Background: There is an unmet need to objectively assess adherence problems that are a common cause of unexplained or unexpected suboptimal outcome. A digital medicine system (DMS) has been developed to address this need in patients with serious mental illness.
Objective: To conduct a quantitative expert consensus survey to (1) assess relative importance of causes of suboptimal outcomes, (2) examine modalities used to assess adherence, (3) provide guidance on when and how to use the DMS in clinical practice once available, and (4) suggest interventions for specific reasons for nonadherence.
Methods: A panel of 58 experts in psychiatry completed a 23-question survey (October 13 through December 23, 2013) and rated their responses on a 9-point Likert scale. A χ2 test of score distributions was used to determine consensus (P < .05).
Results: The panel rated adherence as the most important factor in suboptimal outcomes and yet the least likely to be assessed accurately. All predefined uses of the DMS received high mean first-line ratings (≥ 7.4). The experts recognized the utility of the DMS in managing adherence problems, identified clinical situations appropriate for DMS, and assessed potential benefits and challenges of this technology. Consensus was reached on first-line interventions for 10 of 11 reasons for nonadherence.
Conclusions: The results provide a guide to clinicians on the evaluation of suboptimal outcomes, when and how to use the DMS, and the most appropriate interventions to address detected adherence problems.
Expert Consensus Survey on Medication Adherence
in Psychiatric Patients and Use of a Digital Medicine System
ABSTRACT
Background: There is an unmet need to objectively assess adherence problems that are a common cause of unexplained or unexpected suboptimal outcome. A digital medicine system (DMS) has been developed to address this need in patients with serious mental illness.
Objective: To conduct a quantitative expert consensus survey to (1) assess relative importance of causes of suboptimal outcomes, (2) examine modalities used to assess adherence, (3) provide guidance on when and how to use the DMS in clinical practice once available, and (4) suggest interventions for specific reasons for nonadherence.
Methods: A panel of 58 experts in psychiatry completed a 23-question survey (October 13 through December 23, 2013) and rated their responses on a 9-point Likert scale. A χ2 test of score distributions was used to determine consensus (P < .05).
Results: The panel rated adherence as the most important factor in suboptimal outcomes and yet the least likely to be assessed accurately. All predefined uses of the DMS received high mean first-line ratings (≥ 7.4). The experts recognized the utility of the DMS in managing adherence problems, identified clinical situations appropriate for DMS, and assessed potential benefits and challenges of this technology. Consensus was reached on first-line interventions for 10 of 11 reasons for nonadherence.
Conclusions: The results provide a guide to clinicians on the evaluation of suboptimal outcomes, when and how to use the DMS, and the most appropriate interventions to address detected adherence problems.
J Clin Psychiatry
https://doi.org/10.4088/JCP.16m11252
© Copyright 2017 Physicians Postgraduate Press, Inc.
aOtsuka America Pharmaceutical, Inc, and Otsuka Pharmaceutical Development & Commercialization, Inc, Princeton, New Jersey
bRoss Editorial, Independence, Virginia
cDepartment of Psychiatry, University of Illinois, Chicago
dODH, Inc, Princeton, New Jersey (current affiliation)
*Corresponding author: Ainslie Hatch, PhD, ODH, Inc, 508 Carnegie Center Blvd, Ste 300, Princeton, NJ 08540 ([email protected]).
Psychiatric patients often show variable response to treatment. Numerous factors contribute to suboptimal response, including adherence problems.1 For those with serious mental illness (SMI), including schizophrenia, bipolar I disorder, and major depressive disorder (MDD),2–5 ongoing adherence prevents recurrence, relapse, and rehospitalization. Nonadherence is common in patients with SMI6,7; however, it is very difficult to detect in practice. Mental health practitioners frequently underestimate the extent of nonadherence to prescribed treatments.8,9 The ability to rapidly identify adherence problems as early as possible, however, is very important in preventing suboptimal outcome due to nonadherence leading to relapse or recurrence or to an enduring state of suboptimal outcome. Availability of information about medication disruptions and other differences between prescribed and taken medication is critical. A discrepancy between prescribed and actually ingested medication may lead the physician to flawed assumptions regarding the medication’s efficacy or what appear to be treatment-emergent side effects. Unfortunately, methods currently used to assess adherence have limitations and do not detect actual medication ingestion.10
Various techniques and technologies have been proposed that may provide earlier, more accurate, or objective information about medication adherence. These include theory-based social psychology, such as assessment of patient self-efficacy to predict nonadherence and tailor intervention11–13 and mobile phone video to directly observe medication ingestion.14 A novel digital medicine system (DMS) developed for patients with SMI may offer objective and precise measurement of medication ingestion. The system is a drug-device combination that consists of an ingestible sensor embedded in a medication tablet, a wearable sensor-transmitter, and mobile- and cloud-based software that enables the secure collection and sharing of medication adherence information with health care providers. When the DMS technology becomes available, clinicians will need a practical guide to facilitate their use of this technology and guidance on the use of adherence data. The expert consensus survey methodology was used to address this need.
The survey methodology was developed to quantify and report expert opinion to inform clinical practice in areas not well covered by definitive research.15 It has been applied to 26 areas of clinical practice, including schizophrenia, bipolar disorder, depressive disorders, attention-deficit/hyperactivity disorder, obsessive-compulsive disorder, and dementia, with data presented in more than 75 publications.16,17 An expert consensus survey on adherence problems in SMI published in 2009 also used the expert consensus methodology.17 Efforts to develop the DMS as a new objective method of assessing adherence created the need for an update and extension of a previously published expert survey to address questions that had not been covered.17 The purpose of the study was to gather information from a curated panel of credentialed clinical experts to provide guidance for the psychiatry field in regard to the following 4 questions: Is there a need in psychiatry for this state-of-the-art technology? When could this technology be used in clinical practice? How could this technology be used? and What actions should a clinician take when they have used this technology and detected adherence problems? Results from the survey may be used as recommendations for physicians and other health care providers (psychologists, case managers, social workers) concerning use of a future technology to objectively measure patient medication adherence and as guidance concerning the most appropriate approaches for targeting reasons for adherence problems.
