Objective: Given the chronic, episodic nature of panic disorder, it is important to examine long-term outcomes of patients who respond well to various psychotherapies.
Method: Out of 116 patients with DSM-IV panic disorder who evidenced a ≥ 40% reduction in panic and avoidance symptoms on the Panic Disorder Severity Scale (PDSS) after 12-14 weeks of panic-focused psychodynamic psychotherapy, cognitive-behavioral therapy, or applied relaxation training as part of a 2-site randomized controlled trial conducted between January 2007 and July 2012, 91 patients provided at least 1 PDSS datapoint during follow-up. Patients were assessed at each of the 12 following months using the PDSS, the Sheehan Disability Scale (SDS), and the Hamilton Depression Rating Scale (HDRS) and twice during the follow-up period with the Anxiety Disorders Interview Schedule.
Results: Patients with panic disorder who responded to 1 of 3 treatments maintained their gains on the PDSS, SDS, and HDRS with no differences by condition in rates of change over the follow-up period (all P values ≥ .20). Similarly, 57% of improved patients did not have a panic disorder diagnosis by the end of 1 year, regardless of the treatment received. No differences in rates of panic disorder diagnosis were found across treatment conditions at either 6 months or 12 months (all P values ≥ .78). Results should be interpreted in light of the fact that patients giving data at follow-up experienced lower symptom levels at treatment termination than those not providing data.
Conclusions: Improvement in any of the 3 treatments examined in this trial was largely maintained for a year following the end of therapy. Future treatment development could focus on factors to increase the level of response in the active phase of therapy.
Trial Registration: ClinicalTrials.gov identifier: NCT00353470
Twelve-Month Outcomes Following Successful Panic-Focused Psychodynamic Psychotherapy, Cognitive-Behavioral Therapy, or Applied Relaxation Training for Panic Disorder

ABSTRACT
Objective: Given the chronic, episodic nature of panic disorder, it is important to examine long-term outcomes of patients who respond well to various psychotherapies.
Method: Out of 116 patients with DSM-IV panic disorder who evidenced a ≥ 40% reduction in panic and avoidance symptoms on the Panic Disorder Severity Scale (PDSS) after 12-14 weeks of panic-focused psychodynamic psychotherapy, cognitive-behavioral therapy, or applied relaxation training as part of a 2-site randomized controlled trial conducted between January 2007 and July 2012, 91 patients provided at least 1 PDSS datapoint during follow-up. Patients were assessed at each of the 12 following months using the PDSS, the Sheehan Disability Scale (SDS), and the Hamilton Depression Rating Scale (HDRS) and twice during the follow-up period with the Anxiety Disorders Interview Schedule.
Results: Patients with panic disorder who responded to 1 of 3 treatments maintained their gains on the PDSS, SDS, and HDRS with no differences by condition in rates of change over the follow-up period (all P values ≥ .20). Similarly, 57% of improved patients did not have a panic disorder diagnosis by the end of 1 year, regardless of the treatment received. No differences in rates of panic disorder diagnosis were found across treatment conditions at either 6 months or 12 months (all P values ≥ .78). Results should be interpreted in light of the fact that patients giving data at follow-up experienced lower symptom levels at treatment termination than those not providing data.
Conclusions: Improvement in any of the 3 treatments examined in this trial was largely maintained for a year following the end of therapy. Future treatment development could focus on factors to increase the level of response in the active phase of therapy.
Trial Registration: ClinicalTrials.gov identifier: NCT00353470
J Clin Psychiatry 2018;79(5):17m11807
To cite: McCarthy KS, Chambless DL, Solomonov N, et al. Twelve-month outcomes following successful panic-focused psychodynamic psychotherapy, cognitive-behavioral therapy, or applied relaxation training for panic disorder. J Clin Psychiatry. 2018;79(5):17m11807.
