Background: Almost 50% of women with schizophrenia become pregnant. Little is known about their psychiatric service use patterns during or shortly after pregnancy.
Methods: Using health administrative data, we identified 1,433 women in Ontario, Canada, with schizophrenia (ICD-9, ICD-10, or DSM-IV-TR) who had a live birth delivery from January 2003 through March 2011 and described their use of acute psychiatric care services including hospitalizations, emergency department visits not requiring hospitalization, and self-harm-related emergency department visits during pregnancy and in the first year postpartum. Incidence rates of psychiatric hospitalization during pregnancy, and also within 1 year postpartum, were each compared to the incidence rate of psychiatric hospitalization in the 1-year period before conception. Results are presented as incidence rate ratios (IRRs) and 95% confidence intervals (CIs). Similar comparisons were made for psychiatric emergency department visits not requiring hospitalization, as well as for self-harm-related emergency department visits.
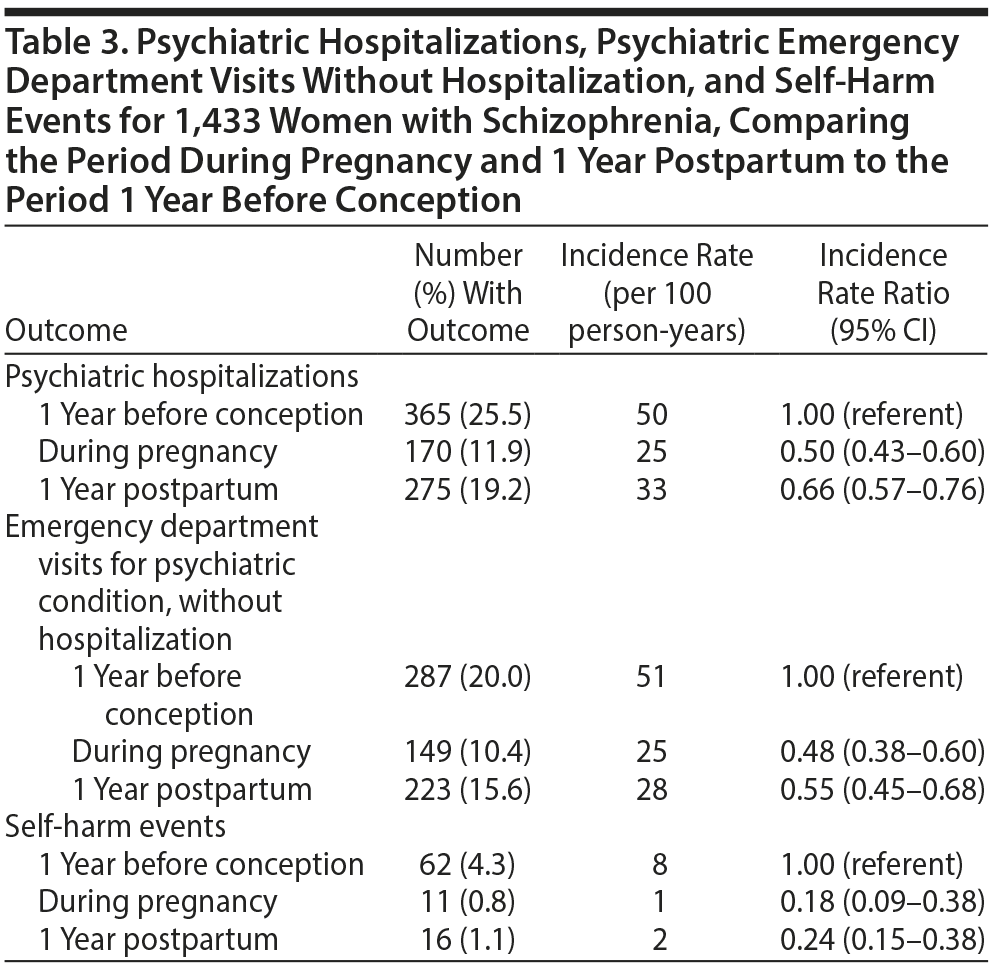
Results: About 12% of the women had at least 1 psychiatric hospitalization during pregnancy, and 19% in the first year postpartum. About 10% had at least 1 psychiatric emergency department visit without hospitalization during pregnancy, and 16% had at least 1 emergency department visit postpartum. Self-harm-related emergency department visits were rarer, affecting only about 1% of the women in each time period. Relative to that in the 1-year period before conception (50 per 100 person-years), the incidence rate of psychiatric hospitalizations was lower during pregnancy (25 per 100 person-years), which is equivalent to an IRR of 0.50 (95% CI, 0.43-0.60). While the IRR of psychiatric hospitalizations was lower over the entire 1-year period postpartum (0.66; 95% CI, 0.57-0.76), it was transiently higher in the first 9 days postpartum (3.59; 95% CI, 2.74-4.69) and then waned by days 10 to 29 postpartum (0.87; 95% CI, 0.56-1.24). Emergency department visits for psychiatric and self-harm reasons were consistently lower during pregnancy and postpartum compared to the year before conception.
Conclusions: Psychiatric hospitalizations and emergency department visits are not uncommon for women with schizophrenia during pregnancy and the postpartum period. However, except for a brief period after delivery, women with schizophrenia are at relatively lower risk of requiring acute psychiatric inpatient and emergency care during and 1 year after pregnancy compared to the 1-year period prior to conception. This is key prognostic information for women with schizophrenia, their families, and providers who counsel them regarding pregnancy, motherhood, and management of schizophrenia. Identifying women at risk of requiring acute psychiatric services in the perinatal period warrants further investigation.
Hospitalizations and Emergency Department Visits
for Psychiatric Illness During and After Pregnancy
Among Women With Schizophrenia
ABSTRACT
Background: Almost 50% of women with schizophrenia become pregnant. Little is known about their psychiatric service use patterns during or shortly after pregnancy.
Methods: Using health administrative data, we identified 1,433 women in Ontario, Canada, with schizophrenia (ICD-9, ICD-10, or DSM-IV-TR) who had a live birth delivery from January 2003 through March 2011 and described their use of acute psychiatric care services including hospitalizations, emergency department visits not requiring hospitalization, and self-harm–related emergency department visits during pregnancy and in the first year postpartum. Incidence rates of psychiatric hospitalization during pregnancy, and also within 1 year postpartum, were each compared to the incidence rate of psychiatric hospitalization in the 1-year period before conception. Results are presented as incidence rate ratios (IRRs) and 95% confidence intervals (CIs). Similar comparisons were made for psychiatric emergency department visits not requiring hospitalization, as well as for self-harm–related emergency department visits.
