ABSTRACT
Objective: To estimate the risk of road traffic crash associated with prescription of antidepressants.
Method: Data were extracted and matched from 3 French national databases: the national health care insurance database, police reports, and the national police database of injurious crashes. A case-control analysis comparing 34,896 responsible versus 37,789 nonresponsible drivers was conducted. Case-crossover analysis was performed to investigate the acute effect of medicine exposure.
Results: 72,685 drivers, identified by their national health care number, involved in an injurious crash in France from July 2005 to May 2008 were included. 2,936 drivers (4.0%) were exposed to at least 1 antidepressant on the day of the crash. The results showed a significant association between the risk of being responsible for a crash and prescription of antidepressants (odds ratio [OR] = 1.34; 95% CI, 1.22-1.47). The case-crossover analysis showed no association with treatment prescription, but the risk of road traffic crash increased after an initiation of antidepressant treatment (OR = 1.49; 95% CI, 1.24-1.79) and after a change in antidepressant treatment (OR = 1.32; 95% CI, 1.09-1.60).
Conclusions: Patients and prescribers should be warned about the risk of crash during periods of treatment with antidepressant medication and about particularly high vulnerability periods such as those when a treatment is initiated or modified.
J Clin Psychiatry 2012;73(8):1088-1094
© Copyright 2012 Physicians Postgraduate Press, Inc.
Submitted: December 29, 2011; accepted April 18, 2012 (doi:10.4088/JCP.11m07624).
Corresponding author: Ludivine Orriols, PhD, Equipe Prévention et Prise en Charge des Traumatismes, Centre de Recherche INSERM U897 “Epidémiologie et Biostatistiques” Université Bordeaux Segalen, Case 11, 146 rue Léo Saignat, 33076 Bordeaux Cedex, France ([email protected]).
Experimental studies conducted on healthy volunteers have shown a deleterious effect on driving of sedating antidepressants such as amitriptyline1,2 (tricyclic antidepressant) and mirtazapine3,4 (tetracyclic antidepressant). Paroxetine,3,5-7 fluoxetine8 (selective serotonin reuptake inhibitors), and venlafaxine9 (serotonin-norepinephrine reuptake inhibitor) are nonsedating antidepressants and appear to have a smaller potential for impairing performance. Driving performance of depressed patients receiving long-term antidepressant treatment has been shown to be impaired,10 but antidepressant treatment can also increase driving safety by improving clinical conditions, including sleepiness.11
A clinical study12 comparing the effects of different antidepressant treatments on driving performance in depressed patients showed an advantage for patients receiving serotonin reuptake inhibitors or mirtazapine compared with tricyclic antidepressants or venlafaxine. Another study13 showed that depressed patients treated with reboxetine (nonsedating antidepressant) or mirtazapine performed better on tasks related to driving than untreated depressive patients. Two epidemiologic studies conducted in the elderly have shown an association between use of tricyclic antidepressants and the risk of traffic crashes (relative risk = 2.2 [95% CI, 1.3-3.5]14 and odds ratio [OR] = 2.3 [95% CI, 1.1-4.8]15). In contrast, Barbone et al16 found no association for tricyclic antidepressants or serotonin reuptake inhibitors in a 3-year study of 19,386 English drivers 18 years or older who were involved in a crash. That study, however, had a case-crossover design that is likely to underestimate the risk in people taking long-term therapies. The most recent study,17 conducted from 2004 to 2006, showed an increased risk for drivers following prescription of sedating or nonsedating antidepressants (standardized incidence ratio [SIR] = 1.4 [95% CI, 1.2-1.6] and SIR = 1.6 [95% CI, 1.5-1.7], respectively), the risk being highest among younger drivers taking sedating antidepressants. The authors of this study discussed their inability to control for the use of alcohol or other medicines such as benzodiazepines, as well as for depression, all of these being factors known to increase the risk of traffic crash.
The results of the few available epidemiologic studies have therefore been inconclusive. For some antidepressants, initiation of treatment seems to be critical for driving performance.4,6,18 Bramness et al17 reported similar risk estimates among incident users and prevalent users of nonsedating antidepressants. The aim of our study was to estimate the risk of road traffic crashes associated with prescription of antidepressants.
- Periods of initiation and change of antidepressant treatment are critical periods with regard to the risk of road traffic crash.
- Prescribers should warn their patients about this risk.
METHOD
This study was approved by the French Data Protection Authority.
The study consisted of extracting data from 3 French nationwide databases, as described previously.19 A case-control analysis comparing responsible versus nonresponsible drivers was conducted. Case-crossover analysis was performed to investigate the acute effect of antidepressant exposure.
Data Sources
Police reports. French police forces are required to fill out a police report for each injurious crash occurring in the country (about 70,000 reports each year). Police reports are scanned and stored as image files. For some of the drivers involved in these injurious road traffic crashes, the national health care ID number (NID) is recorded in the police report. These NIDs were extracted from police report image files for later matching against dispensing records in the national health care insurance database. All 210,818 police reports available over the study period (from July 2005 to May 2008) were compiled.
