Major neurocognitive disorder, including dementia, is a significant cause of functional impairment in the older adult population.1–4 According to the DSM-5, major neurocognitive disorder is diagnosed based on the presence of cognitive deficits that (1) represent a significant decline from baseline in at least 1 cognitive domain, (2) are severe enough to interfere with daily function, (3) do not occur exclusively in the context of delirium, and (4) are not primarily attributable to another mental disorder.1 An initial neurocognitive assessment typically includes a clinical interview with the patient and caregiver/family, mental status examination, brief in-office cognitive screening tests (such as the Mini-Cog,5 Mini-Mental State Examination,6 Montreal Cognitive Assessment [MoCA],7 or Saint Louis University Mental Status Examination8), laboratory testing (such as chemistry panel, thyroid panel, and vitamin B12),2–4,9 and structural brain imaging (such as head magnetic resonance imaging [MRI] or computed tomography [CT]).2,3,9–13 This may be followed by formal neuropsychological testing, which is widely considered the gold standard diagnostic tool and entails a battery of tests to assess an individual’s function across multiple cognitive domains.1,4,9–12,14–20 In complex cases where there is still uncertainty regarding the diagnosis, specialized imaging studies such as positron emission tomography (PET) can provide further diagnostic clarity by helping differentiate between dementia syndromes such as Alzheimer disease and frontotemporal dementia,4,9,11,13,21–25 as well as distinguishing dementia from cognitive impairment due to depression or other etiologies.13,26
Despite the various tools available to assess for major neurocognitive disorder, there is still a risk of inaccurate diagnosis. Formal neuropsychological testing is reported to have greater sensitivity and specificity than brief cognitive screening tests,9,16 although values for sensitivity and specificity can vary significantly depending on cutoff score designations. Standardized neuropsychological interviews for the detection of dementia have been reported to have sensitivity values ranging from 71% to 100% and specificity values from 46% to 90%,27–30 while various neuropsychological test batteries commonly utilized in dementia diagnosis have exhibited a wide range of sensitivity values from 32.1% to 100% and specificity values from 43% to 100%.10,11,17–19,31–44 Given the major social, legal, and financial implications associated with a false-positive diagnosis of dementia, previous literature on the topic of dementia misdiagnosis has primarily focused on the risks of false-positive diagnosis where major neurocognitive disorder does not exist30,39,45–48 and/or the risks of inaccurate diagnosis when only brief cognitive screening tools are utilized without further formal neuropsychological testing.17,19,49–55 In contrast, less attention has focused on the possibility and risks of a false negative diagnosis after formal neuropsychological testing. The risks of a false-negative dementia diagnosis may be especially relevant for patients with milder symptoms,29,56 given that some studies have reported instances of patients who presented with memory complaints but who had normal neuropsychological test results and did not meet criteria for mild cognitive impairment at the time.57,58 Overall, the risks of both false-positive and false-negative diagnoses of dementia are important to consider because neuropsychological screening and testing results can potentially be influenced by the evaluator’s unconscious biases and application of heuristics.59