Results: About 12% of the women had at least 1 psychiatric hospitalization during pregnancy, and 19% in the first year postpartum. About 10% had at least 1 psychiatric emergency department visit without hospitalization during pregnancy, and 16% had at least 1 emergency department visit postpartum. Self-harm–related emergency department visits were rarer, affecting only about 1% of the women in each time period. Relative to that in the 1-year period before conception (50 per 100 person-years), the incidence rate of psychiatric hospitalizations was lower during pregnancy (25 per 100 person-years), which is equivalent to an IRR of 0.50 (95% CI, 0.43–0.60). While the IRR of psychiatric hospitalizations was lower over the entire 1-year period postpartum (0.66; 95% CI, 0.57–0.76), it was transiently higher in the first 9 days postpartum (3.59; 95% CI, 2.74–4.69) and then waned by days 10 to 29 postpartum (0.87; 95% CI, 0.56–1.24). Emergency department visits for psychiatric and self-harm reasons were consistently lower during pregnancy and postpartum compared to the year before conception.
Conclusions: Psychiatric hospitalizations and emergency department visits are not uncommon for women with schizophrenia during pregnancy and the postpartum period. However, except for a brief period after delivery, women with schizophrenia are at relatively lower risk of requiring acute psychiatric inpatient and emergency care during and 1 year after pregnancy compared to the 1-year period prior to conception. This is key prognostic information for women with schizophrenia, their families, and providers who counsel them regarding pregnancy, motherhood, and management of schizophrenia. Identifying women at risk of requiring acute psychiatric services in the perinatal period warrants further investigation.
J Clin Psychiatry
dx.doi.org/10.4088/JCP.14m09697
© Copyright 2016 Physicians Postgraduate Press, Inc.
aQueen’s University, Kingston, Ontario, Canada bDepartment of Family Medicine, University of Alberta, Edmonton, Alberta, Canada cDepartment of Psychiatry, dDepartments of Medicine and Obstetrics and Gynecology, and eFaculty of Nursing, University of Toronto, Toronto, Ontario, Canada fSt Michael’s Hospital, Toronto, Ontario, Canada gInstitute for Clinical Evaluative Sciences, Toronto, Ontario, Canada hWomen’s College Research Institute, Women’s College Hospital, Toronto, Ontario, Canada iInstitute of Health Policy, Management and Evaluation and Department of Medicine, University of Toronto, Toronto, Ontario, Canada jSunnybrook Health Sciences Centre, Toronto, Ontario, Canada kCentre for Addiction and Mental Health, Toronto, Ontario, Canada *Corresponding author: Simone N. Vigod, MD, Women’s Mental Health Program, Women’s College Hospital, 7234-76 Grenville St, Toronto, Ontario, Canada, M5S 1B2
([email protected]).
About half of all women with schizophrenia become pregnant.1–3 Adequate treatment and support can help women with schizophrenia experience healthy parenthood,4 but perinatal psychiatric instability has serious long-term consequences for both women and their children.5 Active psychotic symptoms during pregnancy and the postpartum period have been associated with maternal self-harm, physical harm to the infant,6–9 inadequate maternal-infant attachment, and poorer child development trajectories.10–14 The neuroprotective estrogen hypothesis suggests that higher estrogen levels during pregnancy may protect vulnerable women against psychiatric symptoms.15–18 However, the sudden loss of estrogen following childbirth may increase vulnerability, especially when compounded by sleep deprivation and the psychosocial stress of caring for a newborn.16,19,20 To date, little is known about the psychiatric stability of women with schizophrenia during pregnancy or after delivery, especially in terms of need for acute psychiatric care services such as hospitalization or emergency department visits.21 Further knowledge in this area will help guide the development of supports and services that aim to optimize psychiatric stability for pregnant women and new mothers with schizophrenia.
Two population-based studies have evaluated psychiatric service use postpartum, but not during pregnancy. Howard et al22 found that about half of 40 new UK mothers with schizophrenia or schizoaffective disorder had a psychiatric hospitalization or a visit to a general practitioner for a new psychiatric symptom in the first year postpartum, but that study had no control group. In a larger Danish study,23 15.7% of women with schizophrenia had a psychiatric hospitalization in the first year postpartum, and the highest risk for hospitalization was in the first 60 days after delivery.
The overall objective of our study was to
characterize the use of acute psychiatric care services during pregnancy and postpartum among a
population-based cohort of mothers with preexisting schizophrenia. Our first objective was to describe the use of acute psychiatric care services, including psychiatric hospitalizations, psychiatric emergency department visits without hospitalization, and self-harm-related emergency department visits, both during pregnancy and 1 year postpartum. Our second objective was to compare the incidence rate of psychiatric hospitalization, psychiatric emergency department visits, and self-harm–related emergency department visits during pregnancy and also within 1 year postpartum to the incidence rate of each of these outcomes in the 1-year period before conception.
METHODS
Study Design
A population-based cohort study was conducted that included all women with schizophrenia in Ontario, Canada, who had a live birth from January 1, 2003, through March 31, 2011. Study outcomes were psychiatric hospitalizations, psychiatric emergency department visits without hospitalization, and self-harm–related emergency department visits occurring during pregnancy and in the first year postpartum. Incidence rates for each of these outcomes were compared to rates in the 1 year prior to conception, which served as the referent. This reference period was chosen because it represents a time in which a woman is unaffected by the bio-hormonal and psychosocial impacts of pregnancy or new motherhood. By having each woman serve as her own control, a woman’s sociodemographic factors, family history, substance use, duration and severity of illness, and history of treatment adherence remain generally constant across periods, facilitating a more accurate assessment of the influence of pregnancy and the postpartum period on outcomes.
Data Sources
Health administrative data were accessed from the Institute for Clinical and Evaluative Sciences (ICES), an independent nonprofit research organization that maintains population-level databases to evaluate health care services in Ontario, Canada’s most populous province (population ~13 million). In these databases, personal health identifiers are removed from patient-level inpatient and outpatient records of every Ontario resident. These records are then linked to each other using a unique identifier via the Registered Persons Database (RPDB) that also contains the age, sex, and postal code for all Ontario residents. We used the Canadian Institute for Health Information Discharge Abstract Database (CIHI-DAD) and the Ontario Mental Health Reporting System (OMHRS) database to capture hospitalization data, the National Ambulatory Care Reporting System (NACRS) database for emergency department data, and the Ontario Health Insurance Plan (OHIP) database for physician-based diagnoses and service use. The ICES MOMBABY data file, generated from the CIHI-DAD, provides data on pregnancy outcome (ie, live birth or stillbirth) and gestational age at delivery for all in-hospital births, allowing for accurate estimation of the date of conception.24 Data in these administrative databases have been determined to be complete and accurate.25
Participants
We included all Ontario women who had a live birth during the study period and who met at least 1 of the following criteria within the 5 years prior to conception: (1) at least 1 hospitalization with a medical record discharge diagnosis of “schizophrenia” or “schizoaffective disorder” (ICD-9 code 295 or ICD-10 code F20 or F25 in CIHI-DAD as the most responsible diagnosis or DSM-IV-TR diagnosis in OMHRS) or (2) 2 outpatient OHIP physician service claims for schizophrenia or schizoaffective disorder within a 24-month period.26,27 The requirement of having 2 OHIP outpatient service claims reduces the possibility of a single consultation to “rule out” schizophrenia.28 This approach has yielded a 4-year treatment prevalence of schizophrenia of slightly less than 1.0%, which is in keeping with international prevalence estimates.26 We excluded non-Ontario residents, those with an invalid provincial health card number, and those younger than age 14 years or older than age 50 years at delivery.2,28 If a woman had more than 1 live birth during the study period, 1 of these deliveries was selected at random such that no woman contributed more than 1 delivery to the analysis.
