Repetitive Transcranial Magnetic Stimulation for Treatment-Resistant Depression: A Systematic Review and Meta-Analysis
ABSTRACT
Objective: To evaluate the efficacy of repetitive transcranial magnetic stimulation (rTMS) in patients with major depressive disorder (MDD) and 2 or more prior antidepressant treatment failures (often referred to as treatment-resistant depression [TRD]). These patients are less likely to recover with medications alone and often consider nonpharmacologic treatments such as rTMS.
Data Sources: We searched MEDLINE, EMBASE, the Cochrane Library, PsycINFO, and the International Pharmaceutical Abstracts for studies comparing rTMS with a sham-controlled treatment in TRD patients ages 18 years or older.
Study Selection: We included 18 good- or fair-quality TRD studies published from January 1, 1980, through March 20, 2013.
Data Extraction: We abstracted relevant data, assessed each study’s internal validity, and graded strength of evidence for change in depressive severity, response rates, and remission rates.
Results: rTMS was beneficial compared with sham for all outcomes. rTMS produced a greater decrease in depressive severity (high strength of evidence), averaging a clinically meaningful decrease on the Hamilton Depression Rating Scale (HDRS) of more than 4 points compared with sham (mean decrease = -4.53; 95% CI, -6.11 to -2.96). rTMS resulted in greater response rates (high strength of evidence); those receiving rTMS were more than 3 times as likely to respond as patients receiving sham (relative risk = 3.38; 95% CI, 2.24 to 5.10). Finally, rTMS was more likely to produce remission (moderate strength of evidence); patients receiving rTMS were more than 5 times as likely to achieve remission as those receiving sham (relative risk = 5.07; 95% CI, 2.50 to 10.30). Limited evidence and variable treatment parameters prevented conclusions about which specific treatment options are more effective than others. How long these benefits persist remains unclear.
Conclusions: For MDD patients with 2 or more antidepressant treatment failures, rTMS is a reasonable, effective consideration.
J Clin Psychiatry 2014;75(5):477-489
© Copyright 2014 Physicians Postgraduate Press, Inc.
Submitted: September 27, 2013; accepted February 11, 2014 (doi:10.4088/JCP.13r08815).
Corresponding author: Bradley N. Gaynes, MD, MPH, Department of Psychiatry, CB #7160, 304 MacNider Hall, Room J, University of North Carolina at Chapel Hill, Chapel Hill, NC 27599-7160 ([email protected]).
In any given year, between 13.1 million and 14.2 million US residents experience major depressive disorder (MDD).1 Half seek help for this condition; only 1 in 5 of those seeking help receive adequate acute-phase treatment.2 Even for patients receiving adequate treatment, only 30% (that is, 3% of patients with MDD) reach the treatment goal of remission.3
The remaining 70% of MDD patients will either respond without remission (about 20%) or not respond at all (50%).3 Patients whose depressive disorder does not respond satisfactorily to appropriate therapy clearly have harder-to-treat depression.4 In particular, patients with 2 or more failed attempts with adequate treatment are a common, challenging presentation to psychiatric and primary care clinics.5 Expert consensus considers these patients to have treatment-resistant depression (TRD).5-7
Having 2 adequately dosed but unsuccessful treatment attempts in the same episode predicts a lower likelihood of remission with the next treatment.5 Although the remission rate for depressed patients with a first or, if necessary, a second treatment attempt is approximately 30%, the likelihood of recovery with a subsequent medication treatment decreases to approximately 15%.8 Patients with TRD incur the highest direct and indirect medical costs among those with MDD; these costs increase with the severity of the illness.9
Clinicians and patients need clear evidence to guide treatment decisions in TRD. The wide-ranging choices include both pharmacologic and nonpharmacologic interventions. Somatic treatments, which may involve use of a pharmacologic intervention or a device, are commonly considered for TRD patients. Given the decreasing efficacy of antidepressant medications following 2 unsuccessful treatments and their potentially serious side effects,5,10 clinicians often look for alternative strategies for their TRD patients.
Repetitive transcranial magnetic stimulation (rTMS) is 1 possible option. Repetitive transcranial magnetic stimulation denotes a range of biologic, nonpharmacologic treatment devices that involve magnetic focal stimulation through the scalp. The current elicited by the electromagnetic coil stimulates nerve cells in the region of the brain involved in mood regulation and depression. The treatment may involve different types of coils, coil placements, and frequencies; the field has not yet agreed on one best rTMS method.11 It can be delivered at either low or high frequency and administered in an office setting without anesthesia. Repetitive transcranial magnetic stimulation can be used as a primary treatment or as an augmentation strategy for an already existing antidepressant medication treatment and has been recommended as a consideration for use in acute-phase treatment by the American Psychiatric Association’s Practice Guideline in MDD.12 The US Food and Drug Administration (FDA) first approved a specific rTMS device in October 2008 “for the treatment of major depressive disorder in adult patients who have failed to achieve satisfactory improvement from one prior antidepressant medication at or above the minimal effective dose and duration in the current episode.”13 In 2013, the FDA approved a second device for similar patients “who failed to achieve satisfactory improvement from previous anti-depressant medication treatment in the current episode,”14 not limiting the approval to 1 prior episode failure.
The FDA approvals provide a mixed message about rTMS in TRD; they also reflect the evolving clinical research on rTMS, whose ideal methods for administration continue to be clarified. Repetitive transcranial magnetic stimulation is in many ways a “family” of treatments whose specifics are still being defined. Few insurers cover rTMS for depression in general, let alone TRD.15 Nevertheless, many clinicians and TRD patients consider it a viable treatment option, and at least 1 advisory board recommends its use in TRD.16 To provide an up-to-date synthesis of the available evidence, while acknowledging that rTMS is a general approach rather than a single entity, we report here on the efficacy of rTMS versus sham for patients with TRD.

- The use of nonpharmacologic interventions in patients with treatment-resistant depression is a key clinical issue; clinicians and researchers are only beginning to identify the role of repetitive transcranial magnetic stimulation (rTMS) for this hard-to-treat population.
- Repetitive transcranial magnetic stimulation is a general class of treatments rather than a single entity, much as antidepressants represent a general approach encompassing a variety of compounds with a common therapeutic application but divergent mechanisms for achieving it.
- For patients with major depressive disorder with 2 or more antidepressant treatment failures, rTMS is a reasonable, effective consideration.
METHOD
Data Sources and Searches
As part of a larger comparative effectiveness review on nonpharmacologic interventions for patients with TRD,17 we reviewed evidence (from January 1, 1980, to November 18, 2010) addressing the efficacy of rTMS compared with sham control. This article updates our systematic searches for rTMS through March 20, 2013. We searched MEDLINE, EMBASE, the Cochrane Library, PsycINFO, and the International Pharmaceutical Abstracts. We used Medical Subject Headings (MeSH or MH) as search terms when available and key words when appropriate (Supplementary eTable 1). We combined terms for TRD, including the terms refractory, resistant, and drug resistance. We manually searched reference lists of relevant bibliographies, pertinent review articles, and letters to the editor and used SCOPUS to identify additional articles. Staff of the Agency for Healthcare Research and Quality (AHRQ)-supported Scientific Resource Center (SRC) contacted device manufacturers and invited them to submit dossiers, including citations, for review. In addition, the SRC conducted searches in clinical trial registries and gray literature databases to detect unpublished studies (eg, meeting abstracts, white papers).17
Study Selection
Supplementary eTable 2 outlines study eligibility. We selected randomized controlled trials comparing rTMS with sham. Two independent reviewers assessed article abstracts for inclusion. All titles selected by at least 1 reviewer went on to full-text review by 2 independent reviewers. We applied a more detailed set of inclusion criteria at full-text review, requiring reasons for exclusion (eg, wrong design) and specifics on levels of treatment resistance.
Although we define TRD as 2 or more treatment failures, many trials involving populations with TRD did not use this definition when formulating their inclusion criteria. Some trials limited inclusion to individuals having 2 or more antidepressant treatment failures. Other trials provided analyses on depressed populations that combined subjects with 1 treatment failure and those with 2 or more treatment failures. Yet others reported on groups that most likely met our definition of TRD (eg, those described as “treatment resistant”) but did not specify the number of treatment failures. A priori, we decided that excluding the latter 2 groups would not fairly reflect what was known about treatment for TRD populations.
Accordingly, we focused our synthesis on studies of patients who clearly met our definition of TRD: 2 or more prior antidepressant failures following adequate dose (as reported by the authors) and duration (at least 4 weeks). We called these tier 1 studies (Supplementary eTable 3). We also identified studies of patients with 1 or more antidepressant failures; we called these tier 2 studies. Trials that did not report the number of antidepressant failures but whose clinical context suggested a high probability of patients with 2 or more prior antidepressant failures are called tier 3 studies (eg, undefined treatment resistance).
The core patient population of interest was patients with MDD who met our definition of TRD. With support of a technical expert panel,17 we also included studies in which the patient population included a “mix” of up to 20% of patients with bipolar disorder (ie, 80% or more of patients had only MDD), assuming that this small mix would not substantially alter outcomes seen with MDD-only populations.
Quality Assessment
We assessed the quality (internal validity or risk of bias) of all included studies using predefined criteria based on guidance in the AHRQ Methods Guide for Comparative Effectiveness Reviews17 and the US Preventive Services Task Force definitions18; ratings can be good, fair, or poor. Two independent reviewers assigned quality ratings, resolving any disagreements by consensus discussion or by consulting with a third reviewer. In general terms, a “good” study met all criteria, has the least bias, and provides results considered to be valid. “Fair” studies presumably fulfilled all quality criteria but did not report their methods to an extent that answered all of our questions. A “poor” rating indicates significant bias.