To share: https://doi.org/10.4088/JCP.17m11807
© Copyright 2018 Physicians Postgraduate Press, Inc.
aDepartment of Psychology, Chestnut Hill College, Philadelphia, Pennsylvania
bPerelman School of Medicine, University of Pennsylvania, Philadelphia, Pennsylvania
cDepartment of Psychology, University of Pennsylvania, Philadelphia, Pennsylvania
dGordon F. Derner School of Psychology, Adelphi University, Garden City, New York
eDepartment of Psychiatry, Weill Cornell Medical College, Cornell University, New York, New York
*Corresponding author: Kevin S. McCarthy, PhD, Department of Psychology, Chestnut Hill College, 9601 Germantown Ave, Philadelphia, PA 19118 ([email protected]).
Panic disorder is associated with disability and higher health care costs1-4 and often persists chronically or episodically.5-7 Thankfully, there are effective treatments for panic disorder, including a number of psychotherapies8-11 and medications.8,12,13 However, relapse is likely for many patients,5 suggesting that, for many patients, underlying mechanisms or conflicts predisposing vulnerability to panic may not be successfully resolved. In cognitive-behavioral therapy (CBT), the acquisition of skills to challenge maladaptive thoughts and behaviors may help reduce the recurrence of symptoms, as relapse prevention is often part of the standard protocol.14 A novel treatment for panic disorder, panic-focused psychodynamic psychotherapy (PFPP),15 has been examined in the longer term in only 1 published study.16 In PFPP, termination of treatment is thought to elicit ambivalence in the patient about autonomy and separation, which are the dynamic underpinnings and triggers for panic symptoms. This treatment explicitly engages and addresses these feelings, especially at termination, in order to give the patient a new ability to manage loss and autonomy. Achieving these strengths during PFPP may help patients consolidate their outcomes over time.15
A recent randomized clinical trial17 compared PFPP versus CBT versus applied relaxation treatment (ART), with patients randomized in a 2:2:1 ratio. The majority of patients (58%) responded successfully to 1 of the 3 psychotherapies; however, there were differences in response rates by site. One site (Cornell University) showed higher response rates for CBT and PFPP than for ART, whereas the other site (University of Pennsylvania [Penn]) showed no significant differences among the conditions. ART appeared to be less tolerable to patients, as attrition affected outcome in this condition more than other conditions, especially so among the most severely ill patients. No pretreatment factors that readily accounted for the site-by-treatment interaction have been discerned, although process studies are underway.
We investigated how individuals who responded to 1 of these 3 treatments fared in their symptoms and functioning over the 12 months following the end of therapy (ie, whether responders maintained their gains). It was hypothesized that PFPP would show a faster rate of change over the 1-year follow-up due to the resolution of symptoms emerging as part of the termination phase or the accumulation of gains with the putative benefits of awareness. We focused on responders rather than the entire sample because, for ethical reasons, we offered patients who failed one treatment their choice of another of the protocol treatments rather than retaining them in the follow-up sample.a
METHOD
Patients
Patients were 116 individuals who successfully responded to either PFPP (n = 47), CBT (n = 51), or ART (n = 18). They had a mean (SD) age of 37 (13.1) years, had an annual salary around $45,000 and had some college experience, worked full-time (72%), were female (66%) and of non-minority status (73%), and were not in a committed relationship (55%). They were initially recruited through advertisements and referrals in 2 metropolitan areas. At treatment onset, all had a DSM-IV diagnosis of panic disorder with or without agoraphobia per the Anxiety Disorders Interview Schedule for DSM-IV (ADIS)18 and at least 4 panic attacks in the month prior to intake. Exclusion criteria were DSM-IV psychosis, bipolar disorder, or current substance dependence; age > 70 years; or serious medical conditions. Patients were permitted to be on a stable dose of psychoactive medications through the active phase of treatment and the follow-up period. At one site (Cornell), the number of prescriptions was limited to 1 stable (> 2 months’ prior to entry) antipanic medication only. The institutional review boards at both sites approved and oversaw this trial. All patients gave informed consent. Recruitment, treatment, and follow-up occurred between January 2007 and July 2012. The study was registered with ClinicalTrials.gov (identifier: NCT00353470).