Outcome Measures
The primary study outcome was a psychiatric hospitalization, defined as any admission to a designated psychiatric bed (as recorded in the OMHRS database) or to a nonpsychiatric bed where the most responsible diagnosis was for a mental health condition (ICD-10 codes F00–F99, as recorded in the CIHI-DAD).29 The latter includes admissions to intensive care units, eating disorder units, and non–mental-health beds used due to bed spacing issues and represents approximately 20% of psychiatric admissions in Ontario (source: ICES unpublished data). Secondary outcomes were psychiatric emergency department visits not resulting in hospitalization, as well as deliberate self-harm, representing a severe subset of psychiatric emergency department visits (with or without subsequent hospital admission). Emergency department visits were identified using ICD-10 mental health and addiction codes (F00–F99) in the primary reason for visit field. Self-harm emergency department visits were identified using ICD-10 codes for intentional self-harm (X60–X84) and for sequelae of events of undetermined intent that included a cut/piercing or poisoning (codes Y10–Y19, Y28). The latter were included because they are associated with high rates of future deliberate self-harm and are likely to represent deliberate self-harm episodes.30
Descriptive Variables
We included maternal age at delivery, socioeconomic status (using neighborhood income quintiles), and geographical status (ie, urban vs rural).31 Information on comorbid psychiatric diagnoses (including alcohol and substance use disorders) and mental health service use (defined as visits to a psychiatrist or to a primary care physician for a mental health concern)32 was obtained from inpatient, emergency department, and outpatient databases (ie, OMHRS, CIHI-DAD, NACRS, and OHIP). A usual mental health care provider in the year prior to pregnancy was defined by having more than 2 mental health visits to a physician and at least 50% of these visits to the same physician.33 To estimate medical comorbidity in the 2 years preconception, we used the Johns Hopkins Collapsed Aggregated Diagnosis Groups (CADGs).34 CADG category 5, which groups codes for chronic unstable medical diseases, was used as a marker of significant medical comorbidity.35 Pregnancy outcomes, including preterm birth, poor fetal growth, and infant death, were determined from inpatient and outpatient data sources. These same measures were used in our previous work on women with schizophrenia.28
Statistical Analysis
