Sexual Dysfunction in Male Subjects Receiving Trifluoperazine, Risperidone, or Olanzapine: Rates Vary With Assessment Questionnaire

ABSTRACT
Objective: To assess the rate and typology of sexual dysfunction in male subjects receiving trifluoperazine, risperidone, or olanzapine using the Arizona Sexual Experience Scale (ASEX), the Psychotropic Related Sexual Dysfunction Questionnaire (PRSexDQ), and the sexual function section of the modified Udvalg for Kliniske Unders׸gelser Side Effect Rating Scale (UKU).
Method: The sample included 100 men with psychotic disorders (F2 category of the ICD-10) and receiving trifluoperazine (n = 20), risperidone (n = 30), or olanzapine (n = 50) for at least 3 months’ duration. Subjects with a history of sexual dysfunction prior to antipsychotic intake or chronic medical illness were excluded. A cross-sectional design was employed, and data were collected over a 1½-year period from March 2009 to August 2010.
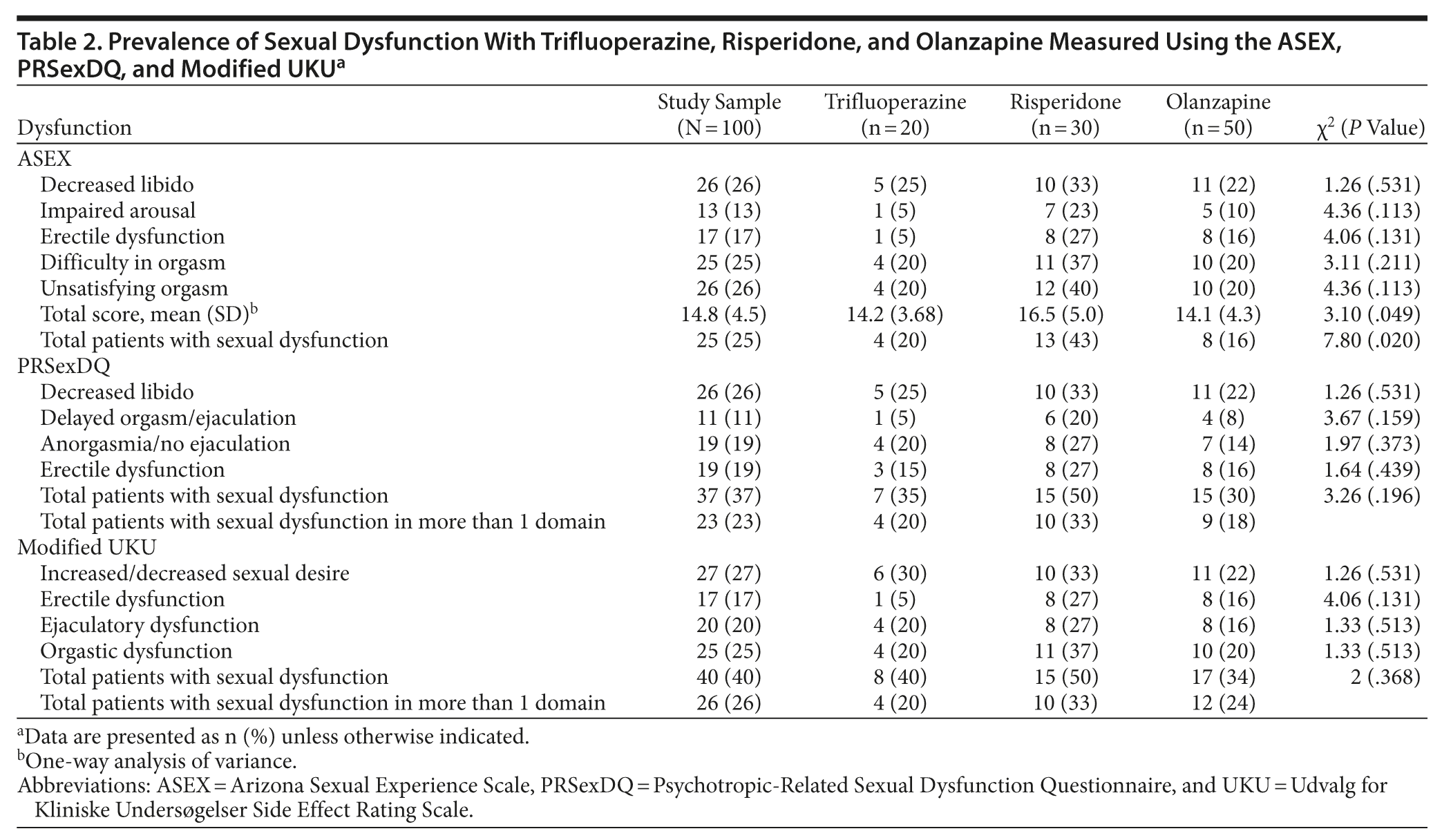
Results: The rate of sexual dysfunction varied from scale to scale among the 100 subjects. The rate of sexual dysfunction was 25% on the ASEX, 37% on the PRSexDQ, and 40% on the UKU. Sexual dysfunction in the trifluoperazine, risperidone, and olanzapine groups was 20%, 43%, and 16%, respectively, on the ASEX; 35%, 50%, and 30%, respectively, on the PRSexDQ; and 40%, 50%, and 34%, respectively, on the UKU. The most common sexual dysfunction as assessed on all scales was decreased libido, except for the risperidone group on the ASEX.