- A number of psychotherapies are helpful in treating panic disorder, but knowing the long-term outcome of these therapies is important given the chronic and episodic nature of the disorder.
- When patients respond to a manualized, well-administered psychotherapy for panic disorder, gains are likely to persist for up 1 year, regardless of the specific type of therapy.
Treatments
PFPP15 helps patients work through unconscious conflict (often loss and anger) expressed as panic using exploration and interpretation, especially in anticipation of the separation around termination. CBT14 challenges maladaptive thoughts associated with panic using psychoeducation, identification of symptomatic thoughts and sensations, cognitive restructuring, interoceptive and situational exposure, and relapse prevention. ART (M. Schwalberg, PhD, and D.L.C, unpublished manuscript, 2006) alleviates anxiety and tension within the body that causes panic symptoms using psychoeducation, tension and relaxation exercises with progressively more muscle groups, and practice paired with anxiety-provoking situations. Treatments were manualized (references 14 and 15 and M. Schwalberg, PhD, and D.L.C, unpublished manuscript, 2006) and were conducted by 31 therapists (MD, PhD, PsyD, or MSW) with an average of 12 years of experience. All received training and periodic supervision in the specific modality of therapy they conducted. Sessions for each treatment were conducted twice weekly for 12 weeks. Twenty-four sessions were delivered over a maximum of 16 weeks’ duration. Adherence to the treatments (both using interventions specific to the delivered treatment and refraining from interventions specific to the competing treatments) was assessed and determined to be adequate.17
Assessment of Response Status and Disposition
Patients underwent a full diagnostic assessment by independent raters blinded to patient condition after their final session of therapy. Response was determined a priori as a decrease of ≥ 40% from baseline19 on the Panic Disorder Severity Scale (PDSS).20,b Designation of responder status did not require patients to no longer meet criteria for panic disorder (achieved by 78% of responders [80/103]) or agoraphobia (achieved by 81% of responders [83/103]), nor did it necessitate the completion of a full treatment protocol (defined as attending ≥ 16/24 sessions, accomplished by 91% of responders [106/116]).
After termination, patients were offered up to 3 booster sessions over the ensuing 12 months if they requested additional treatment or showed a significant increase in symptoms at one of the assessment points. Twelve responders accepted a mean (SD) of 2.25 (0.87) sessions.
During the 1-year follow-up period, patients were requested not to seek outside psychological or pharmacologic treatment unless they or study staff determined that it was necessary. Patients were asked about any potential intervening treatment at each monthly follow-up evaluation, and when patients reported additional nonstudy treatment, the data were censored immediately after the onset of any outside consultation.
Follow-Up Assessments
All patients were followed monthly for 12 months with both self-report questionnaires and interviews with well-trained, reliable diagnosticians blinded to patient condition (for all observer-rated measures, all interrater reliability ÏI values > 0.80).17
The 7-item observer-rated PDSS20 was the primary outcome instrument. It assesses degree of panic symptoms and avoidance as well as functional and interpersonal impairment due to panic. Ratings were taken monthly in the follow-up period. At months 6 and 12, remission status was also determined as having a PDSS score ≤ 5 for at least 3 months.20
The ADIS18 was used to assess the presence or absence of a panic disorder diagnosis. The ADIS was administered at baseline, termination of treatment, and months 6 and 12 of the follow-up period.
The Sheehan Disability Scale (SDS)21 is a 3-item self-report questionnaire given monthly that tracks psychosocial impairment in the areas of work, social, and family life.
The Hamilton Depression Rating Scale (HDRS)22 is a 17-item observer-rated measure of depressive symptoms. It was included as a secondary outcome due to high comorbidity of panic and depression.7 It was administered monthly.
Data Analytic Strategy
First, we compared those responders for whom we had data during follow-up with those for whom we did not for differences in termination PDSS, SDS, and HDRS scores; demographics; and relevant pretreatment factors. For these and all following analyses, comparisons were conducted only with sufficient sample sizes per condition (> 5 patients) to produce estimates of reasonable stability.