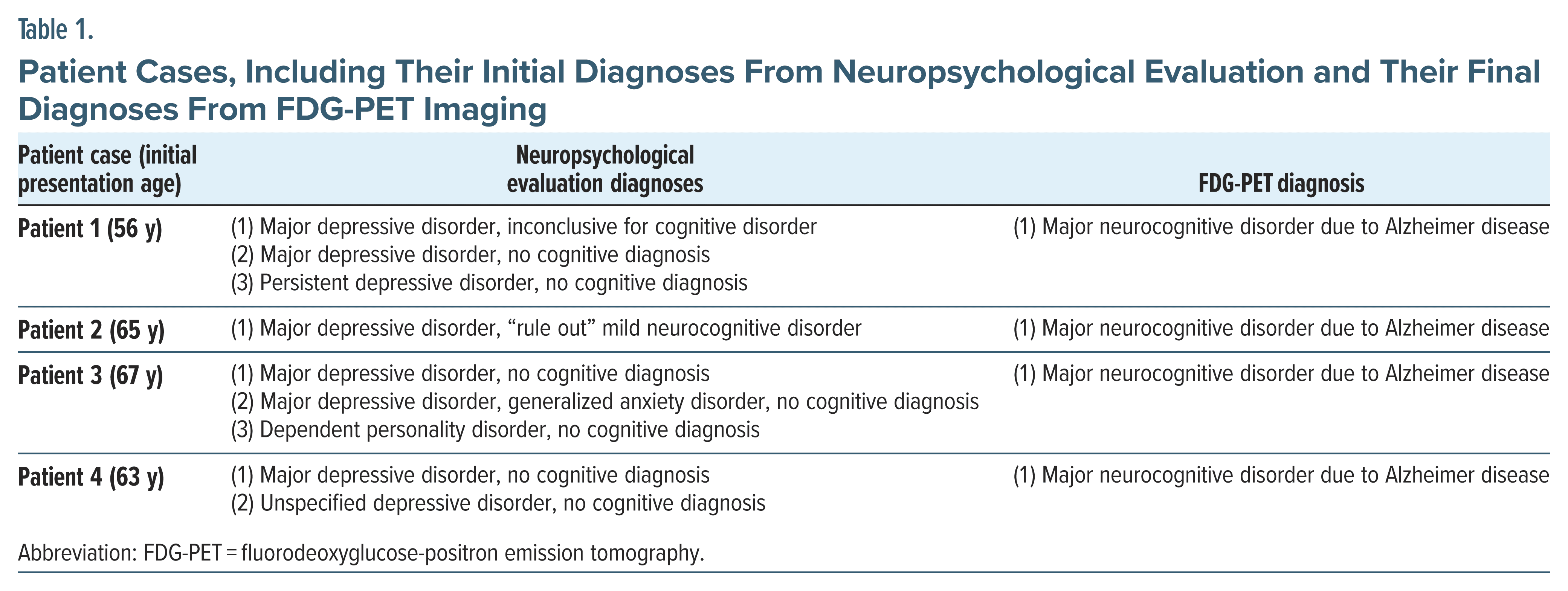
This case series presents several patients who underwent formal neuropsychological testing that did not diagnose dementia, but whose clinical course and neuroimaging findings turned out to be consistent with dementia. These false negative diagnoses following neuropsychological testing for dementia contribute an important perspective to the literature on current standards for dementia assessment.
Case 1
Patient 1 is a male veteran. At age 56 years, he reported falling and striking his head. Shortly afterward, he was referred to the mental health clinic for concerns of memory loss, irritability, headache, and inability to complete his community college classes. At the time, CT of the head was normal. A laboratory panel, which included a comprehensive metabolic panel (CMP), complete blood count (CBC), thyroid-stimulating hormone (TSH) level, and vitamin B12 level, was unremarkable. On psychiatric evaluation, he was diagnosed with postconcussive syndrome, an unspecified cognitive disorder, and an unspecified anxiety disorder.
He and his wife continued to report cognitive and behavioral changes. He underwent formal neuropsychological testing about a year later and was found to have inconsistent effort and motivation, but he scored in the average range on measures of memory and executive function. These results were deemed inconclusive for a neurocognitive disorder and were thought to be consistent with major depressive disorder (MDD). He was treated with a regimen for depression, leading to partial benefit for his depressive symptoms, but no cognitive or functional improvement.
Over the following years, his personal care and hygiene had improved with antidepressant medication, but he continued to exhibit functional deficits and confusion with various activities, such as difficulty using his phone and other electronic devices, doing dishes, playing checkers, and walking without getting lost. About 5 years after initial neuropsychological testing, he underwent neuropsychological reevaluation. He continued to demonstrate reduced engagement with testing, which interfered with the interpretation of other results on testing (such as memory). The only diagnosis given was therefore MDD.
After another 2 years, he underwent a third round of neuropsychological testing. This time, his performance on measures of engagement and motivation was noted to be 17 standard deviations lower than what would be expected for patients with moderate to severe traumatic brain injury, while his performance on measures of memory could not be interpreted due to his observed lack of engagement with testing. He scored in the average range for basic attention and phonemic fluency and high average for naming. It was thought that his performance was not consistent with a known dementia. He was given the diagnosis of persistent depressive disorder without a neurocognitive diagnosis.
However, about a year later, a fluorodeoxyglucose (FDG)-PET scan was done, which showed hypometabolism in the bilateral parietal and temporal lobes, bilateral frontal lobes, posterior cingulate gyrus, and precuneus. Given the imaging findings in a pattern suggestive of Alzheimer disease, combined with the patient’s continued significant decline in cognitive function and cognitive deficits that interfered with everyday activities, the diagnosis was updated to early-onset Alzheimer disease.
