Objective: Lithium is an effective treatment for mood disorders, but lithium level and renal monitoring every 3 months is recommended in older patients treated with lithium to prevent serious adverse events. This study examined lithium monitoring practices in a large geriatric cohort.
Methods: This population-based cohort study (N = 11,006) used linked health care administrative databases. Older lithium users (n = 5,503; mean age = 70.6 years) in Ontario, Canada, enrolled between April 1, 2002, and March 31, 2014, were propensity score matched 1:1 to valproate users (n = 5,503). The frequency with which serum lithium levels were monitored and renal and endocrine laboratory testing was done during a 1-year follow-up period was examined.
Results: The baseline characteristics of the 2 groups were similar. At least 1 serum lithium concentration recorded within 90, 180, and 365 days of follow-up was present in 24.1%, 42.4%, and 66.8% of lithium users, respectively. Corresponding numbers for serum creatinine were 29.6%, 50.4%, and 75.4%, respectively. While serum creatinine monitoring (hazard ratio [HR] = 1.19; 95% CI, 1.12-1.27; P < .001), thyroid-stimulating hormone monitoring (HR = 1.47; 95% CI, 1.37-1.58; P < .001), and calcium testing (HR = 1.15; 95% CI, 1.02-1.29; P = .018) were statistically higher in lithium compared to valproate users, absolute differences between groups were not clinically meaningful.
Conclusions: In a geriatric Canadian community sample, lithium monitoring was infrequent and inconsistent with international standards that call for screening of lithium levels and renal function every 3 months.

CME Background
Articles are selected for credit designation based on an assessment of the educational needs of CME participants, with the purpose of providing readers with a curriculum of CME articles on a variety of topics throughout each volume. Activities are planned using a process that links identified needs with desired results.
To obtain credit, read the article, correctly answer the questions in the Posttest, and complete the Evaluation. A $5 processing fee will apply.
CME Objective
After studying this article, you should be able to:
- Provide serum lithium monitoring that is consistent with international standards
Accreditation Statement
The CME Institute of Physicians Postgraduate Press, Inc., is accredited by the Accreditation Council for Continuing Medical Education to provide continuing medical education for physicians.
Credit Designation
The CME Institute of Physicians Postgraduate Press, Inc., designates this journal-based CME activity for a maximum of 1 AMA PRA Category 1 Creditâ„¢. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Note: The American Academy of Physician Assistants (AAPA) accepts certificates of participation for educational activities certified for AMA PRA Category 1 Creditâ„¢ from organizations accredited by ACCME or a recognized state medical society. Physician assistants may receive a maximum of 1 hour of Category I credit for completing this program.
Release, Expiration, and Review Dates
This educational activity was published in November 2018 and is eligible forAMA PRA Category 1 Creditâ„¢ through December 31, 2020. The latest review of this material was November 2018.
Financial Disclosure
All individuals in a position to influence the content of this activity were asked to complete a statement regarding all relevant personal financial relationships between themselves or their spouse/partner and any commercial interest. The CME Institute has resolved any conflicts of interest that were identified. In the past year, Marlene P. Freeman, MD, Editor in Chief, has received research funding from JayMac and Sage, has been a member of the Independent Data Safety and Monitoring Committee for Janssen, has been medical editor for the Global Organization for EPA and DHA Omega-3s newsletter, and, as a Massachusetts General Hospital (MGH) employee, works with the MGH National Pregnancy Registry, which is sponsored by Alkermes, Otsuka, Actavis, and Sunovion, and works with the MGH Clinical Trials Network and Institute, which receives research funding from multiple pharmaceutical companies and the National Institute of Mental Health. No member of the CME Institute staff reported any relevant personal financial relationships. Faculty financial disclosure appears at the end of the article.
ABSTRACT
Objective: Lithium is an effective treatment for mood disorders, but lithium level and renal monitoring every 3 months is recommended in older patients treated with lithium to prevent serious adverse events. This study examined lithium monitoring practices in a large geriatric cohort.
Methods: This population-based cohort study (N = 11,006) used linked health care administrative databases. Older lithium users (n = 5,503; mean age = 70.6 years) in Ontario, Canada, enrolled between April 1, 2002, and March 31, 2014, were propensity score matched 1:1 to valproate users (n = 5,503). The frequency with which serum lithium levels were monitored and renal and endocrine laboratory testing was done during a 1-year follow-up period was examined.
Results: The baseline characteristics of the 2 groups were similar. At least 1 serum lithium concentration recorded within 90, 180, and 365 days of follow-up was present in 24.1%, 42.4%, and 66.8% of lithium users, respectively. Corresponding numbers for serum creatinine were 29.6%, 50.4%, and 75.4%, respectively. While serum creatinine monitoring (hazard ratio [HR] = 1.19; 95% CI, 1.12-1.27; P < .001), thyroid-stimulating hormone monitoring (HR = 1.47; 95% CI, 1.37-1.58; P < .001), and calcium testing (HR = 1.15; 95% CI, 1.02-1.29; P = .018) were statistically higher in lithium compared to valproate users, absolute differences between groups were not clinically meaningful.
Conclusions: In a geriatric Canadian community sample, lithium monitoring was infrequent and inconsistent with international standards that call for screening of lithium levels and renal function every 3 months.
J Clin Psychiatry 2018;79(6):17m12095
To cite: Rej S, Herrmann N, Gruneir A, et al. Blood lithium monitoring practices in a population-based sample of older adults. J Clin Psychiatry. 2018;79(6):17m12095.