For each of the time-varying outcomes (PDSS, SDS, or HDRS), separate hierarchical linear models were used to predict the follow-up scores on each of the measures with a random intercept and slope of time for patients and fixed effects of treatment, site, treatment ×— site, time ×— treatment, time ×— site ×— treatment, receipt of booster sessions, antidepressant use, and anxiolytic use. Follow-up scores for each measure were residualized on their termination scores to account for the effect of starting values on the ability to show change in the follow-up period. Covariates (site, treatment ×— site, time ×— treatment ×— site, receipt of booster sessions, antidepressant use, anxiolytic use) remained in the model if they were significant or had greater than a small effect (number needed to treat [NNT] < 8.9)23 on the dependent variable.
The effect of interest in these models is the interaction of time and treatment, which, if significant, would suggest that there were differences in the rate of change among conditions over the follow-up period. Effect sizes between conditions are presented as NNT, and small effects are NNT < 8.9, medium are < 3.6, and large < 2.3.23 Using mixed-model simulation methods24 with the number of participants and rate of missingness in this sample, power to detect a large effect between 2 of the treatments in their rate of change over time was 0.96; for a medium effect, 0.61; and for a small effect, 0.16.
For discrete outcomes sampled at months 6 and 12 (panic disorder diagnosis, remission status from PDSS), logistic regressions were performed for each assessment point separately, predicting status from condition, site, and their interaction as well as receipt of booster sessions and psychoactive medication use. Again, covariates (site, booster sessions, medications) were removed from the models if they failed to show significance or greater than a small effect (NNT < 8.9)23 on the dependent variable. Separate models were run for each outcome, and between-condition effect sizes are presented as NNT. A power simulation24 using the same sample size and missingness parameters of this sample indicated a 0.67 likelihood to detect a large effect on dichotomous outcomes between 2 conditions, 0.46 for a medium effect, and 0.16 for a small effect.
RESULTS
Of 116 responders, 78% (n = 91) provided at least 1 PDSS datapoint during follow-up (median = 5; range, 1-12). Those patients giving data had significantly lower PDSS, HDRS, and SDS scores at termination (all P values < .03) than those not providing follow-up data, but there were no differences in diagnostic status at termination, treatment condition received, use of booster sessions, age, sex, race or ethnicity, education, or employment status (all P values > .14).
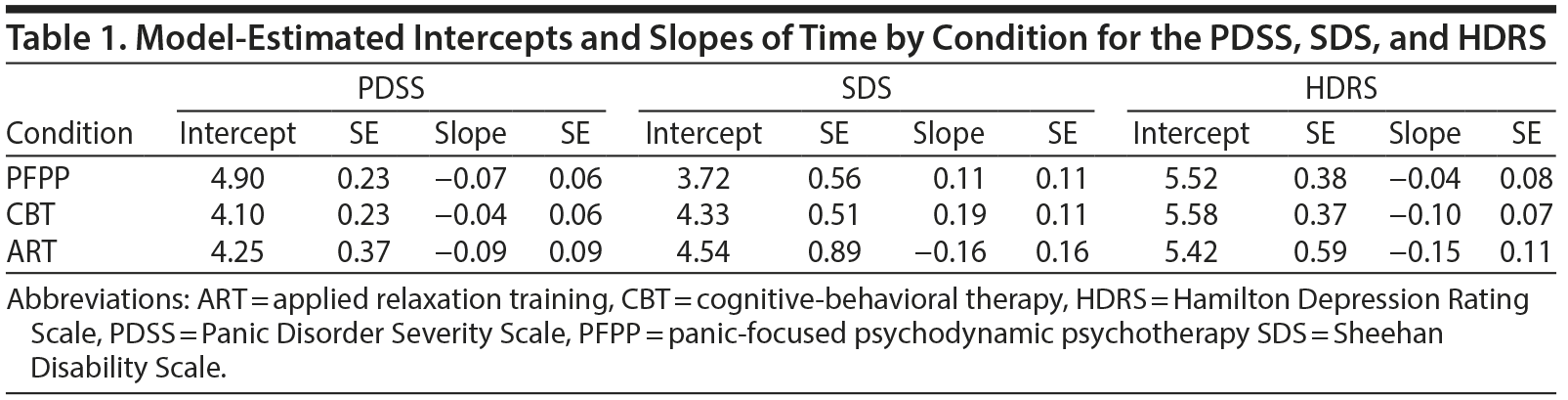