Data Extraction
We used a structured, pilot-tested data abstraction form to ensure consistency of data abstraction.17 Trained reviewers abstracted data on study design, baseline population characteristics, specifications of the intervention, and relevant outcome assessments for both efficacy and harms.
Data Synthesis and Analysis
Our analyses included only those studies assessed as fair or good quality, reported at least 1 key outcome (change in depressive severity, response, or remission), involved patients with a current major depressive episode, and were conducted within a TRD population aged 18 years or older.
Our efficacy outcome measure of choice was the Hamilton Depression Rating Scale (HDRS).19 If a study included a measure other than the HDRS (eg, Montgomery-Asberg Depression Rating Scale [MADRS], Beck Depression Inventory, or Quick Inventory of Depressive Symptomatology), we abstracted those data. We calculated change in depressive severity using the baseline and endpoint data when trials did not present that information. When possible, we used the number of patients randomized rather than only those included in the analysis to show the percentage achieving response (≥ 50% in depressive severity) or remission (as indicated by a score on a validated depressive tool, eg, ≤ 7 on the HDRS) to reflect a true intention-to-treat analysis.
If we found 3 or more studies with similar study populations, treatment interventions, and outcome assessments, we conducted quantitative analyses. We first pooled results for our tier 1 TRD studies. We then ran combined and stratified analyses by tiers to assess how treatment effects differed by tiers. For efficacy, we used 3 outcome measures:
- The weighted mean difference of changes on the HDRS, which estimates the actual differences between intervention and sham in effect sizes.
- The relative risk of being a responder (≥ 50% improvement from baseline) on the HDRS or the MADRS at study endpoint.
- The relative risk of achieving remission on the HDRS or MADRS at study endpoint. The HDRS definition was ≤ 7 for the 17-item version and ≤ 10 for the 21-item version. For the MADRS, the remission definition was a score of ≤ 8. If a trial used a slightly different definition for remission, we noted this difference in the study’s summary table and included the information in our analyses when, in the authors’ judgment, it did not substantially differ from the above definitions.