To share: https://doi.org/10.4088/JCP.17m12095
© Copyright 2018 Physicians Postgraduate Press, Inc.
aDepartment of Psychiatry, Lady Davis Institute/Jewish General Hospital, McGill University, Montreal, Québec, Canada
bDepartment of Psychiatry, Sunnybrook Health Sciences Centre, University of Toronto, Toronto, Ontario, Canada
cWomen’s College Research Institute, Women’s College Hospital, Toronto, Ontario, Canada
dDepartment of Family Medicine, University of Alberta, Edmonton, Alberta, Canada
eInstitute for Clinical Evaluative Sciences, London, Ontario, Canada
*Corresponding author: Soham Rej, MD, MSc, Department of Psychiatry, Lady Davis Institute/Jewish General Hospital, McGill University, 4333 Cote-Ste-Catherine, Rm 144, Montreal, Québec, Canada, H3T 1E4 ([email protected]).
Lithium remains an essential medication for bipolar disorder and treatment-resistant depression.1 However, there has been concern about adverse effects of lithium on the functioning of a variety of organ systems, including endocrine and renal function.2-4 In recent studies, lithium was independently associated with an almost 2-fold higher risk of chronic kidney disease in older mental health service users in the community,2,5 although this finding remains controversial.6 Similar associations have been found for lithium use and a higher risk of acute kidney injury,7 as well as thyroid disease4 and hypercalcemia.8 Lithium has a narrow therapeutic range, so laboratory monitoring is essential; poor monitoring has been associated with acute and chronic lithium level elevations and a higher incidence of most lithium-associated adverse effects.9 These findings suggest that monitoring is critical to the safe usage of lithium.
Poor laboratory monitoring may contribute to lithium-related adverse events in community settings, where monitoring may be less regimented.10,11 Guidelines from the International Society for Bipolar Disorders (ISBD), Canadian Network for Mood and Anxiety Treatments (CANMAT), and the UK National Institute for Clinical Excellence (NICE) recommend that lithium users undergo lithium level and serum creatinine tests at least every 3-6 months1,12,13 and ideally every 3 months in older adults.12,14 Similarly, international guidelines recommend that thyroid and calcium testing be performed every 6-12 months.1,12,13 Despite guidelines, lithium monitoring has been poor in different parts of world, with less than 25% of patients in UK and New Zealand population-based studies receiving lithium monitoring ≥ 4 times a year.13,15,16
Lithium monitoring has not been assessed in older lithium-treated individuals receiving routine health care in a large population-based cohort, even though these individuals are most vulnerable for lithium-associated adverse effects and may benefit from more frequent monitoring.10 To understand how much more monitoring occurs beyond routine testing, it would be useful to assess serum monitoring in lithium users compared to users of another drug such as valproic acid, which is also used for mental illness and does not require renal, thyroid, or calcium monitoring,5 as well as to control statistically for potential confounders that may affect monitoring frequency. However, such research has not previously been done. We conducted this study to address these gaps in knowledge.
Aims of the Study
The aims of our study were to characterize the frequency of relevant laboratory monitoring in a province-wide cohort of older lithium users, specifically (1) lithium level and renal laboratory monitoring and (2) thyroid and calcium monitoring. We compared these monitoring rates to assess whether they differ from those of valproate users, who are similar but do not require renal, thyroid, or calcium monitoring.

- In a Canadian community setting, serum lithium monitoring is not consistent with international standards that call for screening of lithium levels and renal function every 3 months in older adults.
- Rates of testing for serum creatinine, thyroid-stimulating hormone, and calcium levels were statistically higher in the lithium-treated group compared to the valproate-treated group, but the differences did not appear to be clinically meaningful.
- The findings in this sample suggest that clinicians are not necessarily performing additional monitoring in older lithium users.
MATERIALS AND METHODS
Design and Setting
We conducted a population-based cohort study examining adults aged 66 years and older residing in the province of Ontario, Canada, who initiated treatment with either lithium or valproate between April 1, 2002, and March 31, 2014. All Ontario residents receive hospital care and physician services paid for by the Ontario Health Insurance Plan (OHIP), and those aged 65 years and older receive outpatient prescription drug coverage from the Ontario Drug Benefit program (ODB).
We conducted this study at the Institute for Clinical Evaluative Sciences (ICES) according to a prespecified protocol approved by the Research Ethics Board at Sunnybrook Health Sciences Centre (Toronto, Ontario). Participant informed consent was not required for this study. The reporting of this study followed guidelines for observational studies (Supplementary Table 1).
Data Sources
We obtained health care data from multiple linked administrative databases housed at ICES. Data sets were linked using unique, encoded identifiers derived from health card numbers, and patient-level data were analyzed at ICES. Vital statistics were obtained from the Registered Persons Database, which contains demographic data on all Ontario residents with a valid health card. Prescription data were obtained from the ODB database, which contains highly accurate records of all outpatient prescriptions (error rate < 1%) dispensed to patients aged 65 years and older. Diagnostic and procedural information on all hospital admissions and emergency department visits were obtained from the Canadian Institute for Health Information (CIHI) Discharge Abstract Database and CIHI National Ambulatory Care Reporting System database. The OHIP database includes claims for inpatient and outpatient physician services and was used to ascertain covariate information and outpatient laboratory test results. The ICES Physician Database reports prescriber and specialist referral data. For a small proportion of the cohort, outpatient serum creatinine values were provided by a large commercial laboratory in the year before cohort entry, and we determined kidney function in this subpopulation using these data to estimate glomerular filtration rate. Previous studies17-19 have used these databases to study medication use and associated health care use and outcomes.