Model-estimated intercepts and slopes of time for condition on each of the measures are presented in Table 1. All conditions showed a small nonsignificant decline in symptoms on the PDSS across the follow-up period (F1,86 = 2.79, P < .10, NNT = 5.0) and did not vary in their rate of change (F2, 358 = 0.14, P = .87; NNT range, 32.9-63.4). PFPP patients scored somewhat higher across all timepoints (mean [SE] = 4.58 [0.28]) than did CBT (mean [SE] = 3.92 [0.27], F1, 358 = 2.96, P < .09, NNT = 9.8) and ART patients (mean [SE] = 3.81 [0.40], F2, 354 = 2.51, P = .11, NNT = 10.6), but these differences were not statistically significant. CBT and ART patients did not differ from one another (F1, 358 = 0.05, P = .82, NNT = 75.0). There were no differences based on site, booster sessions, or medication use.c
Among patients with follow up data, 10 (39%) of 26 PFPP patients, 19 (56%) of 34 CBT patients, and 8 (67%) of 12 ART patients were remitted at month 6 on the PDSS, with no significant differences among treatments (χ22 = 3.20, P = .20; NNT range, 2.8-5.9). Similarly, at month 12, 12 (50%) of 24 PFPP patients, 19 (59%) of 32 CBT patients, and 9 (64%) of 14 ART patients were remitted, with no significant differences among treatments (χ22 = 2.70, P = .26; NNT range, 7.1-20.0). For analyses at both month 6 and month 12, no differences were found for site, booster sessions, or medication use.
No differences across conditions in rates of panic disorder diagnosis were seen at month 6 (15 [68%] of 22 PFPP patients, 15 [79%] of 19 CBT patients, and 9 [90%] of 10 ART patients continued not to have a diagnosis; χ22 = 0.50, P = .78; NNT range, 4.5-9.1) or at month 12 (16 [80%] of 20 PFPP patients, 13 [72%] of 18 CBT patients, and 10 [91%] of 11 ART patients did not have a diagnosis; χ22 = 0.37, P = .83; NNT range, 5.3-12.5). Again, for analyses at both month 6 and month 12, none of the covariates were significant.
Patients did not have change in scores on the SDS over the follow-up period (F1, 86 = 0.39, P = .53, NTT = 14.4), nor did condition moderate their rate of change on the SDS (F2, 499 = 1.59, P = .21; NNT range, 11.0-42.2). However, they did differ by condition and site in their scores at termination, and these differences persisted in follow-up (F2, 499 = 14.85, P < .0001; NTT range, 5.9-7.3). At Cornell, patients in PFPP were functioning significantly better on average during the follow-up period (mean [SE] SDS score = 2.02 [0.69]) than were those in ART and CBT (combined mean [SE] = [0.77]; F1, 499 = 18.36, P < .0001, NNT = 4.68). Conversely, at Penn, patients in PFPP were not as well off per the SDS over the follow-up period (mean [SE] = 5.43 [0.84]) than those in ART or CBT (mean [SE] = 2.51 [0.64]; F1, 499 = 7.93, P < .005, NNT = 7.07).
Patients did show a small and marginally significantly decline in depressive symptoms over the follow-up period (F1, 89 = 3.74, P < .06, NNT = 4.4) but did not differ in the rate of change on the HDRS by condition (F2, 440 = 0.41, P = .66; NNT range, 21.3-46.7). No differences were observed by site, booster sessions, or medications.