For each meta-analysis, we conducted tests of heterogeneity (I2 index and Cochran Q test) and applied both a random- and a fixed-effects model. To explore reasons for heterogeneity, we used metaregressions. We report the results from random effects models because, in all meta-analyses, the results from random- and fixed-effects models were very similar. If the relative risk was statistically significant, we calculated the number needed to treat (NNT) from the pooled relative risk when variations in baseline risks were small. We assessed publication bias using funnel plots, Egger regression intercept, and Kendall S statistic. Given the small number of trials in some of our meta-analyses, these tests have low sensitivity to detect publication bias.20
We evaluated the strength of evidence (SOE) based on the initial AHRQ guidance for grading SOE.21 Although we provide additional evidence (tiers 2 and 3) for evaluating the outcomes in a TRD population (tier 1), our SOE grades consider tier 1 evidence only.
RESULTS
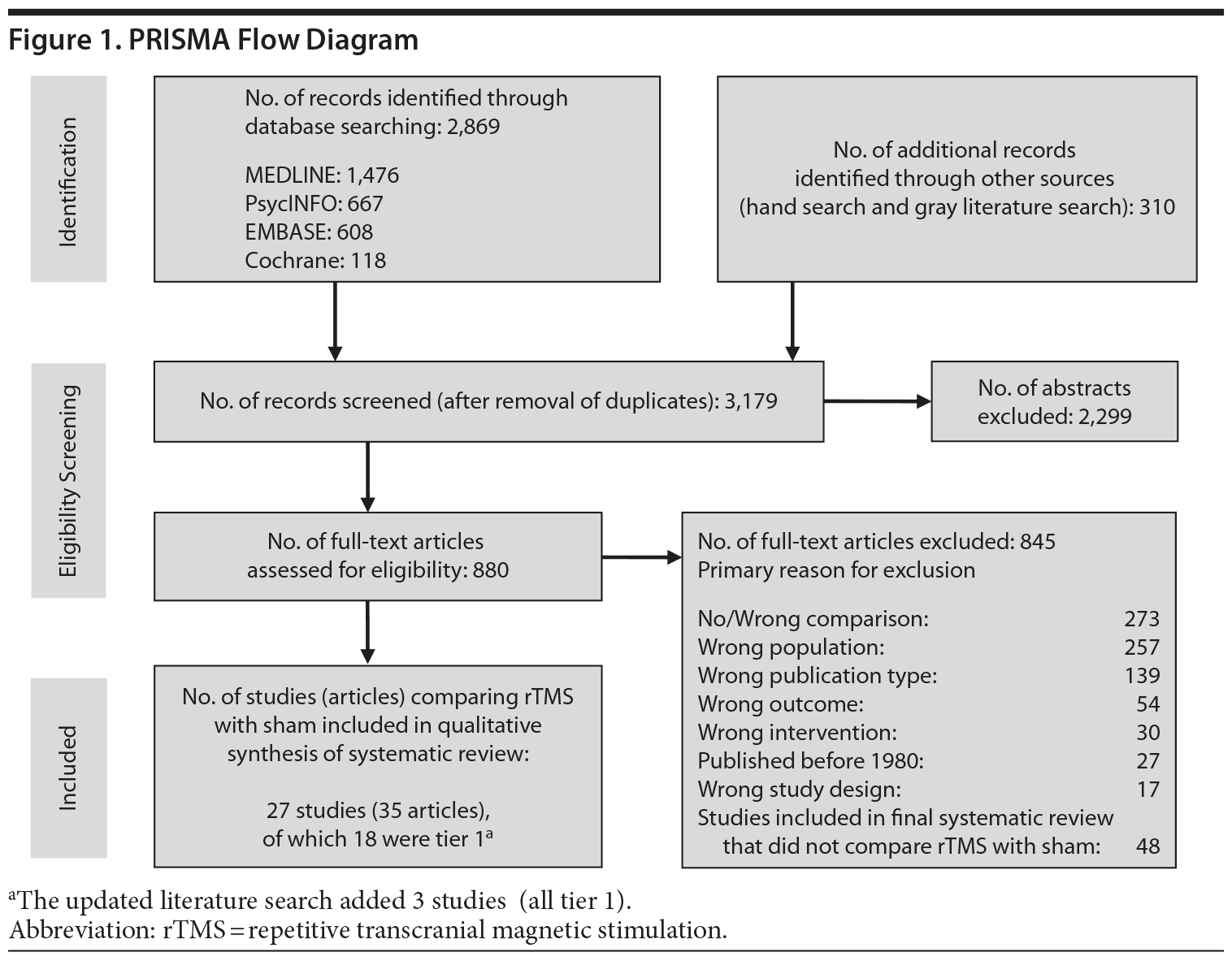
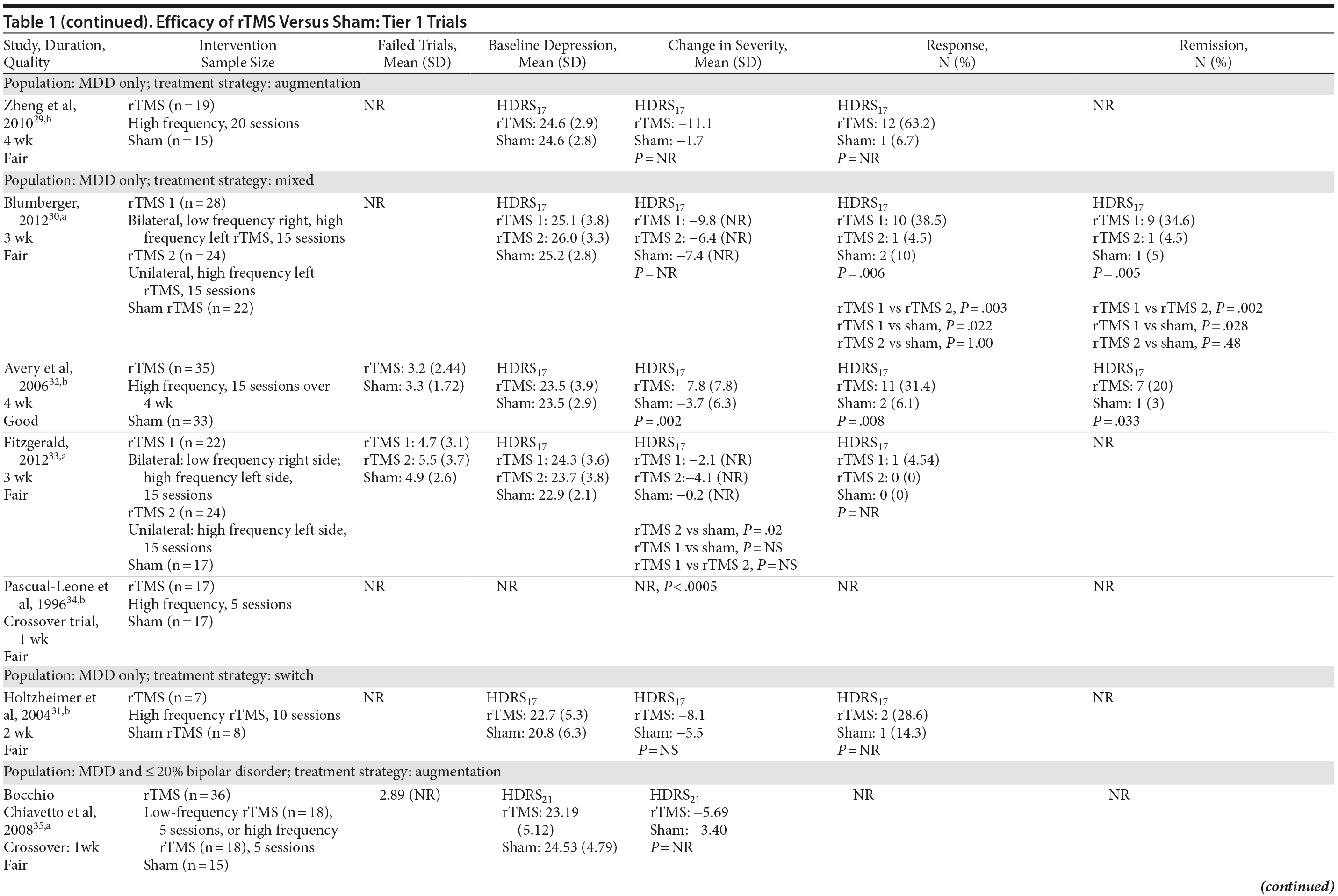
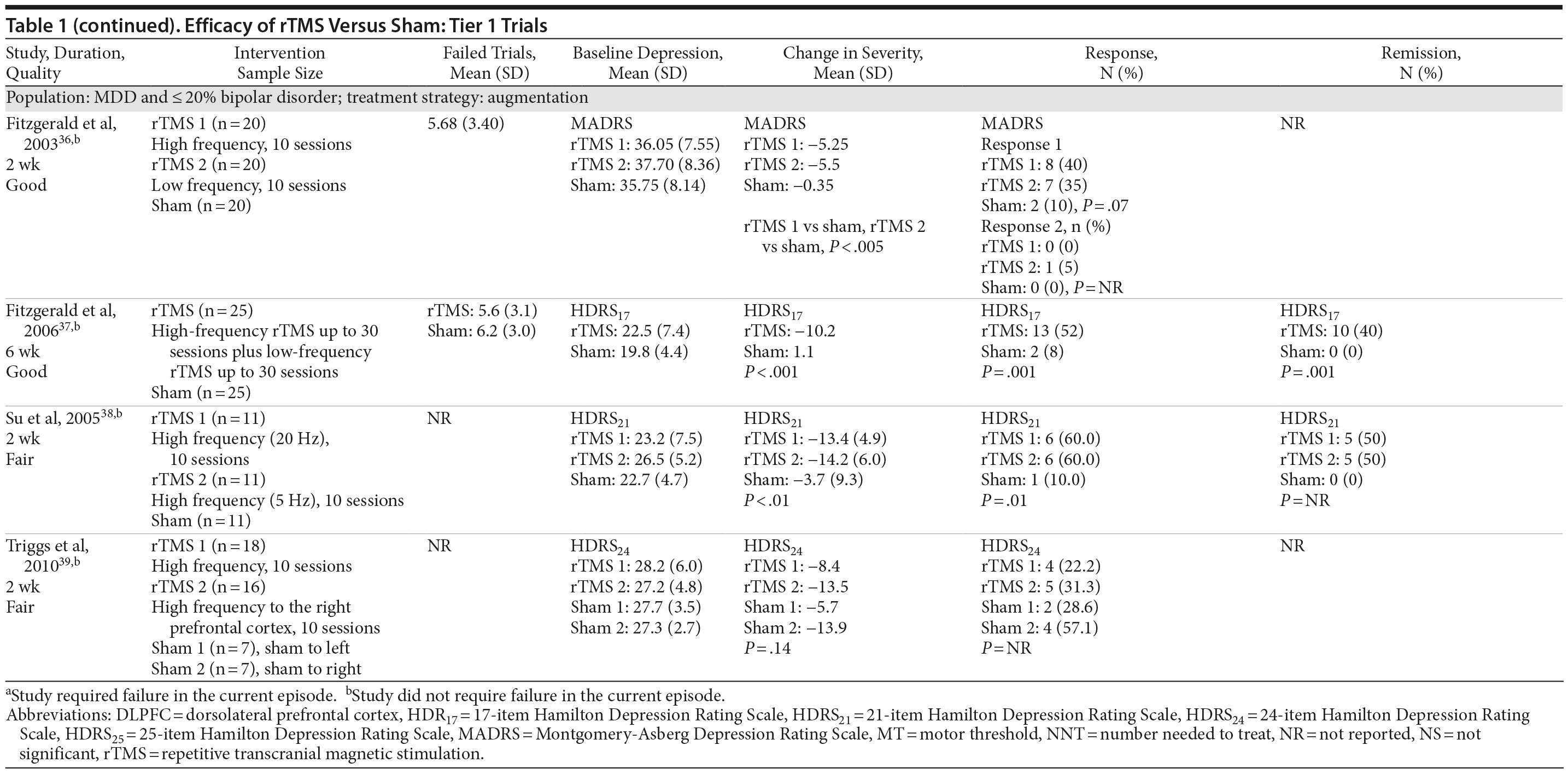
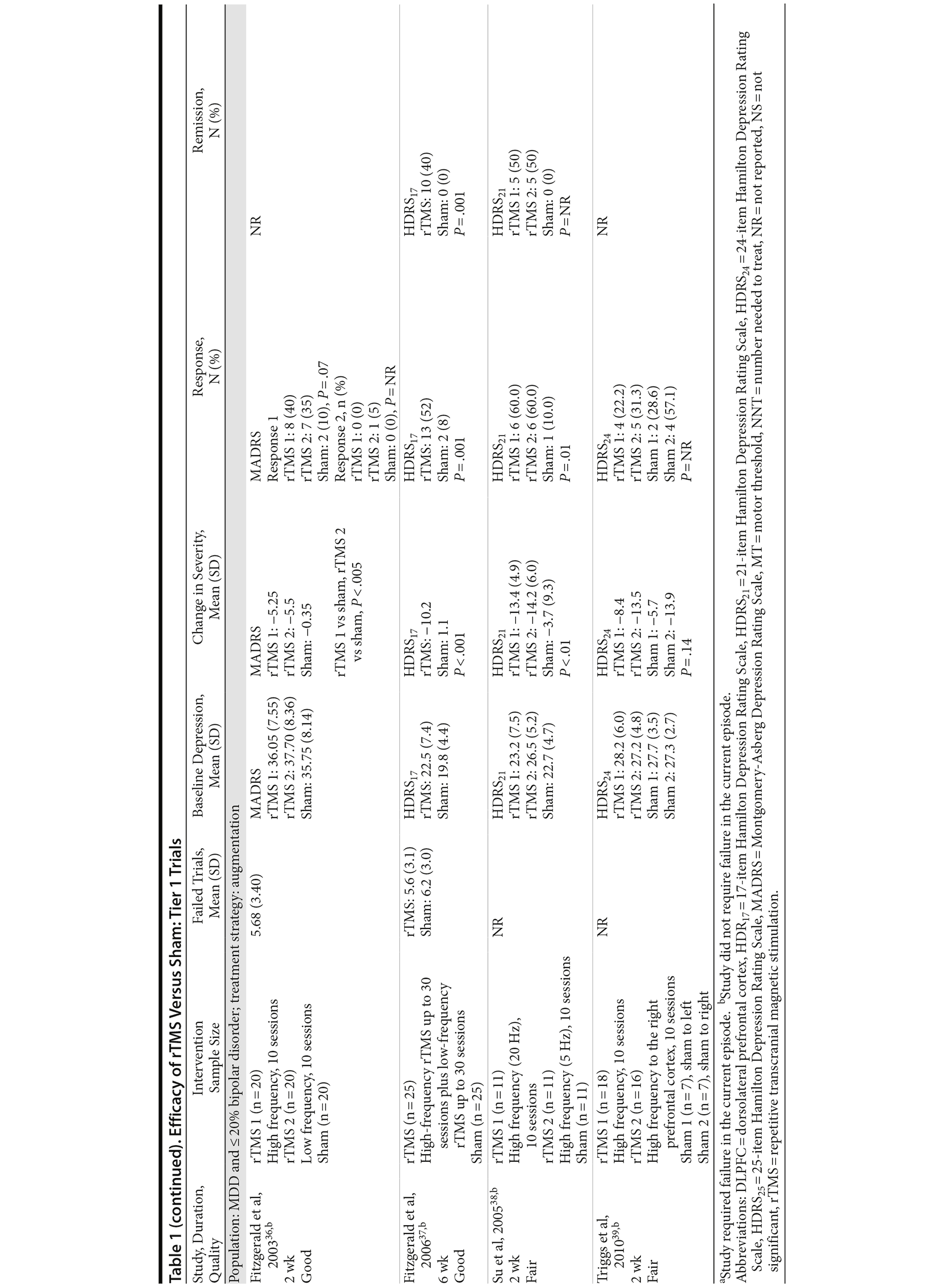
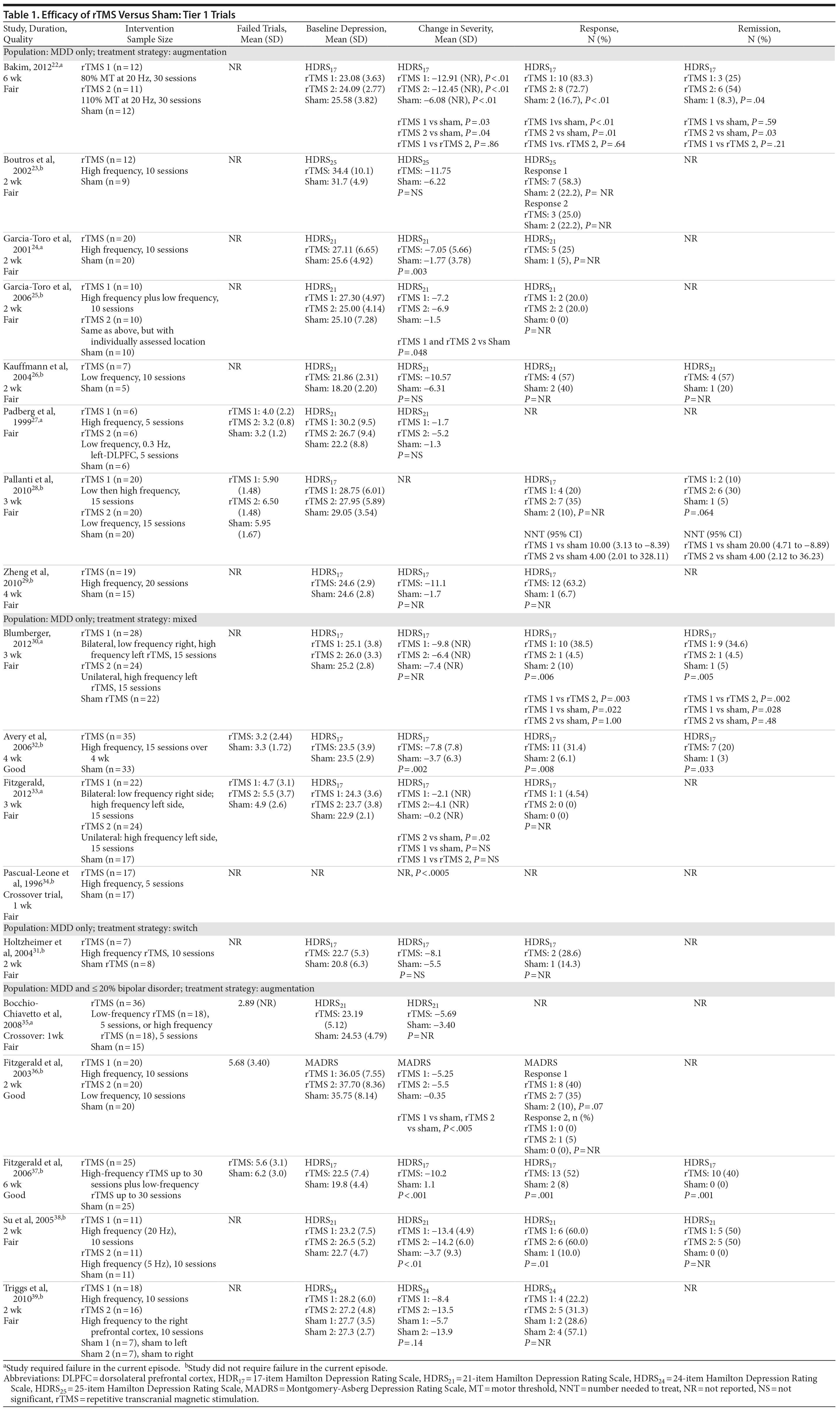
For our full review, we identified 3,179 citations through database and hand searching (Figure 1). For this article on rTMS interventions versus sham control, we included 35 published articles reporting on 27 trials involving all 3 tiers. For tier 1, we identified 18 trials of either good or fair quality: 13 MDD-only22-34 and 5 MDD/bipolar mix studies.35-39 Table 1 lists their characteristics, sorted first by diagnostic population involved (MDD-only or MDD/bipolar mix) and then by treatment strategy (whether rTMS is used as part of an augmentation strategy, a switch strategy, or a mixture of the 2). Data addressing the 3 primary outcomes are provided in the last 3 columns.