Conclusions: Sexual dysfunction is quite prevalent in subjects receiving antipsychotic medications. In our study, rate of sexual dysfunction was highest for risperidone, followed by trifluoperazine and olanzapine. However, the rate of sexual dysfunction varied from scale to scale. Hence, there is a need for a comprehensive instrument to assess sexual dysfunction in patients receiving antipsychotics.
Prim Care Companion CNS Disord 2012;14(2):doi:10.4088/PCC.11m01199
© Copyright 2012 Physicians Postgraduate Press, Inc.
Submitted: April 14, 2011; accepted June 15, 2011.
Published online: April 26, 2012.
Corresponding author: Sandeep Grover, MD, Department of Psychiatry, Postgraduate Institute of Medical Education and Research, Chandigarh 160012, India ([email protected]).
Antipsychotic medications form the mainstay of treatment of schizophrenia; however, one of the major limiting factors in their long-term use is side effects. Studies have shown that 25% to 60% of patients with schizophrenia treated with antipsychotics report sexual dysfunction.1,2 Studies have also shown that sexual dysfunction is an important factor contributing to noncompliance with antipsychotic medications.3,4 It is also suggested that psychiatrists commonly underestimate the presence of sexual dysfunction in patients with schizophrenia.5
Major problems in assessment of sexual dysfunction include the mode of assessment procedure (self-report, questionnaire, direct questioning), type of measurements used (objective versus subjective), and gender differences.
In terms of sexual dysfunction and type of antipsychotic, studies suggest that sexual dysfunction is more frequent with first-generation antipsychotics6,7 and risperidone8-11 and less frequent with quetiapine,8,11,12 olanzapine,9 ziprasidone,13 and aripiprazole.14 Four head-to-head comparison studies of sexual effects of second-generation antipsychotics found that risperidone was associated with greater sexual dysfunction compared with quetiapine and olanzapine; the lowest risk among the 3 antipsychotics was noted with quetiapine.7,8,15,16 In a review of studies published after 2002, the relative risk of sexual dysfunction with various antipsychotics was rated as risperidone > typical antipsychotics (haloperidol) > olanzapine > quetiapine > aripiprazole.17
Unfortunately, there are relatively sparse data from India with respect to sexual dysfunction with antipsychotics. One study assessed sexual dysfunction in patients receiving antipsychotics and reported a very high prevalence of sexual dysfunction (88%-99%) in patients receiving risperidone, olanzapine, or quetiapine compared with healthy controls (23%).18 Sexual dysfunction was regarded to be present in the study when the total score on the Sexual Functioning Questionnaire for a patient was at least 1 standard deviation above the mean for the healthy volunteer group.18 Another study that compared 2 groups of subjects receiving first- and second-generation antipsychotics who were evaluated for sexual dysfunction using the Udvalg for Kliniske Unders׸gelser Side Effect Rating Scale (UKU) reported a higher incidence of disturbance in libido and ejaculation with second-generation antipsychotics and more disturbance in erection and orgasm with first-generation antipsychotics.19
Most of the studies have assessed the prevalence and typology of sexual dysfunction in patients receiving antipsychotics using only 1 rating scale.7-11,13,18-21 In view of the lack of comparisons between various scales, it is difficult to reach a conclusion regarding the true prevalence of sexual dysfunction with antipsychotics.
In view of the above limitations, the primary aim of the present study was to assess the rate and typology of treatment-emergent sexual dysfunction in male subjects receiving antipsychotics using multiple questionnaires: the Arizona Sexual Experience Scale (ASEX),22 the Psychotropic-Related Sexual Dysfunction Questionnaire (PRSexDQ),23 and the sexual side effect section of the modified UKU.24 The secondary aim of the study was to measure the convergent validity of the various scales used for assessment of sexual dysfunction.

- Sexual dysfunction is very common in patients with schizophrenia who are receiving antipsychotics.
- The rate of sexual dysfunction varies with the assessment questionnaire that is used.
- There is a need to develop a valid and relevant instrument for assessing sexual dysfunction associated with use of psychotropics.
METHOD
This study was carried out at a multispecialty teaching tertiary care hospital in north India. The study was approved by the departmental research committee, and written informed consent was obtained from the participants prior to their recruitment into the study. A cross-sectional design was employed, and data were collected over a 1½-year period from March 2009 to August 2010.