Descriptive participant characteristics were presented as means, medians, and proportions. We described the number and proportion of women with 1 or more psychiatric hospitalizations, psychiatric emergency department visits not requiring hospitalization, and self-harm–related emergency department visits during pregnancy and in the postpartum period, as well as the length of stay of psychiatric hospitalizations. We compared the incidence rate of psychiatric hospitalization during pregnancy and also in the 1-year period postpartum to that in the 1-year period prior to conception (the referent). Poisson regression was used to generate incidence rate ratios (IRRs), and 95% confidence intervals (CIs) were used to facilitate comparison across the exposure periods despite their differing lengths. The same analyses were repeated for psychiatric emergency department visits not requiring hospitalization, as well as for self-harm emergency department visits. In secondary analyses, we subdivided each pregnancy into 3 trimesters and the postpartum period into 5 time intervals (days 0–9, 10–29, 30–89, 90–179, and 180–365 after delivery, to be generally consistent with previous research by Munk-Olsen et al23) and repeated the same analyses as above, maintaining the 1-year period before conception as the referent. For privacy reasons, cell sizes of fewer than 6 persons were not reported.
Statistical analyses were performed using SAS statistical software, version 9.3 of the SAS System for Unix (SAS Institute, Cary, North Carolina). Study approval was obtained from the Research Ethics Boards at Women’s College Hospital (Study ID 2012–0013-A) and at Sunnybrook Health Sciences Centre (ICES logged study: 2013 0904 300 000).
RESULTS
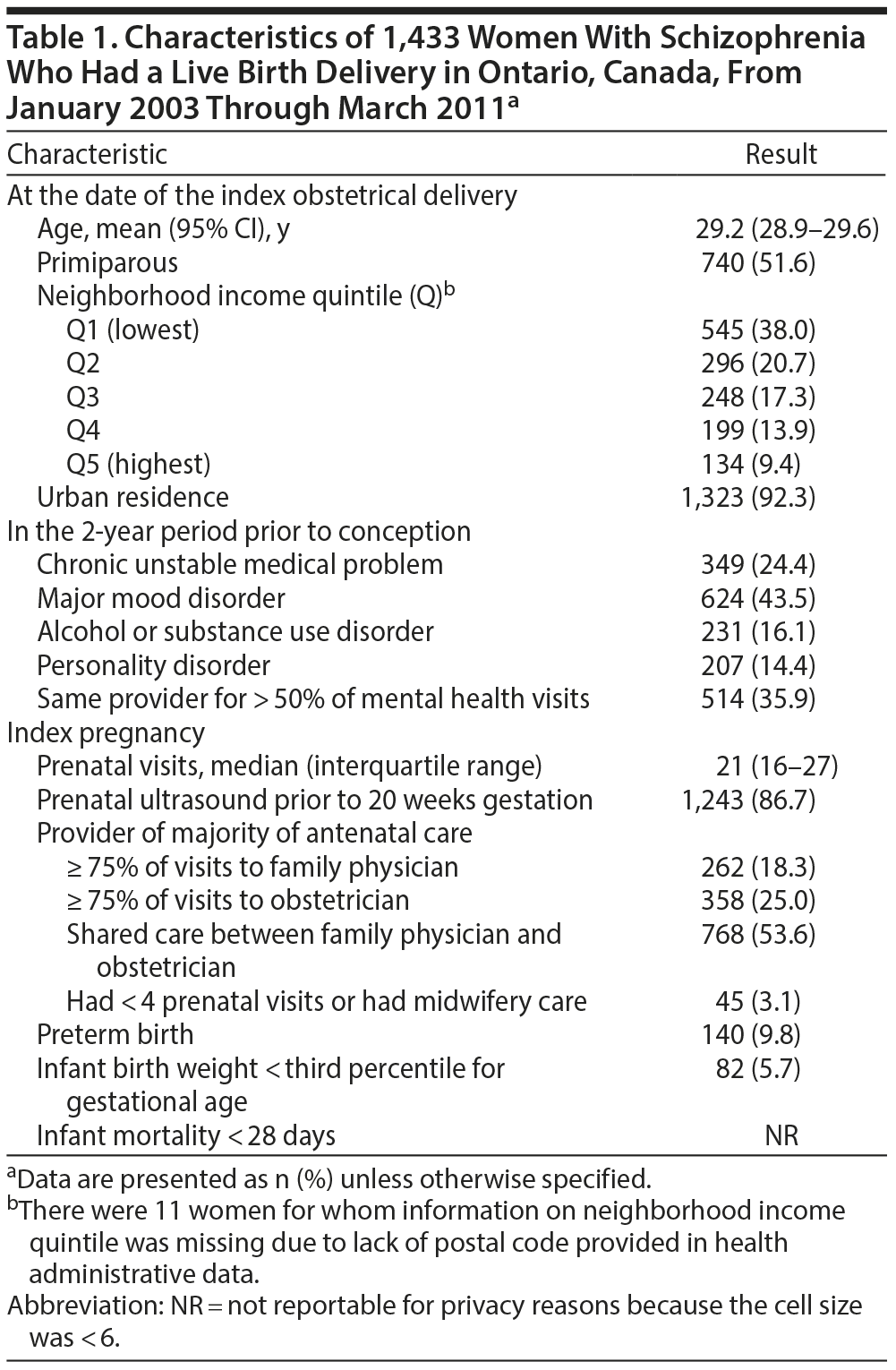
Descriptive characteristics of the 1,433 women with schizophrenia in our cohort are shown in Table 1. About half were primiparous, almost 60% lived within neighborhoods in the lowest 2 income quintiles, and about one-third had a single mental health care provider who provided the majority of their care in the 2 years prior to conception. In addition to their diagnosis of schizophrenia, 43.5% of the women had received a diagnosis of a mood disorder, and 14.4% had received a personality disorder diagnosis. About 10% delivered their infant prematurely (< 37 weeks gestational age), and about 6% of the newborns were very small for gestational age (< third percentile for gestational age).
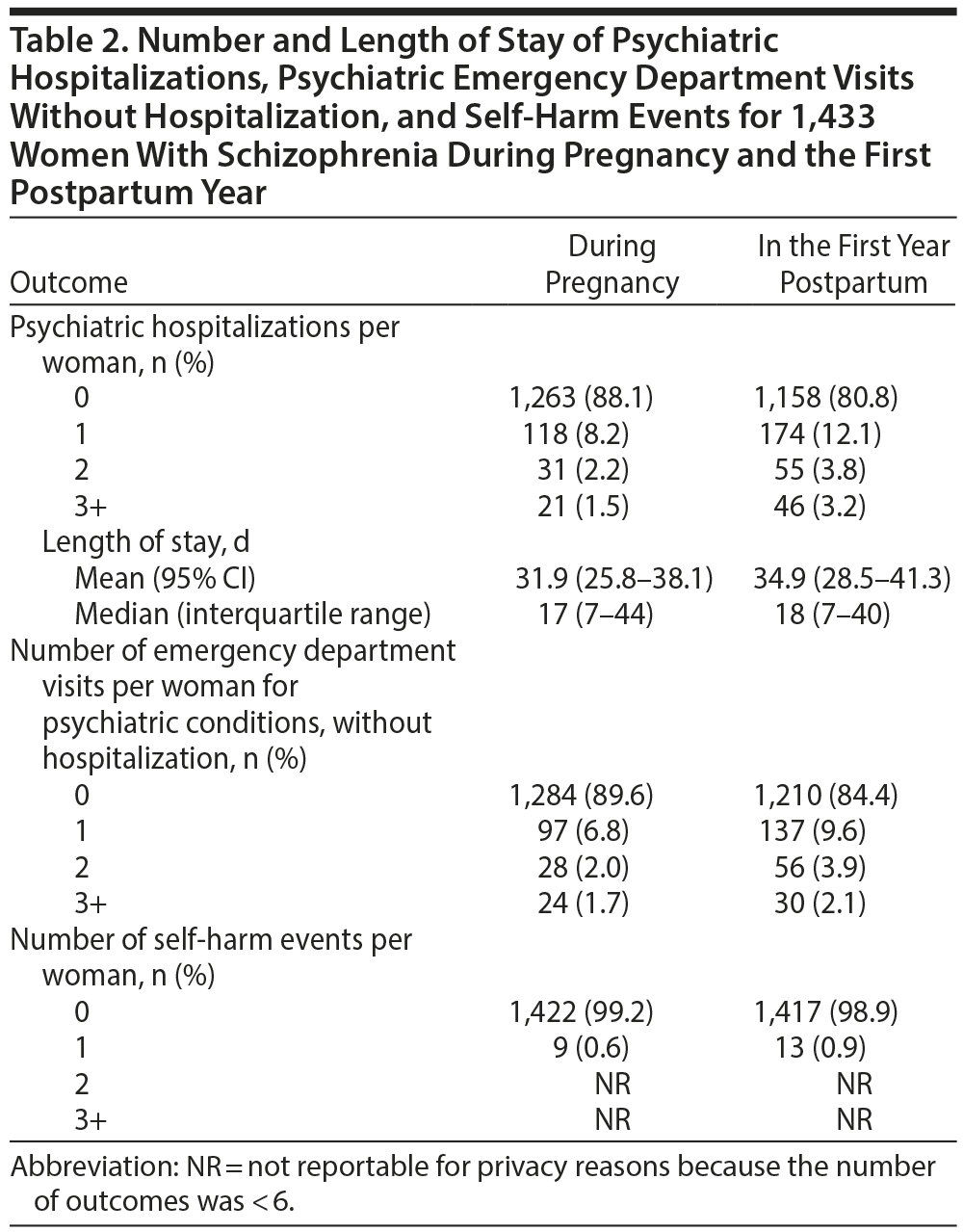
Psychiatric hospitalizations and psychiatric emergency department visits without hospitalization affected between 10% and 20% of women depending on the outcome and time period studied, while self-harm was more rare, affecting < 1% of women in either time period. A minority of women had multiple hospitalizations or emergency department visits (Table 2).
The incidence rate for psychiatric hospitalization was 25 per 100 person-years during pregnancy and 33 per 100 person-years in the first year postpartum, compared to a rate of 50 per 100 person-years in the year prior to conception. The corresponding IRRs were 0.50 (95% CI, 0.43–0.60) and 0.66 (95% CI, 0.57–0.76). A similar pattern was observed for emergency department visits without hospitalization, as well as for self-harm–related emergency department visits (Table 3).