Of these 18 tier 1 trials, we rated 3 as good quality.32,36,37 Sample sizes ranged from 12 to 74 subjects; study duration ranged from 1 week to 6 weeks (mean = 2.67). Nearly three-fourths of the 18 trials (13 studies) used an augmentation strategy22-29,35-39; others (all MDD only) used a switch (1 study)31 or a mixed strategy (4 studies).30,32-34 No study gave information for all 3 outcomes, so the total number of trials providing data for any 1 outcome was fewer than 18.
Seven of these 18 trials reported the mean number of antidepressant treatment failures (range, 3.2-6.5); the remaining 10 trials either did not provide information beyond their eligibility requirement for ≥ 2 antidepressant treatment failures or reported only that all patients had had at least 2 failures. Six trials required failure to be in the current episode (rather than lifetime), 5 with MDD only22,24,27,30,33 and 1 with MDD/bipolar mix.35 Of the 18 trials, all but 1 gave information on baseline depressive severity (HDRS or MADRS): severe to very severe in 15 trials and moderate in 1 trial. Key treatment parameters, including stimulus intensity, pulse frequency, and stimulus duration, differed across the intervention trials.
Efficacy of rTMS Versus Sham for Acute-Phase Treatment of TRD (tier 1)
Results from MDD-only and from MDD/bipolar mix studies were in the same direction and of similar magnitude. Results from combining these 2 populations did not substantially differ from MDD-only results, suggesting that combining these 2 populations was reasonable. Therefore, we report findings based on the combined data.
We excluded 1 trial39 from our meta-analyses because an extensive, supportive social intervention for all treatment groups distinguished it from all other trials. This additional cointervention may have diminished the comparative efficacy of rTMS and sham stimulation, and it introduced considerable heterogeneity to the analyses.
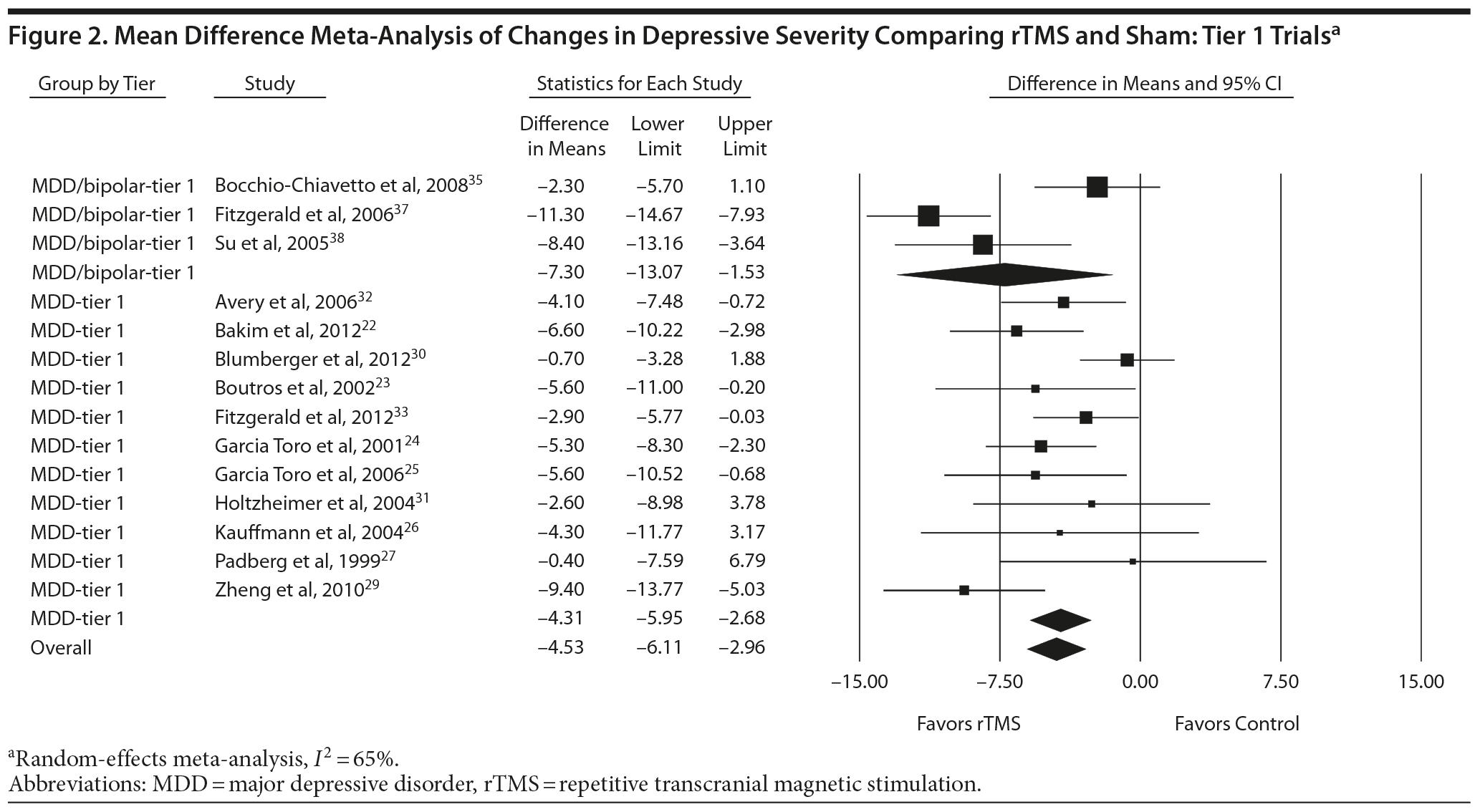
We made strength of evidence assessments for 3 outcomes: change in depressive severity, response rates, and remission rates. For changes in depressive severity, 17 trials of 1 to 6 weeks’ duration (N = 686) provided a high strength of evidence that rTMS produces a greater decrease in depressive severity than sham (Supplementary eTable 4).22-27,29-39 In the meta-analysis of 14 tier 1 trials (N = 564), rTMS produced a decrease in HDRS depressive severity of more than 4 points relative to sham (decrease in HDRS: −4.53; 95% CI, −6.11 to −2.96) (Figure 2).