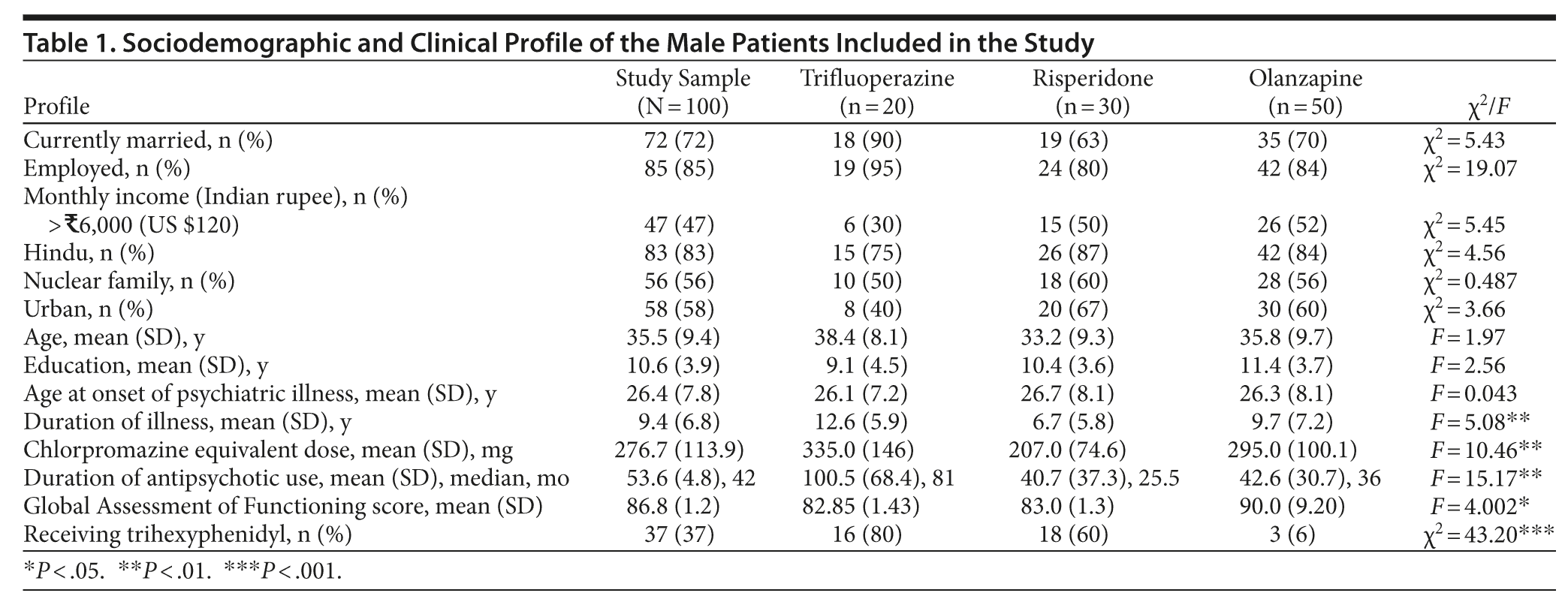
The sample consisted 100 men with psychotic illness (F2 category of the ICD-10) recruited by purposive sampling who were aged 18-50 years, heterosexual, sexually active (presence of common sexual intercourse or masturbatory practices), clinically stable (score on the Global Assessment of Functioning scale [GAF]25 of at least 50; no worsening of clinical symptoms to need an increase in the dose of antipsychotic in the last 6 weeks), and receiving trifluoperazine, risperidone, or olanzapine for at least 3 months’ duration. The use of antipsychotics for at least 3 months ensured a sufficient period of exposure to the antipsychotic before perceiving any sexual dysfunction. Patients receiving these antipsychotics were chosen because our dataset shows that olanzapine and risperidone are the most commonly prescribed second-generation antipsychotics at our center, and trifluoperazine is the most commonly prescribed first-generation antipsychotic.
As per the exclusion criteria, patients should not have had sexual dysfunction prior to starting the antipsychotic. This inclusion criterion was ascertained from the history given by the patient (and the spouse whenever available). Patients with comorbid psychiatric disorders, comorbid diagnosis of substance dependence including nicotine dependence (smoking ≥ 20 cigarettes/day) and consumption of alcohol daily (≥ 30 g/day), organic brain syndrome, and chronic comorbid medical illness that could cause sexual dysfunction (hypertension, diabetes mellitus, thyroid dysfunction, cardiovascular disorders such as angina and myocardial infarction, renal dysfunctions, and neurologic disorders such as stroke and spinal cord lesions) were also excluded. Such conditions were ruled out on the basis of history, physical examination, and investigations as needed. Patients who were receiving more than 1 antipsychotic were excluded. Patients receiving other concurrent medications that are known to cause sexual dysfunction (eg, propranolol, antidepressants, etc) on a regular basis, those taking phosphodiesterase inhibitors or any other drugs that could have increased the desire or improved the level of sexual functioning, and those whose spouse was suffering from sexual dysfunction (on the basis of the history) due to any cause were also excluded.