National police database of injurious crashes. The injurious crashes database contains descriptive variables on the crash characteristics and the vehicles and people involved in the crash. Police forces also conduct additional investigations regarding injury severity from hospital records and categorize the people involved into 4 groups: unhurt, slightly injured, seriously injured (hospitalized more than 24 hours), or killed (died in the 30 days following the crash). All drivers involved in an injurious road traffic crash are supposed to be submitted to a breath alcohol test. If this test is positive (≥ 0.5 g/L), if the driver refuses to take the test, or if the severity of the crash makes the test impossible, then the driver’s blood alcohol concentration is measured. If the breath test is negative, then the driver is registered as not being under the influence of alcohol.
Health care insurance database. The health care insurance database covers the entire French population (64,000,000 in 2008) and includes data on reimbursed prescription medicines. A record is entered into the database each time a prescription medicine is dispensed to an outpatient at the pharmacy, including the NID, the date of dispensing, and the 7-digit code assigned to the medicine at the time of its marketing authorization. Dispensing data were available for the 6 months before the crash. Data on long-term chronic diseases are also registered in this database, with the International Classification of Diseases, Tenth Edition (ICD-10) code and the start and end dates of disease.
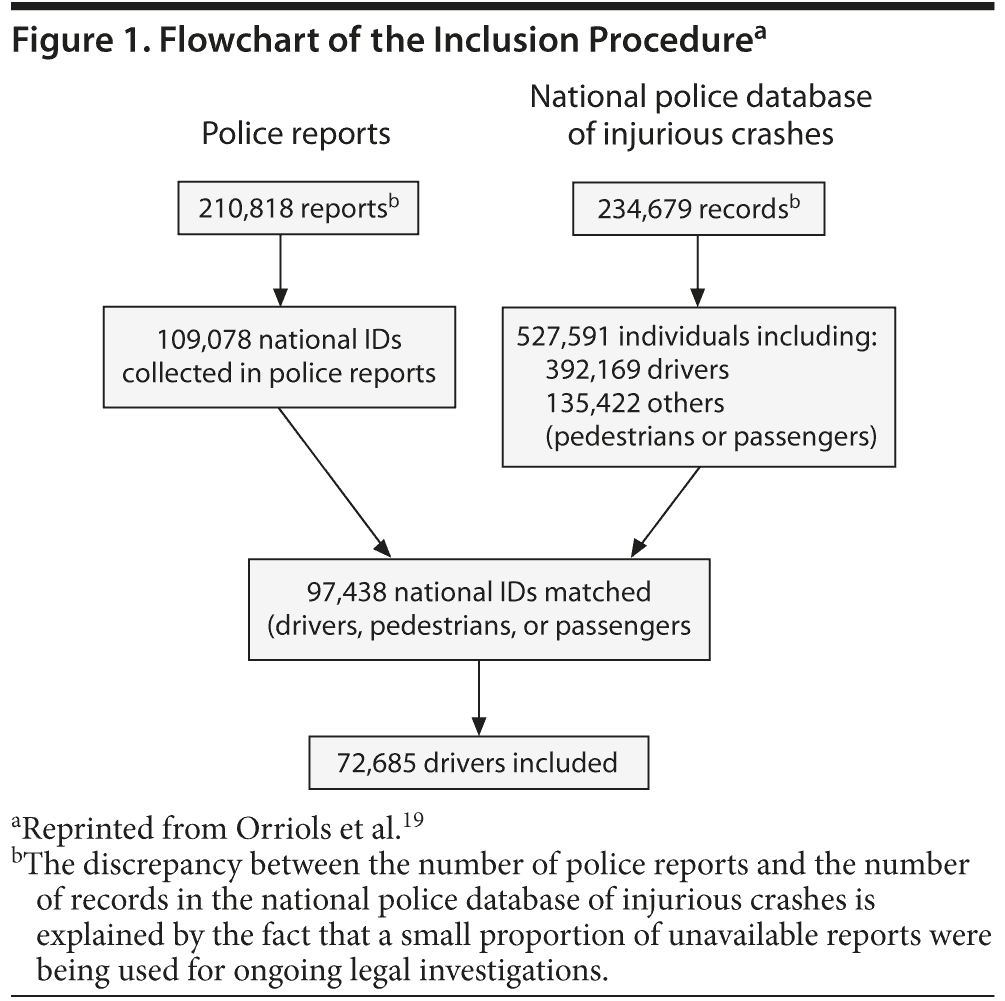
The NIDs of drivers involved in injurious crashes were extracted from the police report database. Police reports were matched with data from the injurious crashes database to provide details of the crash context (Figure 1). NIDs extracted from police reports were matched to the health care insurance database to collect data on the drivers’ exposure to medication. Subjects whose police report did not contain their NID were not included. Confidentiality was ensured by using the personal information anonymization function of the national health care insurance system.20
Cases and Controls
Cases were defined as drivers who were deemed responsible for the crash, while controls were those who were not responsible. Responsibility levels in the crash were determined by a standardized method adapted from Robertson and Drummer.21 This method, previously implemented in France using data from the national police database of fatal crashes,22 takes into consideration the different factors likely to reduce driver responsibility: road environment, vehicle-related factors, traffic conditions, type of accident, traffic rule obedience, and difficulty of the driving task. In each area, a score is assigned from 1 (favorable to driving) to 4 (not favorable to driving), and all 6 scores are subsequently summated into a summary responsibility score. Cases (responsible drivers) are defined as those with a score of less than 15.