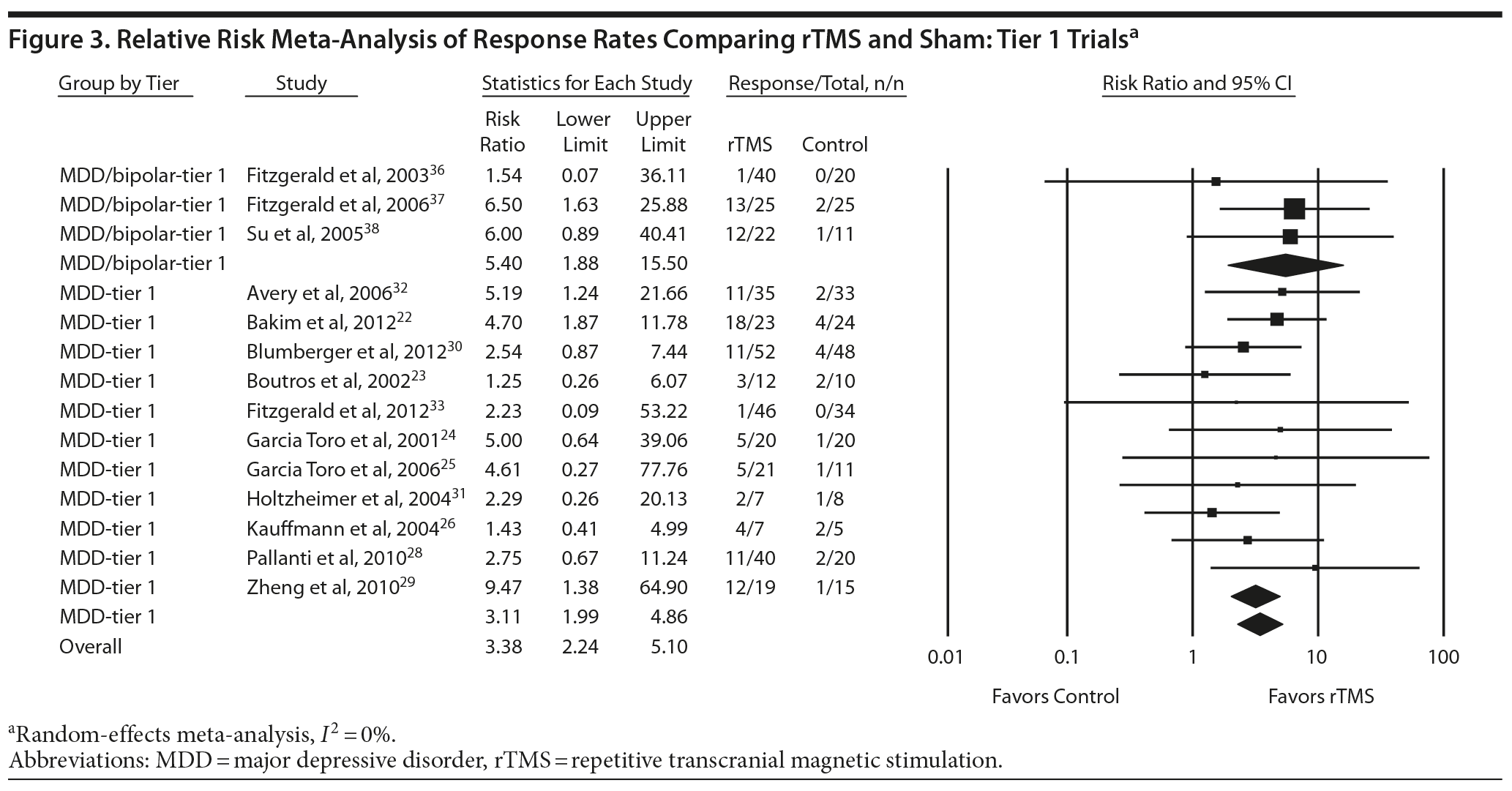
For response rates, 15 rTMS trials of 2 to 6 weeks’ duration (N = 643) provided a high strength of evidence that rTMS is more likely than sham to produce a response.22-26,28-33,36-39 The raw mean response rate was 29% for intervention patients and 8% for control patients. A meta-analysis of 14 tier 1 studies (N = 605) showed that patients receiving rTMS were more than 3 times as likely to achieve a depressive response as patients receiving sham intervention (pooled relative risk for response = 3.38; 95% CI, 2.24 to 5.10) (Figure 3). Given that response rates in the control groups varied widely (most likely because of many small studies), we conservatively calculated the NNT for the mean response rate in the sham groups in the 5 largest trials (sample size from 63 to 87); this control response rate averaged 5% (lower than the overall mean sham rate of 8%). For a population with a control response rate of 5% and study duration between 2 and 6 weeks, the NNT to achieve 1 additional response is 9 (95% CI, 5 to 16). By comparison, if the true control response rate were 10%, the NNT would be 5 (95% CI, 2 to 8).
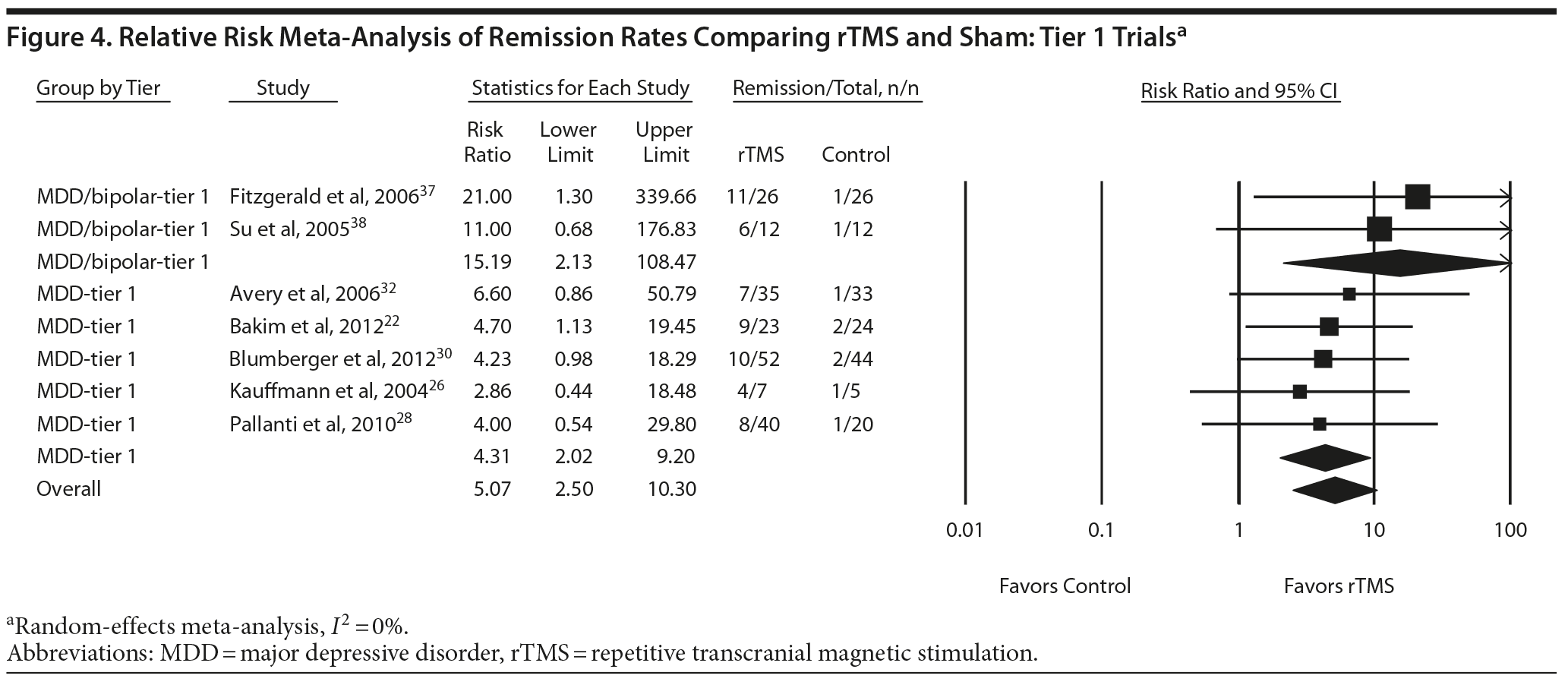
For remission rates, 7 tier 1 rTMS trials (N = 332) provided moderate strength of evidence that rTMS produces greater remission rates than sham.22,26,28,30,32,37,38 The raw mean remission rate was 30% for intervention patients and 6% for sham patients. Meta-analysis (all trials lasting between 2 and 6 weeks) showed that patients receiving rTMS were more than 5 times as likely to achieve remission as patients receiving sham control (pooled relative risk for remission = 5.07; 95% CI, 2.50 to 10.30) (Figure 4). In this population with a 6% spontaneous remission rate, the NNT would be 5 (95% CI, 2 to 11).
We considered how key clinical variables might affect our findings, but various data limitations prevented any firm conclusions. Given the small number and great variability of the tier 1 trials, we were unable to detect clear differences by treatment characteristics (ie, pharmacotherapy strategy, rTMS pulse frequency, stimulus intensity, stimulus duration, or treatment duration). Because nearly all patients were severely depressed, we could not detect any differences by severity of depression. Comparing effect sizes and CIs in the 3 studies that required TRD in the current episode with the effect sizes in trials that included patients with lifetime TRD suggested no differences; the small number of studies, however, prevented us from conducting a formal statistical test of no difference.
We assessed whether date of publication affected the size of the treatment effect. We found a statistically significant decrease in treatment effect of 0.45 HDRS points with each subsequent year of publication over a 17-year period (P < .0001).
Figures 2, 3, and 4 also allow comparison of the MDD-only groups with the MDD/bipolar mix groups. Point estimates for MDD/bipolar mix patients tended to be higher than those for MDD-only. However, the CIs overlapped, suggesting no clear difference between effect sizes in these 2 sets of patient populations. Combining the 2 populations did not affect the directionality of estimates or substantially influence the magnitude of estimates; the combined results were consistent with our findings for the separate tier 1 syntheses.