Patients: Lithium Users and Propensity Score Matched Valproate Comparators
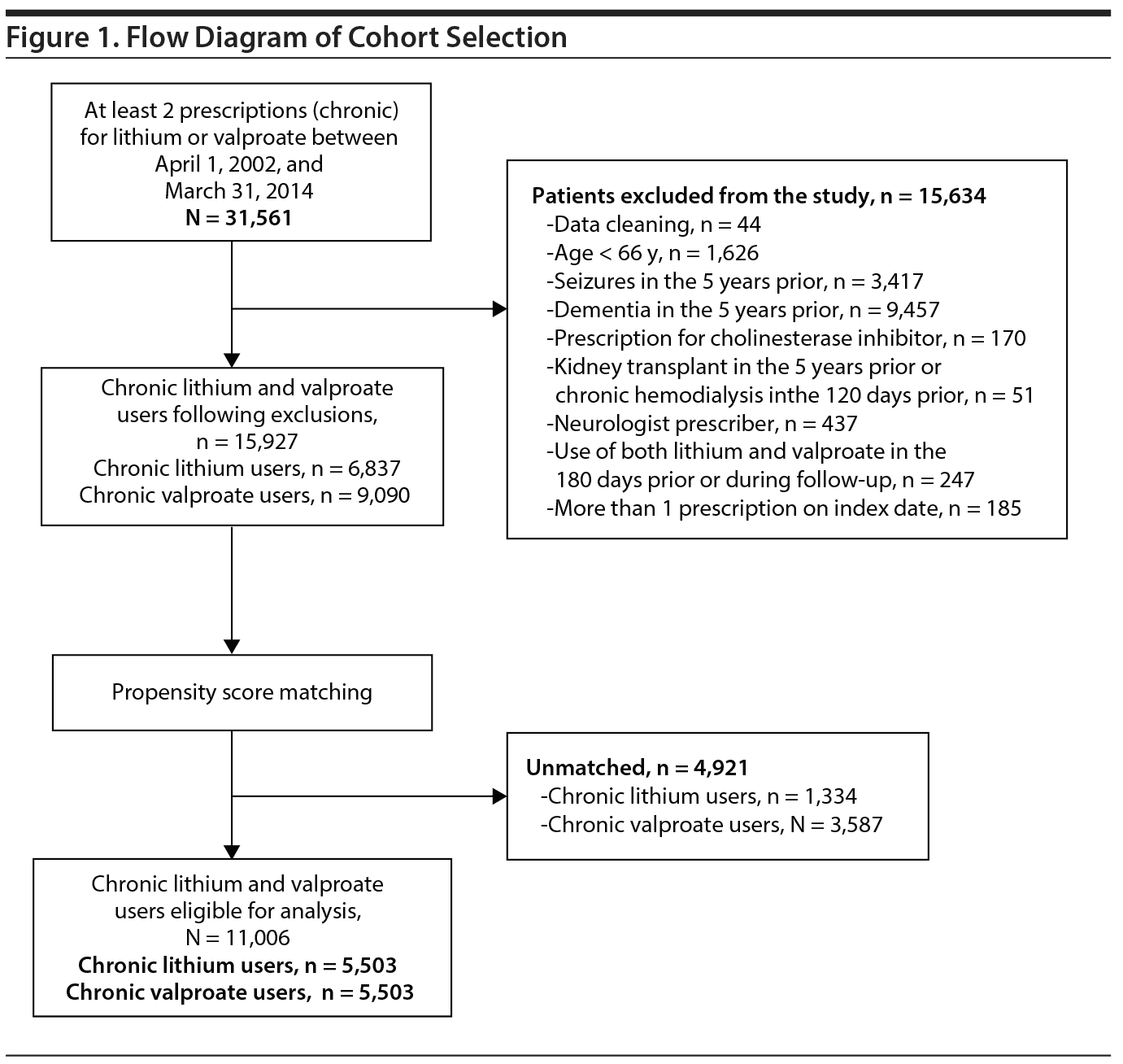
Our cohort included patients aged ≥ 66 years in Ontario who were chronic users of either lithium or valproate (study drug identification numbers outlined in Supplementary Table 2). A flow diagram for cohort selection can be found in Figure 1. We searched for any use of lithium or valproate between April 1, 2002, and March 31, 2014. For a patient to be considered a “chronic user,” we required a second prescription fill within 1.5 times the number of days supplied with the initial prescription (ie, second prescription within 45 days if the initial prescription had 30 days supplied). In the less likely case that a patient received a short lithium prescription (eg, 15 days) and 1 refill, such a person would still be included as a chronic lithium user; however, if they stopped getting lithium prescriptions after the second prescription, we would have stopped their follow-up at that point as well. We then deemed the date of the second prescription fill the index date (start time for follow-up in the cohort). After the index date, we followed individuals as long as they were continuously refilling the medications. Thus, there would be no instances when a patient stopped the drug shortly after index, but then was examined for the entire year of follow-up; such a patient’s follow-up time would have been censored at medication stop. Patients were censored when there was no prescription refill claim within a set period of time relative to the prescription fill immediately prior (1.5 times the number of days supplied). Chronic lithium users were compared to a cohort of chronic valproate users with similar baseline indicators of health. Valproate was chosen as the comparator because (1) compared to the general population, lithium and valproate users both have high rates of physical health comorbidity20 and low medication/monitoring adherence21; and (2) it allowed us to observe whether monitoring was more frequent in lithium users relative to a mental illness cohort exposed to a medication (valproate) that does not specifically require renal, thyroid, or calcium monitoring.
