in the STAR*D study.4 There, patients who achieved remission (QIDS-SR score < 6) with acute treatment after 2 or 3 failed attempts experienced relapse (QIDS-SR score > 11) over the next 12 months at rates of 42.9% (Level 3) and 50.0% (Level 4) under clinician-directed best-choice treatment. A recent meta-analysis37 of long-term outcomes following remission with ECT demonstrated similar difficulty in maintaining benefit.
The more robust the symptom relief obtained during acute treatment, the more favorable the long-term course for patients.38 Results observed in this study are consistent with that general pattern. For instance, among responders or remitters at the end of acute treatment, those who did not subsequently relapse entered long-term follow-up with a lower level of residual symptoms as defined by their end of acute IDS-SR score (did not relapse, n = 79: IDS-SR total score [SD] = 11.3 [6.7] vs relapsed, n = 41: IDS-SR total score [SD] = 15.1 [6.9], P = .0043). Other specific factors that may predict durability of outcome can only be partially addressed here. No pretreatment clinical or demographic features were significantly associated with long-term outcome. Nor was durability of outcome attributable to the number of concurrent medications used for depression or the number of new medication starts or switches during follow-up.
Overall, approximately one-third of patients received TMS reintroduction. In general, prior acute benefit from TMS was associated with a higher likelihood of its usefulness when required for symptom recurrence. Specifically, those patients who achieved the greatest initial clinical improvement from acute treatment with TMS and required subsequent reintroduction of TMS for symptom recurrence experienced the lowest likelihood of symptom relapse at later time points. On the other hand, the observational design of this study cannot answer the question of whether a defined schedule of maintenance TMS can improve long-term outcome as with other antidepressant therapies. Nevertheless, the use of maintenance TMS remains an important topic for future research.
There are limitations to this study. As it had an observational, naturalistic design, there was no concurrent control population. Conclusions regarding the influence of concomitant treatments, including the role of TMS reintroduction, cannot be fully explored. It should be noted that not all patients had unrestricted access to retreatment or continuation TMS. During the study, insurance coverage for patient access to TMS was gradually introduced in the United States, and therefore patients faced variable degrees of personal financial obligation to pay for TMS. Finally, analysis using an LOCF analysis method may exaggerate the consistency of the scores; however, the low overall discontinuation rate and the similar results in the completer sample lessen the significance of this concern.
In summary, TMS demonstrated a sustained durability of effect over 12 months of follow-up in a population receiving minimal to no benefit with antidepressant medications. These clinical outcomes are as good as or superior to those seen with other treatment alternatives.
Author affiliations: Center for Anxiety and Depression, Mercer Island, Washington (Dr Dunner); The Retreat, Sheppard-Pratt Health System, Baltimore, Maryland (Dr Aaronson); Department of Biological Psychiatry, Columbia University, New York, New York (Dr Sackeim); Department of Psychiatry, Rush University Medical Center, Chicago, Ilinois (Dr Janicak); Department of Psychiatry and Human Behavior, Butler Hospital, Providence, Rhode Island (Dr Carpenter); Private Practice, West Chester, Pennsylvania (Dr Boyadjis); Neuronetics, Inc, Malvern, Pennsylvania (Drs Brock, Bonneh-Barkay, and Demitrack); Department of Psychiatry, UCLA Medical Center, Los Angeles, California (Dr Cook); Private Practice, Sacramento, California (Dr Lanocha); and Department of Psychiatry, Stanford University Medical Center, Palo Alto, California (Dr Solvason).