Sexual dysfunction was assessed by the ASEX, PRSexDQ, and sexual side effects section of the modified UKU. The GAF was used to assess the overall level of functioning.25
The ASEX is a 5-item self-report inventory that evaluates sexual functioning of patients taking psychotropic drugs. Each question represents 1 domain, ie, drive, arousal, penile erection/vaginal lubrication, ability to reach orgasm, and satisfaction from orgasm. The subjects are asked to rate their sexual functioning on a 6-point Likert scale. The ASEX is interpreted on the basis of a total score and/or assessment of scores on individual items. A total score ≥ 19, a score ≥ 5 on any 1 item, or a score ≥ 4 on any 3 items are considered to be indicators of sexual dysfunction. Higher ASEX scores reflect greater severity of sexual dysfunction. The ASEX has excellent internal consistency and test-retest reliability and favorable convergent and discriminant validity. The ASEX has shown good psychometric properties (internal reliability, construct and convergent validity) in outpatients with schizophrenia or schizoaffective disorder.26 For the current study, the ASEX was translated to Hindi using the standard World Health Organization methodology.27
The PRSexDQ is a 7-item questionnaire that evaluates the occurrence of sexual dysfunction. The first item is a screening item for the presence of sexual dysfunction of any type and the second item is used to record spontaneous reporting of sexual dysfunction to the interviewer. Items 3 through 6 evaluate 5 dimensions of sexual function according to severity (0 = none/never, 1 = mild/occasionally, 2 = moderate/often, and 3 = severe) or frequency: decrease/loss of libido, delay of orgasm or ejaculation, lack of orgasm or ejaculation, and erectile dysfunction/decrease of vaginal lubrication. The seventh item evaluates the level of tolerance of sexual dysfunction (1 = good, 2 = fair, and 3 = poor). Tolerance is defined as “good” when the patient has no concern even though some type of sexual dysfunction is present, “fair” when the sexual dysfunction provokes concern or distress in the patient and/or partner without any intention to discontinue treatment because of it, and “poor” when the patient is very concerned by the adverse effect with serious intention to discontinue treatment. Sexual dysfunction is defined as present if the patient scores ≥ 1 on any 1 of the third to sixth items of the PRSexDQ that evaluate the various dimensions of sexual function. The PRSexDQ has good psychometric properties for both depressed and psychotic patients.23,28 The internal reliability for research purposes is good (Cronbach α of 0.68), and the questionnaire has good convergent validity and sensitivity to tracking changes in sexual functioning in patients with schizophrenia.28
The items related to sexual function in the modified UKU24 were also used to assess sexual functioning. The items are useful in evaluating sexual side effects (increased or diminished sexual desire; erectile, ejaculatory, or orgasmic dysfunction; and vaginal dryness) and other reproductive side effects (menorrhagia, amenorrhea, galactorrhea, and gynecomastia) of antipsychotics. Each item is rated on a 4-point scale (0 = none or doubtful, 1 = present to a mild degree, 2 = present to a moderate degree, and 3 = present to a severe degree) by the clinician on the basis of information provided by the patient. Any patient who scored ≥ 1 on any of the items was considered to have sexual dysfunction. Studies have shown a very good interclass correlation, with reported reliability ranging from 0.3 to 0.9.24
Procedure
Patients with a diagnosis of a psychotic disorder (F2 of ICD-10) and who were currently clinically stable were approached. The study was explained and written informed consent was obtained. In total, 113 patients were approached by purposive sampling. Of these, 13 patients were excluded from the study because of nonfulfillment of inclusion and/or exclusion criteria (8 patients had sexual dysfunction prior to the start of the current antipsychotic treatment, 4 patients had chronic physical diseases with well-established negative effects on sexual functions, and 1 patient was receiving 2 antipsychotics). The final sample included 100 patients, of whom 50 were receiving olanzapine, 30 were receiving risperidone, and 20 were receiving trifluoperazine.
Statistical Analysis
The data were analyzed using SPSS version 14.0 for Windows (SPSS Inc, Chicago, Illinois). For continuous variables, mean and standard deviation with a range was calculated and for discontinuous variables (nominal and ordinal), frequencies and percentages were calculated. Different groups were compared using χ2 for categorical variables and t test or 1-way analysis of variance for continuous variables.
RESULTS
Sociodemographic and Clinical Profile
The sociodemographic and clinical profile of the study sample is given in Table 1. The mean GAF score was 86.8 (SD = 1.18; range, 50-100). About one-third of the sample (n = 37) were receiving trihexyphenidyl along with the antipsychotic at the time of assessment. There was no significant difference between the 3 groups with respect to any of the sociodemographic variables and the age at onset of the illness. However, those who were receiving trifluoperazine had a significantly longer duration of illness, were receiving higher doses of a chlorpromazine equivalent dose29 of antipsychotic medication, and were on treatment for a significantly longer duration of illness (Table 1). The most common psychiatric diagnosis was schizophrenia (63%), followed by psychosis not otherwise specified (16%).
Self-Perceived and Spontaneous Reporting of Sexual Dysfunction
On the PRSexDQ, which serves as a support for clinician inquiry, the first question, “Have you observed any change in sexual activity (excitement, erection, ejaculation of orgasm)?” and the second question, “Has the patient spontaneously reported this alteration, or was it necessary to expressly question him or her to discover the sexual dysfunction?” (for which the patient makes an impression of yes/no) assessed any change in sexual functioning after starting the medication. Only 8 patients reported sexual dysfunction spontaneously and another 29 reported sexual dysfunction on inquiry.