The rate of psychiatric hospitalization during each trimester of pregnancy was lower than in the preconception year (Figure 1A). However, in the first 9 days postpartum, it was 178 per 100 person-years—an IRR of 3.59 (95% CI, 2.74–4.69). From 10 to 29 days postpartum, there was a decrease to 43 per 100 person-years (an IRR of 0.87 [95% CI, 0.56–1.24]), and then the rate declined thereafter during the first year postpartum (Figure 1A).
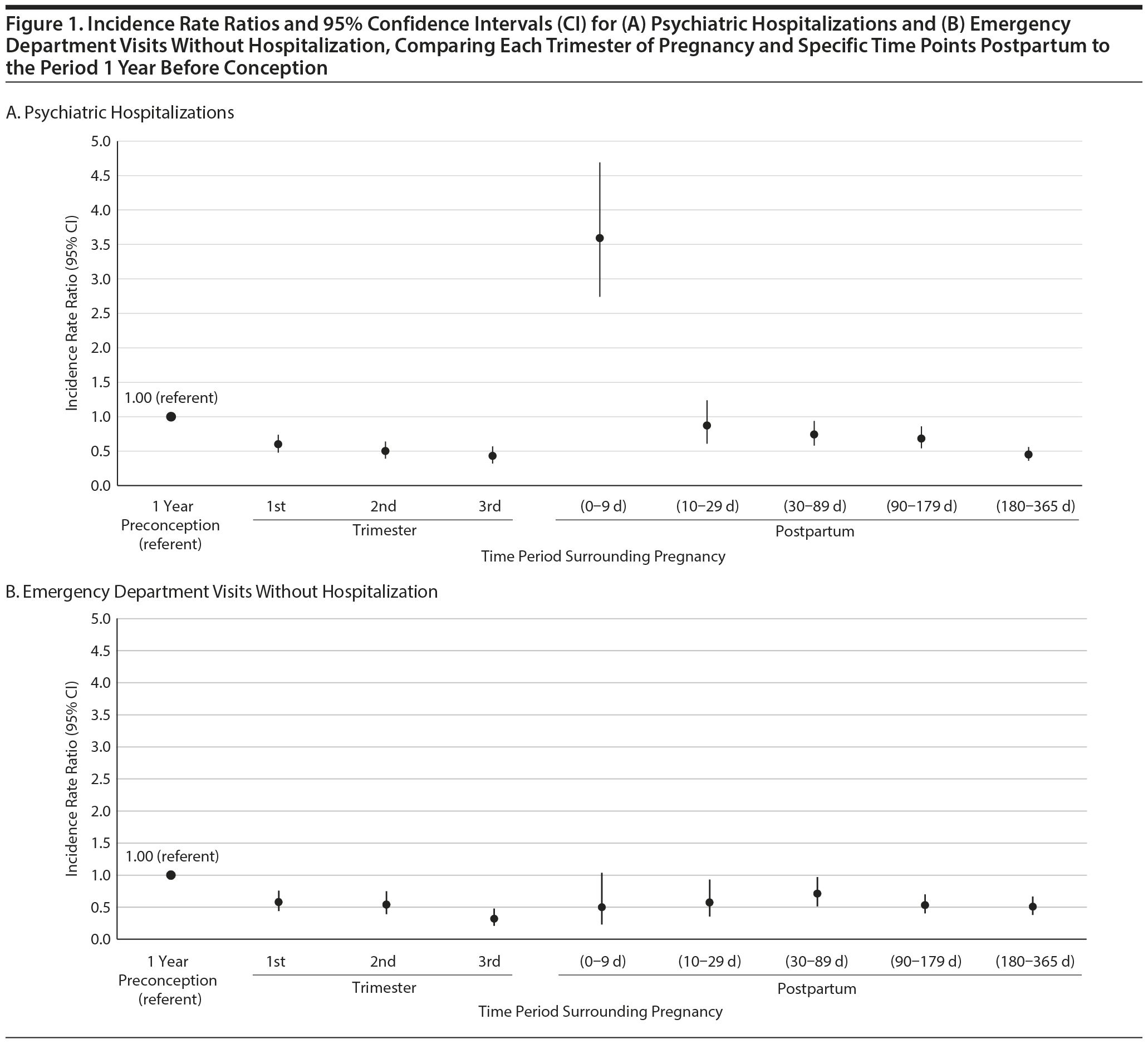
Rates of psychiatric emergency department visits without admission were lower throughout pregnancy and postpartum compared to the preconception year, with minimal difference between any time period during pregnancy or postpartum (Figure 1B). Rates of self-harm–related emergency department visits across subdivided time periods could not be reported for privacy reasons due to small cell sizes.
DISCUSSION
Among women with schizophrenia diagnosed before pregnancy, the risk for psychiatric hospitalization was lower during pregnancy and in the first year postpartum than in the 1 year prior to conception, with the exception of the first month postpartum. Increased risk for hospitalization was observed only in the first 9 days postpartum. The risk for both psychiatric emergency department visits without hospitalization and self-harm emergency department visits was consistently lower during pregnancy and postpartum compared to the prepregnancy year.
To our knowledge, this is the first study to report on population-based acute psychiatric care service use patterns across pregnancy and the postpartum period among women with schizophrenia. Our findings appear to be inconsistent with findings of previous clinical studies reporting prevalence rates for psychotic symptoms during pregnancy that ranged from 66% to 94%.5 However, the presence of psychotic symptoms is not necessarily synonymous with need for hospitalization. Further, previous study samples comprised only women who were hospitalized postpartum, so the participants in these previous studies may have represented subsamples of the population at the more severe end of the spectrum of illness. Two prior population-based studies focused on use of acute psychiatric services for women with schizophrenia in the postpartum period.22,23 About 19% of women in our study were hospitalized in the first year postpartum, a proportion substantially lower than the 55% estimate generated by Howard et al.22 However, the Howard et al study22 also included visits to a general practitioner’s office for new psychiatric symptoms. The proportion of women hospitalized in the 1 year postpartum in our study was closer to the 15.7% observed by Munk-Olsen et al.23 This latter study23 also found that women were most likely to be admitted to a psychiatric hospital during the first 9 days postpartum, but that the risk remained elevated until 60 days postpartum. While we used the preconception year as a comparator, the study by Munk-Olsen and colleagues used days 180 to 360 postpartum as their referent, which, based on our current findings, appears to be a period of relatively lower risk.
