Because this piece does not have an abstract, we have provided for your benefit the first 3 sentences of the full text.
To the Editor: Pediatric obsessive-compulsive disorder (OCD) is a debilitating illness characterized by intrusive thoughts and repetitive behaviors. Patients may exhibit poorer executive function, such as cognitive planning. Substantial overlap exists between brain areas implicated in OCD pathology and those involved in planning (eg, dorsolateral prefrontal cortex, posterior parietal cortex, extrastriate visual cortex, anterior cingulate cortex, inferior frontal gyrus), and functional magnetic resonance imaging (MRI) demonstrates that participants with OCD perform poorly and show less regional brain responsivity as task load increases in comparison to healthy controls. This robust finding could be useful in the domain of precision medicine, as it normalizes after cognitive-behavioral therapy and may be useful in individual activation profiles that could predict treatment response.
This work may not be copied, distributed, displayed, published, reproduced, transmitted, modified, posted, sold, licensed, or used for commercial purposes. By downloading this file, you are agreeing to the publisher’s Terms & Conditions.
Cognitive Planning Neural Correlates in a Pediatric Monozygotic Twin Pair Discordant for Obsessive-Compulsive Disorder: Exploring Potential Application in Precision Medicine
To the Editor: Pediatric obsessive-compulsive disorder (OCD) is a debilitating illness characterized by intrusive thoughts and repetitive behaviors. Patients may exhibit poorer executive function, such as cognitive planning.1 Substantial overlap exists between brain areas implicated in OCD pathology2 and those involved in planning (eg, dorsolateral prefrontal cortex, posterior parietal cortex, extrastriate visual cortex, anterior cingulate cortex, inferior frontal gyrus),3 and functional magnetic resonance imaging (MRI) demonstrates that participants with OCD perform poorly and show less regional brain responsivity as task load increases in comparison to healthy controls.4 This robust finding could be useful in the domain of precision medicine, as it normalizes after cognitive-behavioral therapy5 and may be useful in individual activation profiles that could predict treatment response. However, the finding has never been studied in individual subjects. Here, as a proof of concept, we compare the performance and brain activation of OCD-discordant monozygotic twins during a planning task commonly used in imaging studies,6 the Tower of London (ToL).
Methods. A pair of monozygotic Caucasian twins discordant for pediatric OCD and their parents consented to the study, in compliance with approval from the local institutional review board. All collected data were deidentified to protect anonymity. The Wechsler Abbreviated Scale of Intelligence Second Edition (WASI-II),7 the Cambridge Neuropsychological Test Automated Battery (CANTAB),8 and the Children’s Yale-Brown Obsessive Compulsive Scale (CY-BOCS)9 were administered.
Structural and functional MRI were recorded on a GE MR750 3T MR scanner when the twins were 12 years old. The OCD-affected twin was experiencing severe OCD symptoms (CY-BOCS score = 29/40) and had been taking clomipramine 50 mg and fluvoxamine 100 mg daily for 4 weeks prior to the scan. The unaffected twin reported no OCD symptoms and was taking no medication.
Results. During neurocognitive testing, the OCD-affected twin displayed general cognitive functioning in the superior range (Full Scale Intelligence Quotient, 93rd percentile) and verbal intelligence in the very superior range (98th percentile). Visuospatial information processing and reasoning were average (WASI-II Matrix Reasoning subtest, 70th percentile), as was spatial planning performance (CANTAB Stockings of Cambridge subtest, 75th percentile).
The unaffected twin also displayed general cognitive functioning in the superior range (97th percentile) and verbal intelligence in the very superior range (98th percentile). Consistent with her general cognitive functioning, but in contrast to her monozygotic twin, visuospatial information processing and reasoning were in the superior range (92nd percentile), and spatial planning performance was in the superior range (95th percentile).