Exposure Periods
Exposure was considered to start on the day following dispensing. To ensure that medicines were not prescribed as a consequence of the crash, medicines dispensed on the crash day were not considered.
Antidepressants. Antidepressants were studied according to the following 4 groups: tricyclic antidepressants (imipramine, clomipramine, trimipramine, amitriptyline, doxepin, dosulepin, and amoxapine), selective serotonin reuptake inhibitors (fluoxetine, citalopram, paroxetine, sertraline, fluvoxamine, and escitalopram), serotonin-norepinephrine reuptake inhibitors (venlafaxine, milnacipran, and duloxetine), and other antidepressants (monoamine oxidase inhibitors [iproniazid and moclobemide], mianserin, mirtazapine, viloxazine, and tianeptine). Monoamine oxidase inhibitors were not studied separately because of their low consumption level.
In France, no more than 30 days’ worth of treatment with antidepressants may be dispensed by pharmacies, so 30 days of exposure was attributed following each dispensation.
In order to study treatment initiation, a subanalysis was conducted that considered drivers who had 1 antidepressant prescription during the 6 months before the crash. A change in antidepressant treatment was defined as dispensing of a medicine or combination of medicines different from the previous one.
Concomitant exposure. In France, a 4-level risk classification system has been established for medicines that affect driving abilities.23-25 Comparisons were adjusted for the use of other medicines classified as being in the highest levels of risk (levels 2 and 3). This includes antiepileptics, psycholeptics, all benzodiazepines and derivatives, and analgesic opioids. Antidepressants are level 2 medicines and were thus excluded from the concomitant exposure variable.
Analysis
Descriptive analysis. A logistic regression analysis compared the excluded and included subjects according to age, gender, injury severity, vehicle type, crash location, type of police forces filing the report, alcohol level, and responsibility. Drivers were censored at their first involvement in a road traffic crash in order to mitigate the impact of previous crashes on medicine exposure. Frequencies of exposures to antidepressants were compared according to individual and crash characteristics in a bivariate analysis, using χ2 tests. Multivariate analysis was performed by logistic regression.
Responsibility analysis. Statistical analyses of the case-control study component were conducted using logistic regression, modeling the probability of being responsible for the crash. The associations between responsibility and age, gender, socioeconomic category, time of crash, season, vehicle type, injury severity, blood alcohol concentration, concomitant treatments (level 2 and 3 medicines according to the French classification system), and chronic long-term disorders were initially investigated using univariate analysis; associated variables were included in the multivariate model when the P value was < .20 (χ2 test). This was the case for all variables.
Case-crossover analysis. The case-crossover analysis consisted of a pair-matched analytic approach to compare exposure during a period immediately before the crash (case period) with exposure during an earlier period (control period) for the same subject.26 This method is adapted to the assessment of the short-term effect of exposure while controlling for the potential impact of chronic medical conditions. We compared antidepressant exposure on the crash day with antidepressant exposure during a 1-day control period 30 days earlier. In order to study treatment initiation, only drivers who had 1 prescription during the 6 months before the crash were considered. We also compared exposure to treatment changes in the 30 days before the crash with exposure to treatment changes during a 30-day control period. Odds ratios were estimated by conditional logistic regression, using the PHREG (proportional hazard regression) procedure in SAS.
Data were analyzed using the SAS statistical software package, version 9.1 (SAS Institute Inc, Cary, North Carolina).
RESULTS
We extracted 109,078 NIDs with gender and date of birth from 210,818 police reports available from July 2005 to May 2008, corresponding to any individual involved in an injurious road traffic crash (Figure 1). Ninety percent of these individuals were matched with a corresponding record in the injurious crashes database. The linkage failed for 10% of the individuals, because the ID corresponded either to a driver involved in the crash but not recorded in the police national database or to an individual not involved in the crash (eg, a witness, the owner of a vehicle involved).
The procedure led to the inclusion of 72,685 drivers (34,896 responsible and 37,789 not responsible), that is, 18.5% of the 392,169 drivers registered in the police national database of injurious crashes.
Injury severity was the main factor associated with the probability of being part of the study. The inclusion rate was approximately the same for drivers who were responsible and not responsible (respectively, 18.8% and 18.3%).
The multivariate descriptive analysis showed that exposure to antidepressants was higher among women, drivers aged 45 years or over, and retired or unemployed drivers. Antidepressant exposure was more likely among drivers involved in single-vehicle crashes and among drivers responsible for the crash. The proportion of antidepressant-exposed drivers was higher among those under the influence of alcohol, and it increased with blood alcohol level. Finally, antidepressant users were much more frequently exposed concomitantly to other medicines that can affect driving abilities (level 2 and 3 medicines), particularly to benzodiazepine anxiolytics, benzodiazepine hypnotics, and benzodiazepine-like hypnotics (zolpidem and zopiclone) (Table 1).