Potential conflicts of interest: Dr Dunner has received grants from Neuronetics and Cyberonics and served as a consultant for Neuronetics, Cervel Neurotech, Jazz, Bristol-Myers Squibb, and MedDEV. Dr Aaronson has received grants from Neuronetics and Stanley Medical Research Foundation, has served as a speaker for Neuronetics and Takeda/Lundbeck, has served as a board member for Genomind, and has served as a consultant for Alkermes. Dr Sackeim has served as a board member for Neuronetics and a consultant for Cervel Neurotech, Brainsway, and MECTA. Dr Janicak has received grants from Neuronetics and Cervel Neurotech and served as a consultant and given expert testimony for Neuronetics. Dr Carpenter has served as a consultant for AbbVie, Takeda/Lundbeck, Taisho (Helicon), Magstim, and Naurex and received grant support from National Institute of Mental Health, NeoSync, Cervel Neurotech, and Neuronetics. Dr Boyadjis has received a grant from Neuronetics. Drs Brock, Bonneh-Barkay, and Demitrack are employees of and have received stock options from Neuronetics. Dr Cook has received a grant from and served on speaker and advisory boards for Neuronetics; has served on speaker and advisory boards for Allergan and Pfizer; has received grant support from Covidien, NeoSync, Shire, and National Institutes of Health (NIH); has served as a consultant for Covidien and NeuroSigma; has performed an Interventions Committee for Adult Disorders (ITVA) review for NIH and served on a Data and Safety Monitoring Board for the Department of Veterans Affairs; has received stock options from NeuroSigma; and has received assignment of patents from University of California. Dr Lanocha has received a grant from and served as a speaker for Neuronetics. Dr Solvason has received a grant from Neuronetics.
Funding/support: Supported by a grant from Neuronetics, Inc.
Role of the sponsor: The study sponsor was involved in the design of the study, oversight of administrative aspects of the study documentation, study monitoring, and data analyses. All analyses reported in this study were available for author direct review.
Previous presentations: Preliminary reports of these data were presented at the New Research Poster Session of the American Psychiatric Association Annual Meeting, May 18-23, 2013 and the Institute for Psychiatric Services, October 10-13, 2013; Philadelphia, Pennsylvania.
REFERENCES
1. Moussavi S, Chatterji S, Verdes E, et al. Depression, chronic diseases, and decrements in health: results from the World Health Surveys. Lancet. 2007;370(9590):851-858. PubMed doi:10.1016/S0140-6736(07)61415-9
2. Sackeim HA. The definition and meaning of treatment-resistant depression. J Clin Psychiatry. 2001;62(suppl 16):10-17. PubMed
3. Rush AJ, Thase ME, Dubé S. Research issues in the study of difficult-to-treat depression. Biol Psychiatry. 2003;53(8):743-753. PubMed doi:10.1016/S0006-3223(03)00088-X
4. Rush AJ, Trivedi MH, Wisniewski SR, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163(11):1905-1917. PubMed doi:10.1176/appi.ajp.163.11.1905
5. Fava M, Rush AJ, Trivedi MH, et al. Background and rationale for the Sequenced Treatment Alternatives to Relieve Depression (STAR*D) study. Psychiatr Clin North Am. 2003;26(2):457-494, x. PubMed doi:10.1016/S0193-953X(02)00107-7
6. Trivedi MH, Fava M, Wisniewski SR, et al; STAR*D Study Team. Medication augmentation after the failure of SSRIs for depression. N Engl J Med. 2006;354(12):1243-1252. PubMed doi:10.1056/NEJMoa052964
7. Trivedi MH, Rush AJ, Wisniewski SR, et al; STAR*D Study Team. Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: implications for clinical practice. Am J Psychiatry. 2006;163(1):28-40. PubMed doi:10.1176/appi.ajp.163.1.28
