Rapid Resolution of Obsessions After an Infusion of Intravenous Ketamine in a Patient With Treatment-Resistant Obsessive-Compulsive Disorder
To the Editor: Obsessive-compulsive disorder (OCD) is a leading cause of illness-related disability.1 Identifying more effective treatments with faster onset of action would be a major advance. Medications thought to modulate the glutamate system are promising new agents; for example, open trials of riluzole, memantine, and minocycline as augmentation to serotonin reuptake inhibitors (SRIs) suggest that up to half of OCD patients may experience reductions in symptoms.2-7 We chose to test intravenous ketamine (a noncompetitive N-methyl-d-aspartate [NMDA] receptor antagonist) in an OCD patient for 3 reasons: (1) it is known to modulate the glutamate system, having some mechanistic similarity to memantine8; (2) mice with a genetic deletion in a postsynaptic scaffolding protein (SAPAP3) have OCD-like compulsive behavior thought to be caused by increased NMDA activity, and ketamine decreases NMDA activity9; and (3) ketamine has been safely used in patients with depression and can relieve depressive symptoms in hours.10,11
The following case report describes the rapid resolution of obsessions in an OCD patient when she received ketamine in a double-blind crossover design of intravenous ketamine versus saline. To our knowledge, this is the first report to describe the use of ketamine in an OCD patient.
Case report. Ms A, a 24-year-old woman with DSM-IV OCD (and no other Axis I disorder) presented in May 2010 for treatment of her obsessions about symmetry/exactness (needing to have objects in the right place or else things do not "feel right") and associated repeating/checking compulsions. She spent nearly 8 hours per day managing her OCD symptoms, which interfered with her work and social relationships.
She was medically healthy and was not on medication due to the failure of 3 prior SRI trials: fluoxetine 60 mg/d, escitalopram 30 mg/d, and clomipramine 200 mg/d (each for over 3 months). Augmentation strategies were attempted unsuccessfully (ie, she refused a trial of an antipsychotic due to the possible side effect of weight gain and did not adhere to a trial of cognitive-behavioral therapy with exposure and ritual prevention). At baseline, she had severe OCD (Yale-Brown Obsessive Compulsive Scale12 score = 30) and minimal depressive symptoms (Hamilton Depression Rating Scale13 score = 7). Her sister has OCD, and her mother has depression. She provided written informed consent after a full explanation of the research procedures and their risks. The institutional review board approved the study.