Efficacy of rTMS Versus Sham for Acute-Phase Treatment of TRD (all tiers)
We assessed whether our findings differed if we included studies from all 3 tiers. Adding tier 2 provided 6 additional trials (Supplementary eTable 5); 4 were MDD-only studies (reported in 5 articles)40-44 and 2 were MDD/bipolar mix studies.45,46 Adding tier 3 gave 3 more trials; these were MDD/bipolar mix populations reporting only change in depressive severity47-49 (Supplementary eTable 6).
Combining data from all 3 tiers produced results consistent with those from tier 1 alone but yielded more conservative point estimates and narrower CIs. The weighted mean difference in HDRS depressive severity was −4.81 (95% CI, −6.11 to −3.52). Because sample sizes of individual studies were small and responses to sham procedures varied in the small control groups, heterogeneity was high (I2 = 78%), and our estimates are uncertain with respect to the magnitude of changes on the HDRS.
Considering the additional studies reduced the intervention response rate slightly (to 26% from 29%) and increased the control response rate slightly (to 9% from 8%), the pooled relative risk indicated that patients receiving rTMS were more than twice as likely to respond than those receiving sham (pooled relative risk = 2.62; 95% CI, 1.93 to 3.56) (Supplementary eFigure 1); for a population with a control response rate of 9%, the NNT is 8 (95% CI, 5 to 13).
Remission rates also favored rTMS. Adding in the tier 2 and tier 3 trials decreased the intervention remission rate from 30% to 21%; the control remission rate remained at 6%. The pooled relative risk for remission was 2.76 (95% CI, 1.79 to 4.26) (Supplementary eFigure 2), which translates to a NNT of 10 (95% CI, 5 to 21) for a population with a control remission rate of 6%.
DISCUSSION
Repetitive transcranial magnetic stimulation was beneficial relative to patients receiving a sham procedure for all 3 outcomes—severity of depressive symptoms, response rate, and remission rate. For depressive severity (high strength of evidence), rTMS averaged a decrease in depressive severity measured by the HDRS of more than 4 points relative to sham control; this change meets the minimum threshold of the 3-point HDRS difference that is considered clinically meaningful.50 Response rates (which averaged 29%) were greater with rTMS than with sham (high strength of evidence); those receiving rTMS were more than 3 times as likely to respond. The NNT ranged between 9 (assuming a spontaneous response rate of 5%) and 5 (assuming a spontaneous response rate of 10%); as a comparison, the NNT for atypical antipsychotics in TRD is approximately 9.51 Finally, rTMS was also more likely to produce remission than sham (moderate strength of evidence); patients receiving rTMS had an average remission rate of 30% and were more than 5 times as likely to achieve remission. These response and remission rates are consistent with, if slightly more conservative than, rates seen with pharmacologic antidepressants following 2 antidepressant failures (38%-39% and 22%-27%, respectively).17 Further, the clinically meaningful benefit persists even when the stringent inclusion criteria requiring MDD and 2 treatment failures are relaxed.
A recent systematic review that considered the role of rTMS in TRD reported NNTs for remission similar to ours (an NNT of 5, assuming a spontaneous remission rate of 6%): 6 for a sample with a 9% spontaneous response rate, and 7 for a sample with a 6% spontaneous remission rate.52 Our analyses added 6 tier 1 studies published since then and applied a stricter and more current definition of TRD (2 or more failed antidepressants of adequate dose and duration, not just 1 or more failed antidepressant trials). Our approach thus represents treatment strategies more reflective of current practice. Furthermore, we could distinguish between study populations with MDD only and those with samples of at least 80% MDD patients and no more than 20% bipolar patients. Combining these 2 groups did not substantially affect our overall results, although in each case the MDD-only group had more precise and conservative estimates of benefit.
Our results also extend results from recent systematic reviews that have underscored the efficacy of rTMS as a general treatment for MDD (not just TRD). Compared with placebo, the response and remission rates for low-frequency rTMS were, respectively, 38.2% versus 15.1% (P = .007) and 34.6% versus 9.7% (P < .001)53; for high frequency rTMS, the rates were, respectively, 29.3% versus 10.4% (P < .0001) and 18.6% versus 5% (P < .0001)11; and for bilateral rTMS, they were 24.7% versus 6.8% (P < .0001) and 19% versus 2.6% (P < .006).54 Our results mirror these earlier findings, considering all tiers for response rates (26%) and remission rates (21%). Each of these 3 reviews had produced NNTs between 5 and 7; these estimates are consistent with our NNT of 7 for response (all tiers) but slightly more beneficial than our NNT of 10 for remission (all tiers). Taken together, these findings suggest benefit for populations that have been stringently defined as TRD and for those including patients likely to have TRD.
Accordingly, our analyses add to the evidence base supporting benefit for rTMS as a general class of treatments for patients with TRD. Initial FDA approval for rTMS involved a specific treatment device with an indication for patients who have not responded to 1 adequate trial of antidepressant medication. The evidence for the regulatory approval involved a secondary data analysis of a trial of patients with nonpsychotic MDD; the number of prior treatment failures was the strongest predictor for positive response to acute treatment with TMS.55 In that analysis using change in MADRS as the primary outcome, patients with 1 adequate antidepressant trial (N = 164) had a significantly greater change from baseline depressive severity than the sham group, whereas those who failed 2 to 4 adequate trials (N = 137) could not be distinguished from the sham group.
In contrast, our analyses of rTMS trials involving a variety of treatment parameters for patients having failed 2 or more antidepressant trials showed an rTMS benefit in depressive severity (N = 564, 14 trials in meta-analysis) averaging more than 4 points (a clinically meaningful difference). Moreover, benefits for rTMS versus sham were greater for both rates of response (N = 605, 14 studies) and remission (N = 332, 7 studies). These findings are more in line with information supplied for the second rTMS device for MDD with FDA approval, which did not restrict the indication to patients having failed a single treatment.14
Decisions to use an effective intervention must always be weighed by considerations of harms. Patients tend to tolerate rTMS well.56,57 The greatest concern focuses on the increased risk of seizures with rTMS58; the risk of seizure in individuals with no prior history of seizure disorder may not be greater than that associated with oral antidepressant treatment; the incidence is approximately 0.01% among people with no prior history.59 Our earlier work has shown low or insufficient evidence addressing this issue.17
The available data had some limitations. The adequacy of sham controls in earlier rTMS studies has been a persistent concern. In earlier studies, patients were likely to be able to distinguish between the sham and intervention arms and hence might favor rTMS.44,60 Such an effect might bias results in favor of rTMS in TRD studies. Consequently, more recent trials have made greater efforts to ensure high-quality sham procedures; this improvement is reflected in our findings of a statistically significant decrease in treatment effect of 0.45 HDRS points per year.
Even with this decrease, however, more recent trials remained strongly positive for rTMS benefit. Our response and remission relative risks (3.38 and 5.07, respectively) were consistent with the 3 most recent trial results (ranging from 2.23 to 4.70 and from 4.23 to 4.70, respectively); they were also similar to results from the George et al clinical trial,44 a tier 2 MDD-only study with most likely the best sham control to date (4.6 and 4.2, respectively). Finally, a recent meta-analysis61 of the blinding integrity of sham-controlled rTMS trials for MDD found that commonly used sham methods appear to be adequate for concealing treatment allocation.
In addition, treatment lengths ranged from 1 to 6 weeks (mean = 2.67). Given the few tier 1 trials, we were unable to detect clear differences by rTMS frequency. However, the 4 trials with the 2 longest treatment periods (4 weeks29,32 or 6 weeks22,37) each had response and remission relative risks (ranging from 4.70 to 9.47 and from 4.70 to 21.0, respectively) that exceeded our meta-analytic results. This finding suggests that our results may underestimate rTMS effects. Further, the average number of sessions in our sample (12.75) is equivalent to the 12 to 13 sessions considered standard by other reviews.11,53,54
Ideally, all studies we meta-analyzed would have used the 17-item HDRS version; however, 7 did not. Sensitivity analyses including only the 17-item versions did not change our estimates.
Also, given the limited amount of relevant data in tier 1 trials, we could not explore how other potential effect modifiers such as pharmacotherapy strategy, severity of depression, and treatment resistance in the current episode (vs in previous episodes) might affect outcomes.
Publication bias is a concern for any systematic review. Although we conducted comprehensive searches of the gray literature, we cannot determine conclusively whether we missed any relevant unpublished studies. Statistical tests and funnel plots did not indicate publication bias in our meta-analyses. However, we acknowledge that the power and reliability of such tests are low. To assess the potential impact of publication bias on the results of our meta-analyses, we conducted fail-safe n tests. Findings showed that for all of our meta-analyses it would require more than 20 unpublished studies to change statistically significant findings to nonstatistically significant results.
We uncovered no information about maintenance therapy following completion of rTMS treatment for TRD. No studies assessed outcome beyond a week following the final session of rTMS (or sham) treatment, so no data were available to assess the persistence of the positive effect in this population.
Finally, our results reflect an assessment of the degree to which, in general, rTMS interventions can offer benefit to patients with TRD. Repetitive transcranial magnetic stimulation is an emerging field; in its current stage of development, it is best understood as a general class of treatments whose most effective elements are still being actively researched. The limited number of studies to date prevented us from performing subgroup analyses to determine how specific treatment parameters affect outcomes for different types of TRD patients. Accordingly, our results indicate an overall effect for the general class of rTMS interventions and represent an important first step toward defining the role of rTMS in TRD.