DISCUSSION
We examined maintenance of gains across 12 months of follow-up for those patients who met response criteria19 during treatment. On average, there was no significant deterioration or further improvement in this interval, and no differences were observed in performance among conditions (PFPP, CBT, or ART). Most effect sizes for differences among conditions in change during the follow-up period were small (NNT > 8.9). Some effect sizes were medium, but these cases involved comparisons between 2 small groups of patients and so confidence intervals of these estimates were very wide. Similar findings were found for both dimensional and categorical measures of panic symptoms and for measures of depression and psychosocial functioning. Concurrent medication use and booster sessions did not appear to influence the long-term outcomes of responders. There still may exist some true and meaningful differences among the 3 treatments during follow-up. However, the lack of differences observed in this study was fairly robust in that it occurred across different measures and methods of assessment and was invariant to other events during follow-up.
In previous research, PFPP and CBT have sometimes been shown to be superior to ART for panic disorder recovery at follow-up,10,16,25 largely because in those studies, PFPP and CBT outperformed ART in the treatment phase, and these differences persisted in follow-up. Different results were observed in the present investigation, possibly because patients with successful treatments were examined as opposed to all individuals beginning a treatment. ART proved unacceptable to a great many patients as shown by a dropout rate of 41%,17 and patients with more severe symptoms were more likely to drop out early with ART (69% vs 26% with CBT and 29% with PFPP). Yet for those 46% of patients who persisted in ART and improved, gains were held across the follow-up period as much as those for the patients in the other 2 conditions. This finding shows the need to enhance therapies to be as successful as possible during treatment in terms of both symptom change and acceptability.
Relapse prevention and support were built into each of these treatments. For instance, the last session of treatment in ART and CBT prepared the patient for the period after treatment (reference 14 and M. Schwalberg, PhD, and D.L.C, unpublished manuscript, 2006), including how to employ skills learned in the absence of the therapist. In PFPP, great focus was placed in the final third of therapy on the meaning of termination and the ways in which termination constitutes a recreation of loss and anger experiences that lead to the appearance of panic symptoms.15 This planning and foresight in all treatments could be the reason for the relatively equivalent maintenance of gains as opposed to a preferential rate of change in follow-up for 1 treatment over the others.
When treatments worked, symptoms and functioning remained constant across the follow-up period for all treatments. This finding suggests that it may be critical for clinicians to focus on the improvement during the acute phase of therapy. Because the level of symptoms at which patients ended treatment was how symptomatic they remained for the next year, it is important that patients receive the maximum amount of improvement possible during treatment itself. Candidate mechanisms for change include improvement in catastrophic thinking about panic attacks26 and increases in the ability to reflect on mental states relevant to the experience of panic.27 We are presently studying the process factors in this sample as a guide to methods of improving outcome.28,29
Limitations
Limitations of the study include attrition and the response status of the sample. We examined the long-term outcomes of those who responded well to treatment initially. Those responders who provided data in follow-up were also symptomatically better at termination compared to responders who did not give data, so findings reported here must be interpreted in that light. Furthermore, there were initial site-by-treatment differences in outcome. Power to detect very small effects, such as those seen in rate of change among conditions, may have been low in this sample, although it may be argued that very small effects such as those found in the majority of comparisons in this study are unlikely to be meaningful.
CONCLUSIONS
When patients improve in a well-administered and monitored panic-focused psychotherapy, it appears that gains are maintained over the year following treatment regardless of the specific type of psychotherapy for panic disorder that is delivered. It is encouraging that multiple psychological interventions have sustained effects up to a year after treatment, and targeting symptom reduction in the active phase of these therapies may reduce the health care burden of patients with panic disorder in the longer term.28,29
Submitted: July 13, 2017; accepted March 16, 2018.
Published online: September 11, 2018.