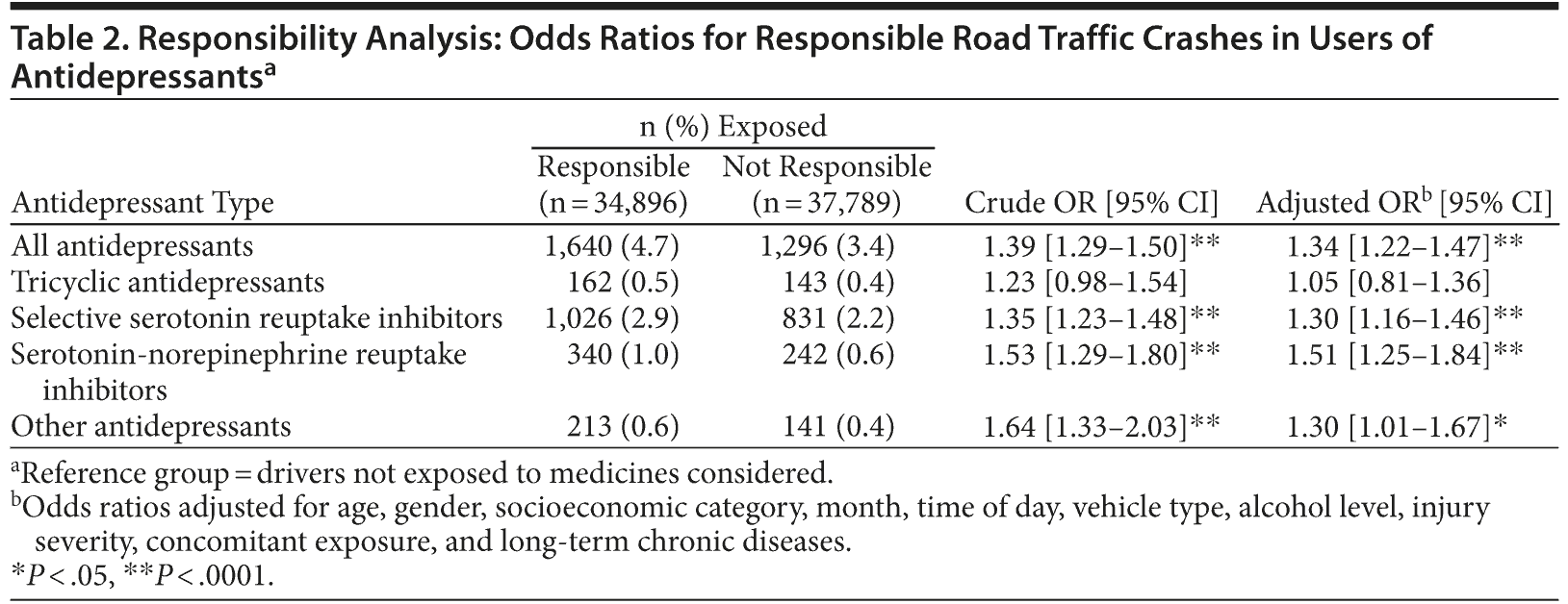
After we adjusted for variables found to be associated with responsibility in the crash, the prescription of antidepressants was associated with the risk of being responsible for a crash (OR = 1.34 [95% CI, 1.22-1.47]). The adjusted odds ratios were 1.30 (95% CI, 1.16-1.46) for selective serotonin reuptake inhibitors and 1.51 (95% CI, 1.25-1.84) for serotonin-norepinephrine reuptake inhibitors. The association with prescription of antidepressants classified in the “other” category was also significant (OR = 1.30 [95% CI, 1.01-1.67]). This includes monoamine oxidase inhibitors, mianserin, mirtazapine, viloxazine, and tianeptine. No significant association was found for tricyclic antidepressants (Table 2). Subgroup analysis showed that, of the 2,936 drivers exposed to antidepressants on the day of the crash, the responsibility OR was 1.28 (95% CI, 0.97-1.68) in the 289 drivers who had 1 prescription over the 6-month period before the crash and 1.35 (95% CI, 1.22-1.48) for the remaining 2,647 drivers who had more than 1 prescription over the same period. There was no interaction of antidepressant use with alcohol consumption. The effect of benzodiazepine anxiolytics was similar in antidepressant users (OR = 1.29 [95% CI, 1.08-1.54]) and nonusers (OR = 1.22 [95% CI, 1.07-1.39]).