Sexual Dysfunction: Rate and Typology
It was found that the rate of sexual dysfunction in the study group varied according to the scale used. The rate of sexual dysfunction was 25% as per the ASEX, 37% as per the PRSexDQ, and 40% as per the modified UKU. However, on all scales, sexual dysfunction was highest in the risperidone group and lowest in the olanzapine group (Table 2). Among the various domains of sexual dysfunction, decreased libido was the most common sexual dysfunction in all 3 groups and on all 3 scales, except for the risperidone group on the ASEX (Table 2). On the ASEX, all patients had sexual dysfunction on more than 1 domain (n = 25), while 62.2% (23 subjects of 37) on the PRSexDQ and 65% (26 subjects of 40) on the UKU had sexual dysfunction on more than 1 domain. On the PRSexDQ, although 37% of patients were found to have sexual dysfunction, only 21.6% (8 of 37 subjects with sexual dysfunction) of the patients reported the same spontaneously. Further, among the 37 patients exhibiting sexual dysfunction on the PRSexDQ, 22 reported good tolerance and 15 reported to have fair tolerance to the disturbance.
It is important to note that on all of the scales, about half of the patients with sexual dysfunction were receiving trihexyphenidyl (ASEX: 13 of 25, PRSexDQ: 19 of 37, and UKU: 19 of 40).
Convergent Validity of Various Sexual Dysfunction Assessment Scales
The PRSexDQ picked up all cases of sexual dysfunction diagnosed using the ASEX; however, an additional 12 patients were diagnosed with sexual dysfunction using the PRSexDQ. Cohen κ for the ASEX and PRSexDQ was 0.724 (P = .000). Similarly, the UKU also picked up all cases of sexual dysfunction diagnosed by using the ASEX, and an additional 15 patients were found to have sexual dysfunction per the UKU. The Cohen κ for the ASEX and UKU was 0.667 (P = .000). In terms of the relationship between the PRSexDQ and UKU, again, the UKU picked up all cases of sexual dysfunction diagnosed by the PRSexDQ. The Cohen κ for the PRSexDQ and UKU was 0.937 (P = .000). The 3 additional cases of sexual dysfunction as per the UKU included 1 case of increased sexual desire.
Difference in Sociodemographic and Clinical Profile of Those With and Without Sexual Dysfunction
When sociodemographic and clinical profiles of subjects with sexual dysfunction were compared with those without sexual dysfunction, no significant difference was found on any of the sociodemographic and clinical variables except for significantly higher mean (SD) GAF scores in those without sexual dysfunction as defined by the ASEX (88.25 [11.08] vs 82.28 [13.22], t = −2.22, P = .029), PRSexDQ (88.85 [11.18] vs 83.18 [12.31], t = −2.35, P = .020), or UKU (88.83 [11.45] vs 83.65 [11.95], t = −2.17, P = .032).
Correlates of Sexual Dysfunction
None of the sociodemographic variables emerged as a correlate of sexual dysfunction as defined by any of the 3 scales. Among the clinical variables, duration of illness was negatively correlated with sexual dysfunction as defined by the ASEX (Spearman rank correlation = −0.201, P = .045), trihexyphenidyl coprescription was positively correlated with sexual dysfunction as defined by the PRSexDQ (Spearman rank correlation = 0.228, P = .023), and GAF scores were negatively correlated with sexual dysfunction as defined by any of the 3 scales (ASEX: Spearman rank correlation = −0.219, P = .029; PRSexDQ: Spearman rank correlation = −0.262, P = .008; UKU: Spearman rank correlation = −0.274, P = .006).
DISCUSSION
This study included a cohort of high-functioning patients with psychotic disorders, of whom 63% had chronic schizophrenia, 72% were married, 85% were employed, and the mean GAF was 86.8. These demographics ensured that information was obtained in a reliable manner. Since most of the patients were married, corroboration of information from the spouse, whenever required, was possible. This sociodemographic profile is different from that reported in some of the studies from our center and was possibly due to our preference for married subjects for this study.
Sexual dysfunction is an important limiting factor in ensuring compliance with antipsychotic medications.3,4 In developing countries such as India, where discussing sexual matters is considered a taboo, patients usually do not report sexual side effects of psychotropics on their own. This fact is exemplified by the finding of this study in which only 8 (21.62%) of 37 subjects who were taking antipsychotics reported sexual dysfunction spontaneously.
Some important limitations of the earlier studies are the lack of information about the baseline sexual functioning, inclusion of subjects with physical comorbidity, and assessment of sexual dysfunction using 1 of the available instruments.7-11,13,18-21 The present study tried to overcome the limitation of using only 1 instrument by assessing sexual dysfunction using 3 standardized scales, which have been extensively used in earlier studies. Our study provides evidence that one of the reasons for such a wide variation in the prevalence of sexual dysfunction reported in the literature may be due to the measures used for the assessment of sexual dysfunction. Although many validated instruments are available for assessment of sexual dysfunction, it is important to choose an appropriate instrument for assessment.
In the present study, only one-third of the sample was receiving anticholinergic medications; this finding is lower when compared to the use of concomitant anticholinergic medications reported in some of the earlier studies.19 It is known that the anticholinergic medications or antipsychotics with high anticholinergic properties can cause erectile dysfunction; however, most of the studies that have assessed sexual dysfunction in patients with schizophrenia have not reported on the confounding effect of anticholinergic medications, while studies that have evaluated prescription patterns in schizophrenia show high rates of concomitant use of anticholinergics.30 In the present study, sexual dysfunction was higher in the risperidone and trifluoperazine groups, in which trihexyphenidyl was a frequent coprescribed medication. Hence, it is possible that coprescription could have confounded the results.