Case 2
Patient 2 is a male veteran. At age 65 years, the patient presented to the mental health clinic with a 2-year history of cognitive and functional decline. He scored 12/30 on the MoCA, which would be expected to correspond to moderate cognitive impairment,28 though at the time, the psychiatrist believed this score did not fully reflect his abilities due to his difficulty engaging with the test. Pertinent laboratory values near this time, including a CMP, CBC, TSH level, and vitamin B12 level, showed stable moderate chronic kidney disease but were otherwise unremarkable. CT of the head showed a moderate degree of diffuse cerebral volume loss and features of chronic ischemic change in the deep brain matter.
Neuropsychological testing was then done and led to the assessment of recurrent MDD, although it was noted that mild neurocognitive disorder had not been completely ruled out. Among the findings was underperformance on validity measures of memory and attention that suggested a degree of impairment surprising for his relative independence in instrumental activities of daily living (IADLs).
Subsequently, despite attempts to treat for depression and encourage independence, the patient continued to experience significant cognitive and functional impairment. He had undergone a decline in some IADLs, including loss of his abilities to drive and manage medications. His wife also reported that he had intermittent confusion about where home was and who she was, and he would sometimes leave home while saying he was “going home.” Less than 2 years after neuropsychological testing, he underwent an FDG-PET scan, which found hypometabolism in a pattern suggestive of Alzheimer disease, including in the anterior and posterior cingulate gyri, bilateral temporal poles, medial temporal lobes, hippocampal and parahippocampal gyri, and precuneus. Given these imaging findings, as well as the patient’s continued significant decline in cognitive function and cognitive deficits that interfered with everyday activities, the diagnosis was updated to a major neurocognitive disorder due to early-onset Alzheimer disease.
Case 3
Patient 3 is a female veteran. At age 67 years, she presented to the mental health clinic due to concerns of short-term memory impairment, depressed and anxious mood, and sleep impairment. She had been independent with activities of daily living (ADLs) and semi-independent with some IADLs, though she was noted to be getting lost more frequently and had experienced a recent fall. The MoCA at the time was scored as 17/30, which would be expected to correspond to moderate cognitive impairment,60 and the Geriatric Depression Scale61 was scored as 10/15, which would be expected to correlate with moderate depression. Head MRI revealed chronic microvascular ischemic changes but no acute abnormalities. She was initially diagnosed with dementia and MDD and then started on low-dose donepezil and an antidepressant.
She also underwent formal neuropsychological testing, which showed impairment in cognitive functioning, particularly attention/ concentration and processing speed. At the time, these neuropsychological findings were thought to be most likely due to MDD instead of dementia, given her concurrent mood symptoms and relative functional independence. Donepezil was then discontinued. The patient was also referred for psychotherapy.
Over the following year, however, her memory symptoms and functional deficits did not improve. About a year after initial neuropsychological testing, reevaluation was conducted by another neuropsychologist, but this was discontinued due to low effort and inconsistent performance, and her symptoms were believed to be due to MDD and generalized anxiety disorder instead of dementia. Afterward, she was switched to another antidepressant due to lack of efficacy. A repeat MRI of the head was also done, which again showed nonspecific chronic microvascular ischemia. Subsequently, her psychiatric diagnosis was updated by her psychologist/psychotherapist to include dependent personality disorder.
About 3 years after the second neuropsychological evaluation, the patient underwent neuropsychological testing again with a third different neuropsychologist, and she was again noted to exhibit low effort and inconsistent performance. Her performance was attributed to dependent personality disorder instead of dementia, with the recommendation for no further neuropsychological testing unless she had a documented medical event.
However, she continued to exhibit memory difficulties, along with worsening functional deficits and increasingly bizarre behaviors, such as not recognizing her own reflection in the mirror, as well as beliefs that inanimate objects were talking to her. A month later, an FDG-PET scan was done and revealed moderate regionally decreased metabolic activity within the bilateral posterior parietal and temporal cortices, posterior cingulate, and precuneus, consistent with Alzheimer disease. With these imaging findings, and the patient’s continued significant decline in cognitive function and cognitive deficits that interfered with everyday activities, her diagnosis was finally changed to major neurocognitive disorder due to Alzheimer disease.