The observed reduction in acute psychiatric service use during pregnancy compared to preconception is consistent with the hypothesis that increased estrogen levels during pregnancy may protect against psychotic symptoms.15,16,36 Other factors may also have contributed to the reduced need for acute psychiatric services, including increased outpatient mental health service and community support, or even receipt of treatment in outpatient or inpatient antenatal care settings. It is also possible that there was increased adherence to treatment stemming from the motivation to have a healthy pregnancy and postchildbirth experience. The elevated risk for psychiatric hospitalization immediately postpartum is not surprising, given that the rapid fall of estrogen following delivery19,20,37 combined with the psychosocial stress of the transition to motherhood and increased sleep deprivation early postpartum may have increased the risk for psychotic symptom relapse.38,39 It may also reflect difficulties in the transition of care from hospital to home and/or a lower threshold for hospitalization out of concern for the well-being of a new mother and her newborn. The lack of a corresponding peak in emergency department visits without hospitalization during the first 9 days postpartum was unexpected. It is possible that some emergency department visits were avoided because women had proper access to ambulatory mental health care or other community service providers, such as Ontario public health nurses who make home visits for at-risk mothers and children.29 It may also be that most new mothers with schizophrenia who presented to the emergency department were subsequently hospitalized, as opposed to discharged home to community-based services. Emergency department encounters that result in hospitalization would be categorized as hospitalizations in our study, which could explain why we observed a peak in hospitalization but not in emergency department visits.
We did not expect to observe a decline in psychiatric hospitalization after the first completed month postpartum, nor to observe reduced rates of emergency department visits without hospitalization and self-harm throughout the perinatal period. This occurred despite the fact that almost 60% of our cohort of women were living in the 2 lowest income quintiles, that exposure to stress related to retaining child custody arises at this time in this population,4,40 and that women with schizophrenia are particularly vulnerable to stress.41,42 It is highly likely that new mothers had substantial interaction with their health care providers and social services in the postpartum period. Therefore, it could be that symptom exacerbations were caught early and treated and/or that professional support helped to mitigate stress. It may also be, as some qualitative studies of mothers with schizophrenia illustrate, that having a child often increases a woman’s pride and her motivation to remain psychiatrically stable. She may, therefore, be more likely to engage in strategies to maintain stability, such as improved self care or possibly relatively better adherence to antipsychotic medications, in order to keep her family intact.43–45 These same studies suggest that motherhood may bring a woman with schizophrenia closer to her extended family,43–45 providing valuable social supports that may protect against stress and symptom exacerbation.46,47 It is also important to consider that psychiatric stability may not be the only reason why hospitalization rates were lower postpartum than prior to conception. For example, concerns about potential loss of custody or a wish to avoid mother-infant separation may lead a woman and her health care providers to avoid acute psychiatric hospitalization whenever possible.
Our study has several strengths. To our knowledge, this is the first study to use a same-control design to investigate the impact of pregnancy and childbirth on use of acute psychiatric care services among women with schizophrenia. This approach is useful because several studies48–51 have shown that mothers with schizophrenia are at lower risk for adverse psychiatric outcomes, such as psychiatric hospitalization, self-harm, and suicide, compared to women with schizophrenia who have never given birth, possibly reflecting a selection process whereby women with schizophrenia who choose to become pregnant (or continue with unplanned pregnancies) have higher baseline stability and/or social supports relative to their same-diagnosis peers. By allowing a woman to serve as her own control, sociodemographic factors, family history, substance use, duration and severity of illness, and history of treatment adherence generally do not change, and the impact of pregnancy and childbirth can be better assessed. Other strengths of this study were our inclusion of a large sample of women with schizophrenia, including those without a previous psychiatric hospitalization. This population most likely represents a broader range of schizophrenia severity than previous study samples.23,52,53 We also examined the outcomes of women both prenatally and postnatally, while other studies were limited only to the postpartum period.3,22,23,52–54 A key study limitation is that we were unable to measure mental health outcomes at a symptom level; hence, while our outcomes serve as a proxy for psychiatric stability, they do not necessarily reflect minor symptom exacerbations that can be managed on an outpatient basis. In addition, our study could not identify particular events or issues that precipitated need for acute psychiatric services, such as financial or housing instability, loss of child custody,55–57 new loss of family or partner support, and/or medication nonadherence.58–62
In summary, except for the first 9 days after delivery, women with schizophrenia had lower use of acute psychiatric care services during pregnancy and postpartum, compared with the 1-year period before pregnancy. This finding is encouraging for women with schizophrenia, their families, and their care providers when making decisions about reproduction. Given that most women engage with the health care system for antenatal care, and that most deliver their infants in a hospital, these times are ideal for psychiatric and social services to establish therapeutic alliances that will enhance long-term recovery and enable a healthy transition to parenthood. Additional knowledge about how to identify those who are at greatest risk for illness exacerbation in the pregnancy or postpregnancy period is an important next step for optimizing psychiatric stability in women with schizophrenia who choose to become mothers.
Submitted: December 1, 2014; accepted May 6, 2015.
Online first: March 15, 2016.
Potential conflicts of interest: Dr Grigoriadis received compensation as a speaker and advisory board member for Eli Lilly, and Dr Seeman received compensation as a consultant for Clera Inc. No other authors have competing interests to declare: no support from any organization for the submitted work, no financial relationships with any organizations that might have an interest in the submitted work in the previous 3 years, and no other relationships or activities that could appear to have influenced the submitted work.
Funding/support: This study was funded by a research operating grant from the Canadian Institutes of Health Research (#120267). In addition, this study was supported by the Institute for Clinical Evaluative Sciences (ICES), which is funded by an annual grant from the Ontario Ministry of Health and Long-Term Care (MOHLTC).
Role of the sponsor: The funding agencies had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; or preparation, review, and approval of the manuscript.
Disclaimer: The opinions, results, and conclusions reported in this paper are those of the authors and are independent from the funding sources. No endorsement by ICES or the Ontario MOHLTC is intended or should be inferred.
Additional information: Data were accessed from the ICES in Toronto, Ontario, Canada. ICES is named as a prescribed entity under Section 45(1) of Ontario’s Personal Health Information Protection Act, 2004 (PHIPA). As a requirement of having this status in PHIPA, ICES policies, practices, and procedures are reviewed and approved by the Ontario Information and Privacy Commissioner. Access to raw data is governed by confidentiality agreements between ICES and independent investigators as per PHIPA guidelines.