Before matching, we excluded a small proportion of patients from both cohorts due to data issues (less than 0.2%, n = 44) who had invalid identifiers (age, sex, or invalid health card number); were non-Ontario residents; or who had a date of death on or before the index date. We also excluded patients with the following criteria: those aged less than 66 years on the index date, to ensure complete medication history; those with evidence of seizure or dementia in the 5 years prior to index date; those with prescriptions dispensed by a neurologist (because valproate is sometimes prescribed for seizure and behavioral symptoms of dementia, whereas we wished to capture valproate users with mental illness); those with evidence of cholinesterase inhibitor use (also characterizes dementia); those with evidence of a kidney transplant in the 5 years prior to index date; those with evidence of dialysis in the 120 days prior to index date, as these treatments dramatically influence renal blood monitoring; and those with evidence of prescriptions for more than 1 study drug, to ensure mutually exclusive groups. Patients could enter the cohort only once, so for patients with multiple eligible prescriptions, we restricted to the first eligible prescription.
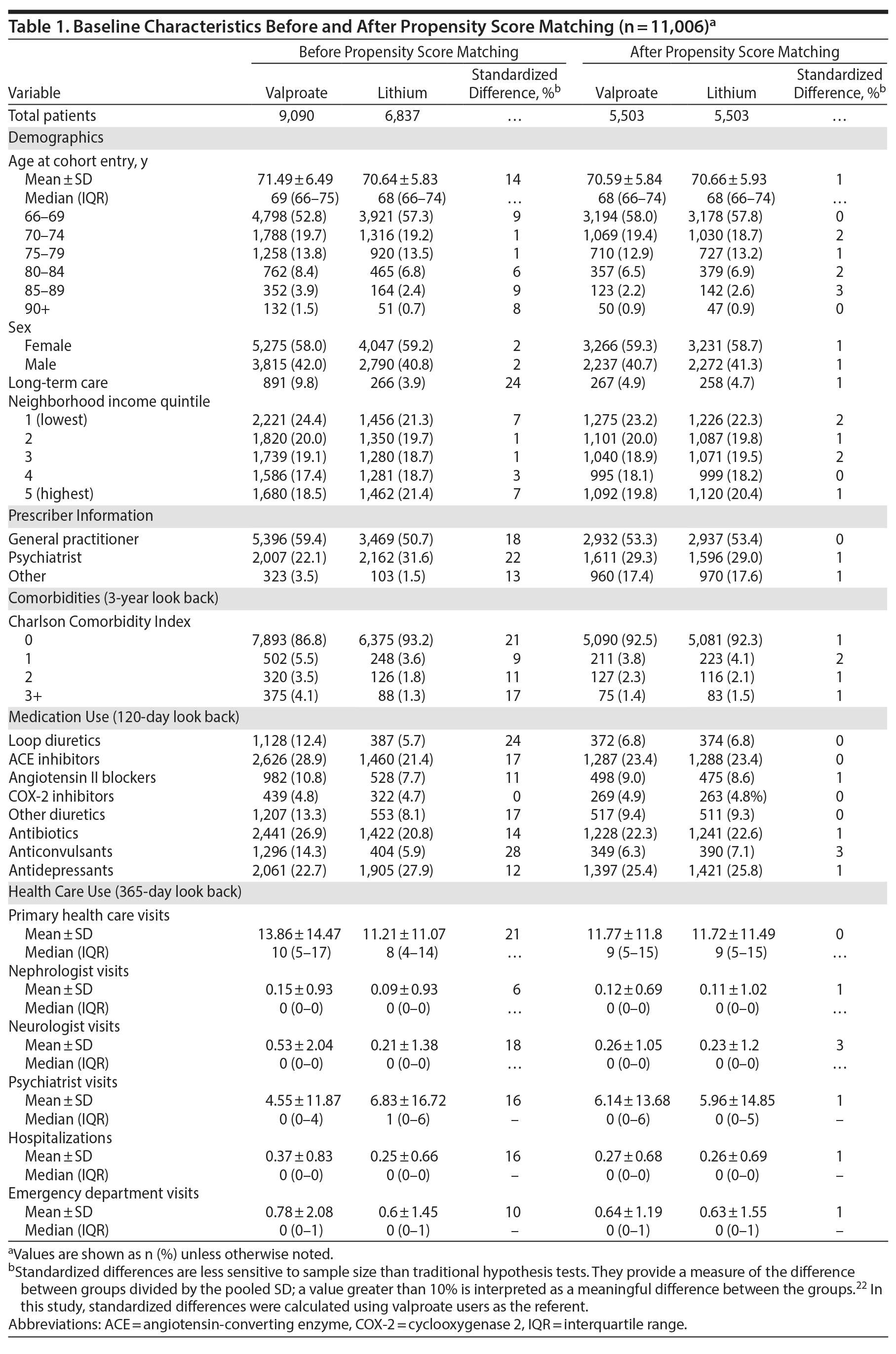
We used propensity score matching to eliminate systematic differences in the measured baseline characteristics of the lithium and valproate groups. Multivariable logistic regression was performed with 35 baseline characteristics selected for their potential influence on outcomes or segregation of patients between groups (listed in Supplementary Table 3). Chronic lithium users were matched 1:1 with chronic valproate users using greedy matching without replacement, within 0.2 standard deviations of the logit of the propensity score. We used standardized differences to assess differences in baseline characteristics between lithium and valproate users. Standardized differences describe differences between group means relative to the pooled standard deviation, with differences greater than 10% considered significant.22
Codes from the International Statistical Classification of Diseases and Related Health Problems, 10th revision (ICD-10) and 9th revision (ICD-9), were used to assess baseline comorbidities in the 5 years prior to the index date (Supplementary Table 4). Medication use was assessed in the 120 days before index date, and health care use, including physician visits and diagnostic and screening tests, was examined in the previous year.