Author contributions: Dr McCarthy conducted all analyses, originated the manuscript, and was Project Manager at the University of Pennsylvania (Penn) site. Dr Chambless was a Principal Investigator (PI) at the Penn site and provided significant conceptual and authorial support. Ms Solomonov cleaned and managed follow-up data at the Cornell University site. Dr Milrod was the PI at the Cornell site, planned the study, was responsible for data collection, provided balance and interpretation of the data, and edited the manuscript. Dr Barber was a PI at the Penn site, was responsible for planning the study and collecting the data, and edited the manuscript.
Potential conflicts of interest: Dr Milrod has received book royalties from Taylor & Francis. Dr Barber received book royalties from Guilford Press. Drs McCarthy and Chambless and Ms Solomonov report no potential conflicts of interest.
Funding/support: Data collection was supported by National Institute of Mental Health (NIMH) grants R01 MH70918 and R01 MH 070664. Portions of Dr Milrod’s time were supported by a fund in the New York Community Trust established by DeWitt Wallace and Taylor & Francis Group book royalties.
Role of the sponsor: The study sponsors had no role in the design and conduct of the study; collection, management, analysis, and interpretation of data; and preparation, review, or approval or the manuscript.
REFERENCES
1. Olatunji BO, Cisler JM, Tolin DF. Quality of life in the anxiety disorders: a meta-analytic review. Clin Psychol Rev. 2007;27(5):572-581. PubMed CrossRef
2. Konnopka A, Leichsenring F, Leibing E, et al. Cost-of-illness studies and cost-effectiveness analyses in anxiety disorders: a systematic review. J Affect Disord. 2009;114(1-3):14-31. PubMed CrossRef
3. Wittchen HU, Jacobi F, Rehm J, et al. The size and burden of mental disorders and other disorders of the brain in Europe 2010. Eur Neuropsychopharmacol. 2011;21(9):655-679. PubMed CrossRef
4. Hendriks SM, Spijker J, Licht CM, et al. Long-term disability in anxiety disorders. BMC Psychiatry. 2016;16(1):248. PubMed CrossRef
5. Bruce SE, Yonkers KA, Otto MW, et al. Influence of psychiatric comorbidity on recovery and recurrence in generalized anxiety disorder, social phobia, and panic disorder: a 12-year prospective study. Am J Psychiatry. 2005;162(6):1179-1187. PubMed CrossRef
6. Spinhoven P, Batelaan N, Rhebergen D, et al. Prediction of 6-yr symptom course trajectories of anxiety disorders by diagnostic, clinical and psychological variables. J Anxiety Disord. 2016;44:92-101. PubMed CrossRef
7. de Jonge P, Roest AM, Lim CC, et al. Cross-national epidemiology of panic disorder and panic attacks in the world mental health surveys. Depress Anxiety. 2016;33(12):1155-1177. PubMed CrossRef
8. Mitte K. A meta-analysis of the efficacy of psycho- and pharmacotherapy in panic disorder with and without agoraphobia. J Affect Disord. 2005;88(1):27-45. PubMed CrossRef
9. Siev J, Chambless DL. Specificity of treatment effects: cognitive therapy and relaxation for generalized anxiety and panic disorders. J Consult Clin Psychol. 2007;75(4):513-522. PubMed CrossRef
10. Sánchez-Meca J, Rosa-Alcázar AI, Mar×n-Mart×nez F, et al. Psychological treatment of panic disorder with or without agoraphobia: a meta-analysis. Clin Psychol Rev. 2010;30(1):37-50. PubMed CrossRef
11. Keefe JR, McCarthy KS, Dinger U, et al. A meta-analytic review of psychodynamic therapies for anxiety disorders. Clin Psychol Rev. 2014;34(4):309-323. PubMed CrossRef
12. Andrisano C, Chiesa A, Serretti A. Newer antidepressants and panic disorder: a meta-analysis. Int Clin Psychopharmacol. 2013;28(1):33-45. PubMed CrossRef
13. Bighelli I, Trespidi C, Castellazzi M, et al. Antidepressants and benzodiazepines for panic disorder in adults. Cochrane Database Syst Rev. 2016;9(9):CD011567. PubMed
14. Craske MG, Barlow DH, Meadows E. Master Your Own Anxiety and Panic: Therapist Guide for Anxiety, Panic, and Agoraphobia (MAP-3). San Antonio, TX: Graywind/Psychological Corporation; 2000.