METHODS
Survey
This survey contained 23 questions and 360 options (see eAppendix 1). The experts were asked to evaluate clinical appropriateness by rating the options on a 9-point Likert scale (eg, 1 = not at all appropriate; 9 = extremely appropriate) that was slightly modified from a format developed by the RAND Corporation (Santa Monica, California) for ascertaining expert consensus.18 One question concerning timing of nonadherence alerts was answered by filling in 3 blank fields. The survey took approximately 1 hour to complete, and respondents were paid a fee ($450) for participating. The survey was conducted from October 13 through December 23, 2013.
The 23 questions in the survey focused on 4 main areas:
(1) Establishing the need for objective data in patients with suboptimal treatment outcomes (6 questions). This section asked about underlying causes of suboptimal clinical outcomes and accuracy of their assessment (4 questions) and the frequency of use and accuracy of 11 methods of assessing adherence used in routine clinical practice (2 questions).
(2) When to use the DMS (5 questions). This section explored the relative importance of assessing nonadherence in patients with schizophrenia, bipolar disorder, and MDD in 10 potential clinical situations (1 question for each disorder); the appropriateness of using the DMS in specific types of patients (1 question); and the usefulness of the DMS as a tool to target different reasons for adherence problems (1 question). In the survey, the DMS was described as “a digital medication platform that will give clinicians access to real-time empirical data concerning whether their patients are actually taking their oral antipsychotic medication.”
(3) How to use the DMS (11 questions). This section was divided into 4 segments: usefulness of the DMS for patients with schizophrenia, bipolar disorder, and MDD (1 question for each disorder); nonadherence alerts generated by the DMS (3 questions); appropriate responses to nonadherence of a rapidly decompensating versus stable patient (1 question for each disorder); and potential benefits and challenges of using the DMS (2 questions).
(4) Interventions in the event of nonadherence (1 question). This section was divided into 11 subsections in which respondents rated the appropriateness of 15 potential interventions as a strategy for targeting different reasons for nonadherence (eg, related to logistic problems or problems with the therapeutic alliance).
Expert Panel