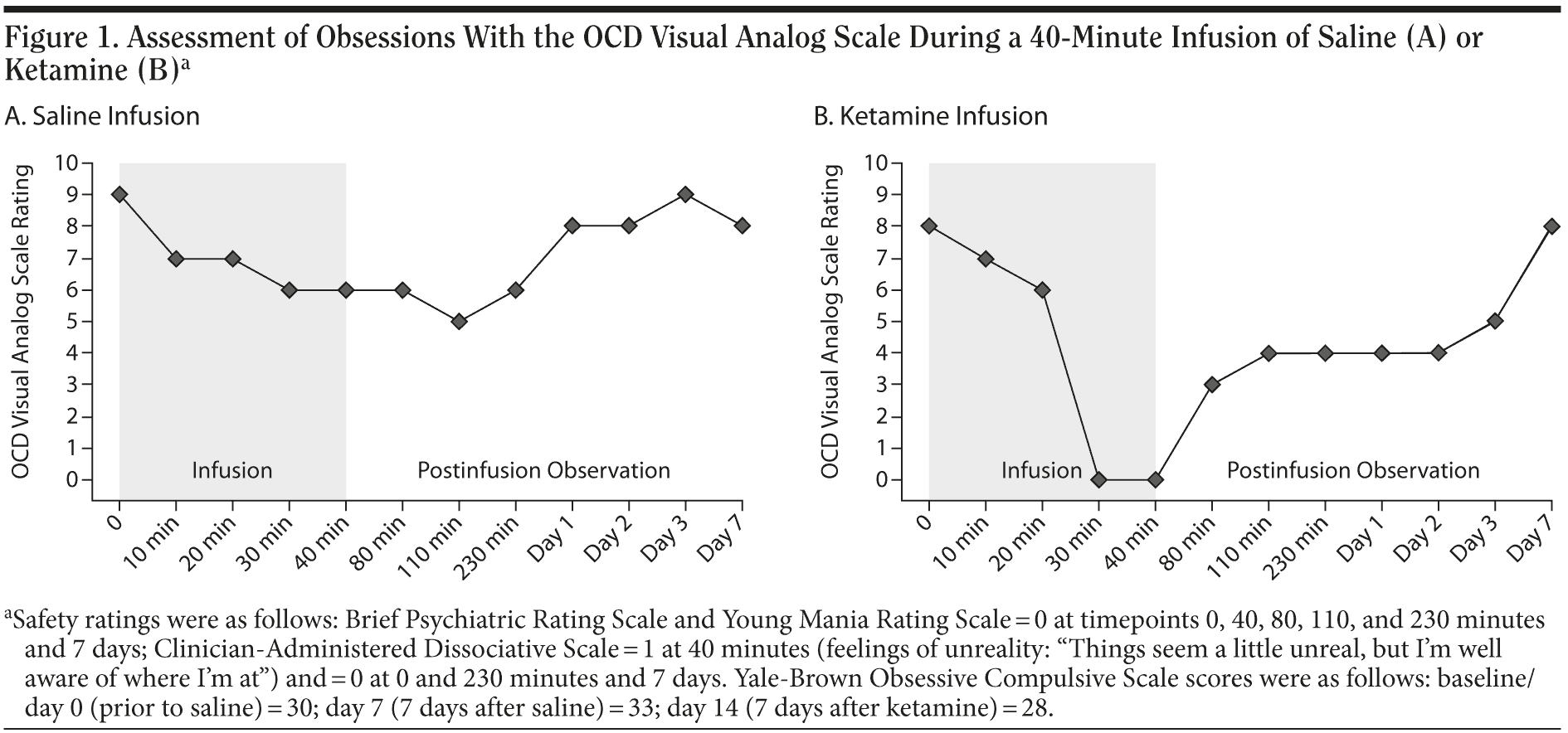
In accordance with the protocol used in a prior depression study,11 she received 2 double-blind intravenous infusions over 40 minutes given 1 week apart of saline or 0.5 mg/kg ketamine hydrochloride. An anesthesiologist provided continuous monitoring during the infusions. The patient’s symptoms were assessed at baseline, every 10 minutes during the 40-minute infusions (to detect rapid changes in symptoms if they occurred), and at several postinfusion time-points (both on the day of the infusion and up to 1 week later) using the OCD Visual Analog Scale (OCD-VAS, a modified self-rating scale used previously to detect rapid changes in OCD symptoms14-16; we focused on obsessions, which are more readily assessed rapidly than compulsions).
Ms A reported minimal reduction in obsessions during the first infusion (placebo/saline) (Figure 1A) but complete cessation of obsessions during the second infusion (ketamine) (Figure 1B). Obsessions partially reemerged 40 to 230 minutes after the infusion (Figure 1B), plateauing until postinfusion day 2; the obsessions did not return to baseline levels until postinfusion day 7.
During both the saline and ketamine infusions, Ms A’s vital signs remained within normal limits. At all time points, she denied symptoms of mania (Young Mania Rating Scale17 score = 0), psychosis (Brief Psychiatric Rating Scale18 score = 0), or intoxication (Visual Analog Scale for Intoxication10 score = 0) using standard scales. During the saline infusion, she reported lightheadedness. During the ketamine infusion, she reported lightheadedness, dry mouth, and feelings of unreality (Clinician-Administered Dissociative States Scale19 score = 1) that resolved 5 minutes after the infusion stopped.
This case report suggests that ketamine might have rapid antiobsessional effects that persist from 1 to 7 days after administration of an infusion, long after the drug has cleared. Limitations include the small sample size and the difficulties of blinding due to the psychoactive effects of ketamine. A larger trial is underway to further evaluate ketamine’s efficacy, safety, duration of effect, and mechanism of effect. In sum, ketamine may provide a useful tool to study the glutamatergic mechanism implicated in OCD and, if proven effective, may help identify novel drug targets for this disabling illness.
References
1. Murray CJL, Lopez AD. The Global Burden of Disease: A Comprehensive Assessment of Mortality and Disability from Diseases, Injuries, and Risk Factors in 1990 and Projected to 2020. Cambridge, MA: Harvard University Press; 1996.