Outcomes
The primary outcomes of our study were outpatient laboratory tests identified in OHIP for serum lithium levels, creatinine, thyroid-stimulating hormone (TSH), and calcium concentrations within 1 year following the index date. The outpatient physician billing codes used to identify laboratory tests are presented in Supplementary Table 5. Frequency of serum lithium laboratory testing was examined in lithium users only; the frequency of the remaining serum laboratory tests and rates of continuous medication use was compared between chronic lithium and valproate users.
Statistical Analysis
We compared the frequency of outpatient laboratory testing between propensity score matched lithium and valproate users. Rates were reported per 1,000 person-years. To ascertain whether laboratory testing followed international guidelines, we also examined the cumulative incidence of a test at 90 days, 180 days, and 365 days following the index date, with cumulative incidences estimated using the “exponential equation.”23 Time to first laboratory test monitoring after index prescription was compared between lithium and valproate users using hazard ratios (HRs), generated from Cox proportional hazard regression models, accounting for matched pairs. Analyses were conducted with SAS version 9.4 (SAS Institute, Cary, North Carolina, 2011) at the ICES Western facility (London, Ontario, Canada).
RESULTS
A total of 5,503 lithium users were matched 1:1 to 5,503 valproate users. In the overall sample (N = 11,006), the mean (SD) age of patients was 70.6 (5.9) years, and 59.0% of patients (n = 6,497) were female. After propensity score matching, lithium and valproate users did not differ with respect to their measured baseline characteristics (standardized difference < 10%). Patients’ baseline characteristics are described in Table 1 and Supplementary Table 6.
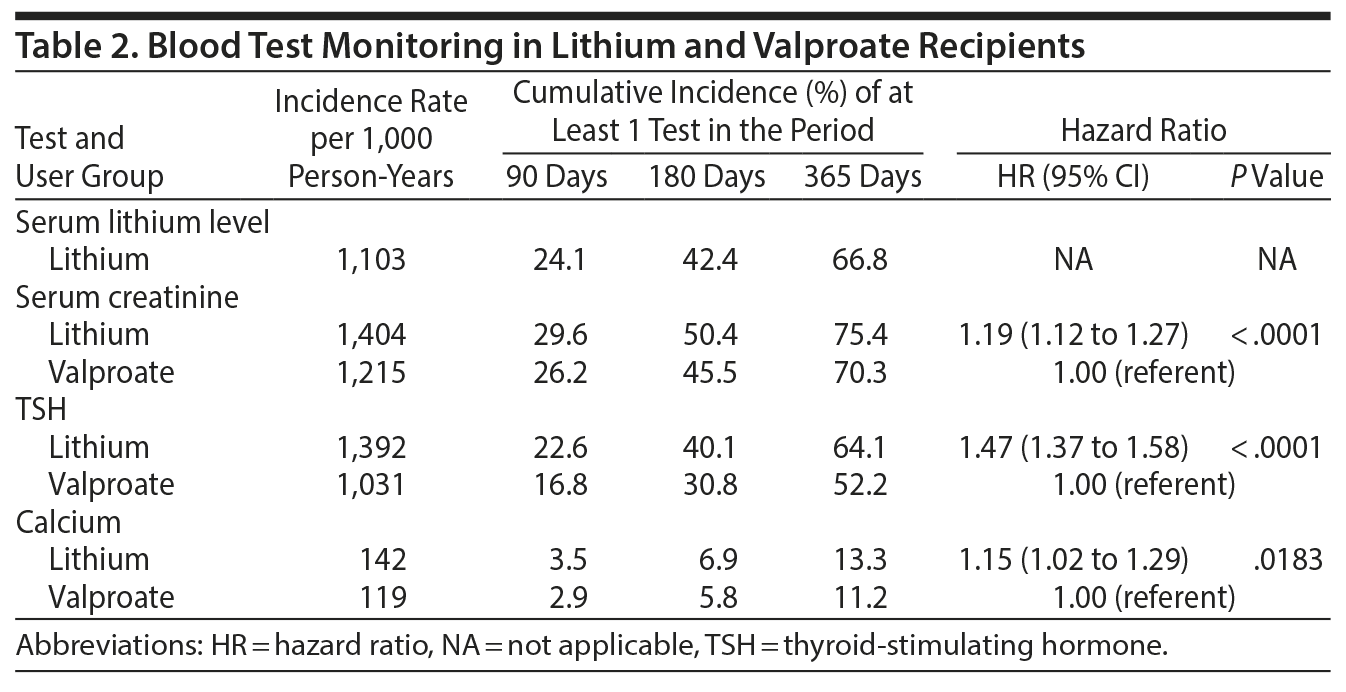
Lithium level monitoring was infrequent in lithium users at 90, 180, and 365 days of follow-up (24.1%, 42.4%, and 66.8%, respectively) (Table 2) in light of international guidelines to monitor every 3 months.12,14 Monitoring rates for serum creatinine were statistically higher in lithium compared to valproate users at 90, 180, and 365 days (lithium vs valproate: 29.6% vs 26.2%, 50.4% vs 45.5%, and 75.4% vs 70.3%, respectively; HR = 1.19; 95% CI, 1.12-1.27; P < .001). Similarly, at 90, 180, and 365 days, TSH and calcium annual testing rates were statistically higher in lithium users compared to valproate users: 22.6%, 40.1%, and 64.1%, respectively, vs 16.8%, 30.8%, and 52.2% for TSH (HR = 1.47; 95% CI, 1.37-1.58; P < .001) and 3.5%, 6.9%, and 13.3%, respectively, vs 2.9%, 5.8%, and 11.2% for calcium levels (HR = 1.15; 95% CI, 1.02-1.29; P = .018).