Case 4
Patient 4 is a male veteran with a history of pacemaker placement. At age 63 years, he presented to the mental health clinic for concerns of worsening memory difficulties over the past 2 months, following a mild concussion that had occurred around a year ago. He had been independent with ADLs and semi-independent with IADLs. At the time of the concussion, CT of the head at an outside facility had been reportedly unremarkable. In his initial workup, CMP and CBC were nonexplanatory. His TSH level was within normal limits, and vitamin B12 was low-normal. Repeat CT of the head showed no acute abnormality.
He then completed a neuropsychological evaluation. Reduced engagement was noted, and he scored within normal limits (low average range or higher) on most measures, including those testing memory. He was given a diagnosis of MDD, and no neurocognitive disorder was diagnosed.
About a year later, repeat neuropsychological testing was conducted by a different neuropsychologist, who diagnosed the patient with an unspecified depressive disorder and again attributed his reported memory difficulties to mental illness. At the time, another CT of the head was unremarkable, and the patient was unable to undergo MRI brain imaging due to his pacemaker.
Given the lack of improvement in his memory deficits, there continued to be clinical suspicion for dementia. Less than 2 years later, an FDG-PET scan was done, which showed significantly decreased FDG uptake in the bilateral precuneus, parietal lobes, posterior cingulate gyrus, middle temporal gyrus, and lateral temporal lobes. These imaging findings, as well as the patient’s continued significant decline in cognitive function and cognitive deficits that interfered with everyday activities, were noted to be consistent with major neurocognitive disorder due to Alzheimer disease, and the patient was finally started on donepezil.
Discussion
As these patient cases illustrate (Table 1), there is a risk that neuropsychological evaluation may result in a false-negative diagnosis for dementia. All 4 of these patients initially presented with memory loss together with mood symptoms, and formal neuropsychological testing showed inconsistent or impaired effort, leading to the attribution of memory loss to depression. However, for these patients, a lack of clinical improvement following depression treatment, combined with findings suggestive of dementia on follow-up imaging studies, ultimately led to dementia diagnosis, consistent with the DSM-5 criteria for neurocognitive disorder due to Alzheimer disease.1 It should be noted that in these patient cases, the time between neuropsychological evaluation and PET imaging was short, making conversion to dementia between the time of neuropsychological testing and imaging less likely. Further, the false-negative dementia diagnoses were not limited to a single neuropsychologist, as these patients were evaluated by several different neuropsychologists.
Although neuropsychological testing is considered a gold standard for major neurocognitive disorder diagnosis, it entails a degree of subjective interpretation of the patient’s cognitive status and function, leading to the potential for false negative misdiagnosis. In this case series, 1 factor that may have influenced the interpretation of neuropsychological testing results was the patients’ relatively young age at the onset of cognitive symptoms. These patients were still in their 50s or 60s at the time of initial presentation and would thus be expected to have a lower baseline probability of dementia compared with the oldest geriatric patients, as dementia risk rises with increasing age.4,12,15,62 Additionally, younger patients with Alzheimer disease may present with a higher prevalence of atypical symptoms,63 and individuals in the early stages of Alzheimer disease may be more difficult to identify if neuropsychological testing overemphasizes certain cognitive abilities, such as immediate recall learning, recognition memory, and intrusions.42 Another consideration for this case series is that patients with dementia and a concurrent psychiatric diagnosis may potentially be at risk of having their memory deficits prematurely attributed to psychiatric illness alone.15 Dementia risk is noted to be higher in individuals with mental health disorder diagnoses,4,15,26,64–67 and thus, it is important for clinicians to note that diagnosing mental illness in a patient with memory deficits does not exclude major neurocognitive disorder (or vice versa). In cases where a diagnosis of dementia versus depression is unclear, imaging studies such as brain PET and MRI can provide additional data to help distinguish between dementia and depression26; however, imaging findings should always be interpreted with caution and as part of a larger clinical picture, given concerns that overreliance on imaging can still present a risk of false-positive dementia misdiagnosis.68,69
Ultimately, this case series adds to the literature on the risk of a false negative dementia diagnosis after formal neuropsychological testing. Although scientific literature on dementia misdiagnosis has focused primarily on the risks of false-positive diagnosis of major neurocognitive disorder,30,39,45–48 the significance of avoiding a false-negative diagnosis has received less scholarly attention. It is important to note that a false-negative dementia diagnosis can lead to a patient not receiving the dementia care and support that they need, given that others around them would expect them to function at a higher level than would be appropriate. This could potentially impact a patient’s safety, access to resources, interactions with family/caretakers, and ability to function in social settings such as conversations. However, the risks and impacts of a false-negative dementia diagnosis can potentially be reduced with repeated neuropsychological testing and/or screening.52
Of note, this case series has several limitations. First, the sample size is small, consisting only of a few cases. In terms of demographics, the individuals in these cases are all veterans who were seen at health care facilities within the Veterans Affairs North Texas Health Care System, limiting generalizability to nonveteran populations. Additionally, although the Veterans Affairs facilities where these patients were seen all had access to PET imaging, not all health care facilities may have PET or other advanced imaging modalities available, and similarly, not all individuals’ health care insurance may cover advanced imaging. Finally, all the patients in this case series were ultimately diagnosed with major neurocognitive disorder due to Alzheimer disease, limiting generalizability to patients with non-Alzheimer dementia.