Experts on schizophrenia, bipolar disorder, or MDD (n = 119) were identified based on recent publication activity, funded research, previous participation in consensus surveys in psychiatry, and/or work on guidelines concerning psychiatric illnesses. Of the 119 invited experts, 21 did not respond and 34 declined to participate. Of the remaining 64 experts, 58 (91%) completed the survey. The mean age of the respondents was 59.9 years. The respondents reported being in practice for a mean of 29.1 years and being involved in research for a mean of 30.3 years. On the basis of the reported proportion of time spent on treatment or supervision of patients with the 3 psychiatric conditions, 24 were considered experts on schizophrenia, 14 on bipolar disorder, and 16 on MDD; 4 respondents were generalists. The majority worked in an academic clinical or research setting and spent > 25% of their time treating or supervising the treatment of patients; 41 (71%) had received federal research funding as principal investigator, and 48 (83%) had been principal investigator for an industry-sponsored grant.
Data Analysis
For each option scored on the 1 to 9 rating scale, the presence or absence of consensus was first defined as a distribution unlikely to occur by chance by performing a χ2 test (P < .05) of the distribution of scores across the 3 ranges of appropriateness (1–3, 4–6, 7–9). Next, the mean and 95% confidence interval (CI) were calculated. A categorical rating of first, second, or third line was designated based on the lowest category in which the CI fell, with boundaries > 6.5 for first-line options and boundaries of 3.5 to 6.5 for second-line options. The 3 write-in options are presented as mean values.
RESULTS
Need for Objective Adherence Data
in Patients With Suboptimal Treatment Outcomes
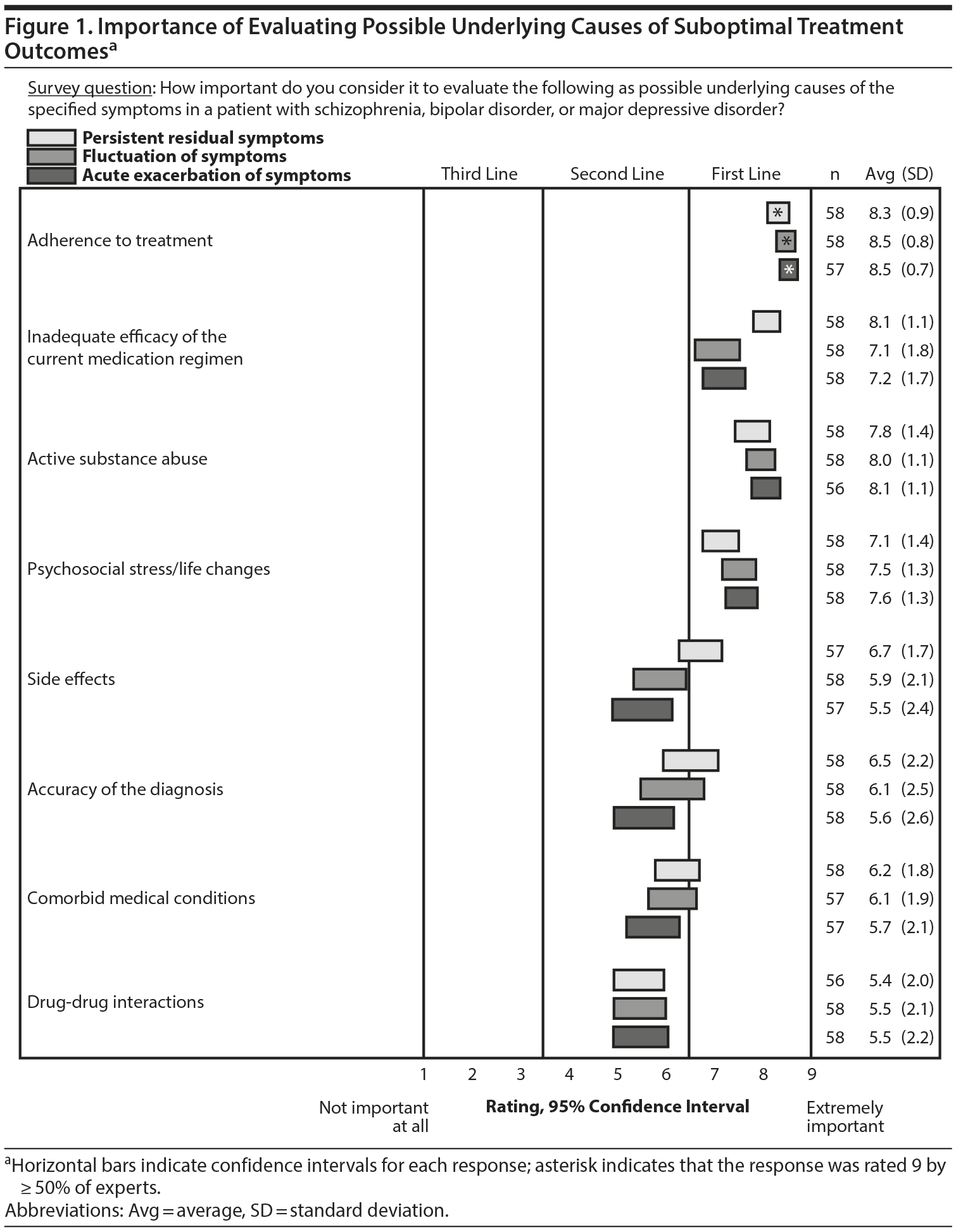
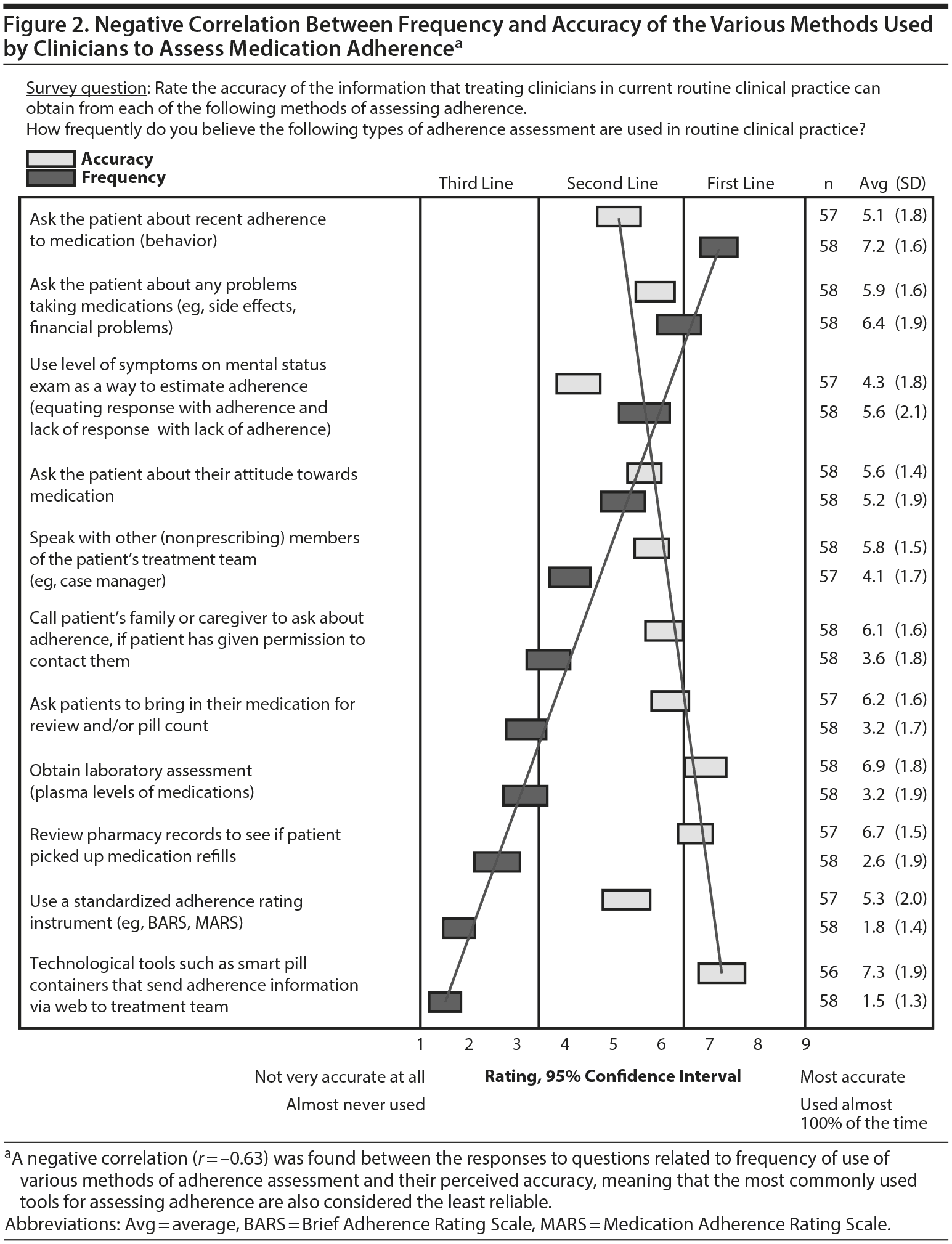
To evaluate whether there was any association between suboptimal outcomes and various underlying causes, suboptimal outcomes were presented as persistent residual symptoms, fluctuation of symptoms, or acute exacerbation of symptoms. The experts rated the importance of 8 possible underlying causes of these outcomes. Regardless of the outcome, adherence was deemed to be the most important factor (Figure 1). Despite this result, adherence was also rated as the causal factor that was least amenable to accurate assessment in current clinical practice. Clinical assessment of adherence was rated as “not accurate at all” by 37% of experts and as “somewhat accurate” by 54%. A negative correlation (r = –0.63) was found between the responses to questions about the frequency with which various methods of assessing adherence are used and their perceived accuracy (Figure 2).