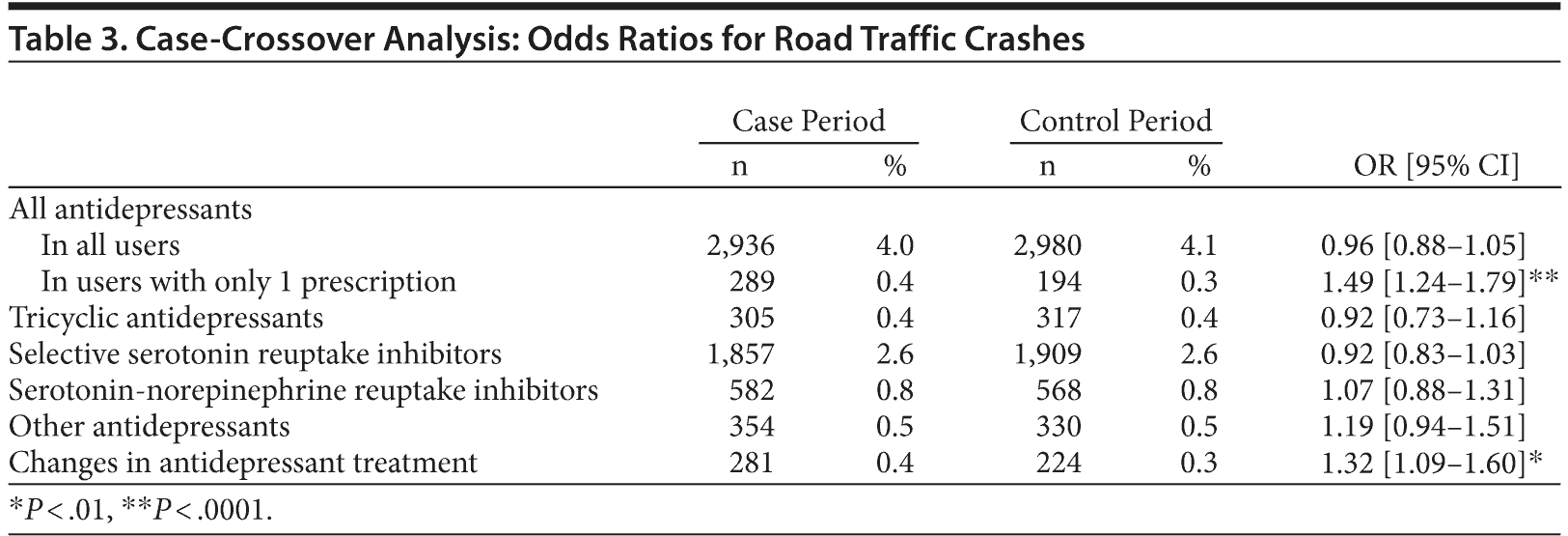
The case-crossover analysis found no association between the risk of crash and exposure to antidepressants (Table 3). Subgroup analysis in drivers who had only 1 prescription in the 6 months before the crash showed that the probability of being exposed to this first prescription was higher just before the crash compared with the control period, showing an increased risk of crash when a treatment was initiated (OR = 1.49 [95% CI, 1.24-1.79]). The case-crossover analysis also showed an increased risk of road traffic crash after a change in antidepressant treatment (OR = 1.32 [95% CI, 1.09-1.60]) (Table 3).
DISCUSSION
The results indicate an increased risk of being responsible for an injurious road traffic crash for drivers with a prescription of antidepressants (OR = 1.34 [95% CI, 1.22-1.47]). The case-crossover analysis showed an increased risk of crash at treatment initiation (OR = 1.49 [95% CI, 1.24-1.79]) and following a change in antidepressant treatment (OR = 1.32 [95% CI, 1.09-1.60]).
The responsibility analysis is a real strength of the study, as cases and controls share some common characteristics: they were all driving a vehicle and, for multiple-vehicle crashes, were on the road at the same time. This is not the case for controls when they are selected from health care or driving license databases. Another option is to select controls on the side of the road. Only low sample sizes, however, are achieved using this method, and this may also lead to selection bias, as participation is on a voluntary basis. The principle of the responsibility analysis is that if a factor contributes to road traffic crash causation, it is expected that it would be overrepresented in the responsible drivers. The method does not capture the risk, for nonresponsible drivers, of being unable to avoid a crash that may be linked to medicine consumption. This would lead to an underestimation of the risk estimate. In a previous study on the impact of illegal drug consumption, using the same police national database but limited to fatal crashes,22 the same method used to determine responsibility was approved by an independent expert evaluation of responsibility. Importantly, responsibility levels were computed independently of alcohol and illicit drug use because of their potential interactions with medicine use.
The results showed a significant association between selective serotonin reuptake inhibitors, serotonin-norepinephrine reuptake inhibitors, and other antidepressants (to a lesser extent) and the risk of being responsible for a crash. The absence of an association with tricyclic antidepressants may be explained by the sedating effects of these antidepressants or advice provided by prescribers that may dissuade patients from driving or make them more cautious at the wheel. This lack of association may also be due to the low prevalence of consumption of tricyclic antidepressants as they were progressively supplanted by newer generations of antidepressants. The analysis was adjusted for other medicines (such as benzodiazepines) and blood alcohol concentration, which are themselves known to increase the risk of crash. Moreover, the descriptive analysis showed that antidepressant users often used these medicines concomitantly and were under the influence of alcohol at the time of the crash. We also controlled for accident type and injury severity, 2 factors that may be related to suicidal gestures.