Article Information
Published Online: December 17, 2024. https://doi.org/10.4088/PCC.24cr03752
© 2024 Physicians Postgraduate Press, Inc.
Prim Care Companion CNS Disord 2024;26(6):24cr03752
Submitted: April 1, 2024; accepted June 24, 2024.
To Cite: Wu R, Maharaj A, Johnson AE, et al. Alzheimer dementia confirmed by FDG-PET after negative neuropsychological testing: a case series. Prim Care Companion CNS Disord. 2024;26(4):24cr03752.
Author Affiliations: University of Texas Southwestern Medical School, Dallas, Texas (Wu, Maharaj, Johnson, Woolbert); Johns Hopkins Hospital, Baltimore, Maryland (Wu); VA North Texas Health Care System, Dallas, Texas (Johnson, Woolbert).
Corresponding Author: Richard Wu, MD, Johns Hopkins Hospital, 1800 Orleans St, Baltimore, MD 21287 ([email protected]).
Relevant Financial Relationships: None.
Funding/Support: None.
Patient Consent: Consent was received from the patients’ caregivers to publish these cases, given the patients’ lack of capacity to consent. Patient information has been de identified to protect privacy.
ORCID: Richard Wu: https://orcid.org/0000-0002-1904-1203
References (69)

- American Psychiatric Association. Neurocognitive disorders. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, Text Revision (DSM-5-TR). American Psychiatric Association Publishing; 2022.
- American Psychological Association Task Force for the Evaluation of Dementia and Age-Related Cognitive Change. APA Guidelines for the Evaluation of Dementia and Age-Related Cognitive Change. 2021. https://www.apa.org/practice/guidelines/guidelinesdementia-age-related-cognitive-change.pdf.
- Gale SA, Acar D, Daffner KR. Dementia. Am J Med. 2018;131(10):1161–1169. PubMed CrossRef
- Hugo J, Ganguli M. Dementia and cognitive impairment: epidemiology, diagnosis, and treatment. Clin Geriatr Med. 2014;30(3):421–442. PubMed CrossRef
- Borson S, Scanlan J, Brush M, et al. The Mini-Cog: a cognitive ‘vital signs’ measure for dementia screening in multi-lingual elderly. Int J Geriatr Psychiatry. 2000;15(11):1021–1027. PubMed CrossRef
- Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189–198. PubMed
- Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695–699. PubMed CrossRef
- Tariq SH, Tumosa N, Chibnall JT, et al. Comparison of the Saint Louis University mental status examination and the mini-mental state examination for detecting dementia and mild neurocognitive disorder—a pilot study. Am J Geriatr Psychiatry. 2006;14(11):900–910. PubMed CrossRef
- Feldman HH, Jacova C, Robillard A, et al. Diagnosis and treatment of dementia: 2. Diagnosis. CMAJ. 2008;178(7):825–836. PubMed CrossRef
- Schroeder RW, Martin PK, Walling A. Neuropsychological evaluations in adults. Am Fam Physician. 2019;99(2):101–108. PubMed
- Weissberger GH, Strong JV, Stefanidis KB, et al. Diagnostic accuracy of memory measures in Alzheimer’s dementia and mild cognitive impairment: a systematic review and meta-analysis. Neuropsychol Rev. 2017;27(4):354–388. PubMed CrossRef
- Fields JA, Ferman TJ, Boeve BF, et al. Neuropsychological assessment of patients with dementing illness. Nat Rev Neurol. 2011;7(12):677–687. PubMed CrossRef
- Small GW, Bookheimer SY, Thompson PM, et al. Current and future uses of neuroimaging for cognitively impaired patients. Lancet Neurol. 2008;7(2):161–172. PubMed CrossRef
- Jónsdóttir MK, Harrison J, Hannesdóttir KI. The ambivalence toward neuropsychology in dementia research, diagnosis, and drug development: myths and misconceptions. Alzheimers Dement. 2023;19(5):2175–2181. PubMed