When to Use the DMS
In considering the most appropriate clinical situations for use of the DMS, the experts rated 10 hypothetical scenarios that trigger concern about adherence for patients with schizophrenia, bipolar disorder, or MDD. Regardless of the disorder, the respondents identified 4 situations in which measuring of adherence is particularly important: (1) patients with a history of recurrent adherence problems, (2) patients in transitional situations (such as recent discharge from the hospital or change from supervised to independent living), (3) previously stable patients showing an increase in symptoms and/or side effects, and (4) patients with a history of serious substance abuse. The usefulness of the DMS may vary depending on the cause of adherence problems; continuing medication after discharge from the hospital, lack of daily routine, and the presence of underlying cognitive deficits were rated as the most appropriate indications (Figure 3).

However, the DMS may not be equally useful for all patients and situations in which treatment nonadherence is a problem. The experts considered patients who are unable to use simple technology as the least appropriate to use the DMS (mean [SD], 4.2 [2.4]) out of 8 specific patient characteristics that might complicate use of the system. However, 3 types of patients received first-line ratings as being appropriate for the DMS: patients with suicidal thoughts or plans (7.9 [1.5]), patients with a history of violent behavior (7.6 [1.5]), and patients who abuse substances (7.2 [1.9]). No consensus was reached on whether the DMS would be appropriate for patients who are homeless, are disorganized, or have illness characterized by persecutory delusions.
How to Use the DMS
The experts rated 4 potential uses for the DMS, all of which received high mean first-line ratings (≥ 7.4) regardless of the treated disorder (schizophrenia, bipolar disorder, MDD): (1) adherence assessment (to provide critical information concerning actual adherence when a patient presents with clinical worsening, in crisis, or with persistent symptoms), (2) monitoring alerts (to generate an alert for the clinician/treatment team after the patient crosses a predefined threshold of consecutive days of missed medication), (3) adherence intervention (to promote adherence in a patient with a pattern of intermittent adherence or premature discontinuations), and (4) routine monitoring (to provide information to be used during regular visits with the patient to integrate adherence assessment and its management into routine clinical care). Adherence assessment and monitoring alerts (1 and 2 above) received the highest rating on the 9-point scale from ≥ 50% of the experts.