DISCUSSION
In our province-wide sample of older lithium users, lithium level and serum creatinine monitoring was relatively infrequent: only 24.1% and 29.6% of lithium users met international standards from the ISBD and NICE guidelines to ideally screen for lithium levels and renal function, respectively, every 3 months in older adults.12,14 While the serum creatinine monitoring rates at 90-, 180-, and 365-day follow-up were statistically higher in lithium compared to valproate users, these differences were not clinically meaningful, even though valproate is not associated with renal disease.5 Annual monitoring rates for TSH and calcium levels were also statistically different in lithium and valproate patients in our sample of 11,006 patients, but these differences may not have necessarily been clinically important. Even though the international guidelines and a recent, important 2014 Health Canada safety advisory recommend annual calcium testing in lithium-treated patients (http://healthycanadians.gc.ca/recall-alert-rappel-avis/hc-sc/2014/37903a-eng.php), only 13.3% of lithium-treated patients in this Canadian sample were tested for serum calcium within 1 year, compared to 11.2% of valproate-treated patients. These initial observations from a Canadian provincial sample suggest that (1) lithium monitoring among older adults may not be meeting current international standards1,12,13 and (2) the monitoring that is taking place may reflect routine laboratory monitoring for older adults (eg, as primary screening or for other medical comorbidities) rather than monitoring specifically for lithium safety.
We suspect a number of physician and patient factors act as barriers to adhering to recommended blood monitoring. It is likely that primary care physicians and many psychiatrists may not necessarily be familiar with lithium monitoring given the decreasing use of this important therapy, as mentioned previously. Monitoring can be a burden on patients since it requires scheduling, taking time for the appointment (and often long waits), and arranging transportation (which may not be trivial for individuals with mobility limitations or who live in remote settings). Patients may also not be aware of why regular monitoring is being recommended. Older lithium users often have significant cognitive, physical, and mental health issues that make it even more difficult to remember, commit to, and arrange for blood monitoring, with caregiver support often inadequate or overwhelmed.14
Our findings are in keeping with those of previous reports in younger adults and mixed-aged lithium samples in the United Kingdom and New Zealand.13,15,16 Lithium level and renal monitoring rates of 4 times/year, approaching the NICE and ISBD guideline recommendations (every 3 months), were similarly < 25%. Our study included novel elements: a larger sample size, calcium and thyroid monitoring frequencies, and a propensity score matched comparator group of patients with mental illness. These elements allowed us to clinically interpret that monitoring rates in lithium users were different, though perhaps not in a clinically meaningful way, compared to valproate users who did not require specific monitoring for renal, thyroid, or calcium disorders.
Since this study focused exclusively on older lithium users, that monitoring rates were low and comparable to those in younger adult/mixed-aged samples in different countries is of particular concern. Older adults are most vulnerable to lithium-associated adverse effects due to a number of factors, including a more narrow therapeutic range (0.4-0.8 mmol/L), physical comorbidity, drug-drug interactions, and decreased renal clearance of lithium with aging.10 Without adequate clinical monitoring, lithium level elevations are more common, putting older adults at higher risk for acute renal injury24 and acute neurotoxicity25,26 as well as chronic kidney disease, hypercalcemia, hypothyroidism, and other chronic physical adverse effects.2-5,8 These effects can then lead to scenarios in which many older patients need to discontinue lithium, which has often been associated with rates of mood episode relapse of > 33%-50%.27,28
Strengths, Limitations, and Future Directions
This study was the first to examine lithium monitoring exclusively in older lithium users, who are most vulnerable to lithium-associated adverse effects.29 This study was also the largest to date examining lithium monitoring practices, 3 times larger than previous studies; previous UK and New Zealand studies have had sample sizes < 1,500,13,16 except for 1 previous study15 with 3,373 patients. We included a valproate comparator group (without a neurologic diagnosis), which allowed us to observe whether monitoring was more frequent relative to a mental illness cohort exposed to a medication that does not require renal, thyroid, or calcium monitoring5 after using propensity score matching to control for important potential confounders (and thereby reduce bias). This study also systematically examined both calcium and thyroid monitoring in addition to lithium level and renal monitoring, which had not previously been done in lithium monitoring studies.
There were some limitations to this study. Although we had data on monitoring frequency, we did not have laboratory values of the lithium levels and renal function tests. Also, the findings of this study can be generalized only to outpatient lithium monitoring; we had data on outpatient laboratory tests, but not all inpatient blood tests since, in Ontario, regular bloodwork performed in hospitalized inpatients can often be paid under a hospital’s global budget. Additionally, poor drug monitoring not only is an issue with lithium treatment, but could also be an issue for health care system(s) more generally or may be specific to older bipolar disorder medications. For example, although monitoring requirements are similar with warfarin, the majority of warfarin-treated patients are not only monitored adequately but also in the therapeutic window.30 Since we used data from Ontario, Canada, which has universal health coverage, it may be difficult to generalize to other jurisdictions and health care contexts with different barriers to or facilitators of regular monitoring. Another limitation is that even with the propensity score, there is still the potential for confounding since control for unmeasured variables is not possible. Future studies could examine how clinical variables, such as medication dosing, affect adherence to laboratory monitoring. It would also be interesting to examine whether provider specialty affected laboratory monitoring rates. At a first glance of our own data, the 3-month, 6-month, and 12-month monitoring rates appear similar in primary care physicians (23.7%, 41.7%, and 66.0%), psychiatrists (24.3%, 42.8%, 67.2%), and other physicians (25.1%, 43.9%, 68.5%). In our sample, > 50% of lithium and valproate prescribing was from primary care physicians, suggesting that this may be an important physician population to target for future interventions. Future population-based studies are also needed to examine the relationship between poor monitoring and lithium-related adverse events; such examination has not been previously performed).