- Tetsuka S. Depression and dementia in older adults: a neuropsychological review. Aging Dis. 2021;12(8):1920–1934. PubMed CrossRef
- Pasternak E, Smith G. Cognitive and neuropsychological examination of the elderly. Handb Clin Neurol. 2019;167:89–104. PubMed CrossRef
- Wang Z, Dong B. Screening for cognitive impairment in geriatrics. Clin Geriatr Med. 2018;34(4):515–536. PubMed CrossRef
- Dalla Barba G, Traykov L, Baudic S. Neuropsychological examination in dementia. Handb Clin Neurol. 2008;89:15–33. PubMed CrossRef
- Jacova C, Kertesz A, Blair M, et al. Neuropsychological testing and assessment for dementia. Alzheimers Dement. 2007;3(4):299–317. PubMed CrossRef
- Boller F, Barba GD. Neuropsychological tests in Alzheimer’s disease. Aging (Milano). 2001;13(3):210–220. PubMed CrossRef
- Ossenkoppele R, Jansen WJ, Rabinovici GD, et al. Prevalence of amyloid PET positivity in dementia syndromes: a meta-analysis. JAMA. 2015;313(19):1939–1949. PubMed CrossRef
- Rabinovici GD, Rosen HJ, Alkalay A, et al. Amyloid vs FDG-PET in the differential diagnosis of AD and FTLD. Neurology. 2011;77(23):2034–2042. PubMed CrossRef
- Foster NL, Heidebrink JL, Clark CM, et al. FDG-PET improves accuracy in distinguishing frontotemporal dementia and Alzheimer’s disease. Brain. 2007;130(Pt 10):2616–2635. PubMed CrossRef
- Ishii K, Sakamoto S, Sasaki M, et al. Cerebral glucose metabolism in patients with frontotemporal dementia. J Nucl Med. 1998;39(11):1875–1878. PubMed
- Pickut BA, Saerens J, Mariën P, et al. Discriminative use of SPECT in frontal lobe-type dementia versus (senile) dementia of the Alzheimer’s type. J Nucl Med. 1997;38(6):929–934. PubMed
- Leyhe T, Reynolds CF 3rd, Melcher T, et al. A common challenge in older adults: classification, overlap, and therapy of depression and dementia. Alzheimers Dement. 2017;13(1):59–71. PubMed CrossRef
- Abbate C, Trimarchi PD, Inglese S, et al. Signs and symptoms method in neuropsychology: a preliminary investigation of a standardized clinical interview for assessment of cognitive decline in dementia. Appl Neuropsychol Adult. 2021;28(3):282–296. PubMed CrossRef
- Galvin JE, Roe CM, Xiong C, et al. Validity and reliability of the AD8 informant interview in dementia. Neurology. 2006;67(11):1942–1948. PubMed CrossRef
- Ellis RJ, Jan K, Kawas C, et al. Diagnostic validity of the Dementia Questionnaire for Alzheimer disease. Arch Neurol. 1998;55(3):360–365. PubMed CrossRef
- Kawas C, Segal J, Stewart WF, et al. A validation study of the Dementia Questionnaire. Arch Neurol. 1994;51(9):901–906. PubMed CrossRef
- Núñez-Fernández S, Rivera D, Arroyo-Anlló EM, et al. Validation of the norma latina neuropsychological assessment battery in patients with Alzheimer’s disease in Mexico. Int J Environ Res Public Health. 2022;19(18):11322. PubMed
- Larner AJ, Mitchell AJ. A meta-analysis of the accuracy of the Addenbrooke’s Cognitive Examination (ACE) and the Addenbrooke’s Cognitive Examination Revised (ACE-R) in the detection of dementia. Int Psychogeriatr. 2014;26(4):555–563. PubMed CrossRef
- Mathews M, Abner E, Kryscio R, et al. Diagnostic accuracy and practice effects in the national Alzheimer’s coordinating center uniform data set neuropsychological battery. Alzheimers Dement. 2014;10(6):675–683. PubMed CrossRef
- Karantzoulis S, Novitski J, Gold M, et al. The Repeatable Battery for the Assessment of Neuropsychological Status (RBANS): utility in detection and characterization of mild cognitive impairment due to Alzheimer’s disease. Arch Clin Neuropsychol. 2013;28(8):837–844. PubMed CrossRef