The most recurring difficulty in all studies of this topic is to disentangle the impact of the medical condition and the impact of medication (confounding by indication). This is the main reason why we also performed a case-crossover analysis. The results showed that prescription of an antidepressant treatment did not significantly increase the risk of road traffic crash. Since the case-crossover approach is unsuited for chronic treatments, this result was not surprising. When we looked at acute exposures such as initiation or change in treatment, significant associations were detected. This effect is likely to be linked to the medication, as depression symptoms may have appeared several weeks before the crash (thus before the control period) and cannot therefore account for the risk measured by the case-crossover analysis. The association between a change in treatment and the risk of crash found in the case-crossover analysis may be due to the medicines but may also be indicative of an aggravation of the depressive state or of a period during which the depressive state was unstable, leading to a higher risk of crash related to the symptoms of the clinical condition. It has been shown that depression is associated with slower reaction time in a driving simulator27 and with lower scores in attention abilities.28 Worrying thoughts in depression seem to predominate over other important information.29 All of these symptoms may lead to impaired driving performance. Risk estimates obtained in the responsibility analysis are a combination of all of these factors: impact of treatment initiation, impact of treatment changes, and impact of depression symptoms. The analysis conducted according to the number of prescriptions showed that there is also an increased responsibility risk in drivers who had more than 1 prescription before the crash. Responsibility risk was associated with the use of several classes of antidepressants and in particular selective serotonin reuptake inhibitors and serotonin-norepinephrine reuptake inhibitors, which are nonsedating antidepressants. Even if such antidepressants have been shown to slightly impair performance in healthy subjects,3,9,18 a driving simulator study suggested that treated depressed patients had better performances on tasks related to driving than untreated depressed patients.13 Long-term treatment with antidepressants may thus reduce the risk, but not to baseline, the residual risk probably being linked to depression symptoms.
Injury severity was associated with the probability of being part of the study. Thus, severely injured drivers were more likely to be included than slightly injured drivers. Killed drivers and uninjured drivers had still lower inclusion rates. This finding can be explained by the fact that injured drivers were more likely to be admitted to the hospital, so their health care number was more frequently noted in the police report. Thus, our study sample slightly overrepresented drivers injured in more severe crashes.
Medicine exposure was ascertained from computerized records of reimbursed prescriptions filled at the pharmacy. These data were not subject to underreporting, a major problem encountered when medicine exposure data are self-reported.30 However, we did not know whether the medicines were actually ingested. Noncompliance, which we were not able to check, would therefore result in exposure misclassification. Nevertheless, the resulting misclassification would be nondifferential between the cases and the controls. While this aspect of the study method may lead to overestimation of exposure prevalence, the association measures remain unbiased. Other studies using patient-derived data and the same dispensation database showed that the health care insurance data are reliable indicators of actual exposure for medicines used over a long time frame.31
In summary, data presented in this study indicate that patients and prescribers should be warned about the risk of crash during periods of antidepressant medication and periods of particularly high vulnerability such as those when a treatment is initiated or changed.
Drug names: citalopram (Celexa and others), clomipramine (Anafranil and others), doxepin (Silenor and others), duloxetine (Cymbalta), escitalopram (Lexapro and others), fluoxetine (Prozac and others), fluvoxamine (Luvox and others), imipramine (Tofranil and others), milnacipran (Savella), mirtazapine (Remeron and others), paroxetine (Paxil, Pexeva, and others), sertraline (Zoloft and others), trimipramine (Surmontil and others), venlafaxine (Effexor and others), zolpidem (Ambien, Edluar, and others).
Disclosure of off-label usage: The authors have determined that, to the best of their knowledge, no investigational information about pharmaceutical agents that is outside US Food and Drug Administration-approved labeling has been presented in this article.
Author affiliations: Université Bordeaux Segalen, Equipe Injury Prevention and Control (PPCT), Institute of Public Health, Epidemiology, and Development (ISPED), Centre INSERM U897-Epidemiologie-Biostatistique, F-33000, Bordeaux (Drs Orriols and Lagarde); P×´le Entre-Deux-Mers, Centre Hospitalier Cadillac, Cadillac (Dr Queinec); National Center for Scientific Research (CNRS), USR 3413 SANPSY, Université Bordeaux Segalen, Bordeaux (Dr Philip); Université de Lyon, F-69000, Lyon; IFSTTAR, UMR T 9405, UMRESTTE, F-69500, Bron; and Université Lyon 1, UMRESTTE, F-69000, Lyon (Ms Gadegbeku); French Health Products Safety Agency (AFSSAPS), Saint-Denis (Dr Delorme); INSERM U657, CIC-P0005, Department of Pharmacology, Université Bordeaux Segalen, Bordeaux (Dr Moore), France; and Department of Epidemiology and Biostatistics, Center for Clinical Epidemiology, McGill University, Montreal, Quebec, Canada (Dr Suissa).