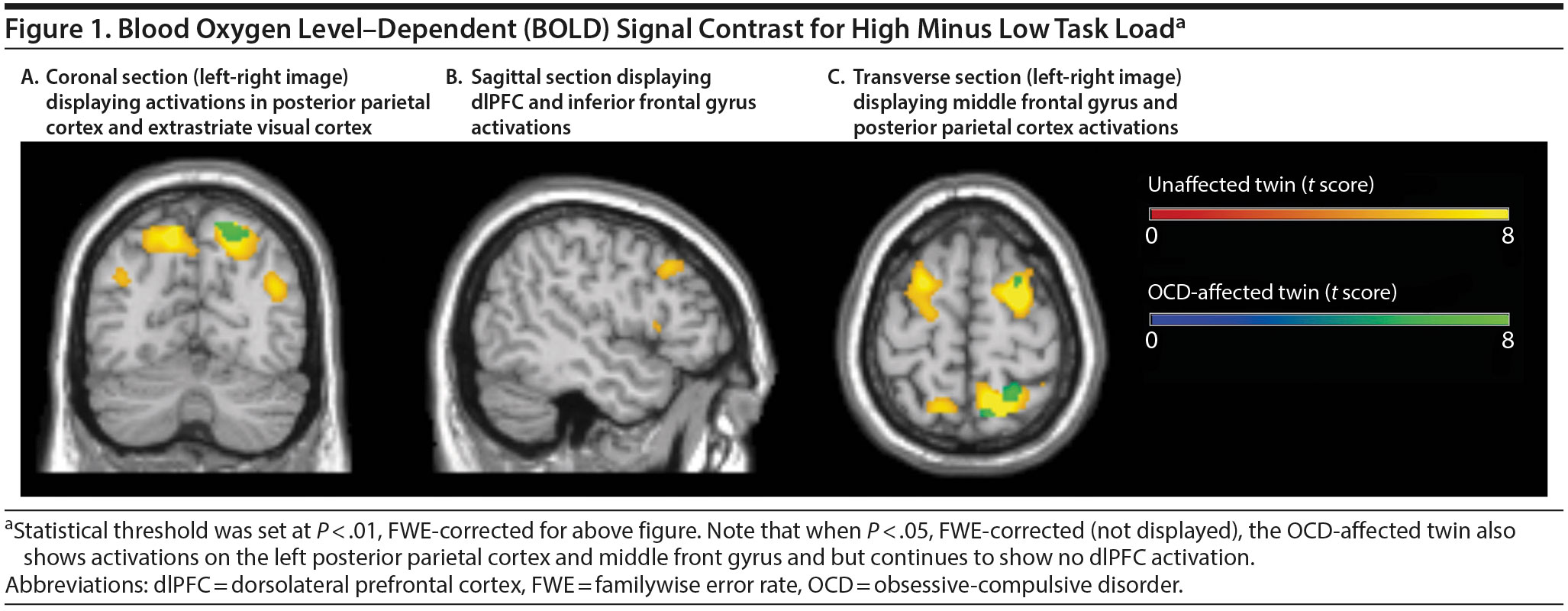
In the scanner, both twins performed well during low load ToL trials (98% accuracy), but the OCD-affected twin performed significantly worse (79% accuracy; 95% confidence interval, 69%-90%) than her unaffected twin (92% accuracy; 95% confidence interval, 85%-100%) during high load trials. As task load increased, the unaffected twin demonstrated activation of bilateral dorsolateral prefrontal cortex (dlPFC), as well as auxiliary areas such as the inferior frontal gyrus (IFG). The OCD-affected twin did not demonstrate increased dlPFC of IFG or compensatory brain activity as task load increased (Figure 1).