CONCLUSIONS AND FUTURE DIRECTIONS
The use of nonpharmacologic interventions in TRD patients is a key clinical issue; clinicians and researchers are only beginning to identify the role of rTMS for this hard-to-treat population. For MDD patients with 2 or more antidepressant treatment failures, rTMS is a reasonable, effective consideration. Further research to clarify rTMS use in TRD patients should reflect improved trial designs and, thus, help to clarify which key treatment parameters may be relevant to TRD. In particular, investigators need to apply a consistent definition of TRD; carefully delineate the number of adequate pharmacologic failures in the current depressive episode; clarify standard parameters of optimal treatment, including stimulus intensity, pulse frequency, stimulus duration, and length of treatment; and specify whether rTMS is used as augmentation or as a switch strategy. Comparative effectiveness designs, which could involve direct comparisons of varying rTMS parameters and of rTMS with other TRD treatments (eg, electroconvulsive therapy or combination medications) would help identify these optimal parameters. Longer trials or follow-up periods will help to clarify whether treatment response is maintained over a period longer than just a few weeks.
Disclosure of off-label usage: The authors have determined that, to the best of their knowledge, while the US Food and Drug Administration has approved Neurostar “for the treatment of major depressive disorder in adult patients who have failed to achieve satisfactory improvement from one prior antidepressant medication at or above the minimal effective dose and duration in the current episode,” findings in this article relate to rTMS devices (including Neurostar) for patients with 2 or more prior antidepressant failures.
Author affiliations: Department of Psychiatry (Dr Gaynes), Cecil G. Sheps Center for Health Services Research (Drs Gaynes and Jonas and Ms Brode), and Department of Medicine (Dr Jonas), University of North Carolina, Chapel Hill; Research Triangle Institute (RTI) International, Research Triangle Park, North Carolina (Drs Garlehner, Viswanathan, and Lohr and Mss Lloyd, Lux, and Evans); Danube University, Krems, Austria (Dr Gartlehner); and Harrison School of Pharmacy, Auburn University, Auburn, Alabama (Dr Hansen). Ms Brode is now with Hayes, Inc, Lansdale, Pennsylvania.
Financial disclosure: Dr Hansen has received research support from Novartis and Daiichi Sankyo and has served as an expert witness for Allergan. Drs Gaynes, Gartlehner, Jonas, Viswanathan, and Lohr and Mss Lloyd, Lux, Brode, and Evans have no personal affiliations or financial relationships with any commercial interest to disclose relative to the article.
Funding/support: This project was funded under Contract No. 290-02-0016I, TO #2 provided to RTI International for the RTI-University of North Carolina (RTI-UNC) Evidence-based Practice Center from the Agency for Healthcare Research and Quality (AHRQ), US Department of Health and Human Services.
Role of the sponsor: The AHRQ participated in formulating the key questions and reviewed and commented on planned methods for the larger comparative effectiveness review. The AHRQ had no role in study selection, quality ratings, strength of evidence grades, or interpretation and synthesis of the evidence, although AHRQ staff reviewed interim and final evidence reports and oversaw distribution of these for external peer review by outside experts. The authors of this report are responsible for its content.
Disclaimer: Statements in the report should not be construed as endorsement by the AHRQ or the US Department of Health and Human Services.
Previous presentation: Presented in part at the 52nd Annual New Clinical Drug Evaluation Unit (NCDEU) Meeting; May 29, 2012; Phoenix, Arizona.
Supplementary material: Available at PSYCHIATRIST.COM.
REFERENCES
1. Kessler RC, Berglund P, Demler O, et al; National Comorbidity Survey Replication. The epidemiology of major depressive disorder: results from the National Comorbidity Survey Replication (NCS-R). JAMA. 2003;289(23):3095-3105. PubMed doi:10.1001/jama.289.23.3095
2. Wang PS, Lane M, Olfson M, et al; Twelve-month use of mental health services in the United States: results from the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62(6):629-640. PubMed doi:10.1001/archpsyc.62.6.629
3. Trivedi MH, Rush AJ, Wisniewski SR, et al; STAR*D Study Team. Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: implications for clinical practice. Am J Psychiatry. 2006;163(1):28-40. PubMed doi:10.1176/appi.ajp.163.1.28
4. Thase ME, Rush AJ. When at first you don’ t succeed: sequential strategies for antidepressant nonresponders. J Clin Psychiatry. 1997;58(suppl 13):23-29. PubMed
5. Rush AJ, Trivedi MH, Wisniewski SR, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163(11):1905-1917. PubMed doi:10.1176/appi.ajp.163.11.1905
6. Berlim MT, Fleck MP, Turecki G. Current trends in the assessment and somatic treatment of resistant/refractory major depression: an overview. Ann Med. 2008;40(2):149-159. PubMed doi:10.1080/07853890701769728
7. Berlim MT, Turecki G. What is the meaning of treatment resistant/refractory major depression (TRD)? A systematic review of current randomized trials. Eur Neuropsychopharmacol. 2007;17(11):696-707. PubMed doi:10.1016/j.euroneuro.2007.03.009
8. Gaynes BN. Identifying difficult-to-treat depression: differential diagnosis, subtypes, and comorbidities. J Clin Psychiatry. 2009;70(suppl 6):10-15. PubMed doi:10.4088/JCP.8133su1c.02
9. Russell JM, Hawkins K, Ozminkowski RJ, et al. The cost consequences of treatment-resistant depression. J Clin Psychiatry. 2004;65(3):341-347. PubMed doi:10.4088/JCP.v65n0309
10. Gartlehner G, Gaynes BN, Hansen RA, et al. Comparative benefits and harms of second-generation antidepressants: background paper for the American College of Physicians. Ann Intern Med. 2008;149(10):734-750. PubMed doi:10.7326/0003-4819-149-10-200811180-00008
11. Berlim MT, van den Eynde F, Tovar-Perdomo S, et al. Response, remission and drop-out rates following high-frequency repetitive transcranial magnetic stimulation (rTMS) for treating major depression: a systematic review and meta-analysis of randomized, double-blind and sham-controlled trials. Psychol Med. 2014;44(2):225-239. PubMed
12. American Psychiatric Association. Practice Guideline for the Treatment of Patients With Major Depressive Disorder. 3rd ed. Washington, DC: American Psychiatric Association; 2010.
13. Melkerson MN. Special Premarket 510(k) Notification for NeuroStar TMS Therapy System for Major Depressive Disorder. 2008. http://www.accessdata.fda.gov/cdrh_docs/pdf8/K083538.pdf. Updated December 16, 2008. Accessed March 19, 2014.
14. FDA. 510(K) Summary Brainsway Deep TMS System. July 10, 2012. www.accessdata.fda.gov/cdrh_docs/pdf12/K122288.pdf. Updated January 7, 2013. Accessed March 19, 2014.
15. Appleby J. Magnetic stimulation helps some people with treatment-resistant depression. Washington Post. August 6, 2012: E1.
16. The New England Comparative Effectiveness Public Advisory Council. Treatment Options for Treatment-Resistant Depression: Repetitive Transcranial Magnetic Stimulation. 2012. http://cepac.icer-review.org/wp-content/uploads/2011/04/rTMS-Coverage-Policy-Analysis.pdf. Accessed March 14, 2014.
17. Gaynes BN, Lux L, Lloyd S, et al. KN L. Nonpharmacologic Interventions for Treatment-Resistant Depression in Adults. Comparative Effectiveness Review No. 33. Rockville, MD: Agency for Healthcare Research and Quality; 2011.
18. US Preventive Services Task Force. US Preventive Services Task Force Grade Definitions. 2008: http://www.uspreventiveservicestaskforce.org/uspstf/grades.htm. Accessed March 19, 2014.
19. Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23(1):56-62. PubMed doi:10.1136/jnnp.23.1.56
20. Sterne JAC, Sutton AJ, Ioannidis JPA, et al. Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomised controlled trials. BMJ. 2011;343(1):d4002. PubMed doi:10.1136/bmj.d4002
21. Owens DK, Lohr KN, Atkins D, et al. AHRQ series paper 5: grading the strength of a body of evidence when comparing medical interventions—agency for healthcare research and quality and the effective health-care program. J Clin Epidemiol. 2010;63(5):513-523. PubMed doi:10.1016/j.jclinepi.2009.03.009
22. Bakim B, Uzun UE, Karamustafalioglu O, et al. The combination of antidepressant drug therapy and high-frequency repetitive transcranial magnetic stimulation in medication-resistant depression. Klinik Psikofarmakoloji Bulteni. 2012;22(3):244-253.
23. Boutros NN, Gueorguieva R, Hoffman RE, et al. Lack of a therapeutic effect of a 2-week sub-threshold transcranial magnetic stimulation course for treatment-resistant depression. Psychiatry Res. 2002;113(3):245-254.
24. Garcia-Toro M, Mayol A, Arnillas H, et al. Modest adjunctive benefit with transcranial magnetic stimulation in medication-resistant depression. J Affect Disord. 2001;64(2-3):271-275. PubMed doi:10.1016/S0165-0327(00)00223-8
25. Garcia-Toro M, Salva J, Daumal J, et al. High (20-Hz) and low (1-Hz) frequency transcranial magnetic stimulation as adjuvant treatment in medication-resistant depression. Psychiatry Res. 2006;146(1):53-57. PubMed doi:10.1016/j.pscychresns.2004.08.005
26. Kauffmann CD, Cheema MA, Miller BE. Slow right prefrontal transcranial magnetic stimulation as a treatment for medication-resistant depression: a double-blind, placebo-controlled study. Depress Anxiety. 2004;19(1):59-62. PubMed doi:10.1002/da.10144
27. Padberg F, Zwanzger P, Thoma H, et al. Repetitive transcranial magnetic stimulation (rTMS) in pharmacotherapy-refractory major depression: comparative study of fast, slow and sham rTMS. Psychiatry Res. 1999;88(3):163-171. PubMed doi:10.1016/S0165-1781(99)00092-X
28. Pallanti S, Bernardi S, Di Rollo A, et al. Unilateral low frequency versus sequential bilateral repetitive transcranial magnetic stimulation: is simpler better for treatment of resistant depression? Neuroscience. 2010;167(2):323-328. PubMed doi:10.1016/j.neuroscience.2010.01.063
29. Zheng H, Zhang L, Li L, et al. High-Frequency rTMS Treatment Increases Left Prefrontal Myo-Inositol in Young Patients With Treatment-Resistant Depression: Progress in Neuro-Psychopharmacology & Biological Psychiatry. Amsterdam, the Netherlands: Elsevier Science; 2010:1189-1195.