Monitoring Alerts
Timing. The DMS can send alerts about patients’ nonadherence between visits. The experts were asked to write in the number of days of missed medication after which they would recommend sending an alert. They suggested 3 days, 2 to 3 days, and 4 to 5 days of missed medication before an alert for patients with schizophrenia, bipolar disorder, and MDD, respectively.
Purpose. The experts indicated that it would be useful to customize alerts based on individual patient characteristics. They also rated the helpfulness of sending alerts between visits for a variety of purposes. The ability to distinguish between inadequate response due to nonadherence versus poor efficacy received the mean rating of 8.2 on the 9-point scale and the highest rating from ≥ 50% of the experts.
Response. The experts rated the appropriateness of various potential responses to nonadherence alerts. Regardless of the disorder, for a patient who rapidly decompensates after missing medication, ≥ 50% of experts gave the highest rating to contacting the patient, the case manager, or a family member. Most experts also agreed that for individuals at high risk of relapsing soon after medication discontinuation, it was important to see the patient as soon as possible for an evaluation rather than wait for the next appointment. In the case of a patient who is stable, reliably takes medications, and is at no immediate risk of harm to self or others, contacting the patient after being alerted about recent medication discontinuation received the highest mean rating.
Potential Benefits and Barriers to Effective Use
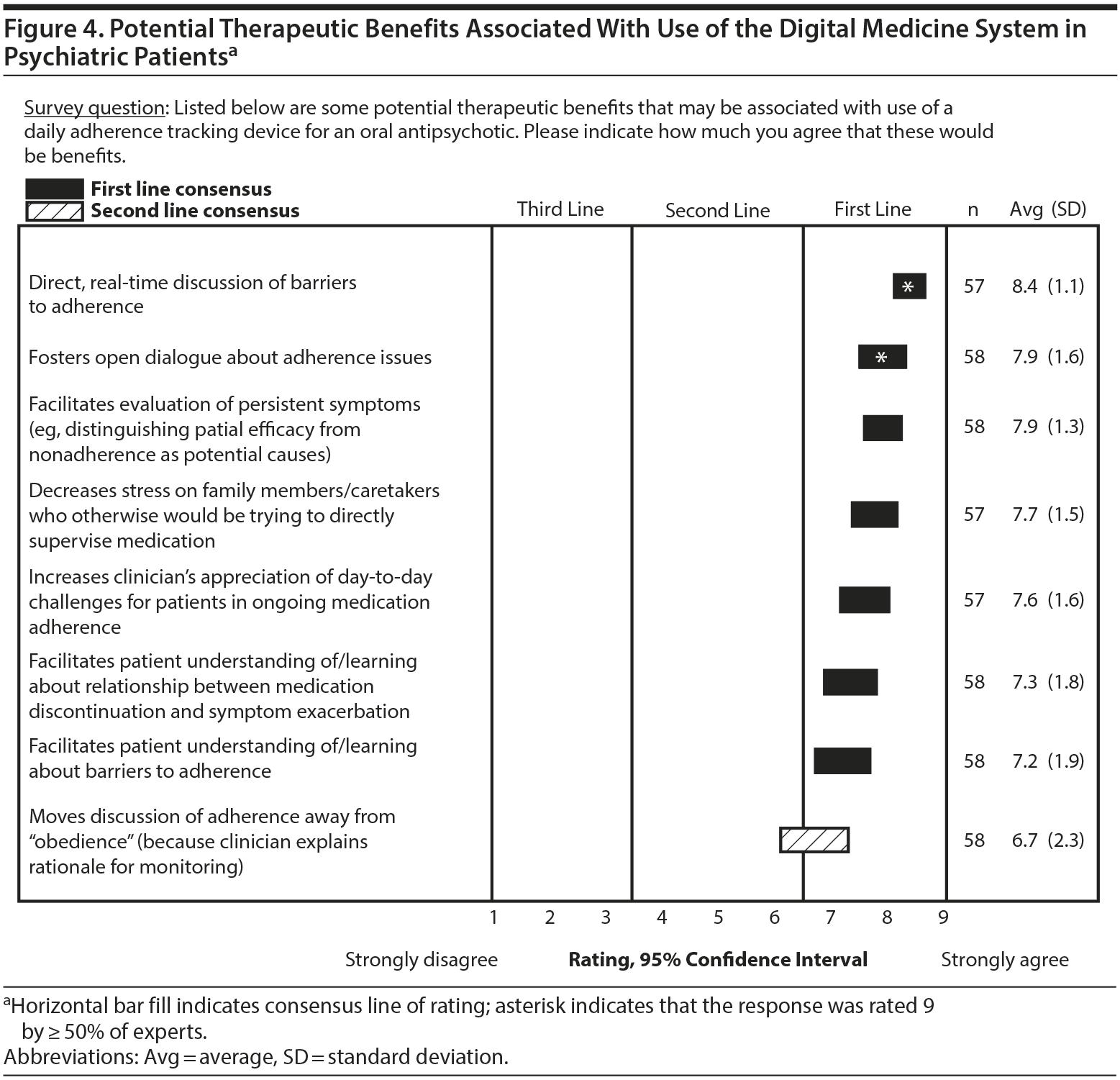
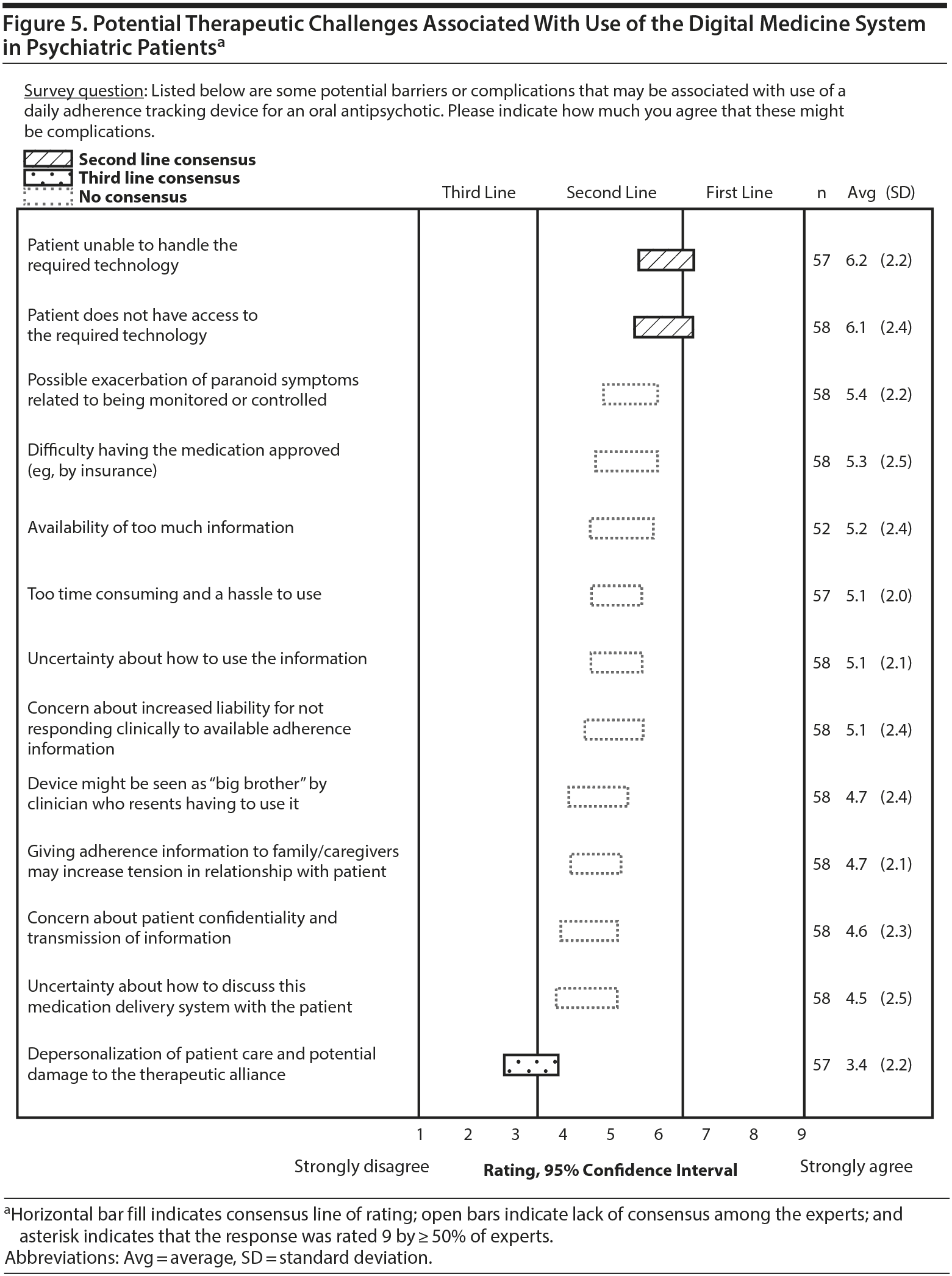
Respondents assessed therapeutic benefits as well as potential risks or challenges associated with using a DMS. Of 8 potential benefits listed in the survey, 7 received a mean first-line rating (Figure 4). The experts found it more difficult to assess challenges or risks of the DMS, as suggested by a lack of consensus on 10 of the 13 barriers or complications about which they were asked. A majority of the experts strongly agreed that patients being unable to handle or not having access to the required technology would be a significant barrier to the use of the DMS (Figure 5). Otherwise, responses tended to be split evenly on the other potential barriers that the experts were asked to rate. For example, when asked about the device being too time consuming and a hassle to use, 33% of the experts strongly agreed, 28% strongly disagreed, and 39% gave ratings in the middle of the scale.