REFERENCES
1. McGrath JJ, Hearle J, Jenner L, et al. The fertility and fecundity of patients with psychoses. Acta Psychiatr Scand. 1999;99(6):441–446. doi:10.1111/j.1600-0447.1999.tb00990.x PubMed
2. Vigod SN, Seeman MV, Ray JG, et al. Temporal trends in general and age-specific fertility rates among women with schizophrenia (1996–2009): a population-based study in Ontario, Canada. Schizophr Res. 2012;139(1–3):169–175. doi:10.1016/j.schres.2012.05.010 PubMed
3. Howard LM. Fertility and pregnancy in women with psychotic disorders. Eur J Obstet Gynecol Reprod Biol. 2005;119(1):3–10. doi:10.1016/j.ejogrb.2004.06.026 PubMed
4. Seeman M. Parenting issues in mothers with schizophrenia. Curr Womens Health Rev. 2010;6(1):51–57. doi:10.2174/157340410790979734
5. Vigod S, Ross L. Epidemiology of psychotic symptoms during pregnancy and postpartum in women with schizophrenia. Curr Womens Health Rev. 2010;6(1):17–21. doi:10.2174/157340410790979725
6. Stewart D. Pregnancy and schizophrenia. Can Fam Physician. 1984;30:1537–1542. PubMed
7. Jablensky AV, Morgan V, Zubrick SR, et al. Pregnancy, delivery, and neonatal complications in a population cohort of women with schizophrenia and major affective disorders. Am J Psychiatry. 2005;162(1):79–91. doi:10.1176/appi.ajp.162.1.79 PubMed
8. Sacker A, Done DJ, Crow TJ. Obstetric complications in children born to parents with schizophrenia: a meta-analysis of case-control studies. Psychol Med. 1996;26(2):279–287. doi:10.1017/S003329170003467X PubMed
9. Nilsson E, Lichtenstein P, Cnattingius S, et al. Women with schizophrenia: pregnancy outcome and infant death among their offspring. Schizophr Res. 2002;58(2–3):221–229. doi:10.1016/S0920-9964(01)00370-X PubMed
10. Hipwell AE, Goossens FA, Melhuish EC, et al. Severe maternal psychopathology and infant-mother attachment. Dev Psychopathol. 2000;12(2):157–175. doi:10.1017/S0954579400002030 PubMed
11. Näslund B, Persson-Blennow I, McNeil T, et al. Offspring of women with nonorganic psychosis: infant attachment to the mother at one year of age. Acta Psychiatr Scand. 1984;69(3):231–241. doi:10.1111/j.1600-0447.1984.tb02490.x PubMed
12. Henriksson KM, McNeil TF. Health and development in the first 4 years of life in offspring of women with schizophrenia and affective psychoses: Well-Baby Clinic information. Schizophr Res. 2004;70(1):39–48. doi:10.1016/j.schres.2003.11.003 PubMed
13. Hawes V, Cottrell D. Disruption of children’s lives by maternal psychiatric admission. Psychiatr Bull. 1999;23(3):153–156. doi:10.1192/pb.23.3.153
14. Schubert EW, McNeil TF. Prospective study of neurological abnormalities in offspring of women with psychosis: birth to adulthood. Am J Psychiatry. 2004;161(6):1030–1037. doi:10.1176/appi.ajp.161.6.1030 PubMed
15. Hendrick V, Altshuler LL, Suri R. Hormonal changes in the postpartum and implications for postpartum depression. Psychosomatics. 1998;39(2):93–101. doi:10.1016/S0033-3182(98)71355-6 PubMed
16. Grigoriadis S, Seeman MV. The role of estrogen in schizophrenia: implications for schizophrenia practice guidelines for women. Can J Psychiatry. 2002;47(5):437–442. PubMed
17. Seeman MV, Lang M. The role of estrogens in schizophrenia gender differences. Schizophr Bull. 1990;16(2):185–194. doi:10.1093/schbul/16.2.185 PubMed
18. Seeman MV. The role of sex hormones in psychopathology: focus on schizophrenia. Prim Care. 2002;29(1):171–182, viii. doi:10.1016/S0095-4543(03)00080-0 PubMed
19. Wieck A, Kumar R, Hirst AD, et al. Increased sensitivity of dopamine receptors and recurrence of affective psychosis after childbirth. BMJ. 1991;303(6803):613–616. doi:10.1136/bmj.303.6803.613 PubMed
20. Meakin CJ, Brockington IF, Lynch S, et al. Dopamine supersensitivity and hormonal status in puerperal psychosis. Br J Psychiatry. 1995;166(1):73–79. doi:10.1192/bjp.166.1.73 PubMed
21. Jones I, Chandra PS, Dazzan P, et al. Bipolar disorder, affective psychosis, and schizophrenia in pregnancy and the post-partum period. Lancet. 2014;384(9956):1789–1799. doi:10.1016/S0140-6736(14)61278-2 PubMed
22. Howard LM, Goss C, Leese M, et al. The psychosocial outcome of pregnancy in women with psychotic disorders. Schizophr Res. 2004;71(1):49–60. doi:10.1016/j.schres.2004.01.003 PubMed
23. Munk-Olsen T, Laursen TM, Mendelson T, et al. Risks and predictors of readmission for a mental disorder during the postpartum period. Arch Gen Psychiatry. 2009;66(2):189–195. doi:10.1001/archgenpsychiatry.2008.528 PubMed
24. Maaten S, Guttman A, Kopp A, et al. Care of women during pregnancy and childbirth. In: Jaakkimainen L, Upshur RE, Klein-Geltink JE, eds. Primary Care in Ontario ICES Atlas, Chapter 2. Toronto, Canada: Institute for Clinical Evaluative Sciences; 2006:15–34.
25. Williams JI, O’Brien BJ, Sellors C, et al. A summary of studies on the quality of health care administrative databases in Canada. In: Goel V, Williams JI, Anderson GM, et al, eds. Patterns of Health Care in Ontario. The ICES Practice Atlas. Ottawa, Canada: Canadian Medical Association; 1996:339–345.
26. Becker T, Hux J. Risk of acute complications of diabetes among people with schizophrenia in Ontario, Canada. Diabetes Care. 2011;34(2):398–402. doi:10.2337/dc10-1139 PubMed
27. Juurlink D, Preyra C, Croxford R, et al. Canadian Institute for Health Information Discharge Abstract Database: A Validation Study. Toronto, Canada: Institute for Clinical Evaluative Sciences; 2006.
28. Vigod SN, Kurdyak PA, Dennis CL, et al. Maternal and newborn outcomes among women with schizophrenia: a retrospective population-based cohort study. BJOG. 2014;121(5):566–574. doi:10.1111/1471-0528.12567 PubMed
29. Canadian Institute for Health Information and Statistics Canada. Health Indicators 2011. Ottawa, Canada: CIHI; 2011. Available at: https://secure.cihi.ca/free_products/health_indicators_2011_en.pdf.