15. Milrod B, Busch F, Cooper A, et al. Manual of Panic-Focused Psychodynamic Psychotherapy. Washington, DC: APA Press; 1997.
16. Milrod B, Busch F, Leon AC, et al. A pilot open trial of brief psychodynamic psychotherapy for panic disorder. J Psychother Pract Res. 2001;10(4):239-245. PubMed
17. Milrod B, Chambless DL, Gallop R, et al. Psychotherapies for panic disorder: a tale of two sites. J Clin Psychiatry. 2016;77(7):927-935. PubMed CrossRef
18. DiNardo PA, Brown TA, Barlow DH. Anxiety Disorders Interview Schedule for DSM-IV: Current Version (ADIS-IV). New York, NY: Graywinds Publications; 1995.
19. Furukawa TA, Katherine Shear M, Barlow DH, et al. Evidence-based guidelines for interpretation of the Panic Disorder Severity Scale. Depress Anxiety. 2009;26(10):922-929. PubMed CrossRef
20. Shear MK, Brown TA, Barlow DH, et al. Multicenter collaborative Panic Disorder Severity Scale. Am J Psychiatry. 1997;154(11):1571-1575. PubMed CrossRef
21. Sheehan DV. The Sheehan Disability Scales. The Anxiety Disease. New York, NY: Charles Scribner and Sons; 1983:151.
22. Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23(1):56-62. PubMed CrossRef
23. Kraemer HC, Kupfer DJ. Size of treatment effects and their importance to clinical research and practice. Biol Psychiatry. 2006;59(11):990-996. PubMed CrossRef
24. Lane SP, Hennes EP. Power struggles: estimating sample size for multilevel relationships research. J Soc Pers Relat. 2018;35(1):7-31. CrossRef
25. Craske MG, Brown TA, Barlow DH. Behavioral treatment of panic disorder: a two-year follow-up. Behav Ther. 1991;22(3):289-304. CrossRef
26. Teachman BA, Marker CD, Clerkin EM. Catastrophic misinterpretations as a predictor of symptom change during treatment for panic disorder. J Consult Clin Psychol. 2010;78(6):964-973. PubMed CrossRef
27. Rudden M, Milrod B, Target M, et al. Reflective functioning in panic disorder patients: a pilot study. J Am Psychoanal Assoc. 2006;54(4):1339-1343. PubMed CrossRef
28. Chambless DL, Milrod B, Porter E, et al. Prediction and moderation of improvement in cognitive-behavioral and psychodynamic psychotherapy for panic disorder. J Consult Clin Psychol. 2017;85(8):803-813. PubMed CrossRef
29. Keefe JR, Milrod BL, Gallop R, et al. What is the effect on comorbid personality disorder of brief panic-focused psychotherapy in patients with panic disorder? Depress Anxiety. 2018;35(3):239-247. PubMed CrossRef
aPatients who completed treatment but did not qualify for responder status were offered a second treatment in one of the conditions they did not initially receive. Twelve of the 84 nonresponders accepted a crossover treatment. Their data are excluded from this analysis.
bDue to the adoption of the last-observation-carried-forward strategy to determine response status, not all patients deemed to be responders will have completed a termination interview. Such patients may still have provided follow-up data (eg, a patient who was unable to be contacted after their last session with their therapist but who was reached for a 6-month follow-up).
cThese and all following analyses were repeated for the entire intent-to-follow sample (n = 147) and for those patients completing at least 16 sessions (n = 135). Results for each of these 2 samples were largely identical to those reported here for responders.
Enjoy this premium PDF as part of your membership benefits!