Studies have shown poorer cognitive planning in OCD patient groups.1,4 Group functional MRI studies of the ToL have linked these impairments to underrecruitment of the dlPFC and compensatory areas that should be involved as task load increases.4 The performance and brain activity of these twins demonstrate results consistent with the literature while using single-subject comparisons, providing proof of concept that the ToL may have a role in future precision medicine efforts. While the use of monozygotic twins ensured optimal matching on many key factors, a limitation of this study is that not all observed findings can be attributed solely to OCD discordance. Future work is necessary to develop tools and refine traditional neural probes for use in precision medicine.10
References
1. Zhang J, Yang X, Yang Q. Neuropsychological dysfunction in adults with early-onset obsessive-compulsive disorder: the search for a cognitive endophenotype. Rev Bras Psiquiatr. 2015;37(2):126-132. PubMed doi:10.1590/1516-4446-2014-1518
2. Menzies L, Chamberlain SR, Laird AR, et al. Integrating evidence from neuroimaging and neuropsychological studies of obsessive-compulsive disorder: the orbitofronto-striatal model revisited. Neurosci Biobehav Rev. 2008;32(3):525-549. PubMed CrossRef
3. Baker SC, Rogers RD, Owen AM, et al. Neural systems engaged by planning: a PET study of the Tower of London task. Neuropsychologia. 1996;34(6):515-526. PubMed CrossRef
4. van den Heuvel OA, Veltman DJ, Groenewegen HJ, et al. Frontal-striatal dysfunction during planning in obsessive-compulsive disorder. Arch Gen Psychiatry. 2005;62(3):301-309. PubMed doi:10.1001/archpsyc.62.3.301
5. Huyser C, Veltman D, Wolters L, et al. Functional magnetic resonance imaging during planning before and after cognitive-behavioral therapy in pediatric obsessive-compulsive disorder. J Am Acad Child Adolesc Psychiatry. 2010;49(12):1238-1248. PubMed
6. Nitschke K, Köstering L, Finkel L, et al. A meta-analysis on the neural basis of planning: activation likelihood estimation of functional brain imaging results in the Tower of London Task. Hum Brain Mapp. 2017;38(1):396-413. PubMed CrossRef
7. Wechsler D. Wechsler Abbreviated Scale of Intelligence-Second Edition (WASI-II). San Antonio, TX: NCS Pearson; 2011.
8. CANTAB [software package]. Cambridge, UK: Cambridge Cognition; 2017.
9. Scahill L, Riddle M, McSwiggin-Hardin M, et al. Children’s Yale-Brown Obsessive Compulsive Scale: reliability and validity. J Am Acad Child Adolesc Psychiatry. 1997;36(6):844-852. PubMed CrossRef
10. Finn ES, Shen X, Scheinost D, et al. Functional connectome fingerprinting: identifying individuals using patterns of brain connectivity. Nat Neurosci. 2015;18(11):1664-1671. PubMed CrossRef
aDepartment of Psychiatry, Faculty of Medicine, University of British Columbia, Vancouver, Canada
bBC Children’s Hospital Research Institute, Vancouver, Canada
cProvincial Health Services Authority, BC Children’s Hospital, Vancouver, Canada.
Potential conflict of interest: None reported.
Funding/support: Dr Jaspers-Fayer is supported by the MSFHR and the BC Children’s Hospital Research Institute, Vancouver, Canada. Dr Stewart has been awarded infrastructure and operating grants from the Michael Smith Foundation for Health Research (MSFHR), Vancouver, Canada; Provincial Health Services Authority, Vancouver, Canada; and the Canadian Institutes of Health Research, Ottawa, Canada.
Role of the sponsor: The funders had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; or the decision to submit the manuscript for publication.
Acknowledgments: The authors thank Donna Lang, PhD (Radiology, University of British Columbia) for help with data acquisition and Manraj K. M. Heran, MD (Radiology, University of British Columbia) for reviewing the structural scans for clinically significant findings. They also thank Odile van den Heuvel, MD, for sharing a computerized version of the Tower of London Task to use in the MR scanner.
J Clin Psychiatry 2017;78(9):e1320-e1321
https://doi.org/10.4088/JCP.16cr10967
© Copyright 2017 Physicians Postgraduate Press, Inc.
This PDF is free for all visitors!