Study group: The authors thank the CESIR research group for their contribution in discussing the analysis strategy and for critical review of the manuscript: Marta Avalos, PhD (INSERM U897, Université Bordeaux Segalen, Bordeaux, France); Fabienne Bazin, PhD (INSERM U657, Bordeaux, France); Sylvie Blazejewski (CIC 0005, Bordeaux, France); Benjamin Contrand, MSc (INSERM U897, Bordeaux, France); Geneviרve Durrieu, PhD (Service de Pharmacologie Médicale et Clinique, CHU Toulouse, INSERM U1027, Toulouse, France); Pierre-Olivier Girodet, MD, PhD (CIC 0005, Bordeaux, France); Marcel Goldberg, MD, PhD (INSERM U687-UVSQ, Villejuif, France); Bernard Laumon, MD, PhD (IFSTTAR, UMRESTTE, Bron, France); Dominique Lauque, MD (CHU Toulouse, Toulouse, France); Nathalie Lecoules, MD (CHU Toulouse, Toulouse, France); Laurence Memes, PharmD (CIC 0005, Bordeaux, France); Louis Merle, MD, PhD (CHU Limoges, Limoges, France); Jean-Louis Montastruc, MD, PhD (Service de Pharmacologie Médicale et Clinique, CRPV, Université de Toulouse, CHU Toulouse, INSERM U1027, Toulouse, France); Pernelle Noize, PhD (INSERM U657, Bordeaux, France); Nathalie Orsoni, MD (CHU Limoges, Limoges, France); Antoine Pariente, MD (INSERM U657, CIC 0005, Bordeaux, France); Régis Ribéreau-Gayon, MD (CHU Bordeaux, Bordeaux, France); Louis-Rachid Salmi, MD, PhD (INSERM U897, Université Bordeaux Segalen, ISPED, CHU Bordeaux, Bordeaux, France); Frantz Thiessard, MD, PhD (LESIM, Bordeaux, France); and Aurore Tricotel, MSc (AFSSAPS, Saint-Denis, France).
Financial disclosure: Dr Delorme is an employee of the French Health Products Safety Agency (AFSSAPS). Drs Orriols, Queinec, Philip, Moore, Suissa, and Lagarde and Ms Gadegbeku have no personal affiliations or financial relationships with any commercial interest to disclose relative to the article.
Funding/support: The project was funded by the French Health Products Safety Agency (AFSSAPS), the French National Research Agency (ANR, DAA 0766CO204), the French National Medical Research Institute (Equipe INSERM), the French Medical Research Foundation (Equipe FRM), and the French General Health Directorate (DGS). Members of AFSSAPS participated in data collection and interpretation and in review of the manuscript.
Acknowledgments: The authors acknowledge the French National Health Insurance (CNAMTS), the Inter-Departmental Observatory on Road Safety (ONISR), and Agira-TransPV for providing health care and road traffic crash data, as well as the Federative Institute of Research on Public Health (IFR 99).
REFERENCES
1. Hindmarch I, Subhan Z, Stoker MJ. The effects of zimeldine and amitriptyline on car driving and psychomotor performance. Acta Psychiatr Scand Suppl. 1983;308:141-146. PubMed
2. Kerr JS, Powell J, Hindmarch I. The effects of reboxetine and amitriptyline, with and without alcohol on cognitive function and psychomotor performance. Br J Clin Pharmacol. 1996;42(2):239-241. doi:10.1046/j.1365-2125.1996.39016.x PubMed
3. Ridout F, Meadows R, Johnsen S, et al. A placebo controlled investigation into the effects of paroxetine and mirtazapine on measures related to car driving performance. Hum Psychopharmacol. 2003;18(4):261-269. doi:10.1002/hup.494 PubMed
4. Wingen M, Bothmer J, Langer S, et al. Actual driving performance and psychomotor function in healthy subjects after acute and subchronic treatment with escitalopram, mirtazapine, and placebo: a crossover trial. J Clin Psychiatry. 2005;66(4):436-443. doi:10.4088/JCP.v66n0405 PubMed
5. Raptopoulos P, McClelland GR, Jackson D. The clinical pharmacology of paroxetine in healthy subjects. Acta Psychiatr Scand Suppl. 1989;80(S350):46-48. doi:10.1111/j.1600-0447.1989.tb07171.x PubMed
6. Robbe HW, O’ Hanlon JF. Acute and subchronic effects of paroxetine 20 and 40 mg on actual driving, psychomotor performance and subjective assessments in healthy volunteers. Eur Neuropsychopharmacol. 1995;5(1):35-42. doi:10.1016/0924-977X(94)00130-4 PubMed
7. Warrington SJ, Dana-Haeri J, Sinclair AJ. Cardiovascular and psychomotor effects of repeated doses of paroxetine: a comparison with amitriptyline and placebo in healthy men. Acta Psychiatr Scand suppl. 1989;80(S350):42-44. doi:10.1111/j.1600-0447.1989.tb07169.x PubMed
8. Kerr JS, Fairweather DB, Hindmarch I. Effects of fluoxetine on psychomotor performance, cognitive function and sleep in depressed patients. Int Clin Psychopharmacol. 1993;8(4):341-344. doi:10.1097/00004850-199300840-00025 PubMed
9. O’ Hanlon JF, Robbe HW, Vermeeren A, et al. Venlafaxine’s effects on healthy volunteers’ driving, psychomotor, and vigilance performance during 15-day fixed and incremental dosing regimens. J Clin Psychopharmacol. 1998;18(3):212-221. doi:10.1097/00004714-199806000-00006 PubMed