In the present study, prevalence of sexual dysfunction was highest with risperidone, followed by trifluoperazine, and lowest with olanzapine. The difference was not significant among the 3 groups when sexual dysfunction was elicited except on the ASEX. This particular finding may lend credence to the findings of earlier studies that evaluated sexual dysfunction by using the Changes in Sexual Functioning Questionnaire31 or self-rated questionnaires32-34 and found no difference in sexual dysfunction with use of different antipsychotics.31-34 However, the significant difference in the rate of sexual dysfunction on the ASEX with the 3 antipsychotics, as found in the current study, is also supported by some of the previous studies.9,20,35-37 The rate of sexual dysfunction with various antipsychotics in the present study is also in the range reported by the earlier studies.7,9,18,31,35
The most common sexual dysfunction was decreased libido in all 3 groups, except for the risperidone group on the ASEX. The prevalence of decreased libido in the present study is again in the range reported by the previous studies.9,18,31,35
An important finding of the present study is that the rate of sexual dysfunction varies with the kind of scale used. This result suggests that there is a need to develop a more useful instrument that can assess sexual dysfunction more accurately. With respect to the concordance between various scales, the Cohen κ for the ASEX and UKU, ASEX and PRSexDQ, and PRSexDQ and UKU were 0.667, 0.724, and 0.937 (P = .000 for each), respectively. When one tries to understand the difference between the 3 scales used, although all have more than 2 options to rate various items, presence or absence of sexual dysfunction on the PRSexDQ and UKU are rated as present if there is mild dysfunction too, whereas on the ASEX, sexual dysfunction is defined as present if the patient rates a 4 or 5 or meets the total cutoff score. Variation in rate of SD as assessed on various scales suggests that the scoring system adapted by the PRSexDQ and UKU may be more useful not only in assessing the rate of sexual dysfunction but also in rating the severity of sexual dysfunction.
None of the sociodemographic variables in the present study had any relationship with presence of sexual dysfunction as defined by any of the scales. Among the clinical variables, only duration of illness was negatively correlated with sexual dysfunction as defined by the ASEX. This finding suggests that with longer duration of illness, patients get used to the sexual dysfunction and underreport the adverse event. It is known that the negative symptoms of schizophrenia and disturbed psychomotor performance can contribute to sexual dysfunction.6 However, psychopathology was not assessed as part of this study.
Trihexyphenidyl coprescription was positively correlated with sexual dysfunction as defined by the PRSexDQ, and GAF scores were negatively correlated with sexual dysfunction as defined by any of the 3 scales. These findings suggest that future studies should evaluate the impact of coprescribed trihexyphenidyl on the prevalence of sexual dysfunction. Further, these findings also suggest that presence of sexual dysfunction may lead to a poor level of functioning. Hence, identification and management of the sexual dysfunction is important for achieving a favorable outcome of the primary disorder. Previous studies have reported an association between sexual dysfunction and the dose of the antipsychotic used6,7,20; however, results of the present study do not support those findings.
Limitations
Our study is limited by a small sample size of male patients attending a tertiary care hospital and receiving trifluoperazine, risperidone, or olanzapine. The cross-sectional design and long treatment duration with the antipsychotics in our study might be associated with a survival bias. There is a significant difference in the duration of treatment between the 3 drugs, and this could influence the reported rates of sexual dysfunction. The study was limited to a unique group of high-functioning subjects with psychotic illnesses. Patients exhibiting the more severe forms of sexual dysfunction would be more prone to discontinue their treatment; hence, the rates of sexual dysfunction found in the present study may actually be underestimations. All of the participants were outpatients and thus not representative of the most severely ill patients who would have been hospitalized. The raters were not blind to the medication received; hence, a potential bias in favor or against a particular drug might have occurred.
CONCLUSIONS
Despite the above-mentioned limitations, our study suggests that about one-third of the patients receiving antipsychotics developed sexual dysfunction. Prevalence of sexual dysfunction was higher in patients receiving risperidone than in those receiving trifluoperazine and olanzapine. The rate of sexual dysfunction varied with the assessment questionnaire, and the modified UKU diagnosed more antipsychotic-associated sexual dysfunction compared to the ASEX and PRSexDQ. There is a need to develop a valid and relevant instrument for assessing antipsychotic use-associated sexual dysfunction.
Drug names: aripiprazole (Abilify), haloperidol (Haldol and others), olanzapine (Zyprexa), propranolol (Inderal, InnoPran, and others), quetiapine (Seroquel), risperidone (Risperdal and others), ziprasidone (Geodon).
Author affiliations: Department of Psychiatry, Postgraduate Institute of Medical Education and Research, Chandigarh, India.
Potential conflicts of interest: None reported.
Funding/sources: None reported.