Interventions When Adherence Problems
Have Been Detected
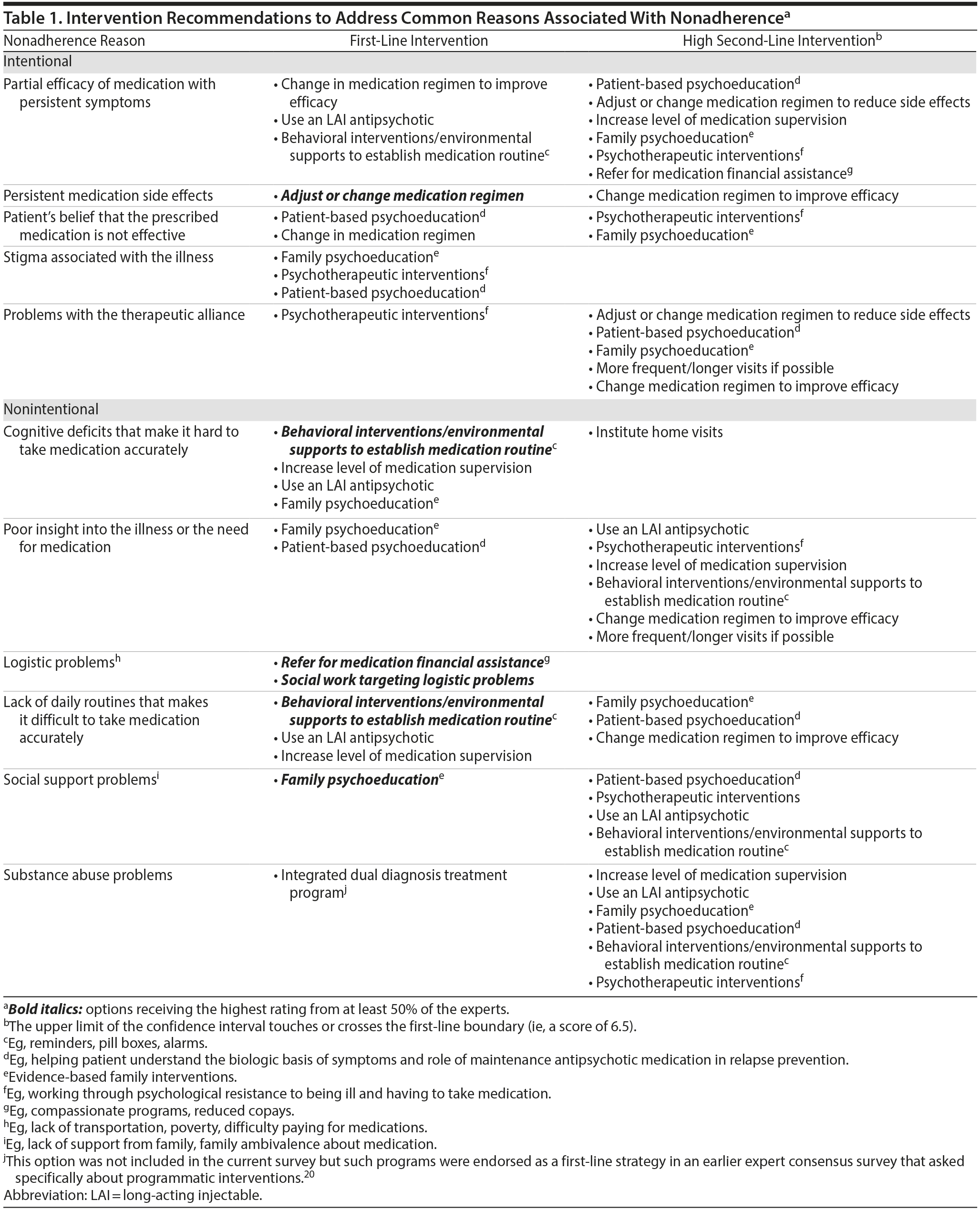
It is important to identify not only adherence problems but also the reasons for these problems. There is no single cause of nonadherence, but rather multiple reasons that can be broadly categorized as intentional or unintentional.19 Intentional nonadherence refers to a conscious decision by the patient to stop taking medication (eg, medication beliefs, side effects, partial efficacy, therapeutic alliance, stigma). Unintentional nonadherence occurs when signs or symptoms or problems in the patient’s environment interfere with medication taking (eg, poor insight, cognitive deficits, substance abuse, logistics, absent daily routines, social support problems). The experts were asked to rate the appropriateness of different interventions as strategies for targeting 11 predefined potential reasons for treatment nonadherence. First-line consensus on interventions was achieved for 10 of the items; the interventions that received first-line or high second-line ratings are summarized in Table 1.20
DISCUSSION
The DMS represents a technological advance in the emerging area of digital psychiatry. Studies evaluating and refining its use and providing general information about its technology have been undertaken in patients with SMI.21–22
In the absence of a study portfolio that addresses the myriad of issues related to the use of a device such as the DMS in practice, clinicians will need guidance facilitating its first-time use. The expert consensus methodology provides a means to develop recommendations in areas where literature is scant.
Suboptimal response to treatment is ubiquitous in schizophrenia, bipolar disorder, and MDD as demonstrated in a number of large, naturalistic studies.23–25 The expert respondents in the present survey indicated that adherence to medication is the most critical to evaluate when a patient is having a suboptimal treatment outcome. The experts also believed that adherence is the causal factor that is the least accurately assessed with the methods currently available in clinical practice. The need for new approaches to adherence assessment is underscored by the finding that the strategies that clinicians most frequently use to assess adherence were perceived as the least accurate. The expert responses about adherence assessment in the 2008 survey were similar and showed that clinicians most often rely on asking patients,17 which often leads to overestimating adherence.9 The experts in the earlier survey had recommended using more objective measures to assess adherence; however, the present survey indicates that a majority of clinicians continue to use patient self-report, likely because of limitations of the more objective methods currently available.10 The fact that nonadherence is still rated as a leading factor in suboptimal response suggests that accurate and clinically practical solutions to measuring adherence are needed.
Assessment of adherence is particularly important in situations that increase the risk of nonadherence. The experts considered accurate assessment of adherence most important in situations known to be associated with poor adherence: in patients with recurrent adherence problems26; in patients in transitional situations, such as recent discharge from hospital27; in patients with increased symptoms and/or side effects28; and in patients with a history of substance abuse.29
The utility of the DMS for assessing adherence, generating adherence alerts, and suggesting the need for interventions to improve adherence was strongly supported in the survey. The experts endorsed most of the potential benefits of the system that they were asked to rate. The majority considered the DMS to be a means to foster an open dialogue about adherence problems that may have a positive influence on the relationship between the patient and the physician. Because therapeutic alliance is associated with a patient’s attitude toward medication,30–32 an improved patient-physician relationship may result in improved medication adherence.