10. Wingen M, Ramaekers JG, Schmitt JA. Driving impairment in depressed patients receiving long-term antidepressant treatment. Psychopharmacology (Berl). 2006;188(1):84-91. doi:10.1007/s00213-006-0471-7 PubMed
11. Shen J, Moller HJ, Wang X, et al. Mirtazapine, a sedating antidepressant, and improved driving safety in patients with major depressive disorder: a prospective, randomized trial of 28 patients. J Clin Psychiatry. 2009;70(3):370-377. doi:10.4088/JCP.08m04234 PubMed
12. Brunnauer A, Laux G, Geiger E, et al. Antidepressants and driving ability: results from a clinical study. J Clin Psychiatry. 2006;67(11):1776-1781. doi:10.4088/JCP.v67n1116 PubMed
13. Brunnauer A, Laux G, David I, et al. The impact of reboxetine and mirtazapine on driving simulator performance and psychomotor function in depressed patients. J Clin Psychiatry. 2008;69(12):1880-1886. doi:10.4088/JCP.v69n1205 PubMed
14. Ray WA, Fought RL, Decker MD. Psychoactive drugs and the risk of injurious motor vehicle crashes in elderly drivers. Am J Epidemiol. 1992;136(7):873-883. doi:10.1093/aje/136.7.873 PubMed
15. Leveille SG, Buchner DM, Koepsell TD, et al. Psychoactive medications and injurious motor vehicle collisions involving older drivers. Epidemiology. 1994;5(6):591-598. doi:10.1097/00001648-199411000-00006 PubMed
16. Barbone F, McMahon AD, Davey PG, et al. Association of road-traffic accidents with benzodiazepine use. Lancet. 1998;352(9137):1331-1336. doi:10.1016/S0140-6736(98)04087-2 PubMed
17. Bramness JG, Skurtveit S, Neutel CI, et al. Minor increase in risk of road traffic accidents after prescriptions of antidepressants: a study of population registry data in Norway. J Clin Psychiatry. 2008;69(7):1099-1103. doi:10.4088/JCP.v69n0709 PubMed
18. Ramaekers JG, Muntjewerff ND, O’ Hanlon JF. A comparative study of acute and subchronic effects of dothiepin, fluoxetine and placebo on psychomotor and actual driving performance. Br J Clin Pharmacol. 1995;39(4):397-404. PubMed
19. Orriols L, Delorme B, Gadegbeku B, et al.; CESIR Research Group. Prescription medicines and the risk of road traffic crashes: a French registry-based study. PLoS Med. 2010;7(11):e1000366. PubMed
20. Trouessin G, Allaert FA. FOIN: a nominative information occultation function. Stud Health Technol Inform. 1997;43 Pt A:196-200. PubMed
21. Robertson MD, Drummer OH. Responsibility analysis: a methodology to study the effects of drugs in driving. Accid Anal Prev. 1994;26(2):243-247. doi:10.1016/0001-4575(94)90094-9 PubMed
22. Laumon B, Gadegbeku B, Martin JL, et al; SAM Group. Cannabis intoxication and fatal road crashes in France: population based case-control study. BMJ. 2005;331(7529):1371. doi:10.1136/bmj.38648.617986.1F PubMed
23. Castot A, Delorme B, Medicinal Products and Driving Working Group. Medicinal products and driving: how to assess the risk? Presented at: P2T Congress; April 15-17, 2009; Marseille, France. Abstract 481.
24. AFSSAPS, on behalf of the Medicinal Products and Driving Working Group. Medicinal Products and Driving. http://www.ansm.sante.fr/var/ansm_site/storage/original/application/e5f2e48d5344bcfef6ca865ac63e7c3d.pdf. Revised March 2009. Accessibility verified June 12, 2012.
25. Arrêté du 8 ao×»t 2008 pris pour l’ application de l’ article R. 5121-139 du code de la santé publique et relatif × l’ apposition d’ un pictogramme sur le conditionnement extérieur de certains médicaments et produits. http://www.legifrance.gouv.fr/affichTexte.do?cidTexte=JORFTEXT000019563838. Revised October 4, 2008. Accessibility verified June 12, 2012.
26. Maclure M. The case-crossover design: a method for studying transient effects on the risk of acute events. Am J Epidemiol. 1991;133(2):144-153. PubMed
27. Bulmash EL, Moller HJ, Kayumov L, et al. Psychomotor disturbance in depression: assessment using a driving simulator paradigm. J Affect Disord. 2006;93(1-3):213-218. doi:10.1016/j.jad.2006.01.015 PubMed
28. Baune BT, Miller R, McAfoose J, et al. The role of cognitive impairment in general functioning in major depression. Psychiatry Res. 2010;176(2-3):183-189. doi:10.1016/j.psychres.2008.12.001 PubMed
29. Joormann J, Lira Yoon K, Zetsche U. Cognitive inhibition in depression. Appl Prev Psychol. 2007;12(3):128-139. doi:10.1016/j.appsy.2007.09.002
30. Honkanen R, Ertama L, Linnoila M, et al. Role of drugs in traffic accidents. BMJ. 1980;281(6251):1309-1312. doi:10.1136/bmj.281.6251.1309 PubMed
31. Noize P, Bazin F, Dufouil C, et al. Comparison of health insurance claims and patient interviews in assessing drug use: data from the Three-City (3C) Study. Pharmacoepidemiol Drug Saf. 2009;18(4):310-319. doi:10.1002/pds.1717 PubMed