REFERENCES
1. Demyttenaere K, De Fruyt J, Sienaert P. Psychotropics and sexuality. Int Clin Psychopharmacol. 1998;13(suppl 6):S35-S41. doi:10.1097/00004850-199807006-00007 PubMed
2. Peuskens J, Sienaert P, de Hert M. Sexual dysfunction: the unspoken side effect of antipsychotics. Eur Psychiatry. 1998;13(supp 1):23s-30s. PubMed
3. Smith S. Effects of antipsychotics on sexual and endocrine function in women: implications for clinical practice. J Clin Psychopharmacol. 2003;23(suppl 1):S27-S32. doi:10.1097/01.jcp.0000084035.22282.31 PubMed
4. Lambert M, Conus P, Eide P, et al. Impact of present and past antipsychotic side effects on attitude toward typical antipsychotic treatment and adherence. Eur Psychiatry. 2004;19(7):415-422. doi:10.1016/j.eurpsy.2004.06.031 PubMed
5. Dossenbach M, Hodge A, Anders M, et al. Prevalence of sexual dysfunction in patients with schizophrenia: international variation and underestimation. Int J Neuropsychopharmacol. 2005;8(2):195-201. doi:10.1017/S1461145704005012 PubMed
6. Smith SM, O’ Keane V, Murray R. Sexual dysfunction in patients taking conventional antipsychotic medication. Br J Psychiatry. 2002;181(1):49-55. doi:10.1192/bjp.181.1.49 PubMed
7. Bobes J, Garc A-Portilla MP, Rejas J, et al. Frequency of sexual dysfunction and other reproductive side-effects in patients with schizophrenia treated with risperidone, olanzapine, quetiapine, or haloperidol: the results of the EIRE study. J Sex Marital Ther. 2003;29(2):125-147. doi:10.1080/713847170 PubMed
8. Knegtering R, Castelein S, Bous H, et al. A randomized open-label study of the impact of quetiapine versus risperidone on sexual functioning. J Clin Psychopharmacol. 2004;24(1):56-61. doi:10.1097/01.jcp.0000106220.36344.04 PubMed
9. Knegtering H, Boks M, Blijd C, et al. A randomized open-label comparison of the impact of olanzapine versus risperidone on sexual functioning. J Sex Marital Ther. 2006;32(4):315-326. doi:10.1080/00926230600666378 PubMed
10. Komossa K, Rummel-Kluge C, Hunger H, et al. Sertindole versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2009;2(2):CD006752. PubMed
11. Westheide J, Cvetanovska G, Albrecht C, et al. Prolactin, subjective well-being and sexual dysfunction: an open label observational study comparing quetiapine with risperidone. J Sex Med. 2008;5(12):2816-2826. doi:10.1111/j.1743-6109.2008.00859.x PubMed
12. Montejo Gonzסlez AL, Rico-Villademoros F, Tafalla M, et al; Spanish Working Group for the Study of Psychotropic-Related Sexual Dysfunction. A 6-month prospective observational study on the effects of quetiapine on sexual functioning. J Clin Psychopharmacol. 2005;25(6):533-538. PubMed doi:10.1097/01.jcp.0000186872.04984.56
13. Montejo AL, Rico-Villademoros F; Spanish Working Group for the Study of Psychotropic-Related Sexual Dysfunction. Changes in sexual function for outpatients with schizophrenia or other psychotic disorders treated with ziprasidone in clinical practice settings: a 3-month prospective, observational study. J Clin Psychopharmacol. 2008;28(5):568-570. doi:10.1097/JCP.0b013e31818501a1 PubMed
14. Hanssens L, L’ Italien G, Loze JY, et al. The effect of antipsychotic medication on sexual function and serum prolactin levels in community-treated schizophrenic patients: results from the Schizophrenia Trial of Aripiprazole (STAR) study (NCT00237913). BMC Psychiatry. 2008;8(1):95. doi:10.1186/1471-244X-8-95 PubMed
15. Kim KS, Pae CU, Chae JH, et al. Effects of olanzapine on prolactin levels of female patients with schizophrenia treated with risperidone. J Clin Psychiatry. 2002;63(5):408-413. doi:10.4088/JCP.v63n0506 PubMed
16. Knegtering H, van der Moolen AE, Castelein S, et al. What are the effects of antipsychotics on sexual dysfunctions and endocrine functioning? Psychoneuroendocrinology. 2003;28(suppl 2):109-123. doi:10.1016/S0306-4530(02)00130-0 PubMed
17. Baggaley M. Sexual dysfunction in schizophrenia: focus on recent evidence. Hum Psychopharmacol. 2008;23(3):201-209. doi:10.1002/hup.924 PubMed
18. Nagaraj AK, Pai NB, Rao SA. A comparative study of sexual dysfunction involving risperidone, quetiapine, and olanzapine. Indian J Psychiatry. 2009;51(4):265-271. doi:10.4103/0019-5545.58291 PubMed
19. Nagaraj AK, Nizamie HS, Akhtar S, et al. A comparative study of sexual dysfunction due to typical and atypical antipsychotics in remitted bipolar I disorder. Indian J Psychiatry. 2004;46(3):261-266. PubMed