Assessing potential challenges of the system was somewhat difficult, as suggested by a lack of consensus on a majority of the rated factors. None of the challenges was rated as a first-line barrier. The experts had no experience with the DMS at the time of the survey, and it is also possible that additional or unanticipated barriers might arise once the technology becomes available in clinical practice. Future research could address potential issues related to provider liability, as well as the feasibility of integrating the DMS into clinical practice. The experts recognized that patient ability to use technology is important for effective use of the DMS. Moreover, as previously mentioned, certain patient and provider characteristics are likely to facilitate successful use of digital health tools in clinical practice. Indeed, a separate expert consensus survey has been conducted to assess patient and provider characteristics and features of the technology that would facilitate use of digital health tools.
Effective detection of nonadherence is a key first step in identifying underlying reasons and designing effective interventions.33 Experts consistently recommended individualized interventions based on the underlying reason for nonadherence, for instance, behavioral interventions or environmental supports to establish medication routine, or the use of long-acting injectable antipsychotics, for patients with cognitive deficits or lacking a daily routine.17 Varied approaches support the notion that there is not a universal intervention for adherence problems. In the present survey, the experts reached a high level of consensus on various first-line strategies for interventions, with the exception of interventions for patients with substance abuse problems.
CONCLUSIONS
Medication adherence was rated as the most important possible underlying cause of suboptimal treatment outcome but considered the least likely to be assessed accurately with current methods. Furthermore, the methods that are most frequently used to assess adherence were rated as being the least accurate. These results suggest an urgent need for more accurate and reliable adherence assessments in routine clinical practice. The experts recognized the potential utility of the DMS in managing medication adherence problems, identified clinical situations particularly appropriate for use of the DMS, and assessed potential benefits and challenges that may be encountered with the intended users of this technology. Detection of adherence problems with the DMS is a first step toward identifying the reasons for these problems. The results of this survey provide expert opinion on the most appropriate interventions to address adherence problems and a guide for clinicians on how to optimize the safe and effective use of the DMS when it becomes available.
Submitted: September 28, 2016; accepted March 8, 2017.
Online first: May 23, 2017.
Potential conflicts of interest: Drs Carpenter, Docherty, and Hatch are employees of ODH, Inc, and were employees of Otsuka America Pharmaceutical, Inc, and Otsuka Pharmaceutical Development & Commercialization, Inc, at the time of the survey development, execution, and interpretation. Ms Ross serves as a consultant for Otsuka and was reimbursed for work on the survey and data analysis. Dr Weiden has received research support and/or consulting honoraria from Allergen (Actavis), Alkermes, Boehringer-Ingelheim, Delpor, Forum, Johnson & Johnson (Janssen Pharmaceuticals), Lundbeck, Neurocrine, Novartis, Otsuka, Reckitt Benckiser Pharmaceuticals, Sunovion, Takeda, Teva, and Vanda and holds stock in Delpor.
Funding/support: Supported by Otsuka Pharmaceutical Development & Commercialization, Inc.
Role of the sponsor: Otsuka America Pharmaceutical, Inc, and Otsuka Pharmaceutical Development & Commercialization, Inc, were responsible for the design and conduct of the study; data collection, management, and analysis; and interpretation of the data. Otsuka Pharmaceutical Development & Commercialization, Inc, also reviewed the manuscript.
Previous presentation: IPS: The Mental Health Services Conference; October 8–11, 2015; New York, NY.
Acknowledgments: Editorial support for development of this manuscript was provided by C4 MedSolutions, LLC (Yardley, PA), a CHC Group company, and was funded by ODH, Inc.
Supplementary material: See accompanying pages.
REFERENCES
1. Trivedi MH, Rush AJ, Wisniewski SR, et al; STAR*D Study Team. Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: implications for clinical practice. Am J Psychiatry. 2006;163(1):28–40. PubMed doi:10.1176/appi.ajp.163.1.28
2. Altman S, Haeri S, Cohen LJ, et al. Predictors of relapse in bipolar disorder: a review. J Psychiatr Pract. 2006;12(5):269–282. PubMed doi:10.1097/00131746-200609000-00002
3. Caseiro O, Pérez-Iglesias R, Mata I, et al. Predicting relapse after a first episode of non-affective psychosis: a three-year follow-up study. J Psychiatr Res. 2012;46(8):1099–1105. PubMed doi:10.1016/j.jpsychires.2012.05.001
4. Morken G, Widen JH, Grawe RW. Non-adherence to antipsychotic medication, relapse and rehospitalisation in recent-onset schizophrenia. BMC Psychiatry. 2008;8:32. PubMed doi:10.1186/1471-244X-8-32
5. Weiden PJ, Kozma C, Grogg A, et al. Partial compliance and risk of rehospitalization among California Medicaid patients with schizophrenia. Psychiatr Serv. 2004;55(8):886–891. PubMed doi:10.1176/appi.ps.55.8.886
6. Sajatovic M, Valenstein M, Blow FC, et al. Treatment adherence with antipsychotic medications in bipolar disorder. Bipolar Disord. 2006;8(3):232–241. PubMed doi:10.1111/j.1399-5618.2006.00314.x
7. Valenstein M, Blow FC, Copeland LA, et al. Poor antipsychotic adherence among patients with schizophrenia: medication and patient factors. Schizophr Bull. 2004;30(2):255–264. PubMed doi:10.1093/oxfordjournals.schbul.a007076