- Hatfield CF, Dudas RB, Dening T. Diagnostic tools for dementia. Maturitas. 2009;63(3):181–185. PubMed CrossRef
- Duff K, Humphreys Clark JD, O’Bryant SE, et al. Utility of the RBANS in detecting cognitive impairment associated with Alzheimer’s disease: sensitivity, specificity, and positive and negative predictive powers. Arch Clin Neuropsychol. 2008;23(5):603–612. PubMed CrossRef
- Mioshi E, Dawson K, Mitchell J, et al. The Addenbrooke’s Cognitive Examination Revised (ACE R): a brief cognitive test battery for dementia screening. Int J Geriatr Psychiatry. 2006;21(11):1078–1085. PubMed CrossRef
- Chandler MJ, Lacritz LH, Hynan LS, et al. A total score for the CERAD neuropsychological battery. Neurology. 2005;65(1):102–106. PubMed CrossRef
- Huppert FA, Jorm AF, Brayne C, et al. Psychometric properties of the CAMCOG and its efficacy in the diagnosis of dementia. Aging Neuropsychol Cogn. 1996;3(3):201–214.
- Cahn DA, Salmon DP, Butters N, et al. Detection of dementia of the Alzheimer type in a population-based sample: neuropsychological test performance. J Int Neuropsychol Soc. 1995;1(3):252–260. PubMed CrossRef
- Monsch AU, Bondi MW, Salmon DP, et al. Clinical validity of the Mattis Dementia Rating Scale in detecting Dementia of the Alzheimer type. A double cross-validation and application to a community dwelling sample. Arch Neurol. 1995;52(9):899–904. PubMed CrossRef
- Welsh K, Butters N, Hughes J, et al. Detection of abnormal memory decline in mild cases of Alzheimer’s disease using CERAD neuropsychological measures. Arch Neurol. 1991;48(3):278–281. PubMed CrossRef
- Bracco L, Amaducci L, Pedone D, et al. Italian Multicentre Study on Dementia (SMID): a neuropsychological test battery for assessing Alzheimer’s disease. J Psychiatr Res. 1990;24(3):213–226. PubMed CrossRef
- Roth M, Tym E, Mountjoy CQ, et al. CAMDEX: a standardised instrument for the diagnosis of mental disorder in the elderly with special reference to the early detection of dementia. Br J Psychiatry. 1986;149(6):698–709. PubMed CrossRef
- Loring DW, Goldstein FC, Chen C, et al. False-positive error rates for reliable digit span and auditory verbal learning test performance validity measures in amnestic mild cognitive impairment and early Alzheimer disease. Arch Clin Neuropsychol. 2016;31(4):313–331. PubMed CrossRef
- Beeri MS, Schmeidler J, Sano M, et al. Age, gender, and education norms on the CERAD neuropsychological battery in the oldest old. Neurology. 2006;67(6):1006–1010. PubMed CrossRef
- Vickery CD, Berry DT, Inman TH, et al. Detection of inadequate effort on neuropsychological testing: a meta-analytic review of selected procedures. Arch Clin Neuropsychol. 2001;16(1):45–73. PubMed
- Fillenbaum G, Heyman A, Williams K, et al. Sensitivity and specificity of standardized screens of cognitive impairment and dementia among elderly black and white community residents. J Clin Epidemiol. 1990;43(7):651–660. PubMed CrossRef
- Seitz DP, Chan CCH, Newton HT, et al. Mini-Cog for the detection of dementia within a primary care setting. Cochrane Database Syst Rev. 2021;7(7):CD011415. PubMed CrossRef
- Ranson JM, Kuźma E, Hamilton W, et al. Predictors of dementia misclassification when using brief cognitive assessments. Neurol Clin Pract. 2019;9(2):109–117. PubMed CrossRef