20. Montejo AL, Majadas S, Rico-Villademoros F, et al; Spanish Working Group for the Study of Psychotropic-Related Sexual Dysfunction. Frequency of sexual dysfunction in patients with a psychotic disorder receiving antipsychotics. J Sex Med. 2010;7(10):3404-3413. doi:10.1111/j.1743-6109.2010.01709.x PubMed
21. Uçok A, Incesu C, Aker T, et al. Sexual dysfunction in patients with schizophrenia on antipsychotic medication. Eur Psychiatry. 2007;22(5):328-333. doi:10.1016/j.eurpsy.2007.01.001 PubMed
22. McGahuey CA, Gelenberg AJ, Laukes CA, et al. The Arizona Sexual Experience Scale (ASEX): reliability and validity. J Sex Marital Ther. 2000;26(1):25-40. doi:10.1080/009262300278623 PubMed
23. Montejo AL, Garc×a M, Espada M, et al. Psychometric characteristics of the Psychotropic-Related Sexual Dysfunction Questionnaire. Spanish Work Group for the Study of Psychotropic-Related Sexual Dysfunctions. Actas Esp Psiquiatr. 2000;28(3):141-150. PubMed
24. Lingjaerde O, Ahlfors UG, Bech P, et al. The UKU Side Effect Rating Scale: A New Comprehensive Rating Scale for Psychotropic Drugs and a Cross-Sectional Study of Side Effects in Neuroleptic-Treated Patients. Acta Psychiatr Scand Suppl. 1987;76(s334):1-100. doi:10.1111/j.1600-0447.1987.tb10566.x PubMed
25. Endicott J, Spitzer RL, Fleiss JL, et al. The Global Assessment Scale: a procedure for measuring overall severity of psychiatric disturbance. Arch Gen Psychiatry. 1976;33(6):766-771. PubMed
26. Byerly MJ, Nakonezny PA, Bettcher BM, et al. Sexual dysfunction associated with second-generation antipsychotics in outpatients with schizophrenia or schizoaffective disorder: an empirical evaluation of olanzapine, risperidone, and quetiapine. Schizophr Res. 2006;86(1-3):244-250. doi:10.1016/j.schres.2006.04.005 PubMed
27. Sartorius N, Kuyken W. Translation of health status instruments. In: Orley J, Kuyken W, eds. Quality of Life Assessment: International Perspectives. Heidelberg, Germany: Springer Veralg; 1994.
28. Montejo AL, Rico-Villademoros F, Spanish Working Group for the Study of Psychotropic Related Sexual Dysfunction. Psychometric properties of the Psychotropic-Related Sexual Dysfunction Questionnaire (PRSexDQ-SALSEX) in patients with schizophrenia and other psychotic disorders. J Sex Marital Ther. 2008;34(3):227-239. doi:10.1080/00926230701866125 PubMed
29. Woods SW. Chlorpromazine equivalent doses for the newer atypical antipsychotics. J Clin Psychiatry. 2003;64(6):663-667. doi:10.4088/JCP.v64n0607 PubMed
30. Paton C, Lelliott P, Harrington M, et al. Patterns of antipsychotic and anticholinergic prescribing for hospital inpatients. J Psychopharmacol. 2003;17(2):223-229. doi:10.1177/0269881103017002012 PubMed
31. Fan X, Henderson DC, Chiang E, et al. Sexual functioning, psychopathology and quality of life in patients with schizophrenia. Schizophr Res. 2007;94(1-3):119-127. doi:10.1016/j.schres.2007.04.033 PubMed
32. Bitter I, Basson BR, Dossenbach MR. Antipsychotic treatment and sexual functioning in first-time neuroleptic-treated schizophrenic patients. Int Clin Psychopharmacol. 2005;20(1):19-21. doi:10.1097/00004850-200501000-00004 PubMed
33. Olfson M, Uttaro T, Carson WH, et al. Male sexual dysfunction and quality of life in schizophrenia. J Clin Psychiatry. 2005;66(3):331-338. doi:10.4088/JCP.v66n0309 PubMed
34. Westheide J, Cohen S, Bender S, et al. Sexual dysfunction in psychiatric inpatients the role of antipsychotic medication. Pharmacopsychiatry. 2007;40(4):140-145. doi:10.1055/s-2007-981480 PubMed
35. Dossenbach M, Dyachkova Y, Pirildar S, et al. Effects of atypical and typical antipsychotic treatments on sexual function in patients with schizophrenia: 12-month results from the Intercontinental Schizophrenia Outpatient Health Outcomes (IC-SOHO) study. Eur Psychiatry. 2006;21(4):251-258. doi:10.1016/j.eurpsy.2005.12.005 PubMed
36. Jayaram MB, Hosalli P. Risperidone versus olanzapine for schizophrenia. Cochrane Database Syst Rev. 2005;(2): CD005237. doi:10.1002/ 14651858.CD005237.
37. Sacristסn JA, Gómez JC, Montejo AL, et al. Doses of olanzapine, risperidone, and haloperidol used in clinical practice: results of a prospective pharmacoepidemiologic study. EFESO Study Group. Estudio Farmacoepidemiologico en la Esquizofrenia con Olanzapina. Clin Ther. 2000;22(5):583-599. doi:10.1016/S0149-2918(00)80046-6 PubMed