8. Rush AJ, Trivedi MH, Wisniewski SR, et al; STAR*D Study Team. Bupropion-SR, sertraline, or venlafaxine-XR after failure of SSRIs for depression. N Engl J Med. 2006;354(12):1231-1242. PubMed doi:10.1056/NEJMoa052963
9. Fava M, Rush AJ, Wisniewski SR, et al. A comparison of mirtazapine and nortriptyline following two consecutive failed medication treatments for depressed outpatients: a STAR*D report. Am J Psychiatry. 2006;163(7):1161-1172. PubMed doi:10.1176/appi.ajp.163.7.1161
10. McGrath PJ, Stewart JW, Fava M, et al. Tranylcypromine versus venlafaxine plus mirtazapine following three failed antidepressant medication trials for depression: a STAR*D report. Am J Psychiatry. 2006;163(9):1531-1541, quiz 1666. PubMed doi:10.1176/appi.ajp.163.9.1531
11. Nierenberg AA, Fava M, Trivedi MH, et al. A comparison of lithium and T(3) augmentation following two failed medication treatments for depression: a STAR*D report. Am J Psychiatry. 2006;163(9):1519-1530, quiz 1665. PubMed doi:10.1176/appi.ajp.163.9.1519
12. Janicak PG, Nahas Z, Lisanby SH, et al. Durability of clinical benefit with transcranial magnetic stimulation (TMS) in the treatment of pharmacoresistant major depression: assessment of relapse during a 6-month, multisite, open-label study. Brain Stimulat. 2010;3(4):187-199. PubMed doi:10.1016/j.brs.2010.07.003
13. Janicak PG, O’ Reardon JP, Sampson SM, et al. Transcranial magnetic stimulation in the treatment of major depressive disorder: a comprehensive summary of safety experience from acute exposure, extended exposure, and during reintroduction treatment. J Clin Psychiatry. 2008;69(2):222-232. PubMed doi:10.4088/JCP.v69n0208
14. George MS, Lisanby SH, Avery D, et al. Daily left prefrontal transcranial magnetic stimulation therapy for major depressive disorder: a sham-controlled randomized trial. Arch Gen Psychiatry. 2010;67(5):507-516. PubMed doi:10.1001/archgenpsychiatry.2010.46
15. Avery DH, Isenberg KE, Sampson SM, et al. Transcranial magnetic stimulation in the acute treatment of major depressive disorder: clinical response in an open-label extension trial. J Clin Psychiatry. 2008;69(3):441-451. PubMed doi:10.4088/JCP.v69n0315
16. Demitrack MA, Thase ME. Clinical significance of transcranial magnetic stimulation (TMS) in the treatment of pharmacoresistant depression: synthesis of recent data. Psychopharmacol Bull. 2009;42(2):5-38. PubMed
17. O’ Reardon JP, Solvason HB, Janicak PG, et al. Efficacy and safety of transcranial magnetic stimulation in the acute treatment of major depression: a multisite randomized controlled trial. Biol Psychiatry. 2007;62(11):1208-1216. PubMed doi:10.1016/j.biopsych.2007.01.018
18. Mantovani A, Pavlicova M, Avery D, et al. Long-term efficacy of repeated daily prefrontal transcranial magnetic stimulation (TMS) in treatment-resistant depression. Depress Anxiety. 2012;29(10):883-890. PubMed doi:10.1002/da.21967
19. Dannon PN, Dolberg OT, Schreiber S, et al. Three and six-month outcome following courses of either ECT or rTMS in a population of severely depressed individuals—preliminary report. Biol Psychiatry. 2002;51(8):687-690. PubMed doi:10.1016/S0006-3223(01)01274-4
20. Dannon PN, Grunhaus L. Repetitive transcranial magnetic stimulation is effective following repeated courses in the treatment of major depressive disorder—a case report. Hum Psychopharmacol. 2003;18(4):313-315. PubMed doi:10.1002/hup.478
21. Demirtas-Tatlidede A, Mechanic-Hamilton D, Press DZ, et al. An open-label, prospective study of repetitive transcranial magnetic stimulation (rTMS) in the long-term treatment of refractory depression: reproducibility and duration of the antidepressant effect in medication-free patients. J Clin Psychiatry. 2008;69(6):930-934. PubMed doi:10.4088/JCP.v69n0607