8. Velligan DI, Wang M, Diamond P, et al. Relationships among subjective and objective measures of adherence to oral antipsychotic medications. Psychiatr Serv. 2007;58(9):1187–1192. PubMed doi:10.1176/ps.2007.58.9.1187
9. Velligan DI, Lam YW, Glahn DC, et al. Defining and assessing adherence to oral antipsychotics: a review of the literature. Schizophr Bull. 2006;32(4):724–742. PubMed doi:10.1093/schbul/sbj075
10. Sajatovic M, Velligan DI, Weiden PJ, et al. Measurement of psychiatric treatment adherence. J Psychosom Res. 2010;69(6):591–599. PubMed doi:10.1016/j.jpsychores.2009.05.007
11. Lynam I, Catley D, Goggin K, et al; MOTIV8. Autonomous regulation and locus of control as predictors of antiretroviral medication adherence. J Health Psychol. 2009;14(4):578–586. PubMed doi:10.1177/1359105309103577
12. Barclay TR, Hinkin CH, Castellon SA, et al. Age-associated predictors of medication adherence in HIV-positive adults: health beliefs, self-efficacy, and neurocognitive status. Health Psychol. 2007;26(1):40–49. PubMed doi:10.1037/0278-6133.26.1.40
13. Simoni JM, Frick PA, Huang B. A longitudinal evaluation of a social support model of medication adherence among HIV-positive men and women on antiretroviral therapy. Health Psychol. 2006;25(1):74–81. PubMed doi:10.1037/0278-6133.25.1.74
14. Hoffman JA, Cunningham JR, Suleh AJ, et al. Mobile direct observation treatment for tuberculosis patients: a technical feasibility pilot using mobile phones in Nairobi, Kenya. Am J Prev Med. 2010;39(1):78–80. PubMed doi:10.1016/j.amepre.2010.02.018
15. Kahn DA, Docherty JP, Carpenter D, et al. Consensus methods in practice guideline development: a review and description of a new method. Psychopharmacol Bull. 1997;33(4):631–639. PubMed
16. Kahn DA; The Expert Consensus Panel for Bipolar Disorder. Treatment of bipolar disorder. J Clin Psychiatry. 1996;57(suppl 12A):3–88. PubMed
17. Velligan DI, Weiden PJ, Sajatovic M, et al; Expert Consensus Panel on Adherence Problems in Serious and Persistent Mental Illness. The expert consensus guideline series: adherence problems in patients with serious and persistent mental illness. J Clin Psychiatry. 2009;70(suppl 4):1–46, quiz 47–48. PubMed doi:10.4088/JCP.7090su1cj
18. Brook RH, Chassin MR, Fink A, et al. A method for the detailed assessment of the appropriateness of medical technologies. Int J Technol Assess Health Care. 1986;2(1):53–63. PubMed doi:10.1017/S0266462300002774
19. Velligan D, Weiden PJ. Interventions to improve adherence to antipsychotic medications. Psychiatr Times. 2006;23(9):1–6.
20. McEvoy JP. The expert consensus guidelines series: treatment of schizophrenia. J Clin Psychiatry. 1999;60(suppl 11):1–80.
21. Rohatagi S, Profit D, Hatch A, et al. Optimization of a digital health feedback system in psychiatry. J Clin Psychiatry. 2016;77(9):e1101–e1107. PubMed doi:10.4088/JCP.16m10693
22. Peters-Strickland T, Pestreich L, Hatch A, et al. Usability of a novel digital medicine system in adults with schizophrenia treated with sensor-embedded tablets of aripiprazole. Neuropsychiatr Dis Treat. 2016;12:2587–2594. PubMed doi:10.2147/NDT.S116029
23. Lieberman JA, Stroup TS, McEvoy JP, et al; Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) Investigators. Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med. 2005;353(12):1209–1223. PubMed doi:10.1056/NEJMoa051688
24. Perlis RH, Ostacher MJ, Patel JK, et al. Predictors of recurrence in bipolar disorder: primary outcomes from the Systematic Treatment Enhancement Program for Bipolar Disorder (STEP-BD). Am J Psychiatry. 2006;163(2):217–224. PubMed doi:10.1176/appi.ajp.163.2.217
25. Rush AJ, Trivedi MH, Wisniewski SR, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163(11):1905–1917. PubMed doi:10.1176/ajp.2006.163.11.1905
26. Ascher-Svanum H, Zhu B, Faries D, et al. A prospective study of risk factors for nonadherence with antipsychotic medication in the treatment of schizophrenia. J Clin Psychiatry. 2006;67(7):1114–1123. PubMed doi:10.4088/JCP.v67n0715
27. Berger A, Edelsberg J, Sanders KN, et al. Medication adherence and utilization in patients with schizophrenia or bipolar disorder receiving aripiprazole, quetiapine, or ziprasidone at hospital discharge: a retrospective cohort study. BMC Psychiatry. 2012;12:99. PubMed doi:10.1186/1471-244X-12-99
28. Karow A, Czekalla J, Dittmann RW, et al. Association of subjective well-being, symptoms, and side effects with compliance after 12 months of treatment in schizophrenia. J Clin Psychiatry. 2007;68(1):75–80. PubMed doi:10.4088/JCP.v68n0110
29. Novick D, Haro JM, Suarez D, et al. Predictors and clinical consequences of non-adherence with antipsychotic medication in the outpatient treatment of schizophrenia. Psychiatry Res. 2010;176(2–3):109–113. PubMed doi:10.1016/j.psychres.2009.05.004
30. McCabe R, Bullenkamp J, Hansson L, et al. The therapeutic relationship and adherence to antipsychotic medication in schizophrenia. PLoS One. 2012;7(4):e36080. PubMed doi:10.1371/journal.pone.0036080
31. Baloush-Kleinman V, Levine SZ, Roe D, et al. Adherence to antipsychotic drug treatment in early-episode schizophrenia: a six-month naturalistic follow-up study. Schizophr Res. 2011;130(1–3):176–181. PubMed doi:10.1016/j.schres.2011.04.030
32. Day JC, Bentall RP, Roberts C, et al. Attitudes toward antipsychotic medication: the impact of clinical variables and relationships with health professionals. Arch Gen Psychiatry. 2005;62(7):717–724. PubMed doi:10.1001/archpsyc.62.7.717
33. Lacro JP, Dunn LB, Dolder CR, et al. Prevalence of and risk factors for medication nonadherence in patients with schizophrenia: a comprehensive review of recent literature. J Clin Psychiatry. 2002;63(10):892–909. PubMed doi:10.4088/JCP.v63n1007
This PDF is free for all visitors!