From both a research and a public health perspective, finding ways to improve lithium monitoring is necessary. A successful example of such an initiative was documented by Kirkham and colleagues13 in Norfolk, UK. Their objective was to ensure that all patients on lithium treatment have access to adequate information, education, and specialist advice and receive regular blood tests following an agreed-upon protocol. To implement their objective, the Norfolk group ensured close communication between the laboratories and primary and secondary care providers. Patients were given automatic reminders for testing lithium levels and renal function every 12 weeks, with additional letters, telephone calls, and family physician alerts if patients did not adhere to schedule. All of this translated to improved monitoring: in 2005, 83% of the sample had 1 renal function test or fewer, while in 2012, approximately 90% of patients received 2 or more creatinine measurements per year.13 Similar centralized jurisdiction-wide monitoring approaches with close communication and education approaches may be helpful across the world.
CONCLUSION
Lithium monitoring in older lithium users was infrequent and did not appear consistent with international standards to screen for lithium levels and renal functioning ideally every 3 months in this population. We recognize the logistical challenges of laboratory monitoring and understand that international recommendations to screen adults (of all ages) at least every 3-6 months is a reasonable clinical compromise that would still be a great improvement from what was observed in this study. It is possible that the observed monitoring may reflect routine laboratory monitoring for older adults rather than monitoring for lithium safety. Previous research has found that suboptimal monitoring may be a possible contributor to increased risk of lithium-related adverse events, although this finding needs further investigation; should it be confirmed, the effects of clinician education strategies and jurisdiction-wide treatment algorithms for improving monitoring could be examined in future studies.
Submitted: December 22, 2017; accepted May 8, 2018.
Published online: November 20, 2018.
Disclosure of off-label usage: The authors have determined that, to the best of their knowledge, no investigational information about pharmaceutical agents that is outside US Food and Drug Administration-approved labeling has been presented in this article.
Financial disclosure: Dr Herrmann has received research funding from Lundbeck, Roche, and Axovant and honoraria from AbbVie, Merck, Mediti, Astellas, and Eli Lilly. Dr Rej receives research funding from Satellite Healthcare. Drs Gruneir, Dixon, and Garg; Ms Jandoc; and Mr McArthur have no personal affiliations or financial relationships with any commercial interest to disclose relative to the article.
Funding/support: This work was supported by an Ontario Mental Health Foundation grant awarded to Dr Herrmann, as well as the Institute for Clinical Evaluative Sciences Kidney, Dialysis and Transplantation (ICES KDT) Program. Dr Herrmann was supported by the Richard Lewar Chair in Geriatric Psychiatry. Dr Rej received salary support from the Canadian Institutes of Health Research (CIHR) Fellowship Award and the Fonds de Recherche Québec-Santé Chercheur Boursier Clinicien Junior 1 Award. Dr Gruneir is supported by a New Investigator Award from CIHR. Dr Garg was supported by a Clinician Investigator Award from the Canadian Institutes of Health Research and the Dr Adam Linton Chair in Kidney Health Analytics. This study was supported by the ICES Western site. ICES is a nonprofit research corporation funded by an annual grant from the Ontario Ministry of Health and Long-Term Care (MOHLTC). Core funding for ICES Western is provided by the Academic Medical Organization of Southwestern Ontario (AMOSO), the Schulich School of Medicine and Dentistry (SSMD), Western University, and the Lawson Health Research Institute (LHRI).
Role of the sponsor: The study design and conduct, opinions, results and conclusions reported in this article are those of the authors and are independent of the funding sources. No endorsement by ICES, AMOSO, SSMD, LHRI, CIHR or the MOHLTC is intended or should be inferred. Parts of this material are based on data and/or information compiled and provided by the Canadian Institute for Health Information (CIHI).
Previous presentation: Some of these data were presented as part of a broader oral presentation at the European Association of Psychosomatic Medicine in Barcelona, Spain, June 30, 2017.
Disclaimer: The analyses, conclusions, opinions and statements expressed in the material are those of the authors and not necessarily those of CIHI.
Acknowledgments: The analysis was conducted by members of the ICES KDT team at the ICES Western facility, who are supported by a grant from CIHR. Authors also thank IMS Brogan Inc for use of their Drug Information Database.
Additional information: The analysis was conducted by members of the ICES Kidney Dialysis & Transplantation team at the ICES Western facility (London, Ontario). Mr McArthur and Dr Garg are responsible for the data analysis. The protocol can be obtained by e-mailing Ms Jandoc at [email protected].