- Tsoi KKF, Chan JYC, Hirai HW, et al. Cognitive tests to detect dementia: a systematic review and meta analysis. JAMA Intern Med. 2015;175(9):1450–1458. PubMed
- Lin JS, O’Connor E, Rossom RC, et al. Screening for cognitive impairment in older adults: a systematic review for the U.S. Preventive services task force. Ann Intern Med. 2013;159(9):601–612. PubMed CrossRef
- Fujii D, Hishinuma E, Masaki K, et al. Dementia screening: can a second administration reduce the number of false positives? Am J Geriatr Psychiatry. 2003;11(4):462–465. PubMed
- Huppert FA, Brayne C, Gill C, et al. CAMCOG—a concise neuropsychological test to assist dementia diagnosis: socio-demographic determinants in an elderly population sample. Br J Clin Psychol. 1995;34(4):529–541. PubMed CrossRef
- Anthony JC, LeResche L, Niaz U, et al. Limits of the ‘Mini-Mental State’ as a screening test for dementia and delirium among hospital patients. Psychol Med. 1982;12(2):397–408. PubMed CrossRef
- Gavett BE, Lou KR, Daneshvar DH, et al. Diagnostic accuracy statistics for seven Neuropsychological Assessment Battery (NAB) test variables in the diagnosis of Alzheimer’s disease. Appl Neuropsychol Adult. 2012;19(2):108–115. PubMed CrossRef
- Nunes T, Fragata I, Ribeiro F, et al. The outcome of elderly patients with cognitive complaints but normal neuropsychological tests. J Alzheimers Dis. 2010;19(1):137–145. PubMed CrossRef
- Saykin AJ, Wishart HA, Rabin LA, et al. Older adults with cognitive complaints show brain atrophy similar to that of amnestic MCI. Neurology. 2006;67(5):834–842. PubMed CrossRef
- Ortega J. Neuropsychological diagnostic accuracy: factors that might lead to a wrong diagnosis and how to avoid them. Rev Iberoam Psicol. 2019;12(2):29–38.
- MoCA Test Inc. MoCA Cognition FAQ. Accessed October 26, 2023. https://mocacognition.com/faq/
- Greenberg SA. The Geriatric Depression Scale (GDS). Best Practices in Nursing Care to Older Adults. 2012;4(1):1–2.
- van der Flier WM, Scheltens P. Epidemiology and risk factors of dementia. J Neurol Neurosurg Psychiatry. 2005;76(suppl 5):v2–v7. PubMed CrossRef
- Smits LL, Pijnenburg YAL, Koedam ELGE, et al. Early onset alzheimer’s disease is associated with a distinct neuropsychological profile. J Alzheimers Dis. 2012;30(1):101–108. PubMed CrossRef
- Elser H, Horváth-Puhó E, Gradus JL, et al. Association of early-, middle-, and late-life depression with incident dementia in a danish cohort. JAMA Neurol. 2023;80(9):949–958. PubMed
- Richmond-Rakerd LS, D’Souza S, Milne BJ, et al. Longitudinal associations of mental disorders with dementia: 30-year analysis of 1.7 million New Zealand citizens. JAMA Psychiatry. 2022;79(4):333–340. PubMed
- Kuring JK, Mathias JL, Ward L. Risk of dementia in persons who have previously experienced clinically significant depression, anxiety, or ptsd: a systematic review and meta-analysis. J Affect Disord. 2020;274:247–261. PubMed CrossRef
- Green RC, Cupples LA, Kurz A, et al. Depression as a risk factor for Alzheimer disease: the MIRAGE study. Arch Neurol. 2003;60(5):753–759. PubMed CrossRef
- Lombardi G, Crescioli G, Cavedo E, et al. Structural magnetic resonance imaging for the early diagnosis of dementia due to Alzheimer’s disease in people with mild cognitive impairment. Cochrane Database Syst Rev. 2020;3(3):CD009628. PubMed CrossRef
- Shipley SM, Frederick MC, Filley CM, et al. Potential for misdiagnosis in community-acquired PET scans for dementia. Neurol Clin Pract. 2013;3(4):305–312. PubMed CrossRef
Enjoy this premium PDF as part of your membership benefits!