22. Fitzgerald PB, Benitez J, de Castella AR, et al. Naturalistic study of the use of transcranial magnetic stimulation in the treatment of depressive relapse. Aust N Z J Psychiatry. 2006;40(9):764-768. PubMed doi:10.1080/j.1440-1614.2006.01881.x
23. Benadhira R, Saba G, Samaan A, et al. Transcranial magnetic stimulation for refractory depression. Am J Psychiatry. 2005;162(1):193. PubMed doi:10.1176/appi.ajp.162.1.193
24. Cohen RB, Boggio PS, Fregni F. Risk factors for relapse after remission with repetitive transcranial magnetic stimulation for the treatment of depression. Depress Anxiety. 2009;26(7):682-688. PubMed doi:10.1002/da.20486
25. Dannon PN, Schreiber S, Dolberg OT, et al. Transcranial magnetic stimulation is effective in the treatment of relapse of depression. Int J Psychiatry Clin Pract. 2000;4(3):223-226. PubMed doi:10.1080/13651500050518118
26. Carpenter LL, Janicak PG, Aaronson ST, et al. Transcranial magnetic stimulation (TMS) for major depression: a multisite, naturalistic, observational study of acute treatment outcomes in clinical practice. Depress Anxiety. 2012;29(7):587-596. PubMed doi:10.1002/da.21969
27. Janicak PG, Dunner DL, Aaronson ST, et al. Transcranial magnetic stimulation (TMS) for major depression: a multisite, naturalistic, observational study of quality of life outcome measures in clinical practice. CNS Spectr. 2013;18(6):322-332. PubMed doi:10.1017/S1092852913000357
28. Guy W, ed. ECDEU Assessment Manual for Psychopharmacology. US Dept Health, Education, and Welfare publication (ADM) 76-338. Rockville, Maryland: National Institute of Mental Health; 1976.
29. Bandelow B, Baldwin DS, Dolberg OT, et al. What is the threshold for symptomatic response and remission for major depressive disorder, panic disorder, social anxiety disorder, and generalized anxiety disorder? J Clin Psychiatry. 2006;67(9):1428-1434. PubMed doi:10.4088/JCP.v67n0914
30. Kroenke K, Spitzer RL, Williams JBW. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16(9):606-613. PubMed doi:10.1046/j.1525-1497.2001.016009606.x
31. Katzelnick DJ, Duffy FF, Chung H, et al. Depression outcomes in psychiatric clinical practice: using a self-rated measure of depression severity. Psychiatr Serv. 2011;62(8):929-935. PubMed doi:10.1176/appi.ps.62.8.929
32. Inventory of Depressive Symptomatology-IDS/QIDS. 2013. www.ids-qids.org. Accessed August 15, 2014.
33. Kornstein SG, Schneider RK. Clinical features of treatment-resistant depression. J Clin Psychiatry. 2001;62(suppl 16):18-25. PubMed
34. Lisanby SH, Husain MM, Rosenquist PB, et al. Daily left prefrontal repetitive transcranial magnetic stimulation in the acute treatment of major depression: clinical predictors of outcome in a multisite, randomized controlled clinical trial. Neuropsychopharmacology. 2009;34(2):522-534. PubMed doi:10.1038/npp.2008.118
35. McDonald WM, Durkalski V, Ball ER III, et al. Improving the antidepressant efficacy of transcranial magnetic stimulation: maximizing the number of stimulations and treatment location in treatment-resistant depression. Depress Anxiety. 2011;28(11):973-980. PubMed doi:10.1002/da.20885
36. Dunner DL, Rush AJ, Russell JM, et al. Prospective, long-term, multicenter study of the naturalistic outcomes of patients with treatment-resistant depression. J Clin Psychiatry. 2006;67(5):688-695. PubMed doi:10.4088/JCP.v67n0501
37. Jelovac A, Kolshus E, McLoughlin DM. Relapse following successful electroconvulsive therapy for major depression: a meta-analysis. Neuropsychopharmacology. 2013;38(12):2467-2474. PubMed doi:10.1038/npp.2013.149
38. Prien RF, Kupfer DJ. Continuation drug therapy for major depressive episodes: how long should it be maintained? Am J Psychiatry. 1986;143(1):18-23. PubMed
This PDF is free for all visitors!