Supplementary material: Available at PSYCHIATRIST.COM.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
REFERENCES
1. Yatham LN, Kennedy SH, Parikh SV, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) collaborative update of CANMAT guidelines for the management of patients with bipolar disorder: update 2013. Bipolar Disord. 2013;15(1):1-44. PubMed CrossRef
2. Shine B, McKnight RF, Leaver L, et al. Long-term effects of lithium on renal, thyroid, and parathyroid function: a retrospective analysis of laboratory data. Lancet. 2015;386(9992):461-468. PubMed CrossRef
3. Marras C, Herrmann N, Fischer HD, et al. Lithium use in older adults is associated with increased prescribing of Parkinson medications. Am J Geriatr Psychiatry. 2016;24(4):301-309. PubMed CrossRef
4. Shulman KI, Sykora K, Gill SS, et al. New thyroxine treatment in older adults beginning lithium therapy: implications for clinical practice. Am J Geriatr Psychiatry. 2005;13(4):299-304. PubMed CrossRef
5. Rej S, Herrmann N, Shulman K, et al. Lithium use, but not valproate use, is associated with a higher risk of chronic kidney disease in older adults with mental illness. J Clin Psychiatry. 2017;78(8):e980-e985. PubMed CrossRef
6. Kessing LV, Gerds TA, Feldt-Rasmussen B, et al. Use of lithium and anticonvulsants and the rate of chronic kidney disease: a nationwide population-based study. JAMA Psychiatry. 2015;72(12):1182-1191. PubMed CrossRef
7. Kirkham E, Skinner J, Anderson T, et al. One lithium level >1.0 mmol/L causes an acute decline in eGFR: findings from a retrospective analysis of a monitoring database. BMJ Open. 2014;4(11):e006020. PubMed CrossRef
8. Lehmann SW, Lee J. Lithium-associated hypercalcemia and hyperparathyroidism in the elderly: what do we know? J Affect Disord. 2013;146(2):151-157. PubMed CrossRef
9. Dols A, Sienaert P, van Gerven H, et al. The prevalence and management of side effects of lithium and anticonvulsants as mood stabilizers in bipolar disorder from a clinical perspective: a review. Int Clin Psychopharmacol. 2013;28(6):287-296. PubMed CrossRef
10. Sylvestre JD, Yu C, Dawson B, et al. Older adults with severe mental illness frequently require acute medical hospitalizations: an increased need for consultation-liaison services in future years? J Psychosom Res. 2015;79(1):87-88. PubMed CrossRef
11. Vestergaard P, Amdisen A. Lithium treatment and kidney function: a follow-up study of 237 patients in long-term treatment. Acta Psychiatr Scand. 1981;63(4):333-345. PubMed CrossRef
12. Ng F, Mammen OK, Wilting I, et al; International Society for Bipolar Disorders. The International Society for Bipolar Disorders (ISBD) consensus guidelines for the safety monitoring of bipolar disorder treatments. Bipolar Disord. 2009;11(6):559-595. PubMed CrossRef
13. Kirkham E, Bazire S, Anderson T, et al. Impact of active monitoring on lithium management in Norfolk. Ther Adv Psychopharmacol. 2013;3(5):260-265. PubMed CrossRef
14. Sajatovic M, Strejilevich SA, Gildengers AG, et al. A report on older-age bipolar disorder from the International Society for Bipolar Disorders Task Force. Bipolar Disord. 2015;17(7):689-704. PubMed CrossRef
15. Collins N, Barnes TR, Shingleton-Smith A, et al. Standards of lithium monitoring in mental health Ttrusts in the UK. BMC Psychiatry. 2010;10(1):80. PubMed CrossRef
16. McKean A, Vella-Brincat J. Is it NICE to monitor lithium routinely? N Z Med J. 2012;125(1355):50-54. PubMed
17. Hwang YJ, Dixon SN, Reiss JP, et al. Atypical antipsychotic drugs and the risk for acute kidney injury and other adverse outcomes in older adults: a population-based cohort study. Ann Intern Med. 2014;161(4):242-248. PubMed CrossRef
18. Juurlink DN, Mamdani MM, Kopp A, et al. Drug-induced lithium toxicity in the elderly: a population-based study. J Am Geriatr Soc. 2004;52(5):794-798. PubMed CrossRef
19. Shulman KI, Rochon P, Sykora K, et al. Changing prescription patterns for lithium and valproic acid in old age: shifting practice without evidence. BMJ. 2003;326(7396):960-961. PubMed CrossRef
20. Rej S, Yu C, Shulman K, et al. Medical comorbidity, acute medical care use in late-life bipolar disorder: a comparison of lithium, valproate, and other pharmacotherapies. Gen Hosp Psychiatry. 2015;37(6):528-532. PubMed CrossRef
21. Sajatovic M, Valenstein M, Blow F, et al. Treatment adherence with lithium and anticonvulsant medications among patients with bipolar disorder. Psychiatr Serv. 2007;58(6):855-863. PubMed CrossRef
22. Austin PC. Using the standardized difference to compare the prevalence of a binary variable between two groups in observational research. Commun Stat Simul Comput. 2009;38(6):1228-1234. CrossRef
23. Rothman KJ. Epidemiology: An Introduction. 2nd ed. New York, NY: Oxford University Press; 2012.
24. Chen KP, Shen WW, Lu ML. Implication of serum concentration monitoring in patients with lithium intoxication. Psychiatry Clin Neurosci. 2004;58(1):25-29. PubMed CrossRef
25. Schou M, Amdisen A, Trap-Jensen J. Lithium poisoning. Am J Psychiatry. 1968;125(4):520-527. PubMed CrossRef
26. Shulman KI, Sykora K, Gill S, et al. Incidence of delirium in older adults newly prescribed lithium or valproate: a population-based cohort study. J Clin Psychiatry. 2005;66(4):424-427. PubMed CrossRef
27. Fahy S, Lawlor BA. Discontinuation of lithium augmentation in an elderly cohort. Int J Geriatr Psychiatry. 2001;16(10):1004-1009. PubMed CrossRef
28. Ross J. Discontinuation of lithium augmentation in geriatric patients with unipolar depression: a systematic review. Can J Psychiatry. 2008;53(2):117-120. PubMed CrossRef
29. Rej S, Elie D, Mucsi I, et al. Chronic kidney disease in lithium-treated older adults: a review of epidemiology, mechanisms, and implications for the treatment of late-life mood disorders. Drugs Aging. 2015;32(1):31-42. PubMed CrossRef
30. Ingram SJ, Kirkdale CL, Williams S, et al. Moving anticoagulation initiation and monitoring services into the community: evaluation of the Brighton and hove community pharmacy service. BMC Health Serv Res. 2018;18(1):91. PubMed CrossRef
This PDF is free for all visitors!