2. Coric V, Taskiran S, Pittenger C, et al. Riluzole augmentation in treatment-resistant obsessive-compulsive disorder: an open-label trial. Biol Psychiatry. 2005;58(5):424-428. doi:10.1016/j.biopsych.2005.04.043 PubMed
3. Aboujaoude E, Barry JJ, Gamel N. Memantine augmentation in treatment-resistant obsessive-compulsive disorder: an open-label trial. J Clin Psychopharmacol. 2009;29(1):51-55. doi:10.1097/JCP.0b013e318192e9a4 PubMed
4. Feusner JD, Kerwin L, Saxena S, et al. Differential efficacy of memantine for obsessive-compulsive disorder vs generalized anxiety disorder: an open-label trial. Psychopharmacol Bull. 2009;42(1):81-93. PubMed
5. Stewart SE, Jenike EA, Hezel DM, et al. A single-blinded case-control study of memantine in severe obsessive-compulsive disorder. J Clin Psychopharmacol. 2010;30(1):34-39. doi:10.1097/JCP.0b013e3181c856de PubMed
6. Rodriguez CI, Bender J Jr, Marcus SM, et al. Minocycline augmentation of pharmacotherapy in obsessive-compulsive disorder: an open-label trial. J Clin Psychiatry. 2010;71(9):1247-1249. doi:10.4088/JCP.09l05805blu PubMed
7. Lafleur DL, Pittenger C, Kelmendi B, et al. N-acetylcysteine augmentation in serotonin reuptake inhibitor refractory obsessive-compulsive disorder. Psychopharmacology (Berl). 2006;184(2):254-256. doi:10.1007/s00213-005-0246-6 PubMed
8. Johnson JW, Kotermanski SE. Mechanism of action of memantine. Curr Opin Pharmacol. 2006;6(1):61-67. doi:10.1016/j.coph.2005.09.007 PubMed
9. Welch JM, Lu J, Rodriguiz RM, et al. Cortico-striatal synaptic defects and OCD-like behaviours in Sapap3-mutant mice. Nature. 2007;448(7156):894-900. doi:10.1038/nature06104 PubMed
10. Berman RM, Cappiello A, Anand A, et al. Antidepressant effects of ketamine in depressed patients. Biol Psychiatry. 2000;47(4):351-354. doi:10.1016/S0006-3223(99)00230-9 PubMed
11. Zarate CA Jr, Singh JB, Carlson PJ, et al. A randomized trial of an N-methyl-d-aspartate antagonist in treatment-resistant major depression. Arch Gen Psychiatry. 2006;63(8):856-864. doi:10.1001/archpsyc.63.8.856 PubMed
12. Goodman WK, Price LH, Rasmussen SA, et al. The Yale-Brown Obsessive Compulsive Scale, 2: validity. Arch Gen Psychiatry. 1989;46(11):1012-1016. PubMed
13. Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23(1):56-62. doi:10.1136/jnnp.23.1.56 PubMed
14. Murphy DL, Mueller EA, Hill JL, et al. Comparative anxiogenic, neuroendocrine, and other physiologic effects of m-chlorophenylpiperazine given intravenously or orally to healthy volunteers. Psychopharmacology (Berl). 1989;98(2):275-282. doi:10.1007/BF00444705 PubMed
15. Greenberg BD, Benjamin J, Martin JD, et al. Delayed obsessive-compulsive disorder symptom exacerbation after a single dose of a serotonin antagonist in fluoxetine-treated but not untreated patients. Psychopharmacology (Berl). 1998;140(4):434-444. doi:10.1007/s002130050787 PubMed
16. Abrantes AM, Strong DR, Cohn A, et al. Acute changes in obsessions and compulsions following moderate-intensity aerobic exercise among patients with obsessive-compulsive disorder. J Anxiety Disord. 2009;23(7):923-927. doi:10.1016/j.janxdis.2009.06.008 PubMed
17. Young RC, Biggs JT, Ziegler VE, et al. Young Mania Rating Scale. Br J Psychiatry. 1978;133:429-435. doi:10.1192/bjp.133.5.429 PubMed
18. Overall JE, Gorham DR. The Brief Psychiatric Rating Scale. Psychol Rep. 1962;10(3):799-812. doi:10.2466/PR0.10.3.799-812
19. Bremner JD, Krystal JH, Putnam FW, et al. Measurement of dissociative states with the Clinician-Administered Dissociative States Scale (CADSS). J Trauma Stress. 1998;11(1):125-136. doi:10.1023/A:1024465317902 PubMed
Author affiliations: New York State Psychiatric Institute and Department of Psychiatry (Drs Rodriguez, Kegeles, and Simpson) and Department of Anesthesia (Dr Flood), College of Physicians and Surgeons, Columbia University, New York. Potential conflicts of interest: None reported. Funding/support: This investigation was supported by grant 5T32 MH015144-31 from the National Institute of Mental Health, the Pisetsky Young Investigator Award, and the Molberger Scholar Award (to Dr Rodriguez). Acknowledgment: The authors acknowledge James Bender Jr, PsyD, for independent evaluations. Dr Bender reports no financial or other relationship relevant to the subject of this letter. Trial registration: clinicaltrials.gov Identifier: NCT01100255.
doi:10.4088/JCP.10l06653
© Copyright 2011 Physicians Postgraduate Press, Inc.