30. Bethell J, Rhodes AE. Identifying deliberate self-harm in emergency department data. Health Rep. 2009;20(2):35–42. PubMed
31. Urquia ML, Frank JW, Glazier RH, et al. Birth outcomes by neighbourhood income and recent immigration in Toronto. Health Rep. 2007;18(4):21–30. PubMed
32. Steele LS, Glazier RH, Lin E, et al. Using administrative data to measure ambulatory mental health service provision in primary care. Med Care. 2004;42(10):960–965. doi:10.1097/00005650-200410000-00004 PubMed
33. Vigod SN, Kurdyak PA, Stewart DE, et al. Depressive symptoms as a determinant of breast and cervical cancer screening in women: a population-based study in Ontario, Canada. Arch Women Ment Health. 2011;14(2):159–168. doi:10.1007/s00737-011-0210-x PubMed
34. Parkerson GR Jr, Harrell FE Jr, Hammond WE, et al. Characteristics of adult primary care patients as predictors of future health services charges. Med Care. 2001;39(11):1170–1181. doi:10.1097/00005650-200111000-00004 PubMed
35. The Johns Hopkins ACG System. Technical Reference Guide Version 9.0 [computer program]. Baltimore, MD: Health Services Research & Development Center, Johns Hopkins University, Bloomberg School of Public Health; 2009.
36. Kapur S. How antipsychotics become anti-“psychotic”—from dopamine to salience to psychosis. Trends Pharmacol Sci. 2004;25(8):402–406. doi:10.1016/j.tips.2004.06.005 PubMed
37. Cookson JC. Post-partum mania, dopamine, and estrogens. Lancet. 1982;2(8299):672. doi:10.1016/S0140-6736(82)92778-7 PubMed
38. Dørheim SK, Bondevik GT, Eberhard-Gran M, et al. Sleep and depression in postpartum women: a population-based study. Sleep. 2009;32(7):847–855. PubMed
39. Goyal D, Gay CL, Lee KA. Patterns of sleep disruption and depressive symptoms in new mothers. J Perinat Neonatal Nurs. 2007;21(2):123–129. doi:10.1097/01.JPN.0000270629.58746.96 PubMed
40. Sands RG, Koppelman N, Solomon P. Maternal custody status and living arrangements of children of women with severe mental illness. Health Soc Work. 2004;29(4):317–325. doi:10.1093/hsw/29.4.317 PubMed
41. Horan WP, Blanchard JJ. Emotional responses to psychosocial stress in schizophrenia: the role of individual differences in affective traits and coping. Schizophr Res. 2003;60(2–3):271–283. doi:10.1016/S0920-9964(02)00227-X PubMed
42. Jansen LM, Gispen-de Wied CC, Kahn RS. Selective impairments in the stress response in schizophrenic patients. Psychopharmacology (Berl). 2000;149(3):319–325. doi:10.1007/s002130000381 PubMed
43. Montgomery P, Tompkins C, Forchuk C, et al. Keeping close: mothering with serious mental illness. J Adv Nurs. 2006;54(1):20–28. doi:10.1111/j.1365-2648.2006.03785.x PubMed
44. Montgomery P. Mothers with a serious mental illness: a critical review of the literature. Arch Psychiatr Nurs. 2005;19(5):226–235. PubMed
45. Ackerson B. Coping with the dual demands of severe mental illness and parenting: the parents’ perspective. Fam Soc. 2003;84(1):109–118. doi:10.1606/1044-3894.69
46. Paykel ES, Emms EM, Fletcher J, et al. Life events and social support in puerperal depression. Br J Psychiatry. 1980;136(4):339–346. doi:10.1192/bjp.136.4.339 PubMed
47. O’Hara MW, Rehm LP, Campbell SB. Postpartum depression. a role for social network and life stress variables. J Nerv Ment Dis. 1983;171(6):336–341. doi:10.1097/00005053-198306000-00002 PubMed
48. Appleby L. Suicide during pregnancy and in the first postnatal year. BMJ. 1991;302(6769):137–140. doi:10.1136/bmj.302.6769.137 PubMed
49. Appleby L, Turnbull G. Parasuicide in the first postnatal year. Psychol Med. 1995;25(5):1087–1090. doi:10.1017/S0033291700037570 PubMed
50. Yarden PEMD, Eisenbach Z. The effect of childbirth on the prognosis of married schizophrenic women. Br J Psychiatry. 1966;112(486):491–499. doi:10.1192/bjp.112.486.491
51. Vesga-López O, Blanco C, Keyes K, et al. Psychiatric disorders in pregnant and postpartum women in the United States. Arch Gen Psychiatry. 2008;65(7):805–815. doi:10.1001/archpsyc.65.7.805 PubMed
52. Davies A, McIvor RJ, Kumar RC. Impact of childbirth on a series of schizophrenic mothers: a comment on the possible influence of oestrogen on schizophrenia. Schizophr Res. 1995;16(1):25–31. doi:10.1016/0920-9964(94)00062-D PubMed
53. Davidson J, Robertson E. A follow-up study of post partum illness, 1946–1978. Acta Psychiatr Scand. 1985;71(5):451–457. doi:10.1111/j.1600-0447.1985.tb05057.x PubMed
54. McNeil TF. A prospective study of postpartum psychoses in a high-risk group, 1: clinical characteristics of the current postpartum episodes. Acta Psychiatr Scand. 1986;74(2):205–216. doi:10.1111/j.1600-0447.1986.tb10607.x PubMed
55. Norman RM, Malla AK. Stressful life events and schizophrenia, I: a review of the research. Br J Psychiatry. 1993;162(2):161–166. doi:10.1192/bjp.162.2.161 PubMed
56. Birley JL, Brown GW. Crises and life changes preceding the onset or relapse of acute schizophrenia: clinical aspects. Br J Psychiatry. 1970;116(532):327–333. doi:10.1192/bjp.116.532.327 PubMed
57. Nuechterlein KH, Dawson ME, Ventura J, et al. The vulnerability/stress model of schizophrenic relapse: a longitudinal study. Acta Psychiatr Scand suppl. 1994;382:58–64. doi:10.1111/j.1600-0447.1994.tb05867.x PubMed
58. Robinson GE. Treatment of schizophrenia in pregnancy and postpartum. J Popul Ther Clin Pharmacol. 2012;19(3):e380–e386. PubMed
59. The Canadian Psychiatric Association. Canadian clinical practice guidelines for the treatment of schizophrenia. Can J Psychiatry. 1998;43(suppl 2):25S–40S. PubMed
60. Cohen LS, Altshuler LL, Harlow BL, et al. Relapse of major depression during pregnancy in women who maintain or discontinue antidepressant treatment. JAMA. 2006;295(5):499–507. doi:10.1001/jama.295.5.499 PubMed
61. Bonari L, Koren G, Einarson TR, et al. Use of antidepressants by pregnant women: evaluation of perception of risk, efficacy of evidence based counseling and determinants of decision making. Arch Women Ment Health. 2005;8(4):214–220. doi:10.1007/s00737-005-0094-8 PubMed
62. Viguera AC, Whitfield T, Baldessarini RJ, et al. Risk of recurrence in women with bipolar disorder during pregnancy: prospective study of mood stabilizer discontinuation. Am J Psychiatry. 2007;164(12):1817–1824, quiz 1923. doi:10.1176/appi.ajp.2007.06101639 PubMed
This PDF is free for all visitors!