30. Blumberger DM, Mulsant BH, Fitzgerald PB, et al. A randomized double-blind sham-controlled comparison of unilateral and bilateral repetitive transcranial magnetic stimulation for treatment-resistant major depression. World J Biol Psychiatry. 2012;13(6):423-435. PubMed doi:10.3109/15622975.2011.579163
31. Holtzheimer PE 3rd, Russo J, Claypoole KH, et al. Shorter duration of depressive episode may predict response to repetitive transcranial magnetic stimulation. Depress Anxiety. 2004;19(1):24-30. PubMed doi:10.1002/da.10147
32. Avery DH, Holtzheimer PE 3rd, Fawaz W, et al. A controlled study of repetitive transcranial magnetic stimulation in medication-resistant major depression. Biol Psychiatry. 2006;59(2):187-194. PubMed doi:10.1016/j.biopsych.2005.07.003
33. Fitzgerald PB, Hoy KE, Herring SE, et al. A double blind randomized trial of unilateral left and bilateral prefrontal cortex transcranial magnetic stimulation in treatment resistant major depression. J Affect Disord. 2012;139(2):193-198. PubMed doi:10.1016/j.jad.2012.02.017
34. Pascual-Leone A, Rubio B, Pallardó F, et al. Rapid-rate transcranial magnetic stimulation of left dorsolateral prefrontal cortex in drug-resistant depression. Lancet. 1996;348(9022):233-237. PubMed doi:10.1016/S0140-6736(96)01219-6
35. Bocchio-Chiavetto L, Miniussi C, Zanardini R, et al. 5-HTTLPR and BDNF Val66Met polymorphisms and response to rTMS treatment in drug resistant depression. Neurosci Lett. 2008;437(2):130-134. PubMed doi:10.1016/j.neulet.2008.04.005
36. Fitzgerald PB, Brown TL, Marston NA, et al. Transcranial magnetic stimulation in the treatment of depression: a double-blind, placebo-controlled trial. Arch Gen Psychiatry. 2003;60(10):1002-1008. PubMed
37. Fitzgerald PB, Benitez J, de Castella A, et al. A randomized, controlled trial of sequential bilateral repetitive transcranial magnetic stimulation for treatment-resistant depression. Am J Psychiatry. 2006;163(1):88-94. PubMed doi:10.1176/appi.ajp.163.1.88
38. Su T-P, Huang C-C, Wei IH. Add-on rTMS for medication-resistant depression: a randomized, double-blind, sham-controlled trial in Chinese patients. J Clin Psychiatry. 2005;66(7):930-937. PubMed doi:10.4088/JCP.v66n0718
39. Triggs WJ, Ricciuti N, Ward HE, et al. Right and left dorsolateral pre-frontal rTMS treatment of refractory depression: a randomized, sham-controlled trial. Psychiatry Res. 2010;178(3):467-474. PubMed doi:10.1016/j.psychres.2010.05.009
40. Manes F, Jorge R, Morcuende M, et al. A controlled study of repetitive transcranial magnetic stimulation as a treatment of depression in the elderly. Int Psychogeriatr. 2001;13(2):225-231. PubMed doi:10.1017/S1041610201007608
41. Moser DJ, Jorge RE, Manes F, et al. Improved executive functioning following repetitive transcranial magnetic stimulation. Neurology. 2002;58(8):1288-1290. PubMed doi:10.1212/WNL.58.8.1288
42. Stern WM, Tormos JM, Press DZ, et al. Antidepressant effects of high and low frequency repetitive transcranial magnetic stimulation to the dorsolateral prefrontal cortex: a double-blind, randomized, placebo-controlled trial. J Neuropsychiatry Clin Neurosci. 2007;19(2):179-186. PubMed doi:10.1176/appi.neuropsych.19.2.179
43. O’ Reardon JP, Solvason HB, Janicak PG, et al. Efficacy and safety of transcranial magnetic stimulation in the acute treatment of major depression: a multisite randomized controlled trial. Biol Psychiatry. 2007;62(11):1208-1216. PubMed doi:10.1016/j.biopsych.2007.01.018
44. George MS, Lisanby SH, Avery D, et al. Daily left prefrontal transcranial magnetic stimulation therapy for major depressive disorder: a sham-controlled randomized trial. Arch Gen Psychiatry. 2010;67(5):507-516. PubMed doi:10.1001/archgenpsychiatry.2010.46
45. Berman RM, Narasimhan M, Sanacora G, et al. A randomized clinical trial of repetitive transcranial magnetic stimulation in the treatment of major depression. Biol Psychiatry. 2000;47(4):332-337. PubMed doi:10.1016/S0006-3223(99)00243-7
46. Bretlau LG, Lunde M, Lindberg L, et al. Repetitive transcranial magnetic stimulation (rTMS) in combination with escitalopram in patients with treatment-resistant major depression: a double-blind, randomised, sham-controlled trial. Pharmacopsychiatry. 2008;41(2):41-47. PubMed doi:10.1055/s-2007-993210
47. Bortolomasi M, Minelli A, Fuggetta G, et al. Long-lasting effects of high frequency repetitive transcranial magnetic stimulation in major depressed patients. Psychiatry Res. 2007;150(2):181-186. PubMed doi:10.1016/j.psychres.2006.04.010
48. George MS, Wassermann EM, Kimbrell TA, et al. Mood improvement following daily left prefrontal repetitive transcranial magnetic stimulation in patients with depression: a placebo-controlled crossover trial. Am J Psychiatry. 1997;154(12):1752-1756. PubMed
49. Möller AL, Hjaltason O, Ivarsson O, et al. The effects of repetitive transcranial magnetic stimulation on depressive symptoms and the P(300) event-related potential. Nord J Psychiatry. 2006;60(4):282-285. PubMed doi:10.1080/08039480600790119
50. National Institute for Clinical Excellence. Depression: Management of Depression in Primary and Secondary Care. Clinical Practice Guideline No. 23. London, UK: National Institute for Clinical Excellence; 2004.
51. Spielmans GI, Berman MI, Linardatos E, et al. Adjunctive atypical antipsychotic treatment for major depressive disorder: a meta-analysis of depression, quality of life, and safety outcomes. PLoS Med. 2013;10(3):e1001403. PubMed doi:10.1371/journal.pmed.1001403
52. Lam RW, Chan P, Wilkins-Ho M, et al. Repetitive transcranial magnetic stimulation for treatment-resistant depression: a systematic review and meta-analysis. Can J Psychiatry. 2008;53(9):621-631. PubMed
53. Berlim MT, Van den Eynde F, Jeff Daskalakis Z. Clinically meaningful efficacy and acceptability of low-frequency repetitive transcranial magnetic stimulation (rTMS) for treating primary major depression: a meta-analysis of randomized, double-blind and sham-controlled trials. Neuropsychopharmacology. 2013;38(4):543-551. PubMed doi:10.1038/npp.2012.237
54. Berlim MT, Van den Eynde F, Daskalakis ZJ. A systematic review and meta-analysis on the efficacy and acceptability of bilateral repetitive transcranial magnetic stimulation (rTMS) for treating major depression. Psychol Med. 2013;43(11):2245-2254. PubMed
55. Lisanby SH, Husain MM, Rosenquist PB, et al. Daily left prefrontal repetitive transcranial magnetic stimulation in the acute treatment of major depression: clinical predictors of outcome in a multisite, randomized controlled clinical trial. Neuropsychopharmacology. 2009;34(2):522-534. PubMed doi:10.1038/npp.2008.118
56. George MS, Aston-Jones G. Noninvasive techniques for probing neurocircuitry and treating illness: vagus nerve stimulation (VNS), transcranial magnetic stimulation (TMS) and transcranial direct current stimulation (tDCS). Neuropsychopharmacology. 2010;35(1):301-316. PubMed doi:10.1038/npp.2009.87
57. Wassermann EM, Zimmermann T. Transcranial magnetic brain stimulation: therapeutic promises and scientific gaps. Pharmacol Ther. 2012;133(1):98-107. PubMed doi:10.1016/j.pharmthera.2011.09.003
58. Belmaker B, Fitzgerald P, George MS, et al. Managing the risks of repetitive transcranial stimulation. CNS Spectr. 2003;8(7):489. PubMed
59. Lee JC, Blumberger DM, Fitzgerald PB, et al. The role of transcranial magnetic stimulation in treatment-resistant depression: a review. Curr Pharm Des. 2012;18(36):5846-5852. PubMed doi:10.2174/138161212803523644
60. Rossi S, Hallett M, Rossini PM, et al; Safety of TMS Consensus Group. Safety, ethical considerations, and application guidelines for the use of transcranial magnetic stimulation in clinical practice and research. Clin Neurophysiol. 2009;120(12):2008-2039. PubMed doi:10.1016/j.clinph.2009.08.016
61. Berlim MT, Broadbent HJ, Van den Eynde F. Blinding integrity in randomized sham-controlled trials of repetitive transcranial magnetic stimulation for major depression: a systematic review and meta-analysis. Int J Neuropsychopharmacol. 2013;16(5):1173-1181. PubMed doi:10.1017